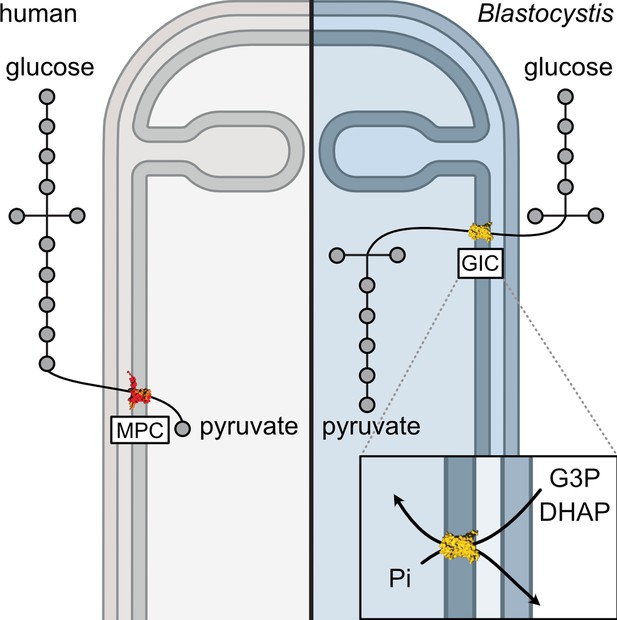
A mitochondrial carrier transports glycolytic intermediates to link cytosolic and mitochondrial glycolysis in the human gut parasite Blastocystis
Figures
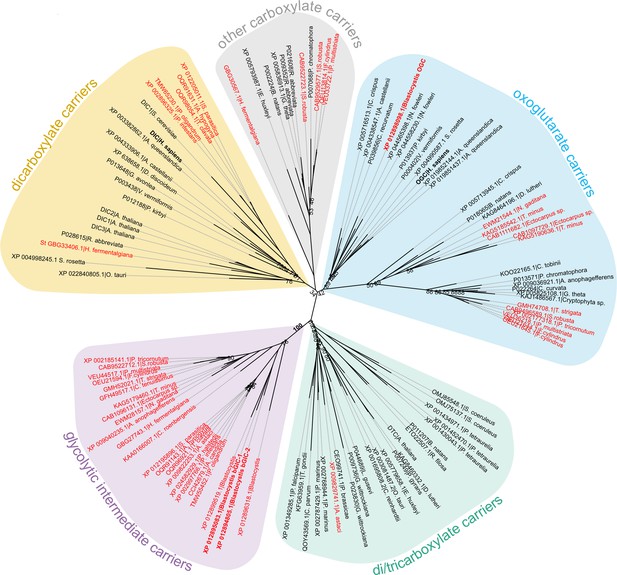
Phylogenetic analysis of carboxylate carriers.
The maximum likelihood tree was generated by IQ-TREE with the LG+F+R7 model suggested by ModelFinder. The support was calculated using ultrafast bootstrap analysis. The unrooted phylogenetic tree shows the uniqueness of the identified putative stramenopile glycolytic intermediate carriers (GIC) among eukaryotes. The stramenopile-specific GIC (pink) are most closely related to di/tricarboxylate carriers (DTC – green), and to a lesser extend to oxoglutarate carriers (OGC – blue) and dicarboxylate carriers (DIC – yellow). Stramenopile proteins are in red, proteins included in this study are in bold. Note the absence of any non-stramenopile sequences in this clade. Bootstrap values less than 90 and a value for the GIC group are indicated.
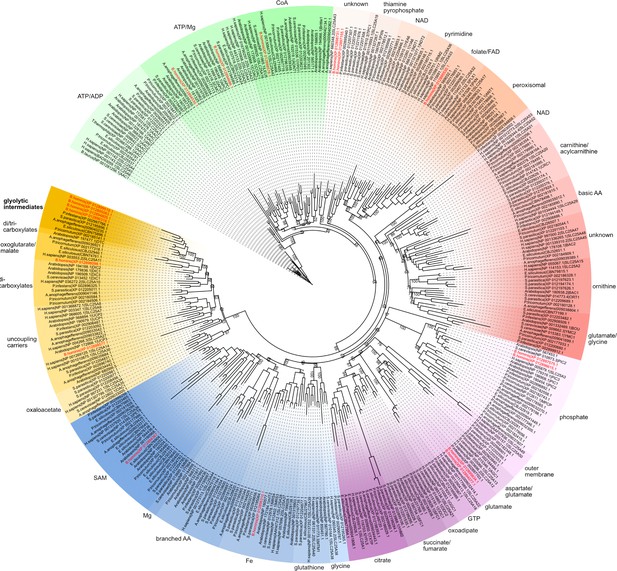
The maximum likelihood tree was generated by IQ-TREE with the LG+F+R9 model suggested by ModelFinder.
