A SUMO E3 ligase promotes long non-coding RNA transcription to regulate small RNA-directed DNA elimination
Figures
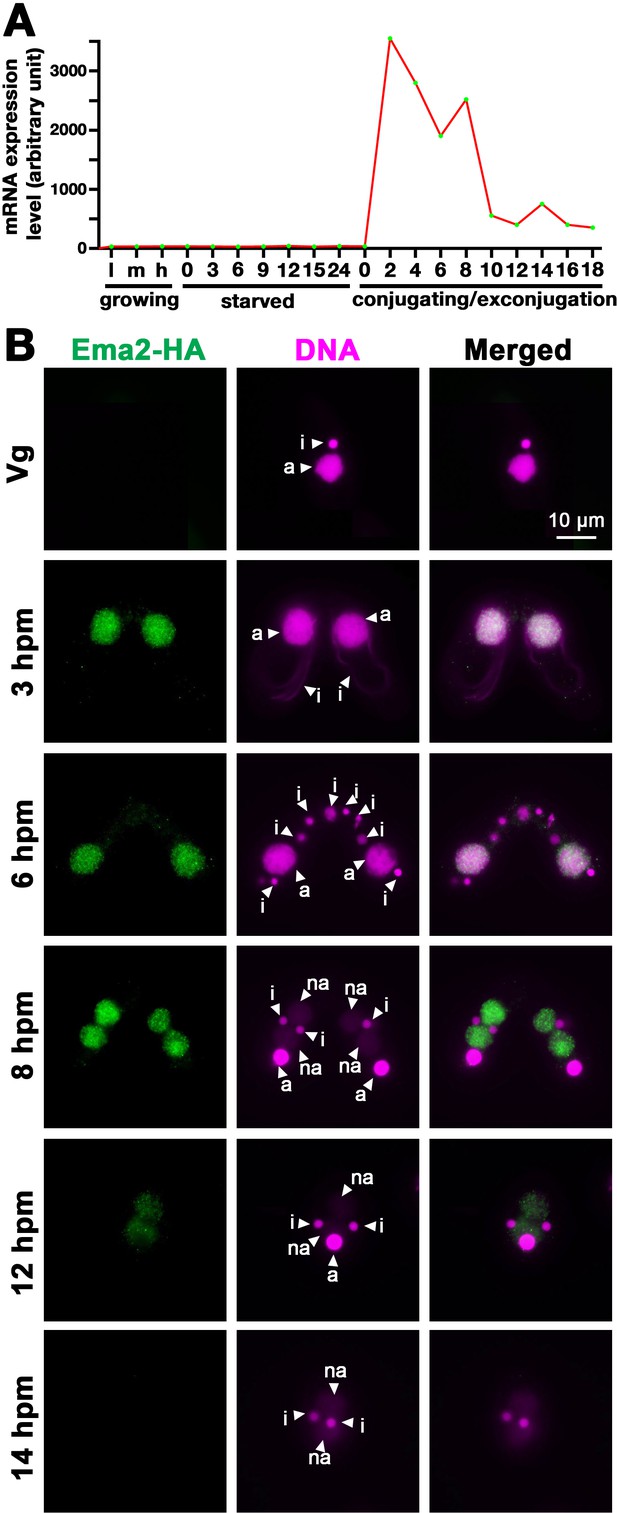
Ema2 is expressed during conjugation and localized in the macronucleus (MAC).
(A) EMA2 mRNA expression levels (in an arbitrary unit) in growing Tetrahymena cells in low (l), middle (m), and high (h) cell concentrations, starved cells from 0 to 24 hr, and cells in the conjugation and post conjugation stages from 0 to 18 hr post-mixing (hpm) are shown. The mRNA expression data were obtained from Miao et al., 2009. (B) Ema2 localization. Two Ema2-HA strains were mated and fixed at the indicated time points (Vg = vegetative cell). An anti-HA antibody was used to localize Ema2-HA (green), and DNA was stained with DAPI (magenta). The micronucleus (MIC), the parental MAC, and the newly formed MAC are marked with arrowheads with ‘i,’ ‘a,’ and ‘na,’ respectively. All pictures share the scale bar.
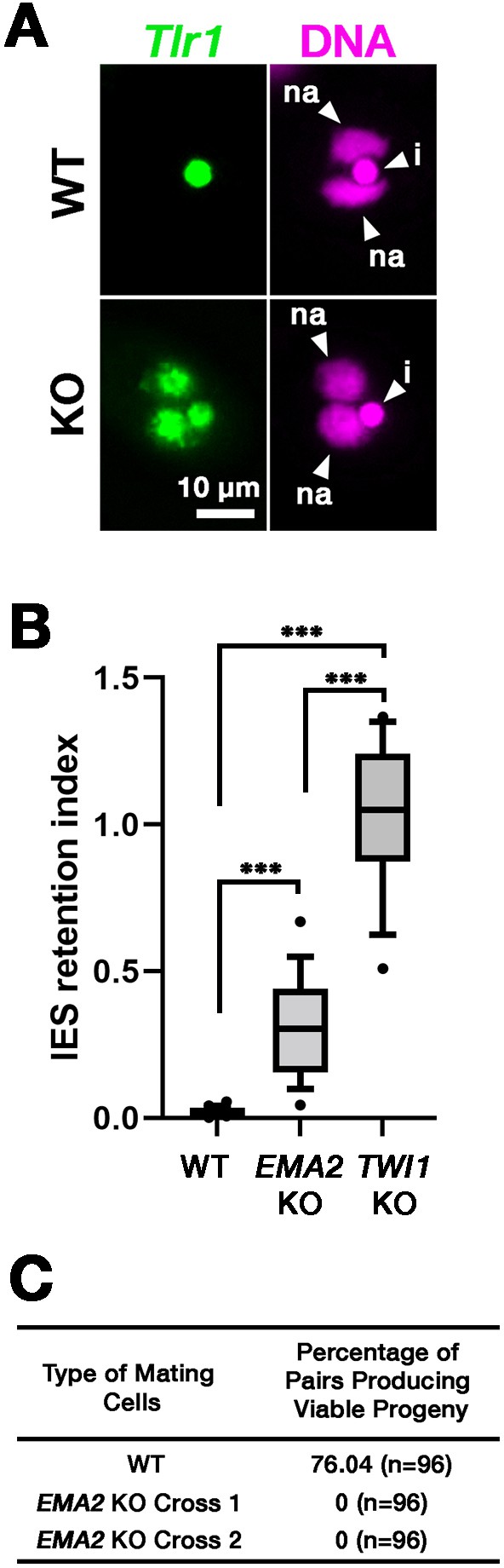
Ema2 is required for completing DNA elimination.
