Characterisation of cell-scale signalling by the core planar polarity pathway during Drosophila wing development
Figures

Planar polarity in the Drosophila wing.
(A) Core proteins form asymmetric intercellular protein complexes at apico-lateral cell membranes mediating intercellular communication. Frizzled (Fz, green), a seven-pass transmembrane protein, and the cytoplasmic proteins Dishevelled (Dsh, dark blue) and Diego (Dgo, magenta) localise to distal cell membranes, while Strabismus (Stbm, orange, also known as Van Gogh [Vang]), a four-pass transmembrane protein and Prickle (Pk, light blue), a cytoplasmic protein, localise to proximal cell membranes. Flamingo (Fmi, red, also known as Starry Night [Stan]), an atypical seven-pass transmembrane cadherin, localises proximally and distally, forming a trans homodimer. (A’) Core protein complexes are thought to interact between themselves through feedback interactions locally on cell junctions (Molecular-scale) to form stable clusters, with positive interactions stabilising complexes of the same orientation (green arrows) or negative interactions destabilising complexes of opposite orientation (black symbols). (B–B’’) Core protein complexes are segregated to opposite cell membranes generating the specific proximo-distal polarised zig-zag core protein localisation pattern which can be promoted by ‘Cell-scale signalling’. In this study, we consider several hypotheses to identify such cell-scale signals, such as being mediated through depletion of a limiting pool of a core protein providing long-range inhibition (red symbols) (B), through oriented microtubules (MTs) transport (MTs in cyan, with motor proteins) (B’), or by a biochemical mechanism such as diffusion of an inhibitor (B’’). (C, C’) In the 28 hr pupal wing, core protein polarity (represented by lines with Stbm in orange and Fz in green) and trichome (black hair) orientation is distal (C’). It is suggested that this results from radial global cues generating an initial asymmetric bias across the tissue. During earlier polarity establishment in the prepupal wing, hinge contraction (brown arrows) induces cell rearrangements and a redistribution of the core proteins, from the radial polarity pattern in the prepupal wing (C) to aligned core protein polarity along the proximo-distal axis of the pupal wing by 28 hr APF (C’).
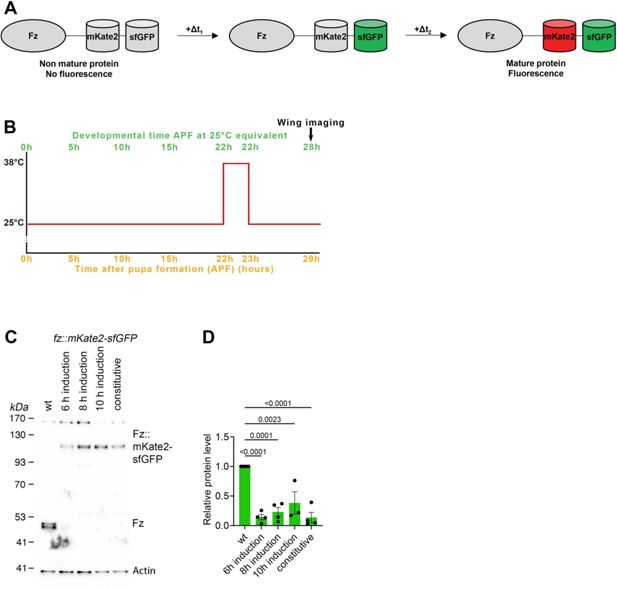
Tools for Fz induction and measurement of stable fractions.
(A) Schematic illustration of evolution over time of fluorescent timer (mKate2 and sfGFP) fused with Fz. (B) Scheme of induced Fz tagged with fluorescent timer expression by heat shock induction, with Fz observation at 28 hr APF. Here is represented the condition with 6 hr between tagged Fz expression induction and its observation. With 8 hr between tagged Fz expression induction and its observation at 28 hr APF, heat shock is performed at 20 hr APF and for 10 hr time lapse, at 18 hr APF. (C) Western blot probed for Fz protein with Actin for loading control. Extracts are from pupal wings at 28 hr APF with wild-type Fz or constitutive or de novo induced (6, 8, and 10 hr induction) Fz::mKate2-sfGFP expression, and two wings were loaded for each sample. (D) Quantification from four biological replicates of Fz and Fz::mKate2-sfGFP levels in conditions described in (C), normalised with wild-type Fz level. Error bars are SEM. ANOVA with Tukey’s multiple comparisons test was used to compare all genotypes, only significant p values are indicated.
-
Figure 1—figure supplement 1—source data 1
zip file containing original uncropped western blots for Figure 1—figure supplement 1.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig1-figsupp1-data1-v1.zip
-
Figure 1—figure supplement 1—source data 2
zip file containing original western blots for Figure 1—figure supplement 1, indicating the relevant bands and treatments.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig1-figsupp1-data2-v1.zip
-
Figure 1—figure supplement 1—source data 3
Excel file containing individual data points for panel D.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig1-figsupp1-data3-v1.xlsx

Dynamics of planar polarity establishment.
