Disruption in CYLC1 leads to acrosome detachment, sperm head deformity, and male in/subfertility in humans and mice
Figures
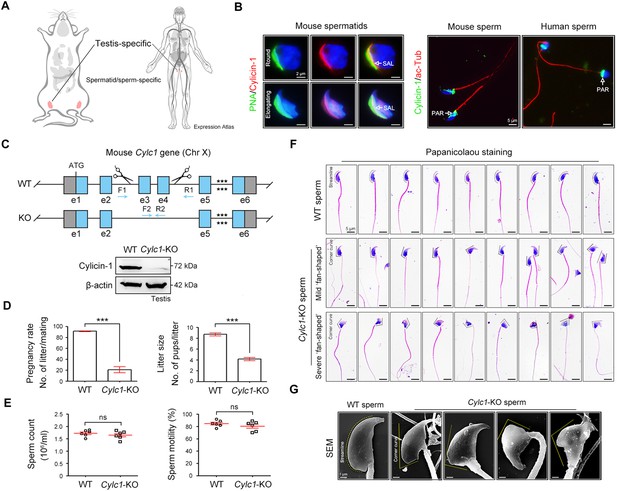
Severe male subfertility with sperm head deformity in Cylc1-KO mice.
(A) According to the Expression Atlas database (https://www.ebi.ac.uk/gxa/home), cylicin-1 was specifically expressed in the testis. Within the testis, cylicin-1 was restricted to the population of early and late spermatids. (B) Costaining of cylicin-1 and peanut agglutinin (PNA)-FITC (fluorescein Isothiocyanate) (an acrosome dye) in mouse round and elongating spermatids (left). Scale bars, 2 μm. Costaining of cylicin-1 and acetylated-tubulin (a flagella marker) in mouse and human sperm (right). Scale bars, 5 μm. Nuclei were counterstained with DAPI (4',6-diamidino-2-phenylindole). SAL, subacrosomal layer; PAR, postacrosomal region. (C) Schematic illustration of the targeting strategy used to generate Cylc1-KO mice. DNA extracted from mouse tails was used for PCR (polymerase chain reaction) genotyping. Knockout efficiency was determined by western blotting using testis protein lysates. β-Actin served as a loading control. (D) Adult Cylc1-KO male mice and their littermate wild-type (WT) mice (n = 3 each group) were continuously coupled with WT female mice at a ratio of 1:2 for 2 months. Data are represented as the mean ± standard error of the mean (SEM), Student’s t test, ***p < 0.001. (E) Sperm counts were determined using a fertility counting chamber under a light microscope, and total motility was assessed via computer-assisted semen analysis (CASA). Data are represented as the mean ± SEM (n = 6 each group), Student’s t test, ns, not significant. (F) Light microscopy analysis of sperm from WT mice and Cylc1-KO mice (n = 3 each) with Papanicolaou staining. The dashed curve indicates the streamline surface at the dorsal side of sperm heads of WT mice. The dashed broken lines point to the corner curve at the dorsal side of sperm heads of Cylc1-KO mice. Scale bars, 5 µm. (G) Scanning electron microscopy (SEM) images of sperm from WT mice and Cylc1-KO mice. Scale bars, 1 µm.
-
Figure 1—source data 1
Original file for the western blot in Figure 1C.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig1-data1-v1.zip
-
Figure 1—source data 2
PDF containing Figure 1C and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig1-data2-v1.zip
-
Figure 1—source data 3
Data used for analysis of Figure 1D.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig1-data3-v1.zip
-
Figure 1—source data 4
Data used for analysis of Figure 1E.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig1-data4-v1.zip
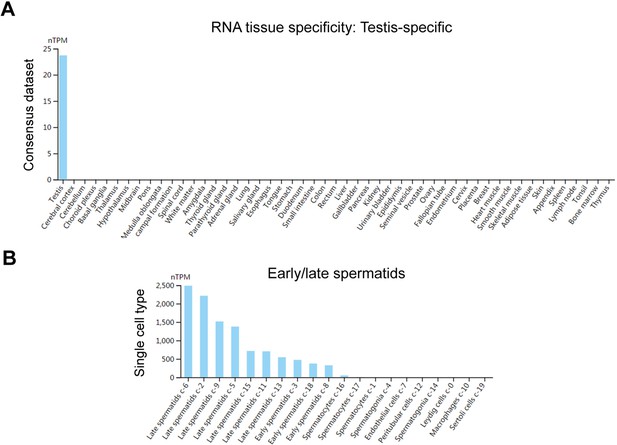
Expression pattern of human CYLC1 mRNA among different tissues and within the testis.
(A) The tissue data for mRNA expression were obtained from the Expression Atlas database (https://www.ebi.ac.uk/gxa). Human CYLC1 mRNA was specifically expressed in testes. (B) According to the Single Cell Expression dataset (https://www.ebi.ac.uk/gxa/sc), human CYLC1 mRNA was restricted to early/late spermatids within the testis.
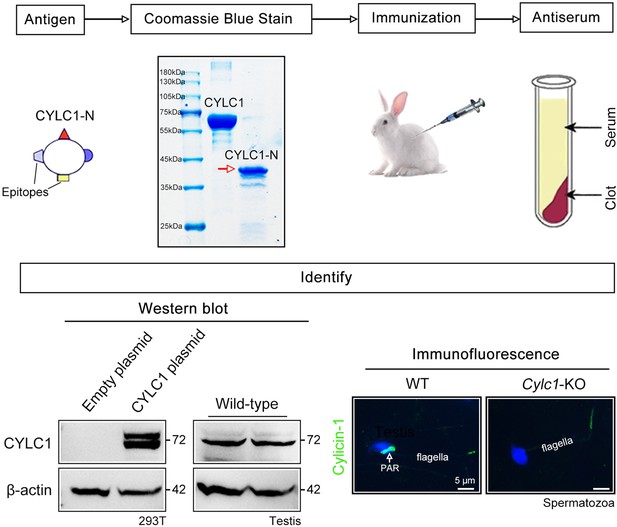
Generation of cylicin-1 antiserum.