(A) Two wild-type (WT) or two EMA2 somatic KO (KO) cell lines were mixed, and their exconjugants at 36 hpm were analyzed by DNA-fluorescent in situ hybridization (FISH) with fluorescent probes complementary to the Tlr1 element (green). DNA was counterstained with DAPI (magenta). The micronucleus (MIC) and the new macronucleus (MAC) are marked with arrowheads with ‘i’ and ‘na’, respectively. All pictures share the scale bar. (B) Exconjugants from wild-type (WT) cells, EMA2 somatic KO cells, and TWI1 KO cells were stained as in (A), the IES retention index was calculated (see Materials and Methods for details) from 20 cells each, and shown as box plots. The whiskers represent 10-90 percentile. Three asterisks (***) indicate a p-value of less than 0.001 in the Welch two-sample t-test. (C) Two wild-type (WT) cells and two independent crosses of EMA2 somatic KO cells (Cross 1 and 2) were mated, and the conjugating pairs were isolated for the viability test. The percentages of pairs that gave rise to viable sexual progeny are shown. ‘n’ represents the number of total pairs tested.
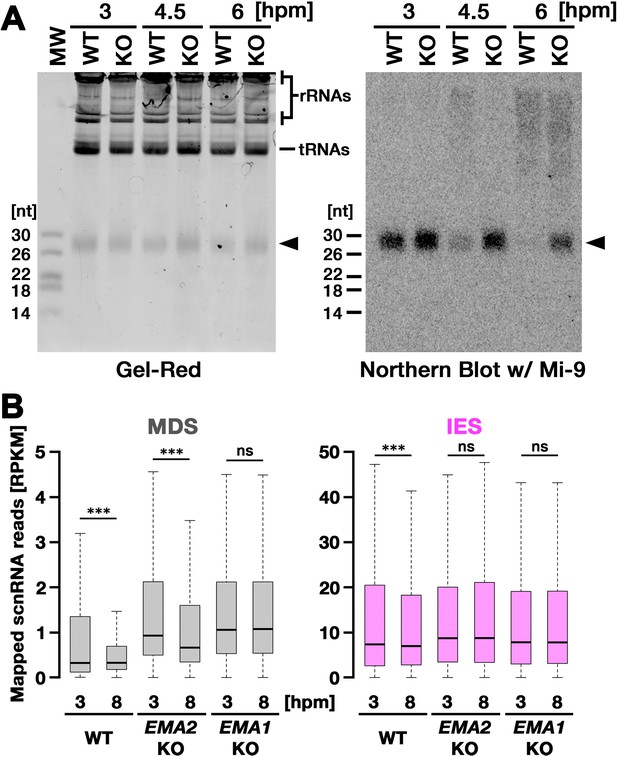
Ema2 is required for target-directed small RNAs (scnRNA) degradation (TDSD).
(A) Total RNA was isolated from conjugating wild-type (WT) and EMA2 somatic KO (KO) cells at 3, 4.5, and 6 hr post-mixing (hpm), separated in denaturing gel and stained with the nucleic acid dye Gel-Red (right). Then, RNA was transferred to a membrane and hybridized with the radioactive Mi-9 probe, which is complementary to a repetitive sequence in MDSs (right). (B) Small RNAs from conjugating wild-type (WT), EMA2 somatic KO, and EMA1 somatic KO strains were isolated at 3 and 8 hpm and analyzed by high-throughput sequencing. Normalized numbers (RPKM [read per kilobase of unique sequence per million]) of sequenced small RNAs (26–32 nt) that uniquely matched to the macronuclear-destined sequences (MDS) (left) or internal elimination sequence (IES) (right) genomic tiles (see Materials and methods) are shown as box plots. The median value is represented by the horizontal bar in the box. The minimum and maximum values are indicated by the bars on top and bottom of the box, respectively, with 1.5 x the interquartile range (IQR). Three asterisks (***) and ‘ns’ respectively indicate a p-value of less than 0.001 and more than 0.05 in the Wilcoxon rank sum test.
-
Figure 3—source data 1
The raw data of northern blot (top) and Gel-red stained gel (bottom) without (Figure_3 A_Original) and with (Figure_3 A_Original-marked) marks of the positions of regions used for Figure 3A.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig3-data1-v2.zip
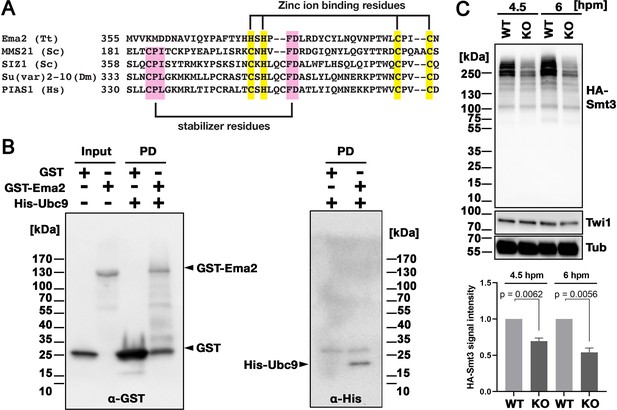
Ema2 acts as a SUMO E3 ligase.
