Identification of drugs associated with reduced severity of COVID-19 – a case-control study in a large population
Figures
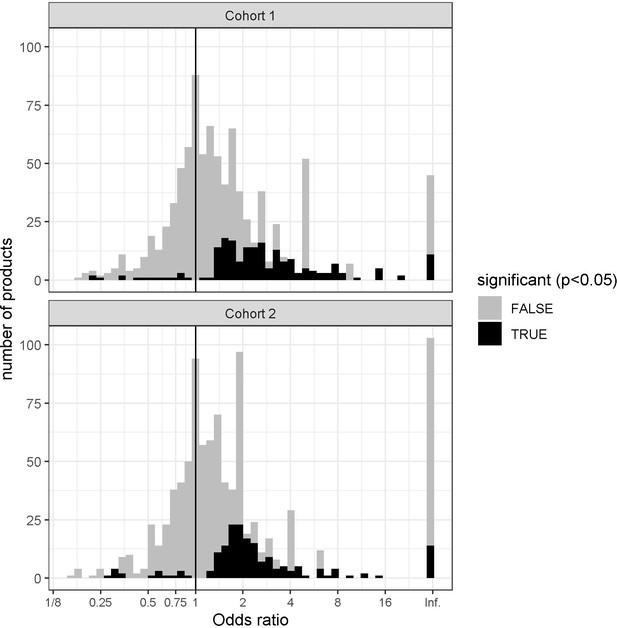
Histogram showing the distribution of the odd ratios (OR) of medication use with the outcome in cohorts 1 and 2.
The overwhelming majority of medications are associated with neutral effect (gray) or increased risk for hospitalization (black, OR>1), only a few are associated with significantly decreased risk (black, OR<1).
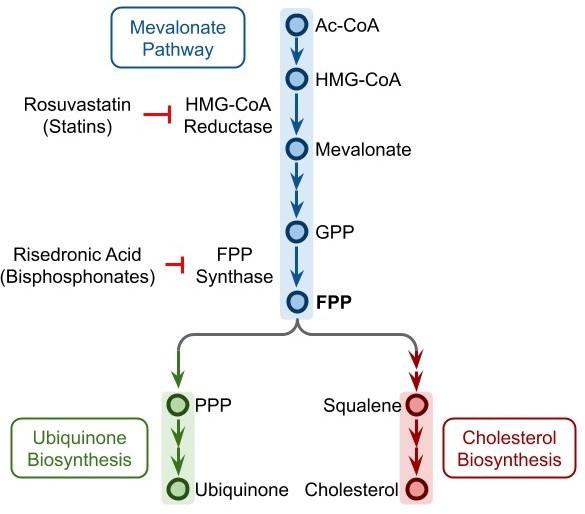
Ubiquinone and cholesterol biosynthesis pathway.
Ubiquinone and cholesterol biosynthesis pathways originate from a branching of the mevalonate pathway at FPP. Rosuvastatin and other statins can inhibit the HMG-CoA reductase, while risedronic acid and other bisphosphonates can inhibit the FPP synthase. Ac-CoA: acetyl coenzyme A, HMG-CoA: hydroxymethylglutaryl coenzyme A, GPP: geranyl pyrophosphate, FPP: farnesyl pyrophosphate, PPP: polyprenyl pyrophosphate.
Tables
Demographics and clinical characteristics of the two matched cohorts of patients (hospitalized versus non-hospitalized).
