Chronically implanted Neuropixels probes enable high-yield recordings in freely moving mice
Figures
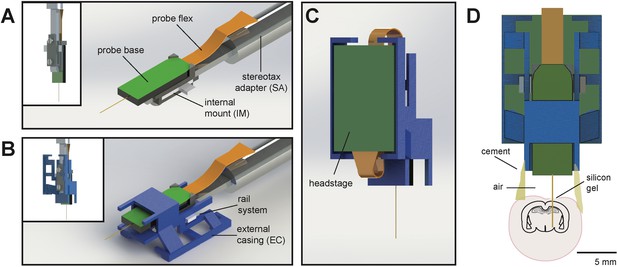
Schematic of Neuropixels AMIE.
(A) Probe base mounted onto 3D printed internal casing and attached to machined metal stereotax adapter. Inset: Rear view, with screws that attach the internal mount (IM) to the stereotax adapter (SA). (B) Entire assembly in a. within 3D printed external casing. Inset: Rear view. (C) The headstage is positioned on the back of the encasing, with the flex wrapped in an ‘S’ shape. (D) Entire assembly in relation to size of mouse brain and skull. The EC is attached to the skull with cement. Silicon gel is used to as an artificial dura to protect the open craniotomy.

Mounting and implanting the Neuropixels probe.
(A) The internal mount (IM) is attached to the stereotax adapter (SA) with two screws, and probe is attached to the internal mount using an epoxy. (B) Medical-grade silicon is added to the base of the shank to add extra support. (C) The external case (EC) is attached to a breadboard, and the IM+probe assembly is carefully guided into the internal compartment of the EC (top view). (D) After cementing the IM to the EC, the entire assembly is ready to be implanted. (E) During surgery, the the shank is lowered into the brain (here at a ~ 16° angle). The ground wire extends down the side of the implant and is attached to the ground screw. (F) The entire encasing is attached to the headbar and skull using Metabond. Tape is added where necessary to add protection between the encasing and the skull. The stereotax adapter (not shown) is removed after this support structure is dry and secure. (G) Image of a mouse with the implant ~48 hr after surgery. The entire assembly is wrapped in Kapton tape to protect the onboard electronics.
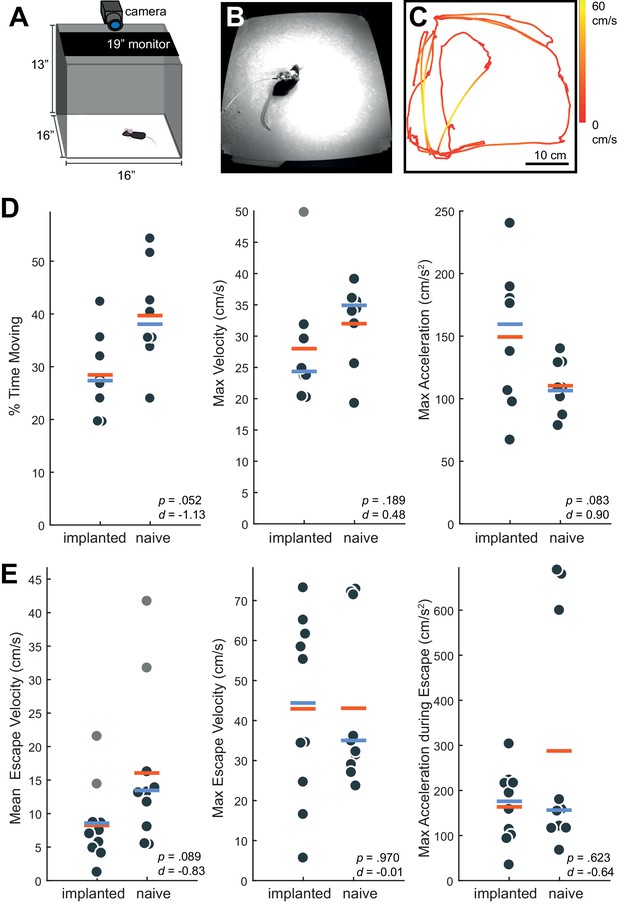
Behavior in implanted mice is comparable to naive mice.