(A) The SP-RING domain of Ema2 is compared with that of MMS21 and SIZ1 in S. cerevisiae, Su(var)2–10 in D. melanogaster and PIAS1 in H. sapiens. The conserved cysteine and histidine residues that are involved in zinc ion binding are highlighted in yellow. The residues that stabilize the domain structure of some SP-RING domain proteins are marked with pink. (B) GST alone (GST), GST-tagged Ema2 (GST-Ema2), and His-tagged Ubc9 (His-Ubc9) were recombinantly expressed in E. coli and purified. GST and GST-Ema2 were immobilized on glutathione beads and incubated with His-Ubc9. Proteins retained on the beads were eluted, and the input and eluted proteins (PD) were analyzed by western blotting using anti-GST (left) and anti-His (right) antibodies. (C) An EMA2 somatic KO strain expressing HA-tagged Smt3 was crossed with a wild-type strain (WT cross, WT) or another EMA2 somatic KO strain (EMA2 KO cross, KO), and their total proteins at the indicated time points were analyzed by western blotting using anti-HA (top), anti-Twi1 (middle) and anti-alpha tubulin (bottom) antibodies. The signal intensities of the anti-HA blots in the individual entire lanes were quantified in three independent experiments. The values in the WT cross were normalized to 1, and their means and standard deviations are presented as a bar graph, with p-values determined by the Welch two-sample t-test.
-
Figure 4—source data 1
The raw data of western blot without (Figure_4B_Original) and with (Figure_4B_Original-marked) marks of the positions of regions used for Figure 4B.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig4-data1-v2.zip
-
Figure 4—source data 2
The raw data of western blot without (Figure_4 C_Original) and with (Figure_4 C_Original-marked) marks of the positions of regions used for Figure 4C.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig4-data2-v2.zip
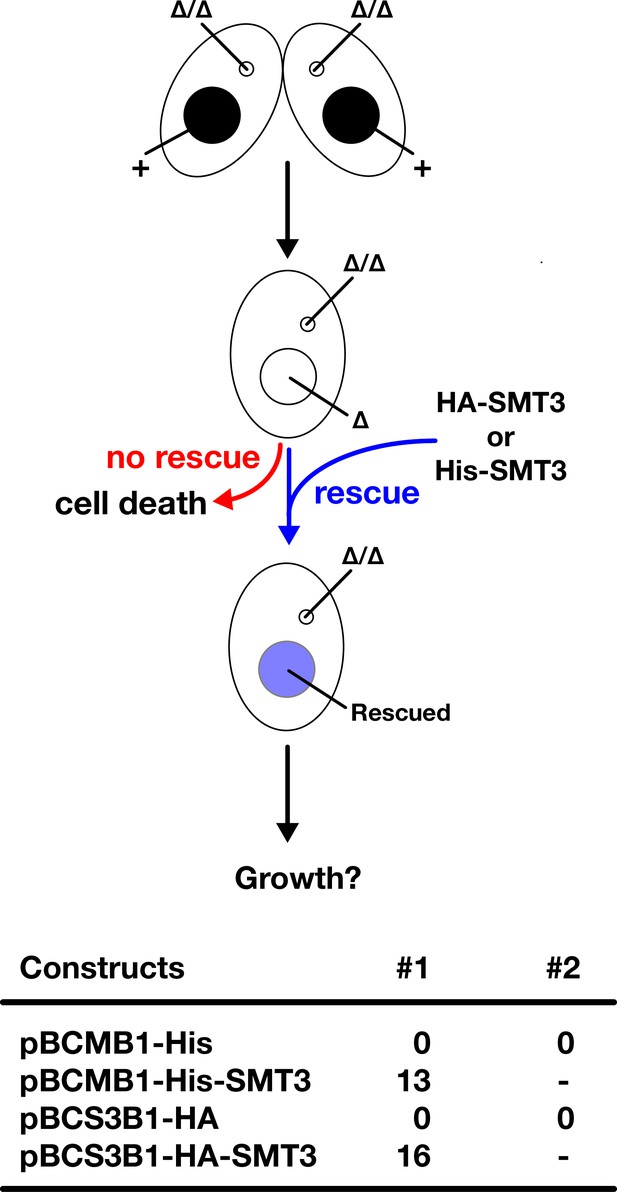
HA-tagged Smt3 (HA-Smt3) and His-Smt3 can replace the essential function of Smt3.
(Top) Schematic representation of genetic rescue experiment of SMT3 KO. Two SMT3 KO homozygous heterokaryon strains were mated and His-SMT3 or HA-SMT3 constructs (or their corresponding vectors without the SMT3 gene) were introduced into the macronucleus (MAC) of their progeny (SMT3 KO homozygous homokaryon). The progeny survives only when the introduced construct can replace the functions of SMT3 in vegetative cells. (Bottom) Number of obtained SMT3 KO homozygous homokaryon strains that were genetically rescued by 10 µg DNA of the indicated constructs are shown. For the empty constructs (pBCMB1-His and pBCS3B1-HA), two transformation experiments (#1 and #2) were done to confirm their inability to restore the cell viability of KO cells.
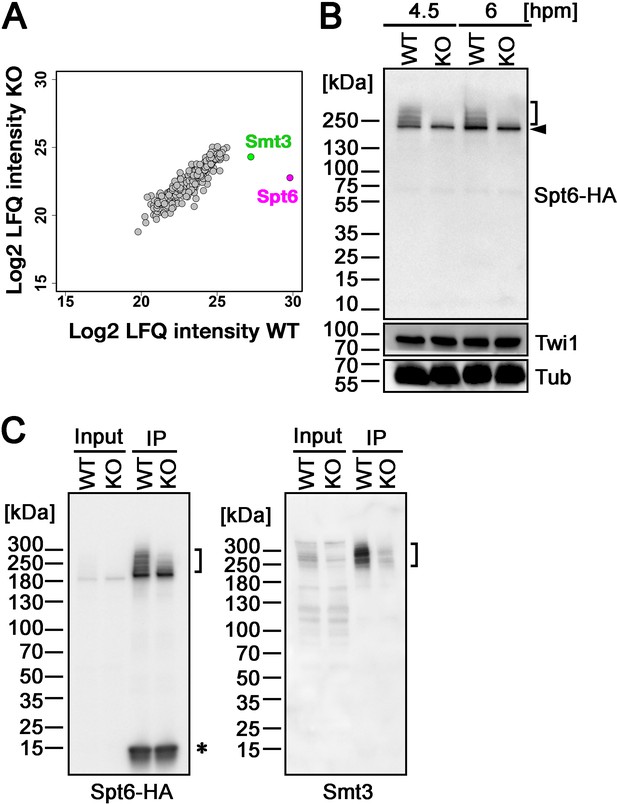
Ema2 promotes SUMOylation of Spt6.