Schematics of localisation of core planar polarity pathway protein complexes in cells of the Drosophila pupal wing at 28 hr APF, in non-polarised cells (A), before polarity induction (B), after polarity induction (6, 8, or 10 hr) (C), or in the control condition with constitutive expression of Fz::mKate2-sfGFP (D), with localisation of Stbm (orange) and Fz (green) at apico-lateral cell junctions indicated. See Figure 1—figure supplement 1B for induction timings. (E–I) Live confocal images of 28 hr APF pupal wing epithelia expressing Fz::mKate2-sfGFP taken below longitudinal vein 4, see Figure 1C’ right-hand box. See Supplementary file 1 for full genotypes. Scale bar, 4 µm and the same hereafter. Native fluorescence for sfGFP (green, E'–I') and mKate2 (red, E''–I''), with de novo condition with 6 hr induction in a dsh1 mutant background (E), with de novo condition with induction for 6 hr (F), 8 hr (G), and 10 hr (H) to establish core protein planar polarity; and in the control condition with constitutive expression of Fz::mKate2-sfGFP which gives a normal core protein proximo-distal polarity (I). See Figure 1—figure supplement 1A for details of Fz tagged to form a fluorescent timer. (E’’’–I’’’) Cell-by-cell polarity pattern of mKate2 fluorescence in pupal wings expressing Fz::mKate2-sfGFP at 28 hr APF. The length and orientation of cyan bars denote the polarity magnitude and angle, respectively, for a given cell. Quantified polarity magnitude based on mKate2 fluorescence (J) or fraction of stable Fz as determined by ratio of mKate2/sfGFP fluorescence (K), in live pupal wings at 28 hr APF, in the conditions described in (E–I). Error bars are standard deviation (SD); n, number of wings. ANOVA with Tukey’s multiple comparisons test was used to compare all genotypes, p values as indicated. (L, M) Images of dorsal surface of mounted adult Drosophila wings, below longitudinal vein 4. Scale bar 50 μm. (L) Control condition of constitutive Fz::mKate2-sfGFP expression, showing uniform distal orientation of trichomes. (M) De novo condition after induction at 22 hr APF, at the equivalent time to the 6 hr induction when imaging at 28 hr APF, showing swirled trichome pattern. Proximal is left, and anterior is up.
-
Figure 2—source data 1
Excel file containing individual data points for panel J.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig2-data1-v1.xlsx
-
Figure 2—source data 2
Excel file containing individual data points for panel K.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig2-data2-v1.xlsx

Effect of core protein dosage during de novo planar polarity establishment.
(A–G) Live confocal images of 28 hr APF pupal wing epithelia expressing Fz::mKate2-sfGFP, in de novo condition taken below longitudinal vein 4. Native fluorescence for sfGFP (green, A’–G’) and mKate2 (red, A’’–G’’), with de novo condition with 6 hr induction to establish core protein planar polarity in an otherwise wild-type background (A), heterozygous mutant backgrounds for dgo (B), pk (C), dsh1 (D), stbm (E), fmi (F), and hemizygous mutant background for dsh1 (G). See Supplementary file 1 for the full genotypes. See Figure 3—figure supplement 2 for live confocal images of equivalent 28 hr APF pupal wing epithelia with constitutive Fz::mKate2-sfGFP expression. (A’’’–G’’’) Cell-by-cell polarity pattern of mKate2 fluorescence in pupal wings expressing Fz::mKate2-sfGFP at 28 hr APF. The length and orientation of cyan bars denote the polarity magnitude and angle for a given cell, respectively. Scale bar, 4 µm. Quantified Fz-mKate2 polarity magnitude based on mKate2 fluorescence (H, I) or fraction of stable Fz as determined by ratio of mKate2/sfGFP (J, K) in live pupal wings at 28 hr APF, in the conditions described in (A–G). Error bars are SD; n, number of wings. ANOVA with Dunnett’s multiple comparisons tests for quantified Fz-mKate2 polarity and ANOVA with Kruskal–Wallis multiple comparisons for fraction of stable Fz were used to compare wild-type to mutant backgrounds, p values as indicated. See also Figure 3—figure supplement 2.
-
Figure 3—source data 1
Excel file containing individual data points for panel H.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-data1-v1.xlsx
-
Figure 3—source data 2
Excel file containing individual data points for panel I.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-data2-v1.xlsx
-
Figure 3—source data 3
Excel file containing individual data points for panel J.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-data3-v1.xlsx
-
Figure 3—source data 4
Excel file containing individual data points for panel K.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-data4-v1.xlsx

Decreasing prickle, flamingo, and diego gene dosage results in lower protein levels.
