Adaptive thermal plasticity enhances sperm and egg performance in a model insect
Figures
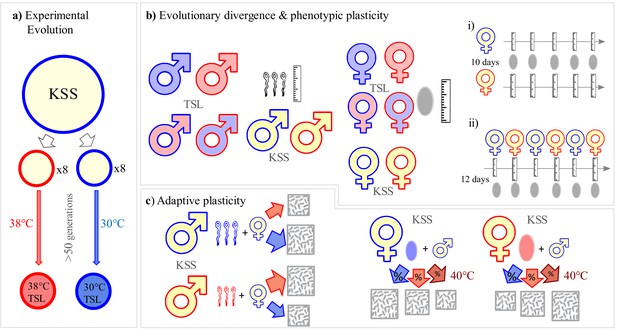
Experimental design.
Overview of experiments investigating thermal adaptation and plasticity in sperm and egg biology and its adaptive significance. (a) Set up of long-term thermal selection lines (TSLs) maintained at 30°C or 38°C for 50+ non-overlapping generations with eight replicate populations per regime. (b) Experimental design to investigate gamete size divergence in 30°C and 38°C TSLs at their long-term evolving environments, short-term plasticity in gamete size measured after a single-generation of novel temperature exposures, and short-term plasticity in individuals from the ancestral KSS (Krakow Super Strain) population. Symbol fill colour represents long-term background (30°C TSLs in blue, 38°C TSLs in red and KSS in yellow) while outline colour represents short-term exposure temperature (30°C blue, 38°C red). bi) and bii) illustrate experiments on temporal patterns of short-term plasticity in egg size (see main text). (c) Experiments investigating the adaptive significance of sperm and egg morphological plasticity in KSS adults. Gametic divergence was achieved by having adults produce gametes at either 30°C or 38°C, whose performance was then tested at 30°C, 38°C or 40°C.
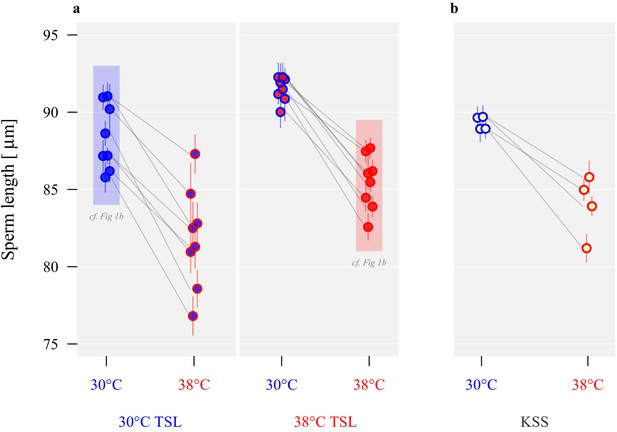
Long-term divergence and short-term plasticity in sperm size.
Symbol fill colour represents long-term background (30°C TSLs in blue, 38°C TSLs in red and KSS in yellow) while outline colour represents short-term exposure temperature (30°C blue, 38°C red; Figure 1b). (a) Sperm length in µm ± SEM of the experimentally evolved TSLs, measured either following production from pupation in their long-term thermal environment (highlighted in shaded boxes) or at the reciprocal temperature. (b) Sperm length of mature KSS males exposed to either 30°C or 38°C from pupation through eclosion.
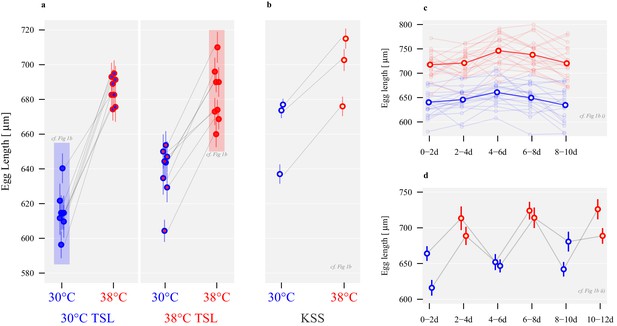
Long-term divergence and short-term plasticity in egg size.