(A) Behavioral testing arena, with a camera to track the position of the mouse and a monitor on top to present visual stimuli. (B) Snapshot of mouse with implant in arena. (C) Sample tracking of 2 min of open field behavior in an implanted mouse. Color of the line indicates the velocity of the mouse. (D) Open field behavior of implanted vs. naive mice. Random 30–180 s exerpts of behavior (N = 8 videos per group, two videos from each mouse) in the open field were used to calculate a percent time moving (>5 cm/s), max velocity, and max acceleration. (E) Visual-looming evoked behavior of implanted vs. naive mice (N = 10 trials, two videos per mouse). A dark dot of linearly increasing diameter (40 cm/s) was presented over the mouse’s head to evoke an escape response. The mean velocity, max velocity, and max acceleration during these responses is presented here. In all panels, orange line indicates the group mean, blue line indicates the median. p-Values (as computed by a two-sided Wilcoxon Rank Sum test) as well as effect sizes (computed by a Cohen’s d) are reported on each panel. Outliers (defined as 1.5*IQR) are marked as light gray points.
-
Figure 3—source data 1
Behavioral data for implanted and naive mice in an open field and in response to a looming stimulus.
- https://doi.org/10.7554/eLife.47188.007
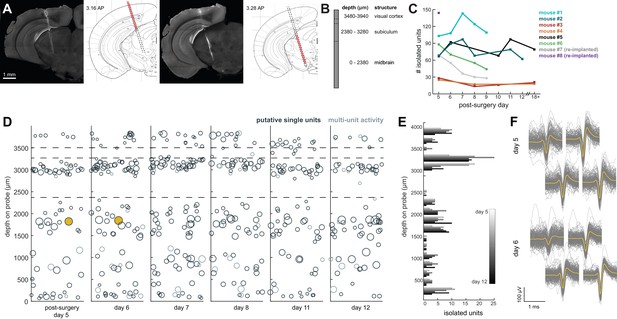
Chronic Neuropixel probe implants in cortex and subcortical regions can record ~20–145 units across multiple days.
(A) Probe location, marked with DiI. Sections from Paxinos and Franklin atlas provided for reference. Mouse #200 was implanted with a probe in visual cortex, hippocampus (subiculum), and the midbrain. (B) Schematic of probe depth in (A). (C) Number of isolated units across recording days for eight different mice. Mouse #3 and #4 were implanted with a probe with fewer recording sites (270 vs. 374). Mouse #2 is featured in the other panels of this figure. Mouse #7 had a probe that was previously implanted in Mouse #5; see Figure 6. (D) Scatter plot of units across days for Mouse #2. Size of circles denotes number of waveforms assigned to that unit. X axis is random for visualization. € Histogram of isolated units across days and brain depth for Mouse #2. (F) Waveforms (n = 200, mean waveform in yellow) recorded from the same four contacts on the probe on day 5 (top) and day 6 (bottom). Units are the same as the yellow filled in circles in (D).
-
Figure 4—source data 1
Number of isolated units for each probe across post-surgery days.
- https://doi.org/10.7554/eLife.47188.009
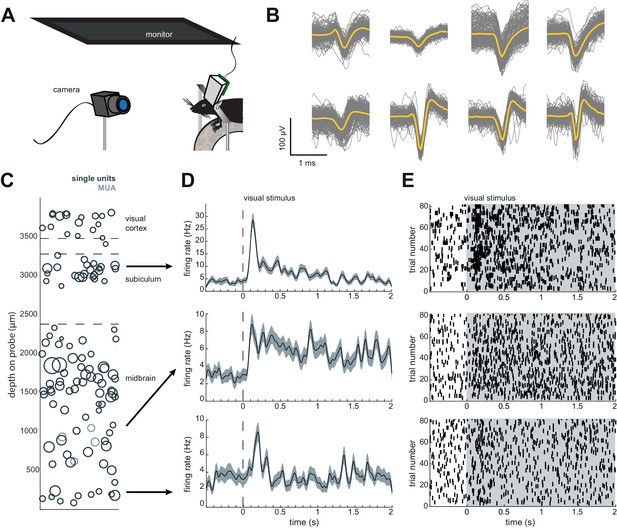
Implant design enables researchers to further characterize brain regions recorded during freely moving behavior.