Flow chart showing major steps to generate the cylicin-1 antiserum. First, the N-terminus of mouse CYLC1 was cloned into the pET-N-His-C-His vector and transfected into the ER2566 E. coli strain. CYLC1-N protein expression was induced by IPTG (isopropyl β-D-thiogalactopyranoside). Second, anti-His beads were added to the E. coli lysates. After incubation and washing, the CYLC1-N protein was eluted with imidazole. Third, recombinant CYLC1-N protein was emulsified with Freund’s complete adjuvant and administered subcutaneously into New Zealand white rabbits four times with an interval of 2 weeks. Fourth, 1 week after the last immunization, blood was collected, and the serum was separated. Fifth, the cylicin-1 antiserum was verified by western blotting and immunofluorescence staining. Cylicin-1 antiserum specifically recognized the cylicin-1 protein (~72 kDa) in the protein lysates of CYLC1-overexpressing HEK293T cells and testis tissues. Immunofluorescence staining of mouse sperm indicated that cylicin-1 was specifically localized to the postacrosomal region (PAR) of heads and that its expression was absent in the sperm of Cylc1-KO mice. Scale bars, 5 μm.
-
Figure 1—figure supplement 2—source data 1
Original file for the western blot in Figure 1—figure supplement 2.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig1-figsupp2-data1-v1.zip
-
Figure 1—figure supplement 2—source data 2
PDF containing Figure 1—figure supplement 2 and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig1-figsupp2-data2-v1.zip
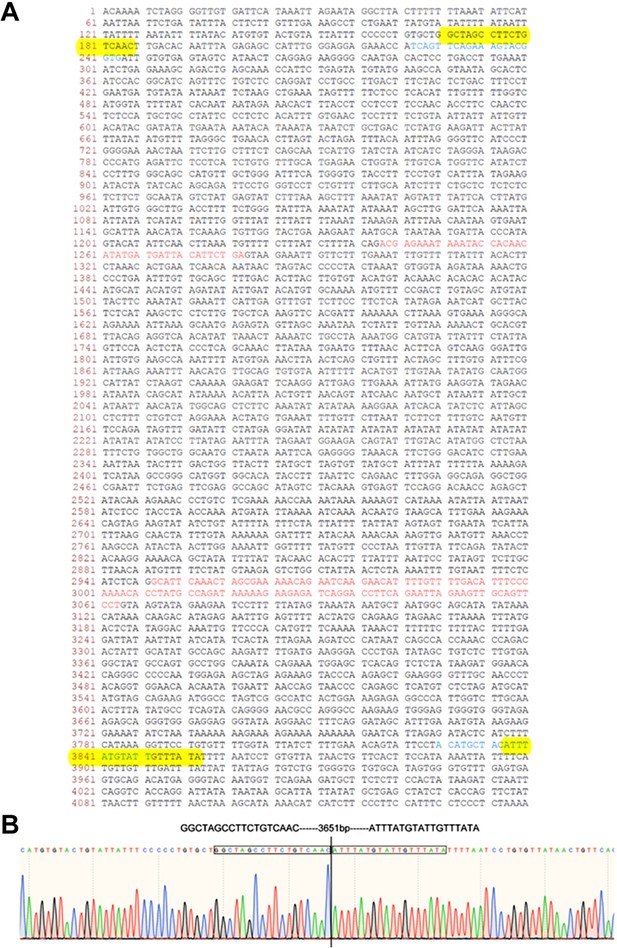
Construction report of Cylc1-KO mice.
(A) Wild-type genomic sequence of the mouse Cylc1 gene. Two gRNAs are marked in blue, and exons are marked in red. The sequence between the two yellow boxes was deleted. (B) Sanger sequencing confirmed that a 3651-bp region containing exons 3 and 4 of the Cylc1 gene was successfully deleted.
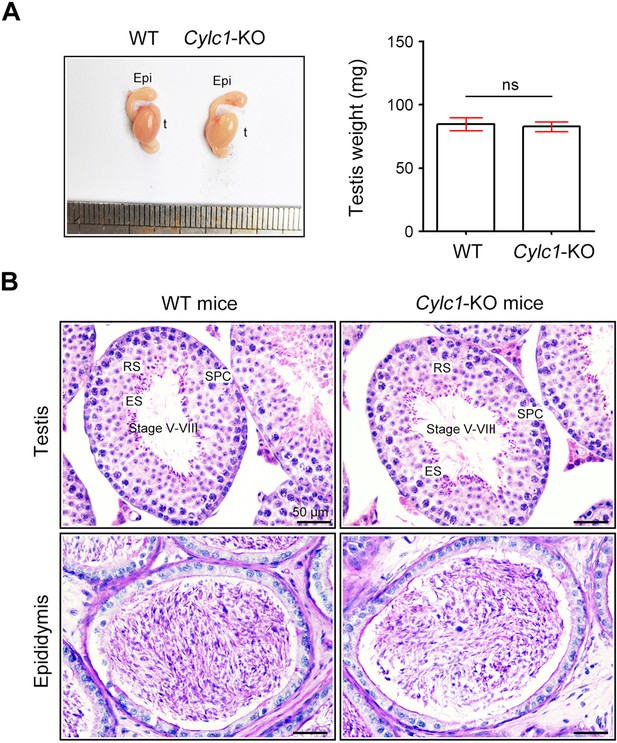
Analysis of the reproductive system of Cylc1-KO male mice.