The support was calculated using ultrafast bootstrap analysis. The tree shows the relatedness of gathered human, Saccharomyces cerevisiae, and Arabidopsis thaliana mitochondrial carriers alongside a variety of stramenopile species. Blastocystis sequences are in red. The putative stramenopile glycolytic intermediate carriers are in dark mustard.
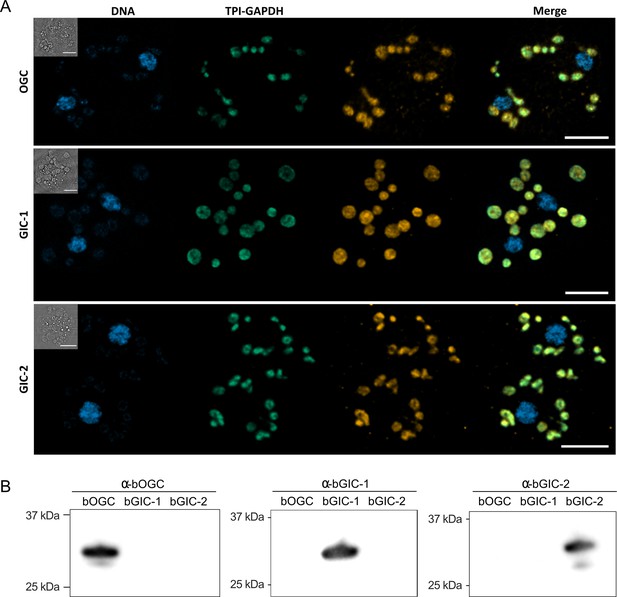
Specific antibodies localise glycolytic intermediate carriers in the mitochondria of Blastocystis.
(A) Localization of glycolytic intermediate carriers (GIC) in Blastocystis ST7-B cells using specific antibodies (orange). The signal colocalizes with the mitochondrial protein TPI-GAPDH (green) (Río Bártulos et al., 2018), demonstrating its mitochondrial localization. DNA is stained with Hoechst 33342 (blue). Inserts top left: corresponding brightfield image. The scale bar is 5 μm. (B) Western blots of Blastocystis OGC, GIC-1, and GIC-2 antibodies against purified protein confirm that there is no cross reactivity.
-
Figure 2—source data 1
Original file for the western blot analysis in Figure 2B.
- https://cdn.elifesciences.org/articles/94187/elife-94187-fig2-data1-v1.pdf
-
Figure 2—source data 2
PDF containing Figure 2B and original scans of the relevant western blot analysis with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/94187/elife-94187-fig2-data2-v1.pdf
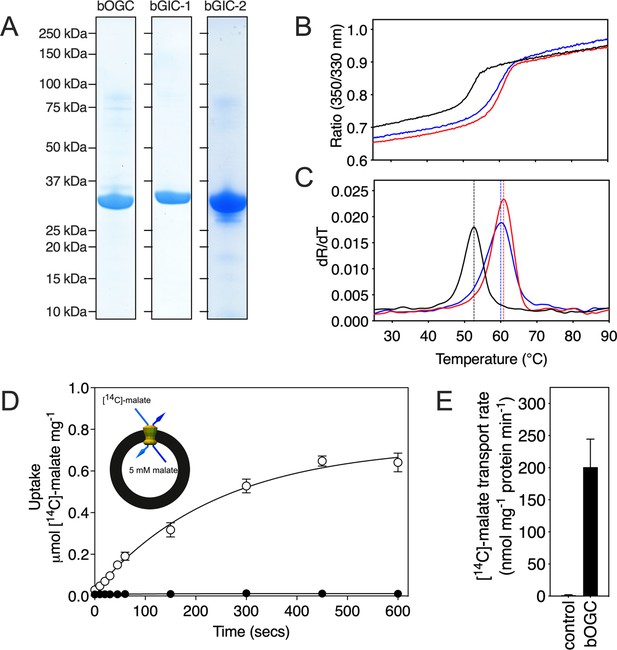
Expression, purification, and biophysical characterization of Blastocystis carriers.