(A) A construct expressing His-tagged Smt3 (His-Smt3) was introduced into a wild-type and an EMA2 somatic KO strain and crossed with another wild-type (WT cross) or EMA2 somatic KO strain (EMA2 KO cross). Proteins were harvested at 6 hpm, and His-Smt3-conjugated proteins were purified with Ni-NTA beads from conjugating cells and analyzed by mass spectrometry. Values of log base 2 of label-free quantification (LFQ) intensities of each identified protein between the WT cross (WT) and EMA2 KO cross (KO) were compared. (B) A construct expressing HA-tagged Spt6 (Spt6-HA) was introduced into an EMA2 somatic KO strain and crossed with a wild-type (WT-cross, WT) or another EMA2 somatic KO (KO-cross, KO) strain. Total proteins were harvested at 4.5 and 6 hpm, and Spt6-HA was detected by western blotting using an anti-HA antibody. Twi1 and alpha-tubulin (Tub) were also analyzed to monitor mating efficiency and loading, respectively. The positions of modified and unmodified Spt6-HA proteins are marked with a bracket and an arrowhead, respectively. (C) Total proteins were harvested from WT and KO crosses at 6 hpm (input), and Spt6-HA was immunoprecipitated using an anti-HA antibody (IP). The purified proteins were analyzed by western blotting using an anti-HA (left) or an anti-Smt3 (right) antibody. SUMOylated Spt6 proteins are marked with brackets. Unidentified protein cross-reacting with the anti-HA antibody is marked with an asterisk.
-
Figure 5—source data 1
The raw data of western blot without (Figure_5B_Original) and with (Figure_5B_Original-marked) marks of the positions of regions used for Figure 5B.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig5-data1-v2.zip
-
Figure 5—source data 2
The raw data of western blot without (Figure_5 C_Original) and with (Figure_5 C_Original-marked) marks of the positions of regions used for Figure 5C.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig5-data2-v2.zip
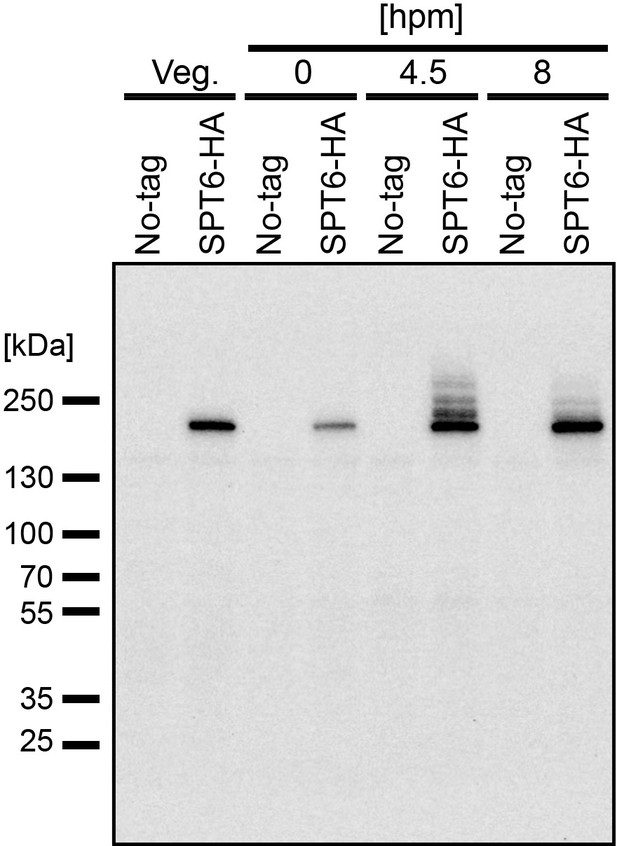
Slower migrating Spt6 species appear during conjugation.
A construct expressing HA-tagged Spt6 (Spt6-HA) was introduced into a wild-type strain to produce a SPT6-HA strain. Total proteins were harvested either from vegetatively growing the SPT6-HA strain (Veg) or from mating SPT6-HA strain with another wild-type strain at 0, 4.5, and 6 hpm. As a control, the same wild-type strain without the Spt6-HA-expressing construct (No-tag) was analyzed similarly. Spt6-HA was detected by western blotting using an anti-HA antibody.
-
Figure 5—figure supplement 1—source data 1
The raw data of western blot without (Figure_5-figure supplement_1_Original) and with (Figure_5-figure supplement_1_Original-marked) marks of the positions of regions used for Figure 5—figure supplement 1.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig5-figsupp1-data1-v2.zip
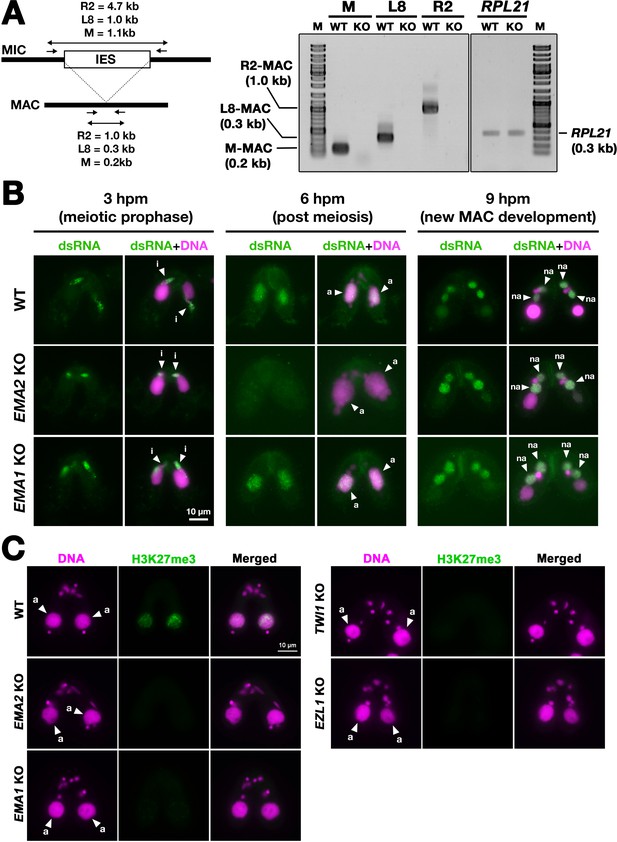
Ema2 is required for the accumulation of long non-coding RNA (lncRNA) transcripts from the parental macronucleus (MAC).