Western blots and quantifications from pupal wings, comparing levels of Pk from pk-sple13/+ heterozygous flies (A), or levels of Fmi from fmiE59/+ heterogygous flies to wild-type (B), or comparing levels of EGFP reflecting Dgo levels from dgo380/EGFP-dgo, dgo380 heterozygous flies to EGFP-dgo, dgo380/EGFP-dgo, dgo380 flies (C). EGFP-Dgo is expressed from a rescue transgene in a dgo380 null mutant background. We used this as a proxy for endogenous Dgo levels, as we do not have an antibody against Dgo that works on western blots. Quantifications are from four biological replicates, unpaired t-test. Two pupal wings were loaded for Pk and Fmi and 8 for the EGFP-Dgo western blot. (D) Western blot showing EGFP band (arrow) in EGFP-Dgo compared to wild-type pupal wings to demonstrate that the correct EGFP band was identified, as the anti-EGFP antibody used also gives several cross-reacting bands. Eight pupal wings were loaded for this blot.
-
Figure 3—figure supplement 1—source data 1
zip file containing original uncropped western blots for Figure 3—figure supplement 1.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-figsupp1-data1-v1.zip
-
Figure 3—figure supplement 1—source data 2
zip file containing original western blots for Figure 3—figure supplement 1, indicating the relevant bands and treatments.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-figsupp1-data2-v1.zip
-
Figure 3—figure supplement 1—source data 3
Excel file containing individual data points for panel A.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-figsupp1-data3-v1.xlsx
-
Figure 3—figure supplement 1—source data 4
Excel file containing individual data points for panel B.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-figsupp1-data4-v1.xlsx
-
Figure 3—figure supplement 1—source data 5
Excel file containing individual data points for panel C.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-figsupp1-data5-v1.xlsx

Core protein polarity patterns under control conditions and in de novo condition with 8 hr induction to establish planar polarity.
(A–G) Live confocal images of 28 hr APF pupal wing epithelia expressing Fz::mKate2-sfGFP taken below longitudinal vein 4, see Figure 1C’. See Supplementary file 1 for the full genotypes. Native fluorescence for sfGFP (green, A'–G') and mKate2 (red, A''–G''), with constitutive expression of Fz::mKate2-sfGFP in an otherwise wild-type background (A), or in heterozygous mutant backgrounds for dgo (B), pk (C), dsh1 (D), stbm (E), fmi (F), and hemizygous mutant background for dsh1 (G). See Figure 1—figure supplement 1B for details of Fz tagged with fluorescent timer. (A’’’–G’’’) Cell-by-cell polarity pattern of mKate2 fluorescence in pupal wings expressing Fz::mKate2-sfGFP at 28 hr APF. The length and orientation of cyan bars denote the polarity magnitude and angle, respectively, for a given cell. Scale bar, 4 µm. Quantified polarity magnitude based on mKate2 fluorescence (H) or fraction of stable Fz as determined by ratio of mKate2/sfGFP (I), in live pupal wings at 28 hr APF, in wild-type or core protein heterozygous mutant backgrounds in de novo condition with 8 hr induction of Fz:mKate2-sfGFP to establish core protein planar polarity. Error bars are SD; n, number of wings. ANOVA with Dunnett’s multiple comparisons tests was used to compare wild-type to mutant backgrounds, p values as indicated.
-
Figure 3—figure supplement 2—source data 1
Excel file containing individual data points for panel H.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-figsupp2-data1-v1.xlsx
-
Figure 3—figure supplement 2—source data 2
Excel file containing individual data points for panel I.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig3-figsupp2-data2-v1.xlsx

Apical microtubules and core proteins are oriented independently of each other.