(A) There was no obvious difference in testis size or weight between adult wild-type (WT) mice and Cylc1-KO mice. Data are presented as the mean ± standard error of the mean (SEM; n = 3 each); Student’s t test; ns, no significance. t, testis; epi, epididymis. (B) Histological morphology of testis and cauda epididymis from WT mice and Cylc1-KO mice by hematoxylin–eosin (H&E) staining. SPC, spermatocytes; RS, round spermatids; ES, elongating/elongated spermatids. Scale bars, 50 μm.
-
Figure 1—figure supplement 4—source data 1
Data used for analysis of Figure 1—figure supplement 4.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig1-figsupp4-data1-v1.zip
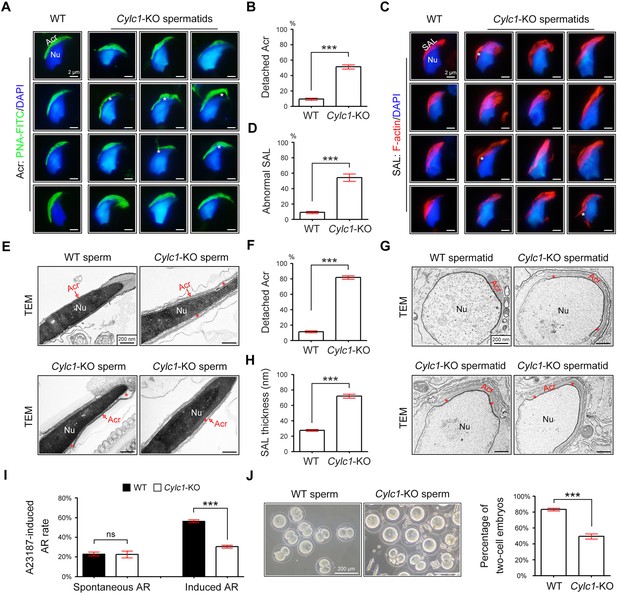
Acrosome detachment in the spermatids and sperm of Cylc1-KO mice.
(A) Visualization of the acrosome (Acr) in spermatids of wild-type (WT) and Cylc1-KO mice using the fluorescent dye peanut agglutinin (PNA)-FITC. The nuclei (Nu) were counterstained with DAPI dye. Asterisks indicate detached acrosomes. Scale bars, 2 µm. (B) Percentage of spermatids with detached Acr in WT and Cylc1-KO mice. (C) Subacrosomal layer of perinuclear theca (PT-SAL) actin bundles in spermatids of WT and Cylc1-KO mice was visible by an F-actin-Tracker Red dye. The asterisk indicates the detachment of Apx from Nu. Scale bars, 2 µm. (D) Percentage of spermatids with abnormal Apx structure in WT and Cylc1-KO mice. In B and D, 100 spermatids in each group (n = 3) were counted. Data are represented as the mean ± standard error of the mean (SEM), Student’s t test, ***p < 0.001. (E) Transmission electron microscopy (TEM) analysis showing detachment of Acr from Nu in the sperm of Cylc1-KO mice. The asterisk indicates the enlarged space area between the Acr and Nu. Scale bars, 200 nm. (F) Percentage of sperm with detached Acr in WT and Cylc1-KO mice as revealed by TEM. Twenty-five sperm in each group (n = 3) were counted. (G) TEM analysis of testis tissues revealing detachment of the developing Acr from Nu in spermatids of Cylc1-KO mice. The asterisk indicates the enlarged space area between the Acr and Nu. Scale bars, 200 nm. (H) Apx thickness in the sperm of WT mice and Cylc1-KO mice. The thickness of the Apx was measured by distance meter software of a Tecnai G2 Spirit electron microscope in TEM images at a magnification of ×30,000. Five spermatids were measured in each group (n = 3). Data in F and H are represented as the mean ± SEM, Student’s t test, ***p < 0.001. (I) Spontaneously underwent and A23187-induced acrosome reaction (AR). PNA-FITC dye was used to label acrosomes, and the sperm nuclei were stained with DAPI. One hundred sperm in each group (n = 3) were counted. Data are represented as the mean ± SEM, Student’s t test, ns, not significant; ***p < 0.001. (J) An in vitro fertilization assay was performed using sperm from Cylc1-KO mice and WT mice (n = 3 for each group). The percentage of two-cell embryos was calculated at 24 hr postfertilization. Data are represented as the mean ± SEM, Student’s t test, ***p < 0.001.
-
Figure 2—source data 1
Data used for analysis of Figure 2B.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig2-data1-v1.zip
-
Figure 2—source data 2
Data used for analysis of Figure 2D.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig2-data2-v1.zip
-
Figure 2—source data 3
Data used for analysis of Figure 2F.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig2-data3-v1.zip
-
Figure 2—source data 4
Data used for analysis of Figure 2H.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig2-data4-v1.zip
-
Figure 2—source data 5
Data used for analysis of Figure 2I.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig2-data5-v1.zip
-
Figure 2—source data 6
Data used for analysis of Figure 2J.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig2-data6-v1.zip
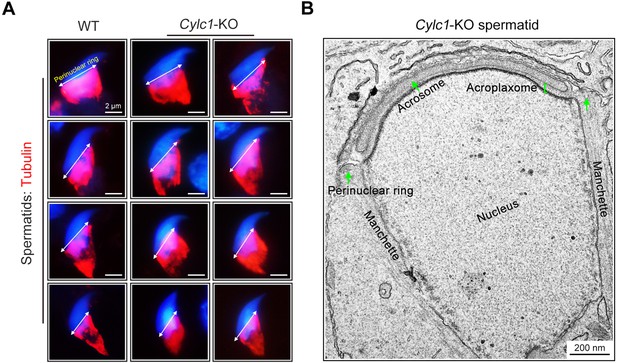
Normal development of manchette in spermatids of Cylc1-KO mice.
