Antimicrobials: A lysin to kill
Being in the right place at the right time is a recipe for success for an antibacterial compound. Streptococcus pyogenes (often simply called Spy) is a bacterium that is responsible for a range of human infections, from the common “strep throat” to necrotizing fasciitis, the life-threatening condition also known as “flesh-eating disease” (Ralph and Carapetis, 2013). Spy infections can often return after treatment because the bacteria can invade human cells, which protect them from both our immune system and antibiotics (Osterlund et al., 1997). Now, in eLife, Daniel Nelson from the University of Maryland and co-workers – including Yang Shen (Maryland) and Marilia Barros (Carnegie Mellon University) as joint first authors – report the unexpected ability of an antimicrobial enzyme called PlyC to find and kill Spy that is hiding inside human cells (Shen et al., 2016).
PlyC is produced by a bacteriophage, a virus that infects bacterial cells. Like all viruses, bacteriophages commandeer their host’s replication machinery to produce copies of themselves. Some bacteriophages also carry genes for enzymes called endolysins or phage lysins that allow them to escape from the bacteria they have invaded by degrading the mesh-like cell wall that protects each bacterial cell.
The medical potential of bacteriophages and phage lysins was recognized shortly after they were discovered over a century ago. In fact, bacteriophage treatment was used for a range of illnesses in the early 20th Century (Chanishvili, 2012), but it was quickly overshadowed by small-molecule antibiotics from the 1940s onwards. The widespread use of antibiotics in medicine and agriculture has, unfortunately and inevitably, led to the spread of antibiotic-resistant bacteria and a possible end to the “Antibiotic Era” (Harrison and Svec, 1998; Boucher et al., 2009). Also, more people are now aware that using broad-spectrum antimicrobial drugs can alter the microbial community in the gut, which in turn can impact on human health (Cotter et al., 2012).
Phage lysins possess a number of good antimicrobial traits. They are potent, fast-acting, specific to a narrow range of bacteria and relatively harmless to other types of cells. They can also kill bacteria that are dormant or not actively growing (Fischetti, 2010), and have been used to cure short-term Spy infections in mice (Gilmer et al., 2013). However, most had assumed that these enzymes were incapable of entering into host cells, and unlikely to be useful for treating chronic Spy infections in humans.
Shen, Barros and colleagues, who are based at various centers across the United States, attempted to overcome this assumed limitation by fusing phage lysins with fragments of proteins that help transport other molecules through cell membranes. Serendipitously, they discovered that PlyC is intrinsically active against intracellular Spy. PlyC is the only known phage lysin that is composed of two subunits. One of these subunits (PlyCA) is enzymatically active and degrades the bacterial cell wall. The other subunit (PlyCB) binds to the Spy cell surface and, as discovered by Shen, Barros et al., is needed to get the enzyme inside a human epithelial cell (Figure 1).
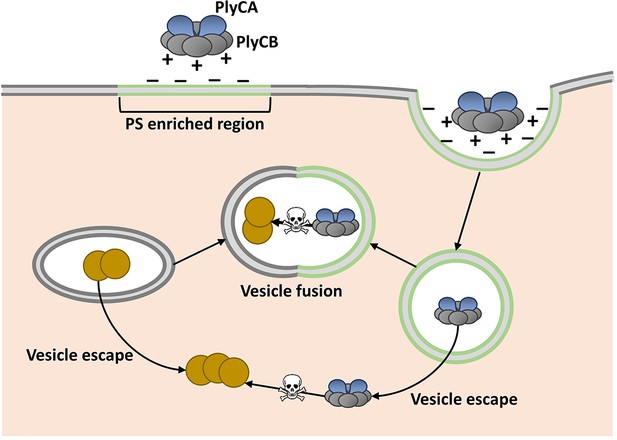
Model showing how the enzyme PlyC can enter human epithelial cells and kill intracellular Streptococcus pyogenes (Spy).