Apical view of cells with deconvolved confocal images of 28 hr APF fixed pupal wing epithelia, distal to the posterior cross vein, below longitudinal vein 4, in a wild-type background (A), in de novo Fz polarity establishment condition in a fz mutant background without polarity induction (B), and with polarity induction for 6 hr (C). Native Fz-eYFP fluorescence (yellow, A’, B', C’), immunolabelling for Stbm (green, A’’, B’’, C’’) and ⍺Tubulin (red, A’’’, B’’’, C’’’). Note there is no Fz-eYFP fluorescence at cell junctions detected in A’, B’. Cell-by-cell polarity pattern of pupal wings in respective conditions (A’’’’, B’’’’, C’’’’). The length and orientation of cyan bars denote the polarity magnitude and angle, respectively, for a given cell. See Supplementary file 1 for the full genotypes. See Figure 1C’ for vein locations and imaged wing area. Scale bar, 4 µm. Polar histograms depicting binned cell orientation relative to the horizontal axis (D–F), Stbm polarity orientation (G–I), and microtubule orientation (J–L) relative to the average cell orientation, in wild-type wings (D, G, J), in de novo condition without (E, H, K) or with (F, I, L) 6 hr Fz-eYFP polarity induction in fixed pupal wings at 28 hr APF. n = 7–9 wings for each condition. See also Figure 4—figure supplement 1A–E. (M) Cartoon of hedgehog expression domain (yellow) in the posterior part of the Drosophila pupal wing at 28 hr APF with a zoom in on the proximal region to the anterior cross vein between longitudinal vein 3 and longitudinal vein 4 (blue dash square). Grey cells are vein cells, white cells are intervein cells and yellow cells are intervein cells with hedgehog expression. Boundary 0 is between intervein cells expressing or not expressing hedgehog and the first row of intervein cells in contact with cells expressing hedgehog across boundary 0 is cell row 0 with anteriorly rows 1–3. Apical view of cells from deconvolved confocal images of 28 hr APF fixed pupal wings, in the region proximal to the anterior cross vein between longitudinal vein 3 and longitudinal 4, in an otherwise wild-type background without Fz repolarisation induction (N) and with Fz repolarisation induction for 6 hr (O). For Fz repolarisation conditions, induced Fz is over-expressed in the hedgehog expression domain (posterior wing part) with the UAS-GAL4 system (M). Immunolabelling as described in A–C, against Fz (yellow, N’, O’), Stbm (green, N’’, O’’), and ⍺Tubulin (red, N’’’, O’’’). Cell-by-cell polarity pattern of pupal wings in respective conditions (N’’’’, O’’’’). The length and orientation of cyan bars denote the polarity magnitude and angle, respectively, for a given cell. See Supplementary file 1 for the full genotypes. See M and Figure 1C’ for vein locations and imaged wing areas. Polar histograms depicting binned cell orientation relative to the horizontal axis (P, Q), Stbm polarity orientation (R, S), and microtubule orientation (T, U) relative to the average cell orientation, without (P, R, T) or with (Q, S, U) Fz repolarisation induction for 6 hr in fixed pupal wings at 28 hr APF. Cells are grouped in rows relative to their location relative to Fz overexpression (hedgehog expression domain) in Fz repolarisation condition or relative to longitudinal vein 4 without Fz repolarisation, with row 0 in contact with the hh-GAL4 overexpression boundary and row 3 furthest away. n = 8 wings for both conditions. See also Figure 4—figure supplement 1F–J.
-
Figure 4—source data 1
Excel file containing individual data points for panels D–L.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig4-data1-v1.xlsx
-
Figure 4—source data 2
Excel file containing individual data points for panels P–U.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig4-data2-v1.xlsx

Quantification of cell shape, Stbm polarity, and microtubule orientation in wild-type and de novo and repolarisation conditions at 28 hr APF.
Quantifications from conditions in Figure 4A–C of cell orientation relative to horizontal axis (A), cell eccentricity (B), Stbm orientation angle variance (C), Stbm polarity magnitude (D), and microtubule orientation (E) relative to the average cell orientation, in wild-type wings, in de novo condition without induction (no de novo) or with (de novo) Fz-eYFP polarisation induction for 6 hr, in fixed pupal wings at 28 hr APF, in the region distal to the posterior cross vein, below longitudinal vein 4. Error bars are SD; n, number of wings. ANOVA with Tukey’s multiple comparisons tests was used to compare all genotypes, p values as indicated. See Supplementary file 1 for the full genotypes. Quantifications from conditions in Figure 4N–O of cell orientation relative to horizontal axis (F), cell eccentricity (G), Stbm orientation angle (H), Stbm polarity magnitude (I), and microtubule orientation (J) relative to the average cell orientation, without (no repol.) or with Fz (repol.) repolarisation induction for 6 hr, in fixed pupal wings at 28 hr APF. Cells are grouped in rows relative to their location relative to Fz overexpression in Fz repolarisation condition or relative to longitudinal vein 4 without Fz repolarisation with row 0 in contact with Fz overexpression boundary and row 3 furthest away. Error bars are SD; n, number of wings. Mann–Whitney tests (F, G, J) and unpaired Mann–Whitney tests (H, I) were used to compare in each condition with or without Fz repolarisation polarisation induction, p values as indicated. See Supplementary file 1 for the full genotypes.
-
Figure 4—figure supplement 1—source data 1
Excel file containing individual data points for panels A–E.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig4-figsupp1-data1-v1.xlsx
-
Figure 4—figure supplement 1—source data 2
Excel file containing individual data points for panels F–J.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig4-figsupp1-data2-v1.xlsx

Reorientation of planar polarity from a boundary under control and de novo conditions.