(A) The manchette of spermatids from wild-type (WT) mice and Cylc1-KO mice was observed by staining using Tubulin-Tracker Red dye. Nuclei were counterstained with DAPI. Double-headed arrows indicate the position of the perinuclear ring. Scale bars, 2 µm. (B) Transmission electron microscopy (TEM) of a spermatid of Cylc1-KO mice showing the generally well-formed manchette structure. Scale bar, 200 nm.

Roles of cylicin-1 in the formation of nuclear envelope (NE)–subacrosomal layer (SAL)–inner acrosomal membrane (IAM) protein complexes.
(A) Myc-tagged CYLC1 was transfected into HEK293T cells with or without FLAG-tagged ACTL7A. Protein lysates were immunoprecipitated with anti-FLAG antibody and then detected with anti-Myc antibody by sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS‒PAGE). (B) Mouse testis lysates were immunoprecipitated with anti-ACTL7A antibody or rabbit IgG and then detected with anti-CYLC1 antibody. (C) The binding affinity between different concentrations of purified CYLC1 and GFP-ACTL7A cell lysates was measured by microscale thermophoresis (MST). (D) Testis protein lysates from wild-type (WT) and Cylc1-KO mice were immunoprecipitated with anti-ACTL7A antibody and then detected with anti-ACTRT1 antibody. (E) Myc-tagged CYLC1 was immunoprecipitated with FLAG-tagged FAM209 in HEK293T cell extracts. (F) Mouse testis protein lysates were immunoprecipitated with anti-FAM209 antibody or rabbit IgG and then detected with anti-CYLC1 antibody. (G) The binding affinity between purified CYLC1 and GFP-FAM209 cell lysates was revealed by an MST assay. (H) Myc-tagged SPACA1 was immunoprecipitated with FLAG-tagged CYLC1 in HEK293T cell extracts. (I) Mouse testis protein lysates were immunoprecipitated with anti-SPACA1 antibody or rabbit IgG and then detected with anti-CYLC1 antibody. (J) Binding between GFP-SPACA1 from HEK293T cell lysates and recombinant CYLC1. (K) Testis protein lysates from WT mice or Cylc1-KO mice were incubated with anti-ACTL7A antibody and then detected with anti-SPACA1 antibody.
-
Figure 3—source data 1
Original file for the western blot in Figure 3A.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data1-v1.zip
-
Figure 3—source data 2
PDF containing Figure 3A and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data2-v1.zip
-
Figure 3—source data 3
Original file for the western blot in Figure 3B.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data3-v1.zip
-
Figure 3—source data 4
PDF containing Figure 3B and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data4-v1.zip
-
Figure 3—source data 5
Original file for the western blot in Figure 3D.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data5-v1.zip
-
Figure 3—source data 6
PDF containing Figure 3D and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data6-v1.zip
-
Figure 3—source data 7
Original file for the western blot in Figure 3E.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data7-v1.zip
-
Figure 3—source data 8
PDF containing Figure 3E and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data8-v1.zip
-
Figure 3—source data 9
Original file for the western blot in Figure 3F.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data9-v1.zip
-
Figure 3—source data 10
PDF containing Figure 3F and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data10-v1.zip
-
Figure 3—source data 11
Original file for the western blot in Figure 3H.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data11-v1.zip
-
Figure 3—source data 12
PDF containing Figure 3H and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data12-v1.zip
-
Figure 3—source data 13
Original file for the western blot in Figure 3I.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data13-v1.zip
-
Figure 3—source data 14
PDF containing Figure 3I and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data14-v1.zip
-
Figure 3—source data 15
Original file for the western blot in Figure 3K.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data15-v1.zip
-
Figure 3—source data 16
PDF containing Figure 3K and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-data16-v1.zip
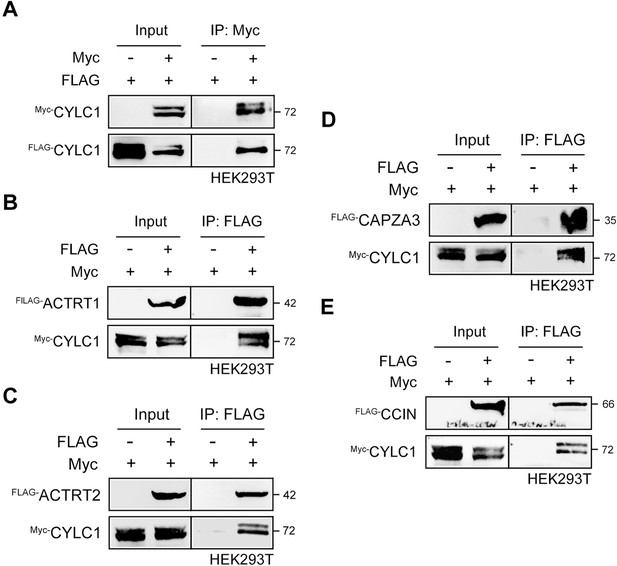
CYLC1 interacts with itself and several other perinuclear theca (PT) proteins.
