Mass Spectrometry: Using proteomics to probe neurons
Deconstructing the immensely complex molecular basis of neuronal networks in the mammalian brain would help us to better understand how neurons develop and form connections (called synapses) with each other. Significant strides in the analysis of gene expression have been made in recent years, revealing the regulatory programs that govern the fate of individual neurons, their interactions with diverse ligands, and their ability to adapt to changes, such as environmental cues and injuries (Schreiner et al., 2014). However, measures of gene expression might not reflect the actual levels of proteins produced in cells, so efforts are being made to study the proteins directly.
Evidence suggests that changes in the abundance or activity of proteins in synapses may lead to defects in neurons that are implicated in neuropsychiatric disorders (Craft et al., 2013). Therefore, uncovering the repertoire of proteins produced by neurons could help us understand the underlying molecular basis of such conditions. The large-scale study of the proteins found in cells—known as proteomics—poses significant challenges to researchers. For example, a single gene in a mammalian cell can be used as a template to make many different forms (or ‘isoforms’) of a protein through a process called alternative splicing. These isoforms can be very similar to each other, but they can play very different roles in cells, so it is important for proteomics techniques to be able to distinguish them. The situation is further complicated by the presence of large protein families encoded by very similar genes.
Mass spectrometry is the leading approach for proteomics investigations, and encompasses both global analyses of all the proteins found in cells, and ‘targeted’ approaches that accurately measure the abundance of particular proteins. Selected reaction monitoring (SRM) mass spectrometry is the most widely used technique for targeted proteomics (Elschenbroich and Kislinger, 2011). Proteins are extracted from cells and digested by enzymes to produce millions of fragments (called peptides). However, only a small fraction of these peptides will be unique to the protein or isoform of interest. SRM mass spectrometry is able to accurately determine protein abundance because it can be used to measure just those peptides we are interested in.
The absolute quantification of proteins by SRM involves ‘spiking’ the samples with known concentrations of labelled synthetic peptides after the enzyme treatment (Barr et al., 1996). However, variations in the efficiency of the enzymes can lead to errors with this approach. Alternative approaches use known concentrations of labelled whole synthetic proteins (Brun et al., 2007; Stergachis et al., 2011), which are added to the sample before the enzyme treatment.
However, the use of SRM mass spectrometry to quantify the isoforms of protein families from complex tissues had not been explored. Now, in eLife, Peter Scheiffele and colleagues at the University of Basel—including Dietmar Schreiner and Jovan Simicevic as joint first authors—have developed SRM assays to quantify the isoforms of the neurexin family of proteins in the mouse brain (Schreiner, Simicevic et al. 2015).
Neurexins are cell adhesion proteins that play important roles in the formation and differentiation of synapses (Zhang et al., 2010). All three of the genes that encode neurexin proteins contain various segments that can be removed from messenger RNA in different combinations by alternative splicing (Chih et al., 2006). The modified messenger RNA molecules are then translated to make the different neurexin protein isoforms. The segments have been shown to regulate the interactions between ligands and their receptors on the surface of neurons and to alter the activity of synapses in a variety of ways (Aoto et al., 2013).
Schreiner, Simicevic et al. made protein standards that contained different neurexin isoforms fused to the fluorescent protein GFP before carrying out SRM assays (Figure 1). They found that, in several different regions of the brain, neurexin isoforms that contain the segments known as AS3 and AS4 were regulated in the same way, but isoforms that contain another segment called AS6 were regulated independently.
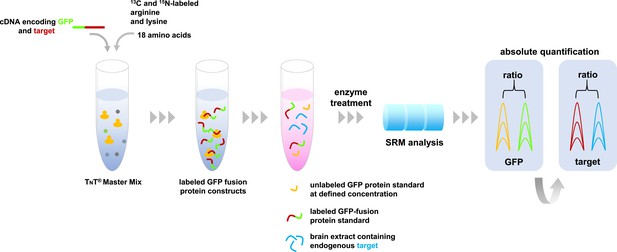
Absolute quantification of proteins by SRM mass spectrometry.