Images of dorsal surface of adult Drosophila wings, taken proximal to the anterior cross vein between longitudinal vein 3 and longitudinal vein 4, showing trichome orientation in control condition with constitutive Fz::mKate2-sfGFP expression (A), in de novo Fz::mKate2-sfGFP induction condition (B), in repolarisation from the hh-GAL4 boundary with constitutive Fz::mKate2-sfGFP expression condition (C), and in repolarisation from the hh-GAL4 boundary under de novo Fz::mKate2-sfGFP induction condition (D). Repolarisation conditions are under UAS-GAL4 control for Fz overexpression in the posterior wing (hedgehog expression domain), with induced Fz expression from UAS-FRT-STOP-FRT-Fz in the presence of hsFLP. Note, the swirling pattern of polarity generated by de novo induction generates a proximo-distal trichome orientation in this analysed wing area (B) but not in surrounding wing regions (Figure 5—figure supplement 1B–D). Trichome polarity is reoriented in the antero-posterior direction in both repolarisation conditions. See Figures 4M and 1C’ for vein locations and imaged wing area. See Supplementary file 1 for the full genotypes. Scale bar, 50 µm (E–H) Planar polarity measurement at the cellular scale in the region proximal to the anterior cross vein, between longitudinal vein 3 and longitudinal vein 4 in fixed pupal wings at 28 hr APF. The length and orientation of cyan bars denote the polarity magnitude and angle for a given cell, respectively. De novo condition with 6 hr induction of Fz::mKate2-sfGFP (E), constitutive expression of Fz::mKate2-sfGFP (F), repolarisation condition with constitutive expression of Fz::mKate2-sfGFP (G), and repolarisation condition with 6 hr de novo induction of Fz::mKate2-sfGFP (H). In repolarisation conditions, induced Fz is over-expressed in the posterior wing, whereas in the anterior wing only Fz::mKate2-sfGFP is expressed. In the posterior wing, the two Fz populations (over-expressed Fz and Fz::mKate2-sfGFP) compete for the same membrane locations and Fz::mKate2-sfGFP signal is not evident. Scale bar, 10 µm (I–L) Circular plots of quantified total Fz (Fz::sfGFP) magnitude and orientation relative to horizontal axis in the region proximal to the anterior cross vein, between longitudinal vein 3 and longitudinal vein 4 at 28 hr APF in fixed pupal wings. Small dots show polarity angle and magnitude for individual wings, arrows show average polarity and magnitude across all wings. Cells are grouped in rows relative to their location relative to the Fz overexpression domain in Fz repolarisation condition or relative to longitudinal vein 4 without Fz repolarisation, with row 0 in contact with Fz overexpression boundary and row 3 furthest away. (I–I’’’) de novo condition with 6 hr to establish core protein polarity, (J–J’’’) control condition with constitutive Fz::mKate2-sfGFP expression, (K–K’’’) repolarisation condition for 6 hr to re-orient core protein polarity, and (L–L’’’) repolarisation under de novo condition for 6 hr induced Fz::mKate2-sfGFP expression; in row 0 (I–L), in row 1 (I’–L’), in row 2 (I’’–L’’), and in row 3 (I’’’–L’’’). Vertical black lines associated with p values on right of each column represent comparisons between different rows of the same polarisation condition. Horizontal pale blue lines associated with p values represent comparison between the two adjacent polarisation conditions for the same cell row. On the far right, horizontal red underlined p values represent comparison between repolarisation in de novo condition versus de novo condition (far left) for the respective rows of cells. Hotelling’s T-square tests were used to compare total Fz polarity (orientation and magnitude).
-
Figure 5—source data 1
Excel file containing individual data points for panels I–L.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig5-data1-v1.xlsx

Trichome and core protein polarity in normal and de novo conditions.
(A) Confocal microscope image proximal to the anterior cross vein (ACV) between the longitudinal vein 3 (LV 3) and the longitudinal vein 4 (LV 4) in a fixed wing at 33 hr APF. Wings stained for GFP revealing Fz localisation (green) (A, A’), and for actin showing trichomes (red) (A, A’’). In cells with Fz repolarisation on horizontal junctions, trichomes emerge repolarised along these same junctions with an antero-posterior orientation, observable in rows 0, 1, 2, and 3. Away from the Fz repolarisation area, trichomes emerge on distal cell edges with a proximo-distal orientation. Orange arrows indicate boundary 1, in row 0 which is the first repolarised row of cells. Scale bar 10 µm. For wing area localisation, see Figure 4M and for cell and boundary localisations, see Figure 6A. See Supplementary file 1 for the full genotype. Microscope images of adult wings surrounding the experimentally analysed region in pupal wings proximal to the anterior cross vein (ACV), for de novo induction conditions with 6 hr (B), 8 hr (C), and 10 hr (D). Scale bar 50 µm. Note similar patterns seen under all induction conditions, with a swirling non-proximo-distal trichome orientation surrounding the proximo-distally oriented region just proximal to the ACV between LV 3 and LV 4. Polar histograms depicting binned Fz-mKate2 polarity orientation, based on mKate2 fluorescence in live pupal wings at 28 hr APF, with de novo condition with induction for 6 hr (E), 8 hr (E’), 10 hr (E’’), and in the control condition (E’’’).