(A) Instant-blue stained sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS–PAGE) gel of purified bOGC, bGIC-1, and bGIC-2. (B) Typical unfolding curves of bOGC (black trace), bGIC-1 (red trace), and bGIC-2 (blue trace) using nanoDSF. (C) The peak in the derivative of the unfolding curve (dR/dT) is the apparent melting temperature (Tm). (D) [14C]-malate uptake curve of bOGC reconstituted into proteoliposomes loaded with (white circles) or without (black circles) 5 mM malate. Transport was initiated by the external addition of 2.5 μM [14C]-malate. (E) Initial transport rates, calculated from panel (D) from the linear part of the uptake curve (first 60 s). The data represent the mean and standard deviation is of n = 4 (two biological repeats, each with two technical repeats).
-
Figure 3—source data 1
Original files for the sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS–PAGE) gels shown in Figure 3A.
- https://cdn.elifesciences.org/articles/94187/elife-94187-fig3-data1-v1.pdf
-
Figure 3—source data 2
PDF containing Figure 3A and original scans of the relevant sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS–PAGE) gels with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/94187/elife-94187-fig3-data2-v1.pdf
-
Figure 3—source data 3
Data used for generating graphs in Figure 3B–E.
- https://cdn.elifesciences.org/articles/94187/elife-94187-fig3-data3-v1.xlsx
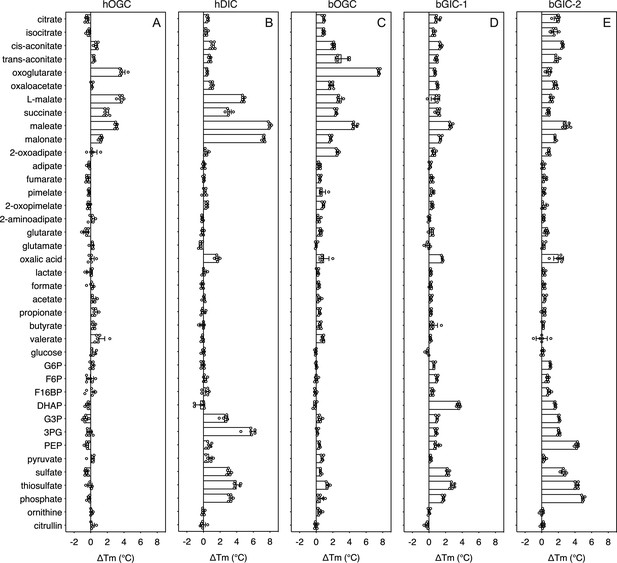
Thermostability screening of detergent-solubilized, purified protein using nanoDSF (Prometheus, Nanotemper).
(A) hOGC, (B) hDIC, (C) bOGC, (D) bGIC-1, and (E) bGIC-2 against 10 mM di- and tricarboxylates, glycolytic intermediates, and inorganic ions. The temperature shift (∆Tm) is the apparent melting temperature in the presence of compound minus the apparent melting temperature in the absence of compound. The data are represented by the mean and standard deviation of two biological repeats, each in triplicate (n = 6 in total). G6P, glucose-6-phosphate; F6P, fructose-6-phosphate; F16BP, fructose 1–6 bisphosphate; DHAP, dihydroxyacetone phosphate; G3P, glyceraldehyde-3-phosphate; 3PG, 3-phosphoglycerate; PEP, phosphoenolpyruvate.
-
Figure 4—source data 1
Data used for generating graphs in Figure 4A–E.
