Cell Cycle: Deciding when to exit
The development and survival of a multicellular organism relies on cells proliferating by passing through a series of events known as the cell cycle. During the first stage of the cycle (G1), each cell makes an important decision: does it progress to the second stage (S-phase) and replicate its genome in preparation for cell division, or does it exit the cycle and become quiescent (Pardee, 1974; Johnson and Skotheim, 2013)?
Cells usually exit the cell cycle in order to differentiate into the distinct cell types of an organism, such as neurons, muscle or fat (Sun and Buttitta, 2017). If the decision to stop proliferating is somehow disrupted, this can affect the normal development or function of tissues and organs and lead to diseases, such as cancer. However, understanding the signals that control this decision can be challenging because a G1 cell preparing to enter S-phase is essentially indistinguishable from a G1 cell that will become quiescent.
One way to overcome this challenge is to visually monitor proteins that are only active in certain phases of the cell cycle (Sakaue-Sawano et al., 2008; Zielke et al., 2014; Grant et al., 2018). For example, a group of proteins called cyclin-dependent kinases (or CDKs for short), which drive cells into S-phase, are active in G1 but are permanently turned off when cells enter a state of quiescence.
In 2013, a group of researchers used this property of CDKs to distinguish cultured mammalian cells in G1 that were preparing to proliferate from those entering quiescence (Spencer et al., 2013). To do this they engineered a fluorescent reporter protein which sits in the nucleus when CDKs are inactive and moves into the cytoplasm when modified by active CDKs (Figure 1A). The nucleus and cytoplasm are easily distinguishable via time-lapse microscopy, making it possible to determine when CDKs are active in individual cells during G1.
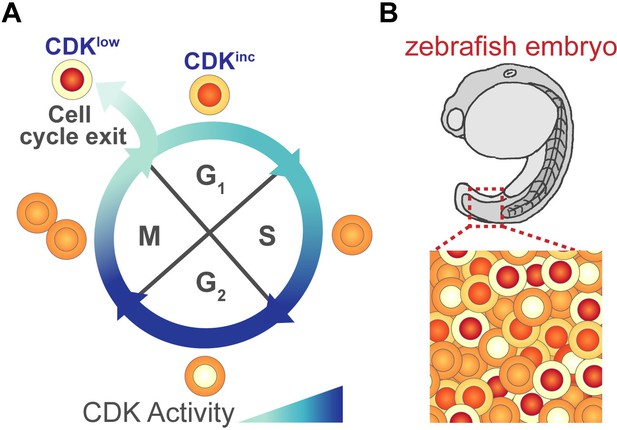
Monitoring cell cycle progression using a fluorescent biosensor.
(A) As cells grow and prepare to divide they pass through four different phases of the cell cycle: G1, S, G2, M. During the course of this cycle, the activity of cyclin-dependent kinases (CDKs) changes. These fluctuations in activity can be monitored using a fluorescent reporter protein that contains a portion of human DNA helicase B (DHB), which moves from the nucleus to the cytoplasm when phosphorylated by active CDKs. Thus, changes in the levels of DHB in the nucleus and cytoplasm (depicted in shades of orange/red) can be used to determine a cell’s CDK activity. At the start of G1, CDK activity is low and DHB remains in the nucleus (shown in red). Some G1 cells maintain this low level of activity (CDKlow) and exit the cell cycle to become quiescent, while cells with increasing levels of CDK activity (CDKinc) commit to another cell cycle and enter S-phase. (B) Adikes et al. showed that the DHB reporter could monitor cell cycle progression of individual cells in live animals, such as the embryo of a zebrafish. It can also identify which cells have exited the cell cycle and which are preparing for division.
Image credit: Joy H Meserve.
Now, in eLife, David Matus from Stony Brook University and co-workers – including Rebecca Adikes, Abraham Kohrman, and Michael Martinez as joint first authors – report how they used this technique to visualize when individual cells decide to stop dividing in living animals (Adikes et al., 2020). To adapt the CDK reporter to animals, the team (who are based at Stony Brook, the University of Colorado, Stanford University, Imperial College and University of Virginia) turned to two well studied experimental organisms: the nematode worm C. elegans and the zebrafish D. rerio.