-
Figure 5—figure supplement 1—source data 1
Excel file containing individual data points for panel E.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig5-figsupp1-data1-v1.xlsx

Distribution of Fz along medio-lateral and horizontal junctions in Fz repolarisation and repolarisation under de novo conditions.
(A) Explanatory diagram for wing region, cell rows and cell boundaries. Visualisation of the anterior part of the anterior cross vein (ACV) between the longitudinal vein 3 (LV 3) and the longitudinal vein 4 (LV 4) in a fixed pupal wing at 28 hr APF. Green area indicates the expression domain of the hh-GAL4 driver used to over-express Fz. E-Cadherin staining is in blue and Stbm staining in magenta. Junctions with horizontal orientation are schematised with grey lines, forming horizontal boundaries with boundary 0 between last row of cells over expressing Fz in LV 4 and the first row of cells without over expression of Fz in the intervein area (row 0). Medio-lateral junctions are schematised in yellow. As in Figure 5, repolarisation conditions are under UAS-GAL4 control for Fz overexpression in the posterior wing (hedgehog expression domain), with induced Fz expression from UAS-FRT-STOP-FRT-Fz in the presence of hsFLP. Scale bar, 10 µm. (B–G) Quantitation of Fz intensity in fixed pupal wings at 28 hr APF, binned by row of cells relative to their location relative to Fz overexpression in Fz repolarisation and Fz repolarisation under de novo conditions, or relative to longitudinal vein 4 without Fz repolarisation induction, with row 0 in contact with Fz overexpression boundary and row 3 furthest away. Boundary 0 is the posterior horizontal junction of row 0 in contact with Fz overexpression area and boundary 1 is the anterior horizontal junction shared between cells in row 0 and row 1. See Supplementary file 1 for the full genotypes. Error bars are SD; n, number of wings, p values as indicated. Total Fz intensity in control condition with constitutive Fz::mKate2-sfGFP expression, along medio-lateral junctions (B) and along horizontal junctions (C). ANOVA with Holm–Sidak’s multiple comparisons test was used to compare all conditions. Comparison of Fz distribution in control condition with constitutive Fz::mKate2-sfGFP expression versus repolarisation condition with 6 hr induction, along medio-lateral junctions (D) and along horizontal junctions (E). Unpaired t-test (D) and Mann–Whitney test (E) were used to compare fluorescence intensities. Comparison of Fz distribution in repolarisation condition versus repolarisation in de novo condition with 6 hr induction, along medio-lateral junctions (F) and along horizontal junctions (G). Unpaired t-test (F) and Mann–Whitney test (G) were used to compare fluorescence intensities. See also Figure 6—figure supplement 1.
-
Figure 6—source data 1
Excel file containing individual data points for panels B–G.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig6-data1-v1.xlsx
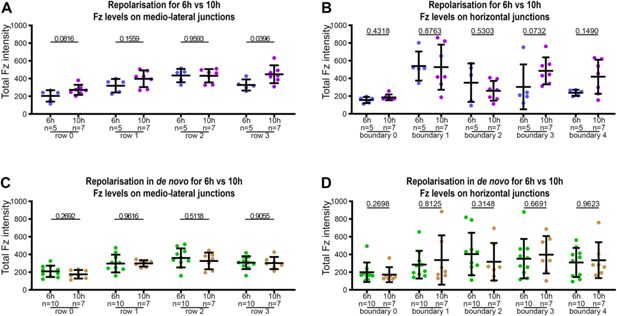
Distribution of Fz along medio-lateral and horizontal junctions in repolarisation and repolarisation under 6 and 10 hr de novo induction conditions.
Quantitation of Fz intensity in fixed pupal wings at 28 hr APF, binned by row of cells relative to their location relative to Fz overexpression in Fz repolarisation and Fz repolarisation under de novo conditions, with row 0 in contact with Fz overexpression boundary and row 3 furthest away. See Supplementary file 1 for the full genotypes. Error bars are SD; n, number of wings. Comparison of Fz distribution in repolarisation condition with 6 hr or with 10 hr to repolarise, along medio-lateral junctions (A) and along horizontal junctions (B). Unpaired t-test (A) and Mann–Whitney test (B) were used to compare fluorescence intensity for 6 versus 10 hr conditions, p values as indicated. Comparison of Fz distribution in repolarisation in de novo condition with 6 or 10 hr induction, along medio-lateral junctions (C) and along horizontal junctions (D). Unpaired t-test (C) and Mann–Whitney test (D) were used to compare fluorescence intensity at 6 versus 10 hr, p values as indicated.