(A) FLAG- and/or Myc-tagged CYLC1 was transfected into HEK293T cells. Protein lysates were immunoprecipitated with anti-Myc antibody and then detected with anti-FLAG antibody by sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS‒PAGE). Myc-tagged CYLC1 was transfected into HEK293T cells with or without FLAG-tagged ACTRT1 (B), ACTRT2 (C), CAPZA3 (D), or CCIN (E). Protein lysates were immunoprecipitated with anti-FLAG antibody and then detected with anti-Myc antibody by SDS‒PAGE.
-
Figure 3—figure supplement 1—source data 1
Original file for the western blot in Figure 3—figure supplement 1A.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data1-v1.zip
-
Figure 3—figure supplement 1—source data 2
PDF containing Figure 3—figure supplement 1A and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data2-v1.zip
-
Figure 3—figure supplement 1—source data 3
Original file for the western blot in Figure 3—figure supplement 1B.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data3-v1.zip
-
Figure 3—figure supplement 1—source data 4
PDF containing Figure 3—figure supplement 1B and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data4-v1.zip
-
Figure 3—figure supplement 1—source data 5
Original file for the western blot in Figure 3—figure supplement 1C.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data5-v1.zip
-
Figure 3—figure supplement 1—source data 6
PDF containing Figure 3—figure supplement 1C and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data6-v1.zip
-
Figure 3—figure supplement 1—source data 7
Original file for the western blot in Figure 3—figure supplement 1D.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data7-v1.zip
-
Figure 3—figure supplement 1—source data 8
PDF containing Figure 3—figure supplement 1D and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data8-v1.zip
-
Figure 3—figure supplement 1—source data 9
Original file for the western blot in Figure 3—figure supplement 1E.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data9-v1.zip
-
Figure 3—figure supplement 1—source data 10
PDF containing Figure 3—figure supplement 1E and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp1-data10-v1.zip
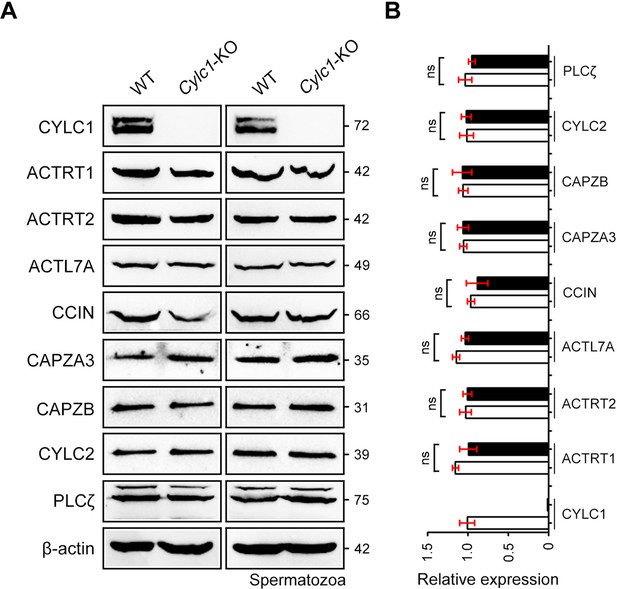
Expression of perinuclear theca (PT) proteins in sperm samples of wild-type (WT) mice and Cylc1-KO mice.
(A) Protein contents of PT proteins, such as CYLC1, ACTRT1, ACTRT2, ACTL7A, CCIN, CAPZA3, CAPZB, CYLC2, and PLCζ, were analysed by western blotting in sperm protein lysates from WT mice and Cylc1-KO mice. β-Actin served as an internal control. (B) Bar graphs representing band intensities, which were analysed by ImageJ software. Data represent the mean ± standard error of the mean (SEM) of three biological replicates. Student’s t test, ns: not significant.
-
Figure 3—figure supplement 2—source data 1
Original file for the western blot in Figure 3—figure supplement 2A.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp2-data1-v1.zip
-
Figure 3—figure supplement 2—source data 2
PDF containing Figure 3—figure supplement 2A and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp2-data2-v1.zip
-
Figure 3—figure supplement 2—source data 3
Data used for analysis of Figure 3—figure supplement 2B.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig3-figsupp2-data3-v1.zip

Identification of a CYLC1 variant in a large cohort of Chinese infertile men with sperm head deformities.
(A) More than 500 patients with sperm head deformities were recruited for WES and subsequent bioinformatic analysis. Nineteen individuals harboured a variant of CYLC1. (B) Sanger sequencing of the 19 individuals who were affected by the c.1377G>T/p.K459N variant in the CYLC1 gene. Sanger sequencing of the wild-type (WT) allele in healthy controls is also shown. (C) This variant was located at the fourth exon of the CYLC1 gene. Conservation analysis was performed by Basic Local Alignment Search Tool (BLAST). The NCBI protein ID for CYLC1: NP_066941.1 (Human), XP_001102833.1 (Macaca mulatta), XP_016799824.1 (Pan troglodytes), NP_776727.1 (Bovin), XP_023489864.1 (Horse), NP_080410.3 (Mouse), and XP_006229368.1 (Rat). The pathogenicity of variants was analysed by Polyphen2 and SIFT.