Previous work using the CDK reporter in cultured mammalian cells showed that not all G1 cells behave the same after cell division: some never activate CDKs and enter quiescence (CDKlow cells), while others begin increasing CDK activity during G1 (CDKinc cells) and ultimately commit to another cell cycle (Figure 1A; Spencer et al., 2013). Adikes et al. found that this bifurcation in G1 cells could also be detected in the tissues of living C. elegans and D. rerio (Figure 1B). They found that the CDKlow and CDKinc phenotypes of G1 cells could be used to predict whether a cell would enter quiescence or would re-commit to another cycle. Further experiments revealed that if a cell had high levels of a protein called p21, which inhibits the activity of CDKs, its daughter cells were more likely to become quiescent following division. This suggests that the decision to proliferate or exit the cell cycle may depend on how p21 levels are regulated in proliferating cells (Overton et al., 2014; Hsu et al., 2019).
The CDK reporter has a number of applications. It could make it easier to study how quiescence is regulated in tissues that are typically difficult to image for long periods of time. It might reveal early steps in tissue regeneration when cells are ramping up to re-enter the cell cycle. It could also be used to sort and recover populations of CDKlow and CDKinc cells for further experiments to identify the pathways regulating entry into quiescence.
The CDK reporter will allow us to tackle many interesting questions in developmental biology. For instance, how might the organization of a tissue influence the decision to stop dividing and enter quiescence? Is it possible to identify cells very early in the differentiation process before genes that demark differentiation turn on? Whatever the application or research question, Adikes et al. demonstrate once again that important new insights into the complexities of biology arise when new tools are developed to visualize living organisms.
References
-
Start and the restriction pointCurrent Opinion in Cell Biology 25:717–723.https://doi.org/10.1016/j.ceb.2013.07.010
-
States of G0 and the proliferation-quiescence decision in cells, tissues and during developmentThe International Journal of Developmental Biology 61:357–366.https://doi.org/10.1387/ijdb.160343LB
Article and author information
Author details
Publication history
- Version of Record published: February 12, 2021 (version 1)
Copyright
© 2021, Meserve and Duronio
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 3,222
- views
-
- 202
- downloads
-
- 1
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Cell Biology
Endogenous tags have become invaluable tools to visualize and study native proteins in live cells. However, generating human cell lines carrying endogenous tags is difficult due to the low efficiency of homology-directed repair. Recently, an engineered split mNeonGreen protein was used to generate a large-scale endogenous tag library in HEK293 cells. Using split mNeonGreen for large-scale endogenous tagging in human iPSCs would open the door to studying protein function in healthy cells and across differentiated cell types. We engineered an iPS cell line to express the large fragment of the split mNeonGreen protein (mNG21-10) and showed that it enables fast and efficient endogenous tagging of proteins with the short fragment (mNG211). We also demonstrate that neural network-based image restoration enables live imaging studies of highly dynamic cellular processes such as cytokinesis in iPSCs. This work represents the first step towards a genome-wide endogenous tag library in human stem cells.
-
- Biochemistry and Chemical Biology
- Cell Biology
Mediator of ERBB2-driven Cell Motility 1 (MEMO1) is an evolutionary conserved protein implicated in many biological processes; however, its primary molecular function remains unknown. Importantly, MEMO1 is overexpressed in many types of cancer and was shown to modulate breast cancer metastasis through altered cell motility. To better understand the function of MEMO1 in cancer cells, we analyzed genetic interactions of MEMO1 using gene essentiality data from 1028 cancer cell lines and found multiple iron-related genes exhibiting genetic relationships with MEMO1. We experimentally confirmed several interactions between MEMO1 and iron-related proteins in living cells, most notably, transferrin receptor 2 (TFR2), mitoferrin-2 (SLC25A28), and the global iron response regulator IRP1 (ACO1). These interactions indicate that cells with high MEMO1 expression levels are hypersensitive to the disruptions in iron distribution. Our data also indicate that MEMO1 is involved in ferroptosis and is linked to iron supply to mitochondria. We have found that purified MEMO1 binds iron with high affinity under redox conditions mimicking intracellular environment and solved MEMO1 structures in complex with iron and copper. Our work reveals that the iron coordination mode in MEMO1 is very similar to that of iron-containing extradiol dioxygenases, which also display a similar structural fold. We conclude that MEMO1 is an iron-binding protein that modulates iron homeostasis in cancer cells.