A master mix containing the components required for protein synthesis, the amino acids arginine and lysine labeled with carbon-13 and nitrogen-15, and all other unlabeled amino acids are mixed with cDNA molecules that encode the proteins of interest fused with green fluorescent protein (GFP). These fusion proteins (indicated in green-red) are combined with a sample containing the target proteins of interest (depicted in blue), and a known concentration of unlabeled GFP as an internal standard (shown in yellow). This protein mixture is digested by enzymes and the resulting peptides are analyzed by SRM mass spectrometry that specifically measures peptides that are unique to the target proteins. The resulting SRM traces contain information about the abundance of these peptides. The known spiked concentration of the unlabeled GFP standard (yellow) is used to determine the absolute amounts of the labeled GFP fusion protein standard (the green part of the fusion protein). In turn, the target protein component of the GFP fusion protein standard (the red section) is used to determine the absolute amounts of the target proteins in the biological sample (shown in blue).
The absolute quantification of the neurexins revealed that two of the isoforms (called alpha and beta) differed in abundance and that, overall, neurexins are present at relatively high levels across the whole mouse brain. The ability of neurexin isoforms to interact with ligands at synapses was associated with specific segments, which demonstrates that alternative splicing can modulate neurexin activity.
The SRM assays developed by Schreiner, Simicevic et al. enabled them to discriminate between neurexin isoforms that only differed in a single amino acid residue. Their simple approach can be applied to other protein families in any accessible tissue, and hence may be applied to a range of biological questions.
References
-
Isotope dilution–mass spectrometric quantification of specific proteins: model application with apolipoprotein A-IClinical Chemistry 42:1676–1682.
-
Isotope-labeled protein standards: toward absolute quantitative proteomicsMolecular & Cellular Proteomics 6:2139–2149.https://doi.org/10.1074/mcp.M700163-MCP200
-
Rapid empirical discovery of optimal peptides for targeted proteomicsNature Methods 8:1041–1043.https://doi.org/10.1038/nmeth.1770
Article and author information
Author details
Publication history
- Version of Record published: July 2, 2015 (version 1)
Copyright
© 2015, Kim and Kislinger
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 1,375
- views
-
- 170
- downloads
-
- 0
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Biochemistry and Chemical Biology
- Cell Biology
Mediator of ERBB2-driven Cell Motility 1 (MEMO1) is an evolutionary conserved protein implicated in many biological processes; however, its primary molecular function remains unknown. Importantly, MEMO1 is overexpressed in many types of cancer and was shown to modulate breast cancer metastasis through altered cell motility. To better understand the function of MEMO1 in cancer cells, we analyzed genetic interactions of MEMO1 using gene essentiality data from 1028 cancer cell lines and found multiple iron-related genes exhibiting genetic relationships with MEMO1. We experimentally confirmed several interactions between MEMO1 and iron-related proteins in living cells, most notably, transferrin receptor 2 (TFR2), mitoferrin-2 (SLC25A28), and the global iron response regulator IRP1 (ACO1). These interactions indicate that cells with high MEMO1 expression levels are hypersensitive to the disruptions in iron distribution. Our data also indicate that MEMO1 is involved in ferroptosis and is linked to iron supply to mitochondria. We have found that purified MEMO1 binds iron with high affinity under redox conditions mimicking intracellular environment and solved MEMO1 structures in complex with iron and copper. Our work reveals that the iron coordination mode in MEMO1 is very similar to that of iron-containing extradiol dioxygenases, which also display a similar structural fold. We conclude that MEMO1 is an iron-binding protein that modulates iron homeostasis in cancer cells.
-
- Cell Biology
- Chromosomes and Gene Expression
Heat stress is a major threat to global crop production, and understanding its impact on plant fertility is crucial for developing climate-resilient crops. Despite the known negative effects of heat stress on plant reproduction, the underlying molecular mechanisms remain poorly understood. Here, we investigated the impact of elevated temperature on centromere structure and chromosome segregation during meiosis in Arabidopsis thaliana. Consistent with previous studies, heat stress leads to a decline in fertility and micronuclei formation in pollen mother cells. Our results reveal that elevated temperature causes a decrease in the amount of centromeric histone and the kinetochore protein BMF1 at meiotic centromeres with increasing temperature. Furthermore, we show that heat stress increases the duration of meiotic divisions and prolongs the activity of the spindle assembly checkpoint during meiosis I, indicating an impaired efficiency of the kinetochore attachments to spindle microtubules. Our analysis of mutants with reduced levels of centromeric histone suggests that weakened centromeres sensitize plants to elevated temperature, resulting in meiotic defects and reduced fertility even at moderate temperatures. These results indicate that the structure and functionality of meiotic centromeres in Arabidopsis are highly sensitive to heat stress, and suggest that centromeres and kinetochores may represent a critical bottleneck in plant adaptation to increasing temperatures.