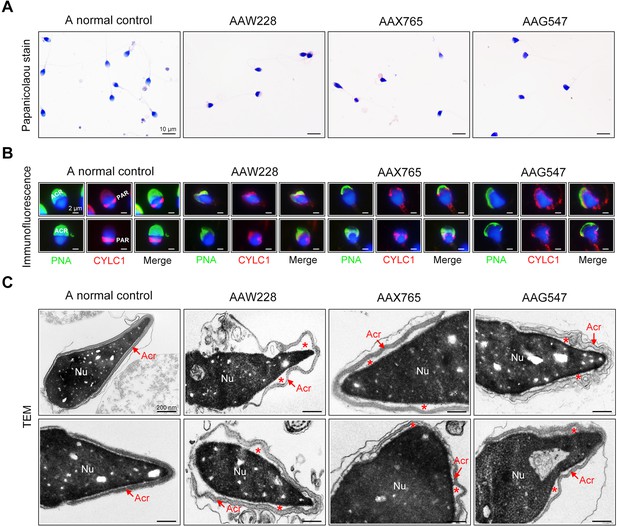
Sperm morphology and ultrastructure analyses for patients harbouring the CYLC1 variant.
(A) Papanicolaou staining of sperm from a fertile individual and three patients (AAW228, AAX765, and AAG547) harbouring the CYLC1 variant. Sperm from patients exhibited deformed heads. Scale bars, 10 µm. (B) Sperm were costained with anti-cylicin-1 antibody and peanut agglutinin (PNA)-FITC (an acrosome dye). In control sperm, Cylicin-1 was specifically localized to the postacrosomal region (PAR) and PNA-FITC-labelled acrosomes covering the anterior of heads. Both cylicin-1 distribution and acrosome structure were altered in the sperm of three patients. Scale bars, 2 µm. (C) Transmission electron microscopy (TEM) analysis of sperm from a normal control and three patients. Detached acrosomes of sperm were frequently observed in patient samples. The asterisk indicates the enlarged space area between the acrosome and the nucleus (Nu). Scale bars, 200 nm.
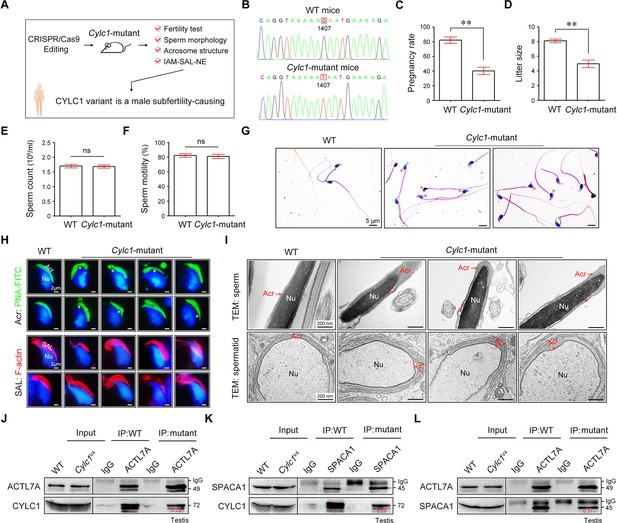
Cylc1-mutant mice show male subfertility with sperm acrosome detachment.
(A) Mice carrying an SNV (c.1407G>T/p.K469N) in the Cylc1 gene equivalent to that in nine patients harboring the CYLC1 variant were generated using CRISPR/Cas9 technology. (B) Sanger sequencing of the Cylc1 gene in wild-type (WT) mice and Cylc1-mutant mice. (C, D) Adult Cylc1-mutant male mice and their littermate controls (n = 3 each group) were continuously coupled with WT female mice at a ratio of 1:2 for 2 months. (E) Sperm count (106/ml) of Cylc1-mutant mice and WT mice. (F) Total sperm motility (%) of Cylc1-mutant mice and WT mice. In C–F, data are all represented as the mean ± standard error of the mean (SEM), Student’s t test, **p < 0.01; ns, not significant. (G) Sperm morphology of Cylc1-mutant mice and WT mice. Scale bars, 5 µm. (H) Visualization of the acrosome (Acr) in spermatids using the fluorescent dye peanut agglutinin (PNA)-FITC (above). Subacrosomal layer (SAL) bundles of spermatids were visible by F-actin-Tracker Red staining (below). The asterisk indicates the enlarged space area between the acrosome and the nucleus. Scale bars, 2 µm. (I) Transmission electron microscopy (TEM) analysis showing detachment of the acrosome from the sperm nucleus in Cylc1-mutant mice (above). TEM analysis of testis tissues revealing the detachment of the developing acrosome from the nucleus in spermatids of Cylc1-mutant mice (below). The asterisk indicates the enlarged space area between the acrosome and the nucleus. Scale bars, 200 nm. (J) Testis protein lysates from WT mice and Cylc1-mutant mice were immunoprecipitated with anti-ACTL7A antibody or rabbit IgG and then detected with anti-cylicin-1 antibody. (K) Testis protein lysates were immunoprecipitated with anti-SPACA1 antibody or rabbit IgG and then detected with anti-cylicin-1 antibody. (L) Testis protein lysates were immunoprecipitated with anti-ACTL7A antibody or rabbit IgG and then detected with anti-SPACA1 antibody.