Symbol fill colour represents long-term background (30°C TSLs in blue, 38°C TSLs in red and KSS in yellow) while outline colour represents short-term exposure temperature (30°C blue, 38°C red; Figure 1b). (a) Egg length in µm ± SEM of the experimentally evolved TSLs, measured either following production in their long-term thermal environment (highlighted in shaded boxes), or at the opposite temperature. (b) Egg length produced by mature KSS females exposed to either 30°C or 38°C for mating and oviposition, measured across three experimental replicates (grey lines). (c) Egg length of KSS females mated to a standard KSS male ovipositing individually either at 30°C or 38°C for 10 days in two-day intervals. The two bold-face lines indicate the averages across all females within a given treatment, while thin lines show average values for individual females. (d) Egg length of groups of 50 females, mated to standard KSS males, and ovipositing alternately at 30°C and 38°C for 12 days in two-day intervals. Egg width did not differ between thermal regimes, demonstrating that oviposition temperature increased egg volumes (Figure 3—figure supplements 1 and 2).
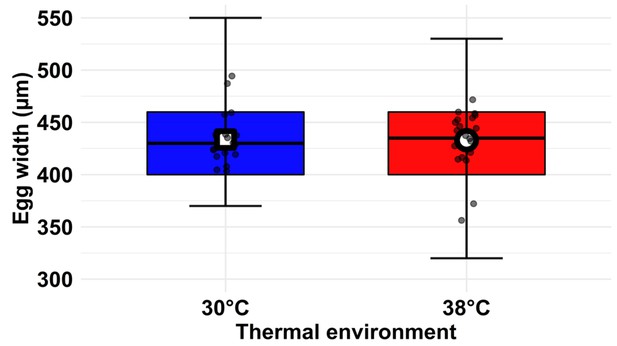
Comparing differences in egg width (µm) of KSS females ovipositing at either 30°C (blue) or 38°C (red).
Additional points in the centre within the boxes indicate the sample means (N = 20 females; five eggs per female; 30°C = square; 38°C = circle). Average egg widths of individual females are indicated as jittered points within the box plots. Box plots indicate the median (horizontal line in the centre) and interquartile ranges (IQR), with whiskers indicating data within 1.5 IQR.
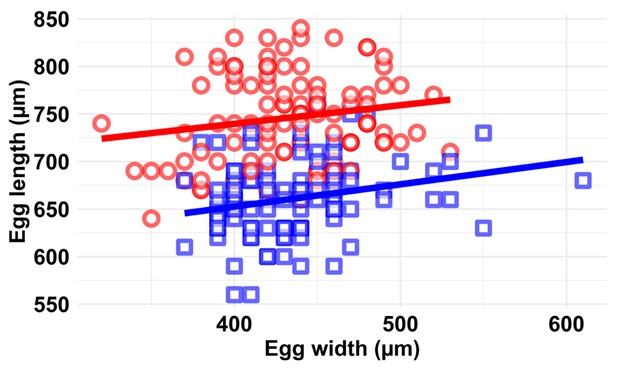
Scatter plot of egg morphology (egg length and width in µm) at the two ovipositing temperatures (blue squares 30°C; red circles 38°C).
There was no significant interaction between the two oviposition temperatures and egg morphology. We measured five eggs from each of 20 individual KSS females that had oviposited for 48 hr at either 30°C or at 38°C. Coloured lines represent linear regression predictions (blue = 30°C and red = 38°C). The volume of the egg (V) was calculated as 0.79 mm3 at 30°C and 1.00 mm3 at 38°C (i.e. a 21% change in egg volume), assuming egg shape is an oblate ellipsoid (formula; V = 4/3 π a2 b; a = length along the long axis [µm] and b = egg width [µm]).
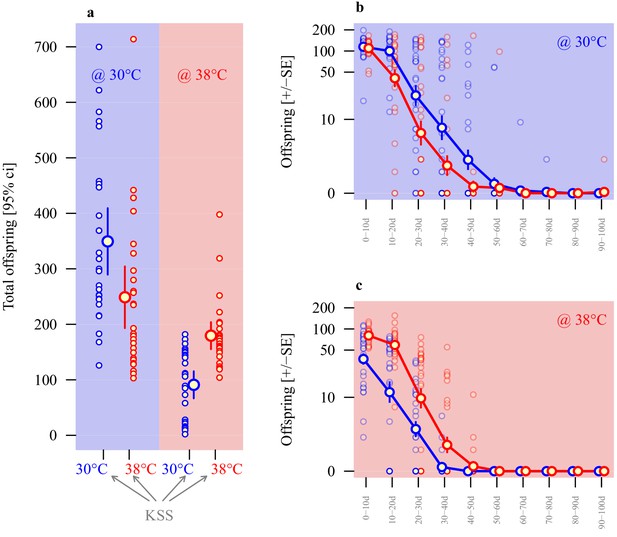
Adaptive thermal plasticity in sperm.