(A) (Left) Schematic representation of the RT‒PCR assay. The black bars and the open box represent the macronuclear-destined sequence (MDS) and internal elimination sequence (IES), respectively. The arrows represent the primers used for RT‒PCR. The lengths of the PCR amplicons are shown with double-sided arrows. (Right) Wild-type (WT) or EMA2 somatic KO (KO) cells were mated, and their total RNAs at 6 hpm were used for RT‒PCR. The positions corresponding to the PCR products of lncRNAs from the MAC M, L8, and R2 loci are marked. RPL21 mRNA was also analyzed as a positive control. (B) Conjugating wild-type (WT), EMA2 somatic KO, and EMA1 somatic KO cells at the indicated time points were analyzed by immunofluorescence staining using the anti-long dsRNA antibody J2 (dsRNA, green). DNA was counterstained with DAPI (magenta). The MIC, the parental MAC, and the new MAC are indicated with arrowheads with ‘i,’, ‘a,’and ‘“na,’ respectively. All pictures share the scale bar. (C) Heterochromatin formation in the parental MAC. Conjugating wild-type (WT), EMA2 somatic KO, EMA1 somatic KO, TWI1 complete (somatic + germline) KO, and EZL1 somatic KO cells at 6 hpm were analyzed by immunofluorescence staining using an anti-H3K27me3 antibody (green). DNA was counterstained with DAPI (magenta). The micronucleus (MIC) and the parental MAC are marked with arrowheads with ‘i’ and ‘a,’ respectively. All pictures share the scale bar.
-
Figure 6—source data 1
The raw data of ethidium bromide-stained PCR products separated in agarose gels without (Figure_6 A_Original) and with (Figure_6 A_Original-marked) marks of the positions of regions used for Figure 6A.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig6-data1-v2.zip
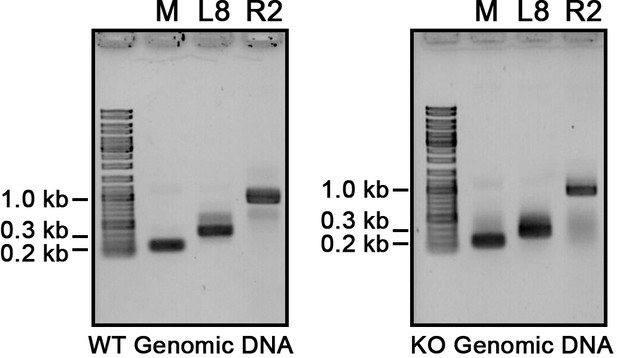
Confirmation of the presence of the corresponding genomic loci that were used to detect long non-coding RNAs (lncRNAs).
Genomic DNA from wild-type (WT) and EMA2 somatic KO (KO) were used for PCR with the same primers used for the RT-PCR assay in Figure 6A.
-
Figure 6—figure supplement 1—source data 1
The raw data of ethidium bromide-stained PCR products separated in agarose gels without (Figure_6-figure supplement 1-Original) and with (Figure_6-figure supplement 1-Original-marked) marks of the positions of regions used for Figure 6—figure supplement 1.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig6-figsupp1-data1-v2.zip
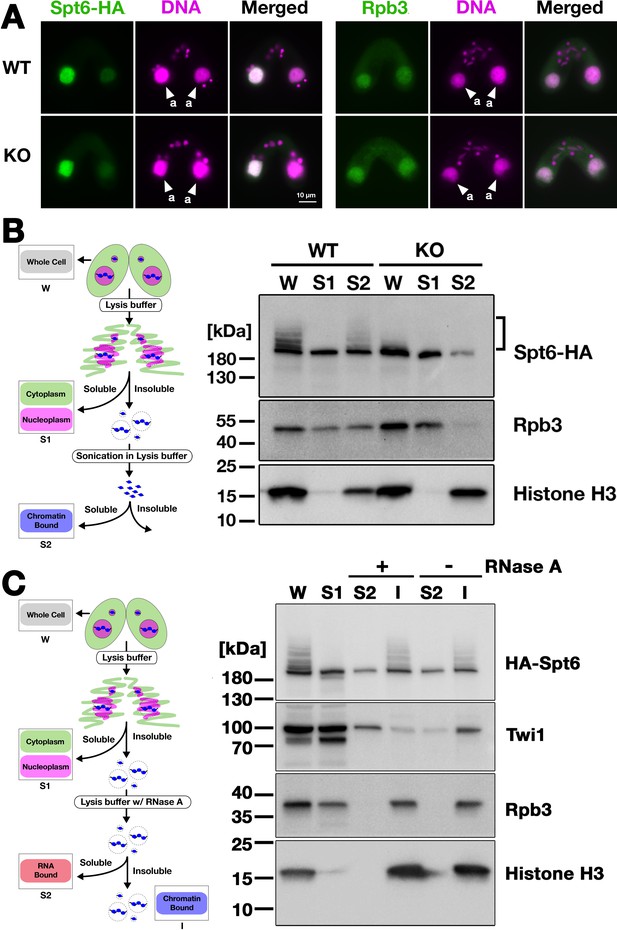
Ema2 facilitates the chromatin association of Spt6 and RNA polymerase II.