-
Figure 6—source data 1
Data used for analysis of Figure 6C.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data1-v1.zip
-
Figure 6—source data 2
Data used for analysis of Figure 6D.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data2-v1.zip
-
Figure 6—source data 3
Data used for analysis of Figure 6E.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data3-v1.zip
-
Figure 6—source data 4
Data used for analysis of Figure 6F.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data4-v1.zip
-
Figure 6—source data 5
Original file for the western blot in Figure 6J.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data5-v1.zip
-
Figure 6—source data 6
PDF containing Figure 6J and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data6-v1.zip
-
Figure 6—source data 7
Original file for the western blot in Figure 6K.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data7-v1.zip
-
Figure 6—source data 8
PDF containing Figure 6K and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data8-v1.zip
-
Figure 6—source data 9
Original file for the western blot in Figure 6L.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data9-v1.zip
-
Figure 6—source data 10
PDF containing Figure 6L and relevant western blot with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-data10-v1.zip
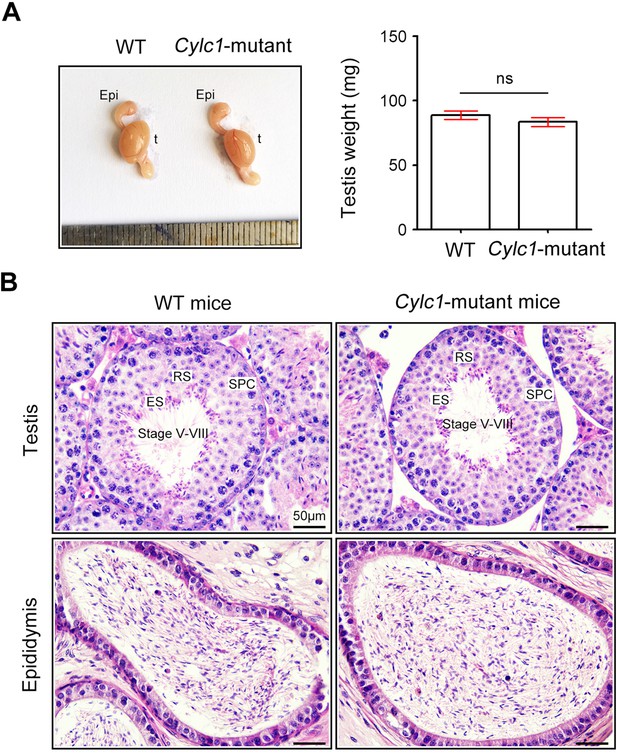
Analysis of the reproductive system of Cylc1-mutant mice.
(A) The testis size and weight were similar between wild-type (WT) mice and Cylc1-mutant mice. Data are presented as the mean ± standard error of the mean (SEM) (n = 3 each); Student’s t test; ns, no significance. t, testis; epi, epididymis. (B) Histological morphology of testis and cauda epididymis from WT mice and Cylc1-mutant mice by hematoxylin–eosin (H&E) staining. SPC, spermatocytes; RS, round spermatids; ES, elongating/elongated spermatids. Scale bars, 50 μm.
-
Figure 6—figure supplement 1—source data 1
Data used for analysis of Figure 6—figure supplement 1.
- https://cdn.elifesciences.org/articles/95054/elife-95054-fig6-figsupp1-data1-v1.zip
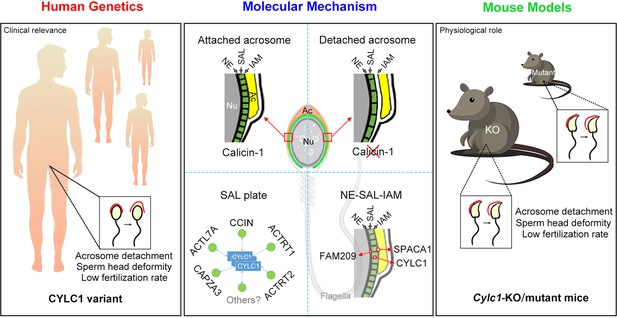
Schematic diagram showing the physiological role of CYLC1 on acrosome attachment in humans and mice.
A CYLC1 variant is identified in a large cohort of >500 Chinese infertile men with sperm head deformities. Both Cylc1-KO mice and Cylc1-mutant mice exhibited severe male subfertility with spermatozoa showing detached acrosome and head deformities. Cylicin-1 is specifically expressed at the acroplaxome (Apx), between nuclear envelope (NE) and inner acrosomal membrane (IAM), of spermatids. Cylicin-1 interacts itself and with other perinuclear theca (PT) proteins (including CCIN, ACTRT1, ACTRT2, ACTL7A, and CAPZA3) in the acroplaxome layer and mediates the acrosome–nucleus connection through interacting with IAM protein SPACA1 and NE protein FAM209.
Tables
Nineteen individuals carrying a CYLC1 variant identified from >500 infertile men with sperm head deformities.
| Variant | |
|---|---|
| cDNA alternation | c.1377G>T |
| Protein alteration | p.K459N |
| Patient(s) | AAG547; BBB654; BBB841; AAX199; AAX765; BBA344; BBB396; AAW228; AAT812; 3086; 3165; 3172; 3190; 3203; 3209; 3307; 3256; 3010; 2431 |
| Allele frequency in human population | |
| gnomAD genome | 0.001386 |
| Functional prediction | |
| SIFT | Deleterious |
| PolyPhen-2 | Damaging |
| CADD | 15.18 |
| | Conservation between mouse and human |
| | Yes |
-
NCBI reference sequence number of CYLC1 is GenBank: NM_021118.3.
Semen characteristics of individuals harbouring the CYLC1 variant.