-
Figure 6—figure supplement 1—source data 1
Excel file containing individual data points for panels A–D.
- https://cdn.elifesciences.org/articles/107947/elife-107947-fig6-figsupp1-data1-v1.xlsx

Summary model of polarity propagation confronted with cell-scale polarity.
(A) Core protein polarisation in control (wild-type) conditions at 28 hr APF. The level of total Fz (green) does not vary significantly on different medio-lateral junctions or on different horizontal junctions. Stbm (orange) levels mirror Fz levels on the same cell junctions. Planar polarity is directed by the integrated effects of an upstream extrinsic global cue and a cell-intrinsic cell-scale polarisation system (blue arrow) with a proximo-distal orientation. (B) Repolarisation of core protein polarity from a boundary. Before repolarisation induction, Fz (green) and Stbm (orange) become polarised following the proximo-distal axis due to the normal extrinsic and intrinsic planar polarity systems (blue arrow). After repolarisation induction by Fz overexpression in the posterior part of the wing, over-expressed Fz (yellow) accumulates on posterior boundary 0. In row 0, Fz is seen to be lost from medio-lateral junctions and to accumulate on the horizontal junction away from the region of overexpression (boundary 1). This could be due to redistribution of existing protein via endocytosis and trafficking (green arrows) or removal and degradation of protein from medio-lateral junctions and net delivery of new protein to horizontal junctions. Stbm redistribution also occurs (orange arrows) with a loss of Stbm from medio-lateral junctions and accumulation on anterior boundary 0. We surmise that Stbm redistribution to boundary 0 is primarily a result of Stbm being sequestered by forming asymmetric complexes with the high levels of over-expressed Fz on the other side of the boundary. In row 1, it is likely that a loss of Stbm located on medio-lateral junctions, and relocation along boundary 1, again occurs via the same potential mechanisms with sequestration into boundary 1 due to accumulation of Fz on the other side of this boundary. We hypothesise that Stbm redistribution to boundaries 0 and 1 results in overall cell repolarisation of rows 0 and 1 through generation of a cell-scale cell-intrinsic polarity signal (magenta arrows) that overrides the original cell polarity cues (blue arrows). Notably, repolarisation does not propagate into row 2, presumably due to weak cell–cell coupling of polarity and the strong cell-intrinsic polarity mechanisms in these cells (blue arrows). (C) Repolarisation of core protein polarity from a boundary in de novo conditions. As in repolarisation of core protein polarity in the control conditions, we surmise that Stbm accumulates on boundary 0 in row 0, due to high Fz levels on the other side of the boundary causing Stbm to be sequestered into asymmetric complexes. This could then result in a cell-scale polarity signal (magenta arrows) on the antero-posterior axis that promotes Fz accumulation on boundary 1 in row 0, rather than on medio-lateral boundaries. Concurrently, cells in rows 1–3 spontaneously polarise and adopt a proximo-distal polarity due to the presence of a strong cell-intrinsic polarisation system (blue arrows). Weak cell–cell coupling of polarity means that the altered polarity in row 0 does not propagate significantly.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Antibody | Mouse monoclonal anti-α-Tubulin DM1A | Sigma | cat#T9026 ; RRID:AB_477593 | (1:500 for immunofluorescence) |
| Antibody | Rabbit polyclonal anti-Stbm | Warrington et al., 2013 | N/A | (1:1000 for immunofluorescence) |
| Antibody | Rat anti-Stbm | Strutt and Strutt, 2008 | N/A | (1:1000 for immunofluorescence) |
| Antibody | Rabbit polyclonal anti-Fz | Bastock and Strutt, 2007 | N/A | (1:300 for immunofluorescence) |
| Antibody | Rabbit polyclonal anti-GFP | Abcam | cat#ab6556 ; RRID:AB_305564 | (1:4000 for immunofluorescence) |
| Antibody | Mouse monoclonal anti-α-Tubulin DM1A | Sigma | cat#T9026 ; RRID:AB_477593 | (1:10000 for immunoblotting) |
| Antibody | Mouse monoclonal anti-Actin AC40 | Sigma | cat#A4700 ; RRID:AB_476730 | (1:5000 for immunoblotting) |