(A) A construct expressing HA-tagged Spt6 (Spt6-HA) was introduced into an EMA2 somatic KO strain and crossed with a wild-type (WT-cross, WT) or another EMA2 somatic KO (KO-cross, KO) strain. The localizations of Spt6-HA and Rpb3, the third largest subunit of RNA polymerase II (RNAPII), were analyzed by immunofluorescence staining at 4.5 hpm using anti-HA (left) or anti-Rpb3 (right) antibodies, respectively. DNA was counterstained with DAPI (magenta). The parental macronucleus (MACs) are marked with arrowheads with ‘a.’ All other structures stained by DAPI are micronucleus (MICs). All pictures share the scale bar. (B) (Left) Schematic representation of the cell fractionation assay. Conjugating cells were incubated with a lysis buffer that releases cytoplasmic (green) and nucleoplasmic (magenta) proteins to the soluble fraction (S1). Then, the insoluble fraction was resuspended in fresh lysis buffer and sonicated. The solubilized fraction after sonication (S2) contains fragmented chromatin. (Right) S1 and S2 fractions as well as total cellular proteins (W) from WT-cross (WT) and EMA2 KO-cross (KO) explained in (A) at 4.5 hpm were analyzed by western blotting using anti-HA (Spt6-HA), anti-Rpb3, and anti-histone H3 antibodies. The position of SUMOylated Spt6-HA is marked with a bracket. (C) (Left) Schematic representation of the cell fractionation assay with RNase treatment. Conjugating cells were incubated with a lysis buffer that releases cytoplasmic (green) and nucleoplasmic (magenta) proteins to the soluble fraction (S1). Then, the insoluble fraction was incubated in fresh lysis buffer with (+) or without (-) RNase A, and the solubilized (S2) and insoluble (I) fractions were obtained. (Right) S1, S2, and I fractions as well as total cellular proteins (W) from SPT6 germline (MAC + MIC) KO strains rescued with the HA-SPT6-WT construct (see Figure 8) were analyzed by western blotting using anti-HA (HA-Spt6), anti-Twi1p, anti-Rpb3, and anti-histone H3 antibodies. The position of SUMOylated HA-Spt6 is marked with a bracket.
-
Figure 7—source data 1
The raw data of western blot without (Figure_7B-Original) and with (Figure_7B-Original-marked) marks of the positions of regions used for Figure_7B.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig7-data1-v2.zip
-
Figure 7—source data 2
The raw data of western blot without (Figure_7C-Original) and with (Figure_7C-Original-marked) marks of the positions of regions used for Figure_7 C.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig7-data2-v2.zip
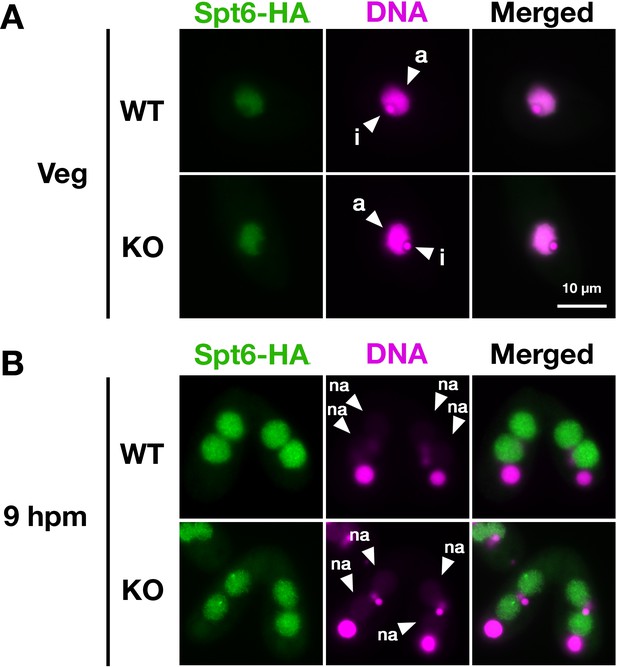
Localization of Spt6 at late conjugation stage.
A construct expressing HA-tagged Spt6 (Spt6-HA) was introduced into an EMA2 somatic KO strain and crossed with a wild-type (WT-cross, WT) or another EMA2 somatic KO (KO-cross, KO) strain. The localization of Spt6-HA was analyzed by immunofluorescence staining at 9 hpm using an anti-HA antibody. DNA was counterstained with DAPI (magenta). The new macronucleus (MACs) are marked with arrowheads with ‘na.’ All pictures share the scale bar.
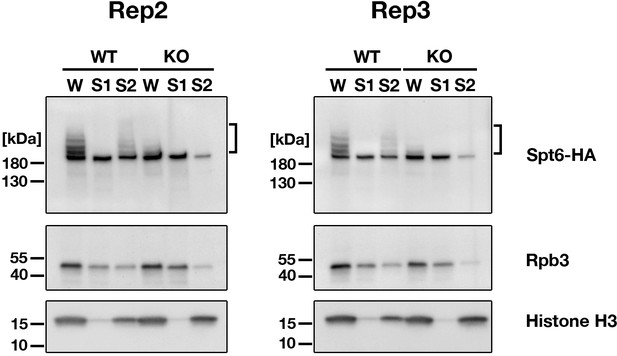
Replicated cell fractionation assay examining the chromatin association of proteins.
Conjugating cells were incubated with a lysis buffer that releases cytoplasmic (green) and nucleoplasmic (magenta) proteins to the soluble fraction (S1). Then, the insoluble fraction was resuspended in fresh lysis buffer and sonicated. The solubilized fraction after sonication (S2) contains fragmented chromatin. S1 and S2 fractions as well as total cellular proteins (W) from wild-type (WT)-cross (WT) and EMA2 KO-cross (KO) at 4.5 hpm were analyzed by western blotting using anti-HA (Spt6-HA), anti-Rpb3 and anti-histone H3 (bottom) antibodies. The position of SUMOylated Spt6-HA is marked with a bracket.
-
Figure 7—figure supplement 2—source data 1
The raw data of western blot without (Figure_7-figure supplement 2-Original) and with (Figure_7-figure supplement 2-Original-marked) marks of the positions of regions used for Figure 7—figure supplement 2.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig7-figsupp2-data1-v2.zip

Replicated cell fractionation assay examining proteins associated with the chromatin in an RNA-dependent manner.