Reproductive output of ancestral KSS males following a 24 hr mating bout with a single female. Symbol outline colour represents male short-term exposure temperature (30°C blue, 38°C red) while background colour indicates fertilisation and offspring development temperature (30°C blue, 38°C red; see Figure 1c). (a) Total offspring produced over a 100d period across ten 10 day blocks from sperm produced in either 30°C or 38°C conditions when functioning in either 30°C or 38°C reproductive environments. Temporal patterns in 30°C and 38°C environments are illustrated in (b and c), respectively (note the log-scale of the Y-axis). Analyses of individual male reproductive performance and average sperm length across a range of thermal regimes indicate a longer-sperm advantage in this system (Figure 4—figure supplement 1).
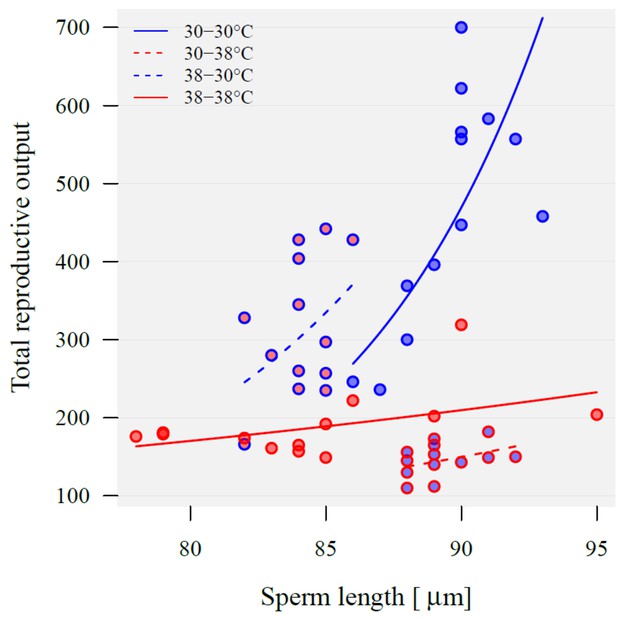
Sperm length (µm) and total reproductive output of KSS males used for the adaptive plasticity experiment (see Figure 4a).
Point fill colour represents the temperature at which males were reared from pupation while outline and regression line colour represent fertilisation and offspring development temperature (30°C blue, 38°C red). Regression lines show back-transformed predictions from a linear model on log-transformed reproductive output with sperm length, unique combination of rearing and fertilisation temperature (four level factor) and their interaction as predictors (sperm length: F1,44 = 68.4, p<0.001; temperature: F3,44 = 68.4, p<0.001; interaction: F3,44 = 4.4, p=0.009).
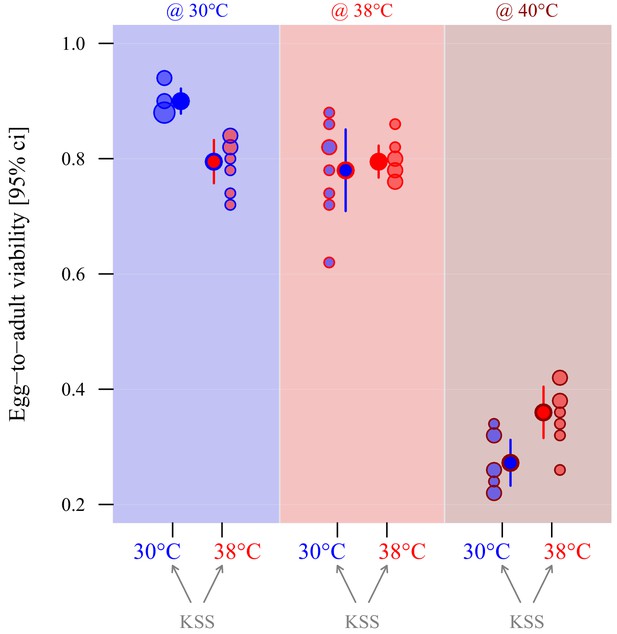
Adaptive thermal plasticity in eggs.
Symbol fill colour represents production and oviposition temperature (30°C blue, 38°C red) for ancestral KSS females while outline and background colour indicate egg incubation, offspring hatching and development temperature (30°C blue, 38°C red, 40°C dark red; see Figure 1c). Egg-to-adult viability was measured in a fodder medium with 0% yeast, with eight replicate groups of 50 eggs per treatment combination. Point surface area is proportional to the number of observations with identical outcomes.