| Cohort 1 | Cohort 2 | |||
|---|---|---|---|---|
| COVID-19 hospitalized (cases) | Not hospitalized (controls) | COVID-19 hospitalized (cases) | Not hospitalized (controls) | |
| n | 6530 | 32,650 | 6953 | 13,906 |
| Age (mean, SD) | 64.6 (16.1) | 64.8 (15.8) | 65.7 (16.0) | 65.7 (15.8) |
| Sex, female (%) | 3259 (49.9) | 16,295 (49.9) | 3381 (48.6) | 6762 (48.6) |
| Hospitalization severity (n, %) | ||||
| Mild condition | 3008 (46.1) | 2676 (38.5) | ||
| Serious condition | 851 (13.0) | 1043 (15.0) | ||
| Severe condition | 1621 (24.8) | 1903 (27.4) | ||
| Deceased | 1050 (16.1) | 1331 (19.1) | ||
| Smoking status (%) | ||||
| Never smoker | 5012 (76.8) | 24,218 (74.2) | 5156 (74.2) | 10,312 (74.2) |
| Past smoker | 1115 (17.1) | 5808 (17.8) | 1338 (19.2) | 2676 (19.2) |
| Current smoker | 403 (6.2) | 2624 (8.0) | 459 (6.6) | 918 (6.6) |
| Nb visits at primary doctor in last year (mean, SD) | 8.1 (7.9) | 7.9 (7.3) | 8.2 (8.4) | 7.5 (7.1) |
| Comorbidity (%) | ||||
| Arrhythmia | 887 (13.6) | 4242 (13.0) | 1278 (18.4) | 2221 (16.0) |
| Asthma | 527 (8.1) | 2941 (9.0) | 650 (9.3) | 1376 (9.9) |
| Congestive heart failure (CHF) | 228 (3.5) | 1140 (3.5) | 784 (11.3) | 851 (6.1) |
| Chronic obstructive pulmonary disease (COPD) | 148 (2.3) | 740 (2.3) | 603 (8.7) | 776 (5.6) |
| Diabetes | 2976 (45.6) | 14,880 (45.6) | 3425 (49.3) | 5549 (39.9) |
| Hypertension | 3850 (59.0) | 19,062 (58.4) | 4396 (63.2) | 8102 (58.3) |
| Ischemic heart disease (IHD) | 1464 (22.4) | 7320 (22.4) | 1838 (26.4) | 3113 (22.4) |
| Malignancy | 1087 (16.6) | 5435 (16.6) | 1280 (18.4) | 2766 (19.9) |
| Chronic kidney disease (CKD) | 102 (1.6) | 510 (1.6) | 1086 (15.6) | 1117 (8.0) |
| Obesity (documented diagnosis) | 3761 (57.6) | 17,837 (54.6) | 3975 (57.2) | 7950 (57.2) |
| Body mass index (BMI) (mean, SD) | 28.7 (5.7) | 28.6 (6.5) | 29.1 (6.3) | 28.5 (5.7) |
| BMI group (%) | ||||
| <18.5 | 17 (0.3) | 85 (0.3) | 51 (0.7) | 93 (0.7) |
| 18.5 to 25 | 1070 (16.4) | 5350 (16.4) | 1244 (17.9) | 2471 (17.8) |
| 25 to 30 | 2295 (35.1) | 11,475 (35.1) | 2264 (32.6) | 4870 (35.0) |
| 30 to 35 | 2053 (31.4) | 10,265 (31.4) | 2005 (28.8) | 4267 (30.7) |
| 35 to 40 | 761 (11.7) | 3805 (11.7) | 886 (12.7) | 1562 (11.2) |
| >40 | 334 (5.1) | 1670 (5.1) | 503 (7.2) | 643 (4.6) |
| Glomerular filtration rate (GFR) (mean, SD) | 85.7 (21.6) | 85.8 (20.3) | 78.7 (28.2) | 83.4 (22.4) |
| Chronic kidney disease (CKD) staging (n, %) | ||||
| G1 | 3047 (46.7) | 15,145 (46.4) | 2837 (40.8) | 6090 (43.8) |
| G2 | 2679 (41.0) | 13,747 (42.1) | 2535 (36.5) | 5722 (41.1) |
| G3a | 558 (8.5) | 2817 (8.6) | 689 (9.9) | 1257 (9.0) |
| G3b | 203 (3.1) | 836 (2.6) | 391 (5.6) | 571 (4.1) |
| G4 | 41 (0.6) | 89 (0.3) | 186 (2.7) | 160 (1.2) |
| G5 | 63 (0.9) | 28 (0.2) | ||
| Dialysis | 2 (0.0) | 16 (0.0) | 252 (3.6) | 78 (0.6) |
Most significant associations for medications acquired in the 35 days preceding the index date in two matched cohorts.