Conjugating cells were treated with a lysis buffer to release cytoplasmic and nucleoplasmic proteins into the soluble fraction (S1). Subsequently, the insoluble fraction was subjected to incubation in fresh lysis buffer with (+) or without (-) RNase A, and the resulting solubilized (S2) and insoluble (I) fractions were obtained. These fractions, along with total cellular proteins (W) from SPT6 germline (macronucleus, MAC + micronucleus, MIC) KO strains rescued with the HA-SPT6-WT construct (refer to Figure 8), were analyzed by western blotting using anti-HA (HA-Spt6), anti-Twi1p, anti-Rpb3, and anti-histone H3 (bottom) antibodies. The position of SUMOylated HA-Spt6 is marked with a bracket.
-
Figure 7—figure supplement 3—source data 1
The raw data of western blot without (Figure_7-figure supplement 3-Original) and with (Figure_7-figure supplement 3-Original-marked) marks of the positions of regions used for Figure 7—figure supplement 3.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig7-figsupp3-data1-v2.zip
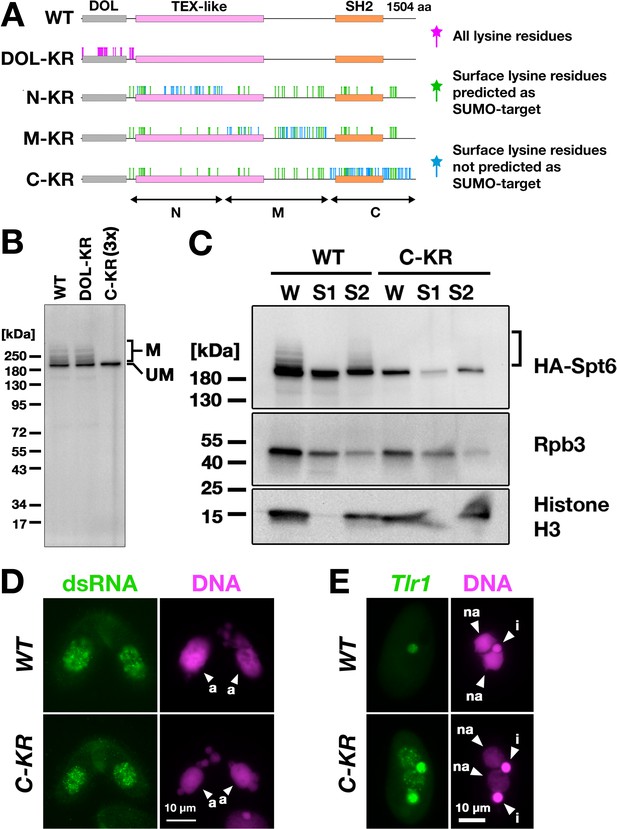
Spt6 SUMOylation is dispensable for parental macronucleus (MAC) long non-coding RNA (lncRNA) transcription.
(A) Schematic representation of SPT6 mutant constructs with lysine (K)-to-arginine (R) substitutions. Spt6 has a domain of low complexity (DOL) region followed by conserved TEX-like and SH2 domains. The DOL-KR mutant has K to R substitutions for all lysine residues in the DOL region. N-KR, M-KR, and C-KR mutants have K to R substitutions for lysine residues that were on the surface of a predicted Spt6 structure in one of the three non-DOL regions (N, M, and C-regions) in addition to those surface lysine residues that were also predicted to be SUMOylatable by an algorithm in all three regions. (B) Total proteins were harvested at 4.5 hpm from conjugating SPT6 germline (MAC + MIC) KO cells rescued with wild-type (HA-SPT6-WT), HA-SPT6-DOL-KR, or HA-SPT6-C-KR constructs. Because mating was low in the HA-SPT6-C-KR rescued cells, three times (3 x) more total protein sample was loaded for these cells. HA-Spt6 was detected by western blotting using an anti-HA antibody. The positions of modified and unmodified HA-Spt6 proteins are marked with ‘M’ and ‘UM,’ respectively. (C) HA-SPT6-WT (WT) and HA-SPT6-C-KR (C–KR) rescued cells at 4.5 hpm were analyzed by cell fractionation as described in Figure 7B. The position of SUMOylated HA-Spt6 is marked with a bracket. (D) Accumulation of long dsRNAs in HA-SPT6-WT (WT) and HA-SPT6-C-KR (C–KR) rescued cells at 4.5 hpm was analyzed by immunofluorescence staining using the J2 antibody as described in Figure 6B. The parental MACs are marked with arrowheads with ‘a.’ All other structures stained by DAPI are micronucleus (MICs). All pictures share the scale bar. (E) HA-SPT6-WT (WT) and HA-SPT6-C-KR (C–KR) rescued cells at 36 hpm were analyzed by DNA- FISH with fluorescent probes complementary to the Tlr1 element (red). DNA was counterstained with DAPI (magenta). The MIC and the new MAC are marked with arrowheads with ‘i’ and ‘a,’ respectively. All pictures share the scale bar.
