Plant-associated CO2 mediates long-distance host location and foraging behaviour of a root herbivore
Figures
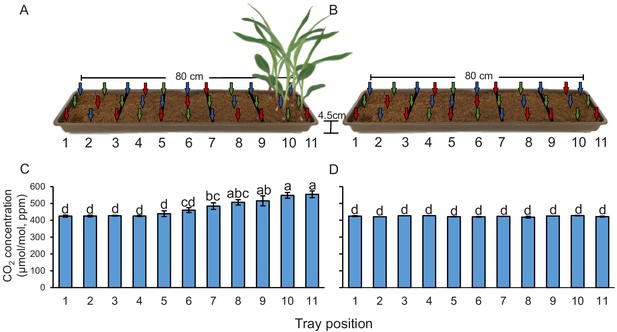
Plants create CO2 gradients in the soil.
(A, B) CO2 levels were determined in soil-filled trays at different distances from young maize seedlings (A) before and (B) after removing the seedlings from the system. Arrows indicate air sampling points. Different colours indicate sampling positions within three individual trays that were assayed (n = 3). Red arrows indicate samplings points in tray 1, green arrows indicate samplings points in tray 2, and blue arrows indicate samplings points in tray 3. (C, D) Mean (± SEM) CO2 levels at different distances from the plant (C) before and (D) after removing the seedlings from the system. Different letters indicate significant differences in CO2 levels in each tray position (p < 0.05 by two-way ANOVA with Holm’s multiple-comparisons test). For details regarding the statistical results, refer to Supplementary file 1. Raw data are available in Figure 1—source data 1.
-
Figure 1—source data 1
Raw data for Figure 1.
- https://cdn.elifesciences.org/articles/65575/elife-65575-fig1-data1-v1.xlsx
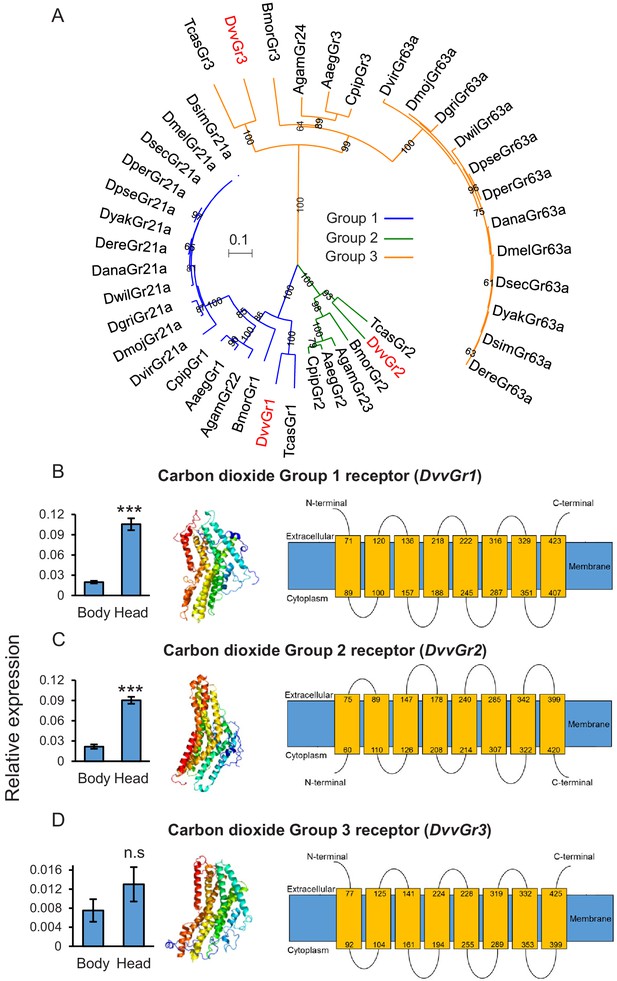
The western corn rootworm (WCR) genome contains three putative carbon dioxide (CO2) receptors.
(A) Phylogenetic relationships between putative CO2 receptors based on protein sequences of different insects. Dmel: Drosophila melanogaster; Dsim: Drosophila simulans; Dsec: Drosophila sechellia; Dyak: Drosophila yakuba; Dere: Drosophila erecta; Dana: Drosophila ananassae; Dper: Drosophila persimilis; Dpse: Drosophila pseudoobscura; Dwil: Drosophila willistoni; Dgri: Drosophila grimshawi; Dmoj: Drosophila mojavensis; Dvir: Drosophila virilis; Agam: Aedes gambiae; Aaeg: Aedes aegypti; Cqui: Culex quinquefasciatus; Bmor: Bombyx mori; Tcas: Tribolium castaneum; Dvv: Diabrotica virgifera virgifera (WCR). Evolutionary relationships were inferred using the neighbor-joining method. The optimal tree with the sum of branch length = 4.44068889 is shown. The percentage of replicate trees in which the associated taxa clustered together in the bootstrap test (100 replicates) are shown next to the branches. The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances were computed using the Poisson correction method and are in the units of the number of amino acid substitutions per site. A total of 242 amino acid positions were included in the final data set. (B–D) Mean (± SEM) relative gene expression levels of group 1 (DvvGr1) (B), group 2 (DvvGr2) (C), and group 3 (DvvGr3) (D) CO2 receptors in the bodies (thorax and abdomen) or heads of second instar WCR larvae (n = 10). Asterisks indicate statistically significant differences between tissue types within genes (***p < 0.001 by Student’s t test; n.s.: not significant). For details regarding the statistical results, refer to Supplementary file 1. Raw data are available in Figure 2—source data 1. (B–D) Predicted protein tertiary structure (left) and transmembrane protein topology (right) of (B) DvvGr1; (C) DvvGr2, and (D) DvvGr3 according to the Phyre2 algorithm.