| ATC code and class | Use in case | Use in contr. | Case % | Contr. % | Odds ratio (95% conf. int.) | p-Value | FDR |
|---|---|---|---|---|---|---|---|
| (A) Cohort 1 (N = 6530 hospitalization cases, N=32,650 controls taken from the general population) | |||||||
| C10AA07 Rosuvastatin | 328 | 2380 | 5.02 | 7.29 | 0.673 (0.596 to 0.758) | <0.0001 | <0.001 |
| C10AX09 Ezetimibe | 73 | 740 | 1.12 | 2.27 | 0.488 (0.377 to 0.622) | <0.0001 | <0.001 |
| A16AX30 Ubiquinone (CoQ-10) | 6 | 165 | 0.09 | 0.51 | 0.181 (0.065 to 0.403) | <0.0001 | <0.001 |
| C01BC04 Flecainide | 7 | 116 | 0.11 | 0.36 | 0.301 (0.118 to 0.641) | 0.00039 | 0.005 |
| J07AL02 Pneumococcus vaccine conjugate | 21 | 220 | 0.32 | 0.67 | 0.476 (0.288 to 0.746) | 0.00049 | 0.006 |
| C09BA05 Ramipril-hydrochlorothiazide | 127 | 859 | 1.95 | 2.63 | 0.734 (0.603 to 0.887) | 0.00099 | 0.011 |
| A10BD07 Sitagliptin-metformin | 243 | 1501 | 3.72 | 4.60 | 0.802 (0.696 to 0.922) | 0.00159 | 0.017 |
| C10AA03 Pravastatin | 52 | 385 | 0.80 | 1.18 | 0.673 (0.493 to 0.902) | 0.00659 | 0.060 |
| N06AB10 Escitalopram | 216 | 1302 | 3.31 | 3.99 | 0.824 (0.708 to 0.955) | 0.00930 | 0.078 |
| M01AC01 Piroxicam | 24 | 205 | 0.37 | 0.63 | 0.584 (0.365 to 0.894) | 0.00980 | 0.082 |
| C09CA06 Candesartan | 65 | 451 | 1.00 | 1.38 | 0.718 (0.544 to 0.934) | 0.01237 | 0.100 |
| M05BA07 Risedronic acid | 56 | 396 | 0.86 | 1.21 | 0.705 (0.522 to 0.935) | 0.01319 | 0.103 |
| G04CB02 Dutasteride | 30 | 240 | 0.46 | 0.74 | 0.623 (0.411 to 0.914) | 0.01367 | 0.105 |
| A11CC05 Cholecalciferol | 660 | 3634 | 10.11 | 11.13 | 0.898 (0.821 to 0.980) | 0.01600 | 0.119 |
| C09AA08 Cilazapril | 17 | 153 | 0.26 | 0.47 | 0.554 (0.315 to 0.918) | 0.01743 | 0.124 |
| G04BE08 Tadalafil | 29 | 229 | 0.44 | 0.70 | 0.632 (0.413 to 0.933) | 0.01862 | 0.132 |
| S01ED61 Timolol-travoprost | 8 | 90 | 0.12 | 0.28 | 0.444 (0.186 to 0.913) | 0.02068 | 0.142 |
| A10BH01 Sitagliptin | 33 | 250 | 0.51 | 0.77 | 0.658 (0.443 to 0.950) | 0.02461 | 0.162 |
| J07BB02 Influenza vaccine inac | 392 | 2205 | 6.00 | 6.75 | 0.882 (0.787 to 0.986) | 0.02548 | 0.165 |
| N06DX02 Ginkgo folium | 2 | 42 | 0.03 | 0.13 | 0.238 (0.028 to 0.915) | 0.02552 | 0.165 |
| A12CC04 Magnesium citrate | 31 | 237 | 0.48 | 0.73 | 0.652 (0.433 to 0.952) | 0.02597 | 0.166 |
| A10BK01 Dapagliflozin | 35 | 255 | 0.54 | 0.78 | 0.685 (0.466 to 0.978) | 0.03283 | 0.193 |
| (B) Cohort 2 (N = 6953 hospitalization cases, N=13,906 controls taken from patients SARS-CoV-2 positive) | |||||||
| C10AA07 Rosuvastatin | 354 | 950 | 5.09 | 6.83 | 0.732 (0.643 to 0.831) | <0.0001 | 0.000 |
| C10AX09 Ezetimibe | 92 | 303 | 1.32 | 2.18 | 0.602 (0.471 to 0.764) | 0.00001 | 0.000 |
| J07AL02 Pneumococcus vaccine conjugate | 20 | 95 | 0.29 | 0.68 | 0.419 (0.245 to 0.685) | 0.00021 | 0.003 |
| M05BA07 Risedronic acid | 47 | 165 | 0.68 | 1.19 | 0.567 (0.400 to 0.789) | 0.00042 | 0.005 |
| A16AX30 Ubiquinone (CoQ-10) | 9 | 56 | 0.13 | 0.40 | 0.321 (0.139 to 0.653) | 0.00052 | 0.006 |
| N06AB10 Escitalopram | 236 | 610 | 3.39 | 4.39 | 0.766 (0.654 to 0.894) | 0.00061 | 0.007 |
| C09BA05 Ramipril-hydrochlorothiazide | 121 | 342 | 1.74 | 2.46 | 0.702 (0.565 to 0.869) | 0.00082 | 0.009 |
| C01BC04 Flecainide | 7 | 43 | 0.10 | 0.31 | 0.325 (0.123 to 0.729) | 0.00253 | 0.023 |