(A) Schematic of headfixed setup. The mouse was implanted with a headbar (see Materials and methods) enabling it to be restrained above a wheel. Visual stimuli was presented on a monitor above the mouse’s head (similar to the unrestrained condition). The mouse’s pupil can be tracked with a high resolution IR camera, and movement can be tracked using a rotary encoder on a 3D printed wheel. (B) Eight sample waveforms (n = 200, mean in yellow) from a restrained recording, same mouse as Figure 4D–F (Mouse #2). (C) Distribution of sorted units across the probe, same mouse as in Figure 4D–F (Mouse #2). (D) Peristimulus time histograms for three example neurons from different locations on the probe. The stimuli were a pseudorandomized set of 2 s full contrast sinusoidal drifting gratings in eight different directions. Shaded region is standard error of the mean. Stimuli began at the dotted line. (E) Raster plots for the neurons in (D). Shaded area indicates the duration of the stimulus.
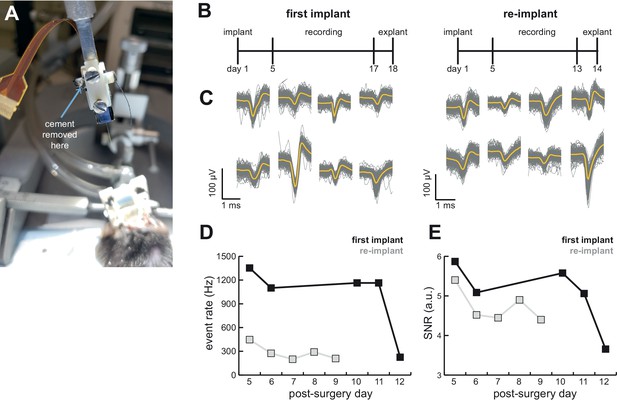
AMIE design allows for probe explantation and subsequent re-implantation with the same Neuropixels probe.
(A) Example successful probe explanation. Cement is drilled away from the wings of the internal casing in order to remove the internal mount from the external casing. (B) Outline of experiment timing. The same probe was used in the first implant and re-implant. (C) Sample mean waveforms (n = 200, mean in yellow) from each mouse. (D) Detected event rate of the first implant versus the re-implanted probe across days of recording. (E) Median signal-to-noise ratio (SNR) for first implant and re-implanted probe across days (see Materials and methods.).
-
Figure 6—source data 1
Computed event rate and SNR ratio for the initial implant and re-implant of the same probe across post-surgery days.
- https://doi.org/10.7554/eLife.47188.012
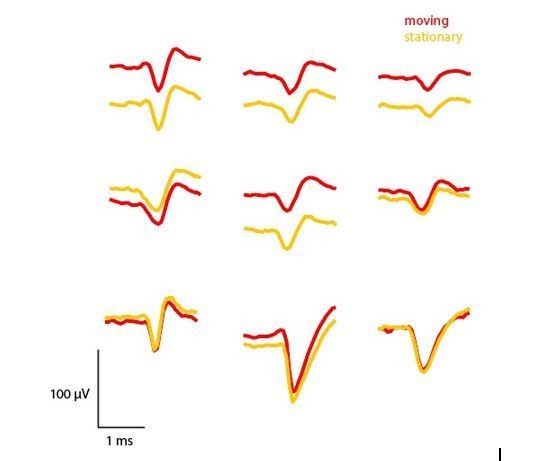
Comparison of mean waveforms (n=50) across periods where the mouse was moving (>15 cm/sec) or stationary (<5 cm/sec).
https://doi.org/10.7554/eLife.47188.016Videos
3D rendering of the AMIE device demonstrating the configuration of internal mount (IM), external casing (EC), and stereotax adapter.
https://doi.org/10.7554/eLife.47188.003Behavior of mouse implanted with Neuropixels AMIE.
Mouse was free to move around a 16”x16’ arena while implanted and tethered. Video is shown at 2x speed.
Tables
Overview of experiments, with the Neuropixels probe option used and the outcome of the experiment.
For each of these experiments, even the unsuccessful explants, neural data was obtained from the initial implant and recording sessions. For an explanation of the probe options, see Materials and methods. Starred mice are included in the paper; + sign indicates the experiment was sorted with Kilosort2; M = mean; SD = standard deviation.