PlyC is composed of two subunits. PlyCB forms an eight-part ring that normally binds to the Spy cell surface. PlyCA is the enzymatically active subunit that degrades the bacterial cell wall. The PlyCB surface is normally positively charged, and Shen, Barros et al. show that it interacts with phosphatidylserine (PS), which is negatively charged. They propose that PlyCB binds to lipid rafts (shown in green) that are enriched in PS; this causes the membrane to fold around PlyC, which ultimately enters the cell inside a vesicle. These vesicles may fuse with bacteria-containing vesicles, giving PlyC access to intracellular Spy (depicted as brown circles). Alternatively, PlyC may escape the vesicle and interact with and kill Spy that is free in the host cell.
The PlyCB subunit was known to have a positively charged surface (McGowan et al., 2012), and so Shen, Barros et al. hypothesized that it interacts with negatively charged components of the cell membrane. Indeed, they then went on to discover that PlyCB binds to a negatively charged molecule called phosphatidylserine that is common in eukaryotic cell membranes. Their results also suggested that PlyCB can only penetrate membranes that contain at least 30% phosphatidylserine.
Regions of the cell membrane called lipid rafts have high levels of phosphatidylserine, and are involved in the uptake of numerous molecules from the cell’s exterior via a process called endocytosis (Pike, 2009). This suggested that PlyC might enter cells via an endocytic mechanism, and Shen, Barros et al. provided further support for this idea by showing that PlyCB co-localizes with another protein that binds to lipid rafts. Based on these data and some molecular modeling, they proposed that PlyCB binds to phosphatidylserine, causes the membrane to fold around it, and ultimately enters the cell via endocytosis (Figure 1).
These new findings demonstrate the potential of phage lysins, and possibly bacteriophage-based therapy, to treat challenging bacterial infections. Specifically, they also highlight the potential of PlyC to reduce or even prevent infection with Spy, which remains a major health concern in low- to middle-income countries and disproportionately targets children (Ralph and Carapetis, 2013). Further work is needed to confirm if PlyC enters cells via lipid raft-mediated endocytosis, and to determine how PlyC ultimately meets the intracellular Spy in order to kill it. However, insights provided by Shen, Barros et al. open up the possibility of engineering new endolysins based on PlyCB to target other disease-causing microbes that evade the immune system by hiding in an infected person’s cells.
References
-
Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of AmericaClinical Infectious Diseases 48:1–12.https://doi.org/10.1086/595011
-
The impact of antibiotics on the gut microbiota as revealed by high throughput DNA sequencingDiscovery Medicine 13:193–199.
-
Bacteriophage endolysins: a novel anti-infective to control Gram-positive pathogensInternational Journal of Medical Microbiology 300:357–362.https://doi.org/10.1016/j.ijmm.2010.04.002
-
Novel bacteriophage lysin with broad lytic activity protects against mixed infection by Streptococcus pyogenes and methicillin-resistant Staphylococcus aureusAntimicrobial Agents and Chemotherapy 57:2743–2750.https://doi.org/10.1128/AAC.02526-12
-
The beginning of the end of the antibiotic era? Part I. The problem: Abuse of the "miracle drugs"Quintessence International 29:151–162.
-
X-ray crystal structure of the streptococcal specific phage lysin PlyCProceedings of the National Academy of Sciences of the United States of America 109:12752–12757.https://doi.org/10.1073/pnas.1208424109
-
The challenge of lipid raftsJournal of Lipid Research 50 Suppl:S323–328.https://doi.org/10.1194/jlr.R800040-JLR200
-
Group a streptococcal diseases and their global burdenCurrent Topics in Microbiology and Immunology 368:1–27.https://doi.org/10.1007/82_2012_280
Article and author information
Author details
Publication history
Copyright
© 2016, Gaca et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 2,046
- views
-
- 260
- downloads
-
- 3
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 3
- citations for umbrella DOI https://doi.org/10.7554/eLife.16111