-
Figure 8—source data 1
The raw data of western blot without (Figure_8B-Original) and with (Figure_8B-Original-marked) marks of the positions of regions used for Figure 8B.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig8-data1-v2.zip
-
Figure 8—source data 2
The raw data of western blot without (Figure_8C-Original) and with (Figure_8C-Original-marked) marks of the positions of regions used for Figure 8C.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig8-data2-v2.zip
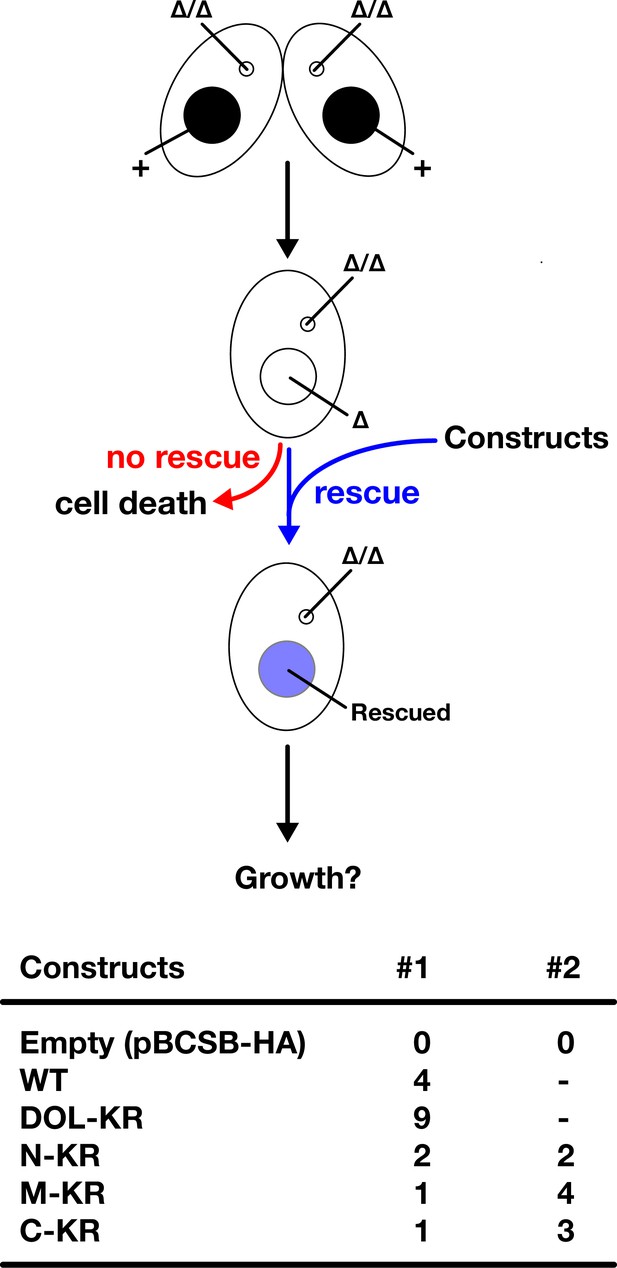
Genetic rescue of SPT6 KO.
(Top) Schematic representation of genetic rescue experiment of SPT6 KO. Two SPT6 KO homozygous heterokaryon strains were mated and different SPT6 expressing constructs (or corresponding pBCSB-HA vector without any of the SPT6 gene) were introduced into the macronucleus (MAC) of their progeny (SPT6 KO homozygous homokaryon = SPT6 germline (MAC + MIC) KO). The progeny survives only when the introduced construct can replace the essential functions of SPT6 in vegetative cells. (Bottom) Number of obtained SPT6 KO homozygous homokaryon strains that were genetically rescued by 10 µg DNA of the indicated constructs in two independent attempts (#1 and #2) are shown.
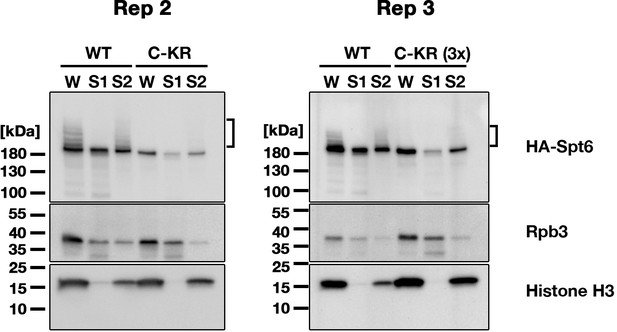
Replicated cell fractionation assay examining the effect of the SUMOylation-defective Spt6 mutation.
SPT6 germline KO cells rescued with HA-SPT6-WT (WT) or HA-SPT6-C-KR (C–KR) at 4.5 hpm were analyzed by cell fractionation as described in Figure 7B. In the experiment shown in right (Rep3), three times (3 x) volume of the fractions obtained from HA-SPT6-C-KR (C–KR) rescued cells were loaded. Spt6-HA, Rpb3, and histone H3 were detected by western blotting using anti-HA, anti-Rpb3, and anti-histone H3 antibodies, respectively. The position of SUMOylated Spt6-HA is marked with a bracket.
-
Figure 8—figure supplement 2—source data 1
The raw data of western blot without (Figure_8-figure supplement 2-Original) and with (Figure_8-figure supplement 2-Original-marked) marks of the positions of regions used for Figure 8—figure supplement 2.
- https://cdn.elifesciences.org/articles/95337/elife-95337-fig8-figsupp2-data1-v2.zip
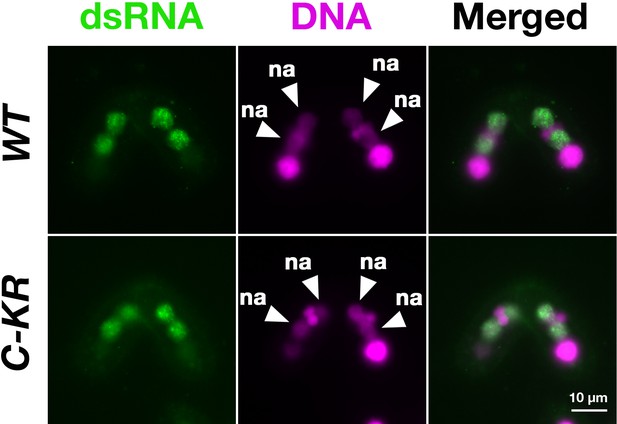
Localization of long non-coding RNA (lncRNA) transcripts in the SUMOylation-defective SPT6 mutant at late conjugation stage.
Accumulation of long dsRNAs in HA-SPT6-WT (WT) and HA-SPT6-C-KR (C–KR) rescued cells at 9 hpm was analyzed by immunofluorescence staining using the anti-long dsRNA antibody J2 (dsRNA, green) as described in Figure 6B. DNA was counterstained with DAPI (magenta). The new macronucleus (MACs) are marked with arrowheads with ‘na.’ All pictures share the scale bar.
Additional files
-
MDAR checklist
- https://cdn.elifesciences.org/articles/95337/elife-95337-mdarchecklist1-v2.docx
-
Supplementary file 1
List of DNA oligos and synthethic DNAs.
- https://cdn.elifesciences.org/articles/95337/elife-95337-supp1-v2.docx