- https://cdn.elifesciences.org/articles/94187/elife-94187-fig4-data1-v1.xlsx
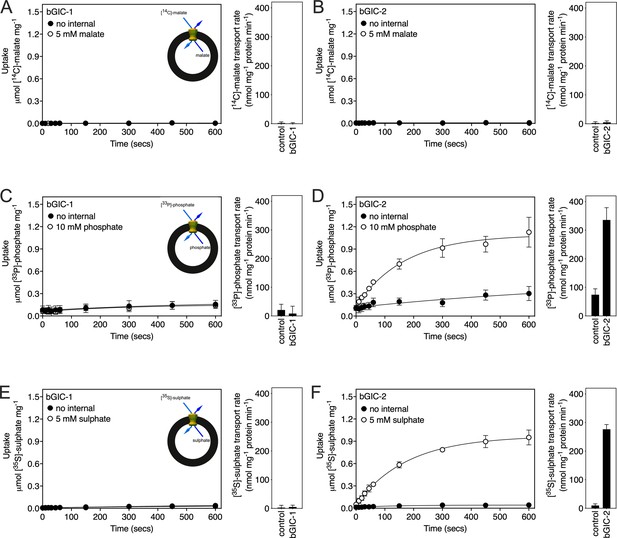
Proteoliposomes containing Blastocystis putative glycolytic intermediate carriers 1 (bGIC-1; A, C, E) and 2 (bGIC-2; B, D, F) were loaded with either (A, B) 5 mM malate, (C, D) 10 mM phosphate, or (E, F) 5 mM sodium sulphate, and transport initiated by the external addition of (A, B) 2.5 μM [14C]-malate, (C, D) 200 μM [33P]-phosphate, or (E, F) 25 μM [35S]-sulphate.
Controls with no internalized substrate were also tested (black circles). Initial transport rates were calculated from the linear part of the uptake curve (60 s). The data represent the mean and standard deviation of two biological repeats, each with two technical repeats (n = 4 in total).
-
Figure 5—source data 1
Data used for generating graphs in Figure 5A–F.
- https://cdn.elifesciences.org/articles/94187/elife-94187-fig5-data1-v1.xlsx
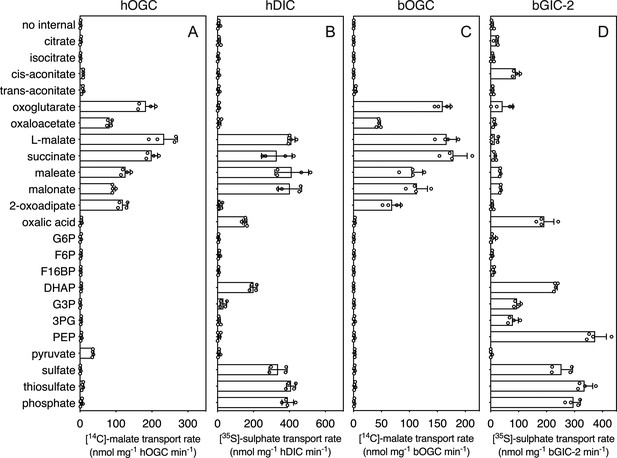
Hetero-exchange reactions catalysed by reconstituted carriers.
(A) Proteoliposomes containing human oxoglutarate carrier (hOGC, SLC25A11), (B) human dicarboxylate carrier (hDIC, SLC25A10), (C) Blastocystis oxoglutarate carrier (bOGC), and (D) Blastocystis putative glycolytic intermediate carrier 2 (bGIC-2) were loaded with 5 mM compound, and transport initiated by the external addition of 2.5 μM [14C]-malate (hOGC and bOGC) or 25 μM [35S]-sulphate (hDIC and bGIC-2). Initial transport rates were calculated from the linear part of the uptake curve (60 s). The data represent the mean and standard deviation of two biological repeats, each with two technical repeats (n = 4 in total). G6P, glucose-6-phosphate; F6P, fructose-6-phosphate; F16BP, fructose 1–6 bisphosphate; DHAP, dihydroxyacetone phosphate; G3P, glyceraldehyde-3-phosphate; 3PG, 3-phosphoglycerate; PEP, phosphoenolpyruvate.
-
Figure 6—source data 1
Data used for generating graphs in Figure 6A–D.
- https://cdn.elifesciences.org/articles/94187/elife-94187-fig6-data1-v1.xlsx