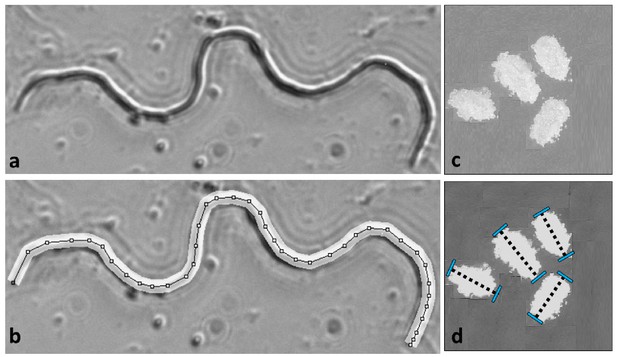
Gamete measurements were performed on mature sperm dissected from males and eggs following oviposition.
Sperm length was measured at 600X magnification in Image J by drawing segmented lines along the backbone of the cell (a and b). Egg lengths were measured along the long axis of each ovoid at 30X magnification (c and d). Further details in Godwin et al. (2017).
Tables
Sperm and egg length in relation to long-term selection and short-term exposure.
https://doi.org/10.7554/eLife.49452.004| Line | Temperature | Sperm length | N | Egg length | N |
|---|---|---|---|---|---|
| 30°C TSL | 30°C | 88.4 ± 5.2 | 40 | 615.4 ± 51.5 | 240 |
| 30°C TSL | 38°C | 81.9 ± 7.9 | 40 | 685.5 ± 42.2 | 240 |
| 38°C TSL | 30°C | 91.5 ± 4.9 | 40 | 638.4 ± 48.3 | 240 |
| 38°C TSL | 38°C | 85.5 ± 4.3 | 40 | 682.7 ± 42.7 | 240 |
| KSS | 30°C | 89.3 ± 4.1 | 26 | 662.6 ± 39.4 | 180 |
| KSS | 38°C | 84.3 ± 4.8 | 26 | 697.9 ± 48.4 | 180 |
-
Shown are mean, standard deviation and sample size (sperm: number of males; eggs: number of eggs) for sperm length and egg length measured in individuals from temperature selection lines (TSL) and the ancestral population (KSS), exposed to different temperatures from pupation onward (see main text and Figures 1–3).
Adaptive thermal plasticity in sperm and eggs improves gamete performance.
https://doi.org/10.7554/eLife.49452.010| Sperm | Eggs | |||||
|---|---|---|---|---|---|---|
| Line | Gamete production | Gamete performance | Offspring | N | Viability | N |
| KSS | 30°C | 30°C | 349.5 ± 151.5 | 27 | 90 ± 2% | 8 |
| KSS | 38°C | 30°C | 248.8 ± 140.9 | 27 | 80 ± 5% | 8 |
| KSS | 30°C | 38°C | 91.0 ± 61.8 | 27 | 78 ± 8% | 8 |
| KSS | 38°C | 38°C | 179.6 ± 61.8 | 27 | 80 ± 3% | 8 |
| KSS | 30°C | 40°C | 27 ± 5% | 8 | ||
| KSS | 38°C | 40°C | 36 ± 5% | 8 | ||
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Tribolium castaneum) | Krakow Super Strain ancestral stock and Thermal Selection Lines at 30°C and 38°C | KSS: Dickinson, 2018 Sales, 2019 TSL: this paper and Dickinson, 2018 Sales, 2019 | KSS & TSL30 or TSL38 | Live beetles |
| Biological sample (Tribolium castaneum) | Spermatozoa and ova | This paper | Sperm and eggs | Sperm recovered from sacrificed live males, eggs recovered from oviposition food medium |
| Software, algorithm | R Studio | R Studio (RStudio Team, 2016) in R (R Development Core Team, 2017) (version 3.4.1) | R Studio version 1.1.463 and R version 3.4.1 |
Additional files
-
Supplementary file 1
Table model summary of the best glmmTMB model (lowest AIC value; see Supplementary file 1 Table 2) for reproductive output of males reared at 30°C or 38°C (Treatment) with offspring developing at 30°C or 38°C (Regime).
- https://doi.org/10.7554/eLife.49452.013
-
Supplementary file 2
Table overview of models for adaptive significance of sperm plasticity.
Models were run using glmmTMB and were sorted along ascending AIC values. All conditional models additionally included random intercepts for ten-day blocks as well as random intercepts and random slopes for individual males.
- https://doi.org/10.7554/eLife.49452.014
-
Transparent reporting form
- https://doi.org/10.7554/eLife.49452.015