| Mouse | Probe option | Recordable channels | M ± SD Isolated units | Silicone on shank | Outcome | ||
|---|---|---|---|---|---|---|---|
| NP6* (Figure 4; Mouse #3) | 4 | 276 | 19.8 ± 6.23 | No | Shank broke during explant | ||
| NP7* (Figure 4; Mouse #4) | 4 | 276 | 20.0 ± 4.36 | no | Shank broke during freely moving recording | ||
| NP8* (Figure 4; Mouse #2) | 1 | 384 | 77.2 ± 13.7 | no | Shank broke during explant | ||
| NP9* (Figure 4; Mouse #1) | 1 | 384 | 117.4 ± 16.3 | no | Shank broke during explant | ||
| NP11 | 1 | 384 | - | no | Shank broke during freely moving recording | ||
| NP12 | 3 | 384 | - | no | Mouse didn’t recover from surgery, probe successfully explanted and re-implanted in NP13 | ||
| NP13 (Figure 4; Mouse #8) | 3 | 384 | 145+ | yes | Ground wire issues after surgery; one session successfully recorded. Successful explant | ||
| NP14* (Figure 4 and 6; Mouse #5) | 3 | 384 | 80.6 ± 13.6 | yes | Successful explant, re-implanted in NP16 | ||
| NP15* (Figure 4; Mouse #6) | 3 | 384 | 64.3 ± 19.1 | yes | Successful explant | ||
| NP16* (Figure 4 and 6; Mouse #7) | 3 | 384 | 43.6 ± 17.8 | yes | Successful explant | ||
| Reagent type/resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Chemical compound/drug | Medical-grade clear silicon adhesive | Mastersil | 912MED | |
| Chemical compound/drug | Loctite Instant Adhesive 495 | ULINE | S-7595 | |
| Chemical compound/drug | Medigel CPF | Clear H20 | 74-05-5022 | |
| Chemical compound/drug | Isoflurane | Allivet | 50562 | |
| Chemical compound/drug | C and B Metabond 'B' Quick Base | Parkell | S398 | |
| Chemical compound/drug | C and B Metabond 'C' Quick Base | Parkell | S371 | |
| Chemical compound/drug | C and B Metabond Radiopaque L-Power | Parkell | S396 | |
| Chemical compound/drug | Optibond Solo Plus | Kerr | 31514 | |
| Chemical compound/drug | Vetbond | Santa Cruz Biotechnology | sc-361931 | |
| Chemical compound/drug | Charisma A1 Syringe | Net32 | 66000085 | |
| Chemical compound/drug | Eye Ointment | Rugby | 370435 | |
| Chemical compound/drug | Dental Cement | Stoelting | 5217307 | |
| Chemical compound/drug | DiI | ThermoFisher Scientific | D282 | |
| Chemical compound/drug | Silicone Gel Kit | Dow Coning | 3–4860. | |
| Chemical compound/drug | Bleach | Amazon | B01K8HT54G | Any brand bleach ok |
| Other | Neuropixel Probe | Neuropixel Stock Center (Neuropixels.org) | Neuropixel 1.0 Probe | |
| Other | 3D Printed Internal Mount | ‘this paper’ - Github repository | IM_Neuropixel1.stl | Internal mount design file (.stl) can be downloaded from the following github repository: https://github.com/churchlandlab/ChronicNeuropixels |
| Other | 3D Printed External Casing | ‘this paper’ - Github repository | EC_Neuropixel1.stl | same as above |
| Other | Sterotax Adapter | ‘this paper’ - Github repository | stereotax adapter v4.ipt | same as above |
| Other | 2-56A Screws | Amazon | B00F34U238 | |
| Other | Silver Wire | WPI | AGW1010 | |
| Other | 4’ post holder with thumbscrew | Thorlabs | PH4 | |
| Other | Slim right angle bracket | Thorlabs | AB90B | |
| Other | Aluminum Breadboard | Thorlabs | MB624 | |
| Other | M6 Cap Screw | Thorlabs | SH6MS20 | |
| Other | M6 Nut | Thorlabs | HW-KIT2/M | |
| Other | Kapton Tape | ULINE | S-7595 | |
| Other | Kimwipes | Kimtech | 34120 | |
| Other | Oxygen Cylinders | Airgas | OX USP300 | |
| Other | Mouse Anesthesia System with Isoflurance Box | Parkland Scientific | V3000PK | |
| Other | Small rodent sterotax fitted with anesthesia mask | Narishige | SG-4N | |
| Other | Dental Drill | Osada | EXL-M40 | |
| Other | 0.9 mm burrs for micro drill | Fine Science Tools | 19007–09 | |
| Other | T/Pump Warm Water Recirculator | Kent Scientific | TP-700 | |
| Other | Warming Pad for warm water recirculator | Kent Scientific | TPZ | |
| Other | Cotton Applicators | Fisher Scientific | 19-062-616 | |
| Other | Surgical Spears | Braintree Scientific Inc. | SP 40815 |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.47188.014