| S01XA40 Hydroxypropyl-methylcellulose (tears) | 67 | 203 | 0.96 | 1.46 | 0.657 (0.490 to 0.871) | 0.00273 | 0.025 |
| A11CC05 Cholecalciferol | 737 | 1669 | 10.60 | 12.00 | 0.869 (0.792 to 0.954) | 0.00280 | 0.025 |
| B01AE07 Dabigatran etexilate | 37 | 124 | 0.53 | 0.89 | 0.595 (0.400 to 0.866) | 0.00543 | 0.042 |
| C09AA08 Cilazapril | 15 | 64 | 0.22 | 0.46 | 0.468 (0.247 to 0.831) | 0.00579 | 0.044 |
| N02CC04 Rizatriptan | 1 | 17 | 0.01 | 0.12 | 0.118 (0.003 to 0.750) | 0.01065 | 0.075 |
| A12CC04 Magnesium citrate | 33 | 108 | 0.48 | 0.78 | 0.609 (0.399 to 0.908) | 0.01191 | 0.080 |
| S01KA01 Hyaluronic acid (artificial tears) | 5 | 31 | 0.07 | 0.22 | 0.322 (0.098 to 0.836) | 0.01249 | 0.083 |
| C09DB01 Valsartan-amlodipine | 227 | 549 | 3.27 | 3.95 | 0.821 (0.698 to 0.963) | 0.01445 | 0.094 |
| A10BD07 Sitagliptin-metformin | 233 | 560 | 3.35 | 4.03 | 0.826 (0.704 to 0.967) | 0.01721 | 0.108 |
| B03BA51 Vit.B12 combinations | 31 | 100 | 0.45 | 0.72 | 0.618 (0.399 to 0.934) | 0.01979 | 0.119 |
| G03CA03 Estradiol | 18 | 67 | 0.26 | 0.48 | 0.536 (0.300 to 0.914) | 0.02047 | 0.122 |
| C09DA01 Losartan-hydrochlorothiazide | 124 | 315 | 1.78 | 2.27 | 0.783 (0.630 to 0.969) | 0.02424 | 0.140 |
| S01ED01 Timolol | 20 | 70 | 0.29 | 0.50 | 0.570 (0.328 to 0.949) | 0.02492 | 0.143 |
| G04BD12 Mirabegron | 22 | 74 | 0.32 | 0.53 | 0.593 (0.351 to 0.967) | 0.02998 | 0.163 |
| S01XA02 Retinol (eye ointment) | 3 | 21 | 0.04 | 0.15 | 0.285 (0.054 to 0.956) | 0.03015 | 0.163 |
| Z01CE01 Eye care wipes | 3 | 21 | 0.04 | 0.15 | 0.285 (0.054 to 0.956) | 0.03015 | 0.163 |
| N06AX12 Bupropion | 6 | 30 | 0.09 | 0.22 | 0.399 (0.136 to 0.976) | 0.03385 | 0.177 |
| N06BA04 Methylphenidate | 8 | 36 | 0.12 | 0.26 | 0.444 (0.178 to 0.972) | 0.03656 | 0.186 |
| A12AX05 Calcium-zinc CD | 0 | 10 | 0.00 | 0.07 | 0.000 (0.000 to 0.892) | 0.03696 | 0.186 |
| A11JC02 Multivitamins for ocular use | 25 | 81 | 0.36 | 0.58 | 0.616 (0.376 to 0.976) | 0.03827 | 0.191 |
-
Numbers are of patients from the group who have acquired a medication from the class in the last month before the index date.
p-Values are calculated according to Fisher's exact test. Medications are sorted by increasing order of p-values.
-
OR: odds ratio; [95% CI]: 95% confidence interval; FDR: false discovery rate calculated according to Benjamini-Hochberg (BH) procedure.
Shown in this table are anatomical therapeutic chemical (ATC) classes for which the p-value is less than 0.05, and for which the FDR is less than 0.20 (about 80% of entries are expected to be true positive).
Additional files
-
Source code 1
R source code, producing Figure 1.
- https://cdn.elifesciences.org/articles/68165/elife-68165-code1-v2.zip
-
Supplementary file 1
Supplementary tables and figures.
Suppl Tab 1 Multivariable logistic regression for hospitalization status according to ethnicity and medication consumption in Cohort 1. Suppl Tab 2 Multivariable logistic regression for hospitalization status according to ethnicity and medication consumption in Cohort 2. Figure supplements Forest plot showing association between drug use and hospitalization risk in each of the cohorts, divided by body mass index (BMI) category.
- https://cdn.elifesciences.org/articles/68165/elife-68165-supp1-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/68165/elife-68165-transrepform-v2.docx