| Individuals | Infertile for | Semen characteristics | ||||
|---|---|---|---|---|---|---|
| Volume (ml) | Concentration (106/ml) | Motility sperm (%) | Progressive motility (%) | Morphological normality (%) | ||
| AAG547 | 5 y | 1.8 | 50.2 | 49.2 | 43.1 | 3 |
| BBB654 | 3 y | 2.7 | 50.4 | 54.9 | 51 | 0 |
| BBB841 | 5 y | 4 | 52.3 | 81.1 | 62.3 | 1–2 |
| AAX199 | 3 y+ | 2.7 | 55 | 55.2 | 40.8 | 2 |
| AAX765 | 5 y | 3.2 | 4.4 | 35.9 | 19.2 | 0 |
| BBA344 | 3 y | 3 | <2 | 41.9 | 36.5 | 1 |
| BBB396 | 6 y | 1.6 | 12.8 | 30.8 | 23.1 | 0 |
| AAW228 | 5 y | 3.9 | 7.31 | 71.6 | 39.4 | 3 |
| AAT812 | 2 y | 3 | 58.7 | 21.8 | 7.6 | 3.6 |
| 3086 | 1 y | 2 | 0.9 | 30.8 | 23.1 | 0 |
| 3165 | 1 y | 1.2 | 58 | 0 | 0 | 0 |
| 3172 | 2 y | 3 | 151.5 | 14.1 | 9.6 | 1 |
| 3190 | 2 y | 2 | 22.6 | 29.5 | 26.2 | 0 |
| 3203 | 4 y | 1.2 | 24.1 | 8.9 | 6.2 | 1 |
| 3209 | 5 y | 6.4 | 10.6 | 14.2 | 4.1 | 0 |
| 3307 | 6 y | 5 | 59.9 | 77.6 | 57.1 | 4 |
| 3256 | 1 y | 1.4 | 66.6 | 67.5 | 53 | 3 |
| 3010 | 2 y | 3.4 | 54.4 | 13 | 10.2 | 1 |
| Reference values | >1.5 | >15 | >40 | >32 | >4 | |
Clinical outcomes of assisted reproductive technologies (ART) using sperm from men harbouring the CYLC1 variant.
| Individual | IVF/ICSIcycles | IVF | ICSI | ICSI + AOA | Transfer cycles (embryos) | Live birth | |||
|---|---|---|---|---|---|---|---|---|---|
| Fertilization rate | Good-quality embryo rate | Fertilization rate | Good-quality embryo rate | Fertilization rate | Good-quality embryo rate | ||||
| AAW228 | 2 | 0% (0/10) | - | 30% (3/10) | 0% (0/3) | 91.3% (21/23) | 66.7% (14/21) | 2 (4) | 1 |
| AAG547 | 4 | 10.5% (2/19) | 50% (1/2) | 78.9% (15/19) | 46.7 (7/15) | N/A | N/A | 3 (6) | 1 |
| AAX199 | 4 | 0% (0/2) | - | 83.3% (5/6) | 100% (5/5) | 100% (4/4) | 100% (4/4) | 5 (8) | 0 |
| BBB654 | 2 | 0% (0/14) | - | 88.9% (8/9) | 100% (8/8) | N/A | N/A | 2 (4) | 0 |
| BBB841 | 5 | 0% (0/5) | - | 50% (3/6) | 100% (3/3) | 75% (3/4) | 100% (3/3) | 2 (3) | 0 |
| AAT812 | 3 | 35.7% (5/14) | 0% (0/5) | 80% (8/10) | 50% (4/8) | 80% (4/5) | 75% (3/4) | 2 (4) | 1 |
| 3307 | 2 | 0% (1/13) | - | 100% (8/8) | 37.5% (3/8) | N/A | N/A | 1 (1) | P |
| 3256 | 1 | 0% (0/3) | - | 100% (5/5) | 60% (3/5) | N/A | N/A | 1 (1) | P |
| 3172 | 1 | 33.3% (1/3) | 0% (0/1) | N/A | N/A | N/A | N/A | 1 (1) | 0 |
| AAX765 | 7 | N/A | N/A | 72.7% (8/11) | 0% (0/8) | 100% (6/6) | 50% (3/6) | 2 (3) | 1 |
| BBA344 | 1 | N/A | N/A | N/A | N/A | 94.1% (16/17) | 37.5% (6/16) | 2 (3) | 1 |
| BBA396 | 1 | N/A | N/A | 100% (1/1) | 100% (1/1) | 75% (3/4) | 100% (3/3) | 1 (2) | 1 |
| 3086 | 1 | N/A | N/A | 100% (12/12) | 66.7% (8/12) | N/A | N/A | 2 (2) | 1 |
| 3165 | 1 | N/A | N/A | 66.7% (4/6) | 75% (3/4) | N/A | N/A | 1 (2) | 1 |
| 3190 | 1 | N/A | N/A | 93.7% (15/16) | 33.3% (5/15) | N/A | N/A | 1 (1) | 1 |
| 3203 | 1 | N/A | N/A | 83.3% (5/6) | 60% (3/5) | N/A | N/A | 1 (1) | 1 |
| 3209 | 1 | N/A | N/A | 71.4% (5/7) | 20% (1/5) | N/A | N/A | 2 (2) | 0 |
| 3010 | 1 | N/A | N/A | 70% (7/10) | 57.1% (4/7) | N/A | N/A | 1 (2) | 1 |
-
N/A, not available; P, pregnant.
Additional files
-
Supplementary file 1
WES reveals variants in known sperm head deformity-associated genes in infertile men.
- https://cdn.elifesciences.org/articles/95054/elife-95054-supp1-v1.docx
-
Supplementary file 2
Intracytoplasmic sperm injection (ICSI) experiments using sperm from wild-type (WT) mice or Cylc1-KO mice.
- https://cdn.elifesciences.org/articles/95054/elife-95054-supp2-v1.docx
-
Supplementary file 3
Primers for mouse genotyping.
- https://cdn.elifesciences.org/articles/95054/elife-95054-supp3-v1.docx
-
Supplementary file 4
Primers for Sanger sequencing of CYLC1 in humans.
- https://cdn.elifesciences.org/articles/95054/elife-95054-supp4-v1.docx
-
Supplementary file 5
Antibodies used in this study.
- https://cdn.elifesciences.org/articles/95054/elife-95054-supp5-v1.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/95054/elife-95054-mdarchecklist1-v1.pdf