| Antibody | Mouse monoclonal anti-Fmi 74 | DSHB Usui et al., 1999 | RRID:AB_2619583 | (1:2000 for immunoblotting) |
| Antibody | Rabbit polyclonal anti-Fz | Bastock and Strutt, 2007 | N/A | (1:250 for immunoblotting) |
| Antibody | Rabbit polyclonal anti-GFP | Abcam | cat#ab6556; RRID:AB_305564 | (1:2000 for immunoblotting) |
| Antibody | Rat polyclonal anti-Pk | Strutt et al., 2013 | N/A | (1:200 for immunoblotting) |
| Genetic reagent (D. melanogaster) | stbm6 | Wolff and Rubin, 1998 | FlyBase: FBal0062423 | |
| Genetic reagent (D. melanogaster) | dsh1 | Perrimon and Mahowald, 1987 | FlyBase: FBal0003138 | |
| Genetic reagent (D. melanogaster) | dgo380 | Feiguin et al., 2001 | FlyBase: FBal0141190 | |
| Genetic reagent (D. melanogaster) | pkpk-sple13 | Gubb et al., 1999 | FlyBase: FBal0060943 | |
| Genetic reagent (D. melanogaster) | fmiE59 | Usui et al., 1999 | FlyBase: FBal0101421 | |
| Genetic reagent (D. melanogaster) | fzP21 | Jones et al., 1996 | FlyBase:FBal0004937 | |
| Genetic reagent (D. melanogaster) | P[w+, Actin5C-FRT-PolyA-FRT-fz-eYFP] | Strutt, 2001 | FlyBase: FBtp0017633 | |
| Genetic reagent (D. melanogaster) | ActP-FRT-polyA-FRT-fz-mKate2-sfGFP | Ressurreição et al., 2018 | FlyBase: FBal0361851 | |
| Genetic reagent (D. melanogaster) | P[w+, Act >STOP > fz-mKate2-sfGFP] | Ressurreição et al., 2018 | FlyBase: FBal0361851 | |
| Genetic reagent (D. melanogaster) | hsFLP[attP2] | Bloomington Drosophila Stock Center | FlyBase: FBti0160508 | |
| Genetic reagent (D. melanogaster) | hsFLP1 | Bloomington Drosophila Stock Center | FlyBase: FBti0002044 | |
| Genetic reagent (D. melanogaster) | hsFLP22 | Bloomington Drosophila Stock Center | FlyBase: FBti0000785 | |
| Genetic reagent (D. melanogaster) | P[w+, hh-GAL4] | Tanimoto et al., 2000 | FlyBase: FBal0121962 | |
| Genetic reagent (D. melanogaster) | P[w+, UAS >STOP > FzNL] | This work (on III) | P[w+, UAS>STOP>FzNL] flies can be obtained by request from the corresponding author | |
| Genetic reagent (D. melanogaster) | w1118 | Bloomington Drosophila Stock Center | FlyBase: FBal0018186 | |
| Genetic reagent (D. melanogaster) | P[acman]-EGFP-Dgo | Ressurreição et al., 2018 | FlyBase: FBrf0237330 | |
| Chemical compound, drug | 16% paraformaldehyde solution (methanol free) | Agar Scientific | cat#R1026 | |
| Chemical compound, drug | Triton X-100 | VWR | cat#28817.295 ; CAS: 9002-93-1 | |
| Chemical compound, drug | Tween-20 | VWR | cat#437082Q ; CAS: 9005-64-5 | |
| Chemical compound, drug | Glycerol | VWR | cat#284546F ; CAS: 56-81-5 | |
| Chemical compound, drug | DABCO | Fluka | cat#33480 ; CAS: 280-57-9 | |
| Chemical compound, drug | Vectashield Mounting Medium H-1000 | Vector Labs | cat#H-1000-10 ; RRID:AB_2336789 | |
| Chemical compound, drug | Normal Goat Serum | Jackson Labs | cat#005-000-121 ; RRID:AB_2336990 | |
| Chemical compound, drug | Alexa568-Phalloidin | Thermo | cat#A12380 ; RRID:AB_3096418 | (1:1000) |
| Software, algorithm | ImageJ 1,54f | https://imagej.nih.gov/ij/ Schneider et al., 2012 | ||
| Software, algorithm | MatLab R2019b | https://uk.mathworks.com/products/matlab.html | ||
| Software, algorithm | Puncta selection script (MatLab) | Strutt et al., 2016 | ||
| Software, algorithm | Microtubules orientation script (MatLab) | Ramírez-Moreno et al., 2023 | ||
| Software, algorithm | GraphPad Prism v9 | https://www.graphpad.com | ||
| Software, algorithm | QuantifyPolarity | Tan et al., 2021 | ||
| Software, algorithm | NIS Elements AR 4.60 | Nikon | ||
| Software, algorithm | Tissue Analyzer | https://grr.gred-clermont.fr/ labmirouse/software/WebPA/ Etournay et al., 2016 |
Additional files
-
Supplementary file 1
Genotypes used in each experiment.
Excel file listing genotypes of Drosophila strains used for each experiment.
- https://cdn.elifesciences.org/articles/107947/elife-107947-supp1-v1.xlsx
-
Supplementary file 2
Matlab scripts.
PDF file containing new Matlab scripts used for data analysis.
- https://cdn.elifesciences.org/articles/107947/elife-107947-supp2-v1.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/107947/elife-107947-mdarchecklist1-v1.docx