-
Figure 2—source data 1
Raw data for Figure 2.
- https://cdn.elifesciences.org/articles/65575/elife-65575-fig2-data1-v1.xlsx
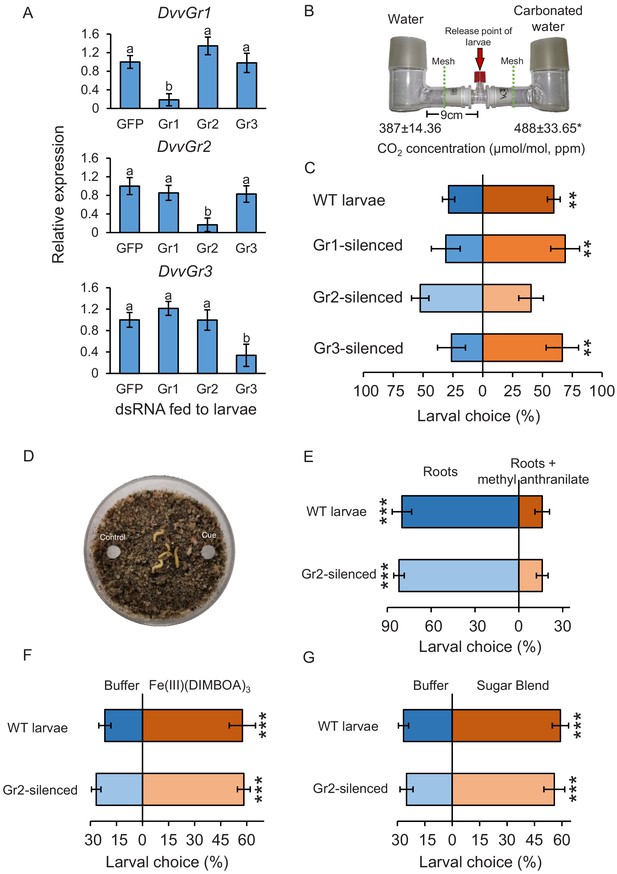
The carbon dioxide group 2 receptor (DvvGr2) is specifically required for the attraction of western corn rootworm (WCR) towards CO2.
(A) Mean (± SEM) relative gene expression levels of group 1 (DvvGr1), group 2 (DvvGr2), and group 3 (DvvGr3) CO2 receptors after WCR larvae were fed with dsRNA-expressing bacteria targeting green fluorescent protein (GFP, herein referred to as WT), DvvGr1, DvvGr2, or DvvGr3 genes (n = 11–13). Different letters indicate significant differences of gene expression levels (p < 0.05 by one-way ANOVA with Holm’s multiple-comparisons test). (B) Mean (± SEM) CO2 levels in each L-shaped pot of the two-arm belowground olfactometers used to test the attractive and repellent effects of CO2 to WCR larvae (n = 4–8). Asterisk indicates significant differences in CO2 levels (*p < 0.05 by Student’s t test). For detailed data on CO2 levels, refer to Figure 3—figure supplement 1. (C) Mean (± SEM) proportion of WCR larvae observed in the olfactometer arms with higher CO2 levels (carbonated water side) or in control arms (distilled water side). Larvae were considered to have made a choice when they were found at a distance of 1 cm or less from the wire mesh (indicated by dashed green lines). Seven olfactometers with six larvae each were assayed (n = 7). (D) Petri plates used to test insect responses to methyl anthranilate, to Fe(III)(DIMBOA)3, and to sugars. (E) Mean (± SEM) proportion of WCR larvae observed on roots or on roots placed next to filter paper discs impregnated with methyl anthranilate, (F) on filter paper discs impregnated with buffer or with Fe(III)(DIMBOA)3, and (G) on filter paper discs impregnated with buffer or with a blend of glucose, fructose, and sucrose. For (E), 10 choice arenas with five larvae each were assayed (n = 10). Larvae were considered to have made a choice when they made physical contact with the roots or the filter paper discs. For (F, G), 20 choice arenas with six larvae each were assayed (n = 20). Asterisks indicate statistically significant differences in larval choices between treatments (***p < 0.001 by generalized linear model followed by False discovery rate (FDR)-corrected post hoc tests). Note that the number of replicates across experiments varied depending on the availability of insects. For details regarding the statistical results, refer to Supplementary file 1. Raw data are available in Figure 3—source data 1.
-
Figure 3—source data 1
Raw data for Figure 3.
- https://cdn.elifesciences.org/articles/65575/elife-65575-fig3-data1-v1.xlsx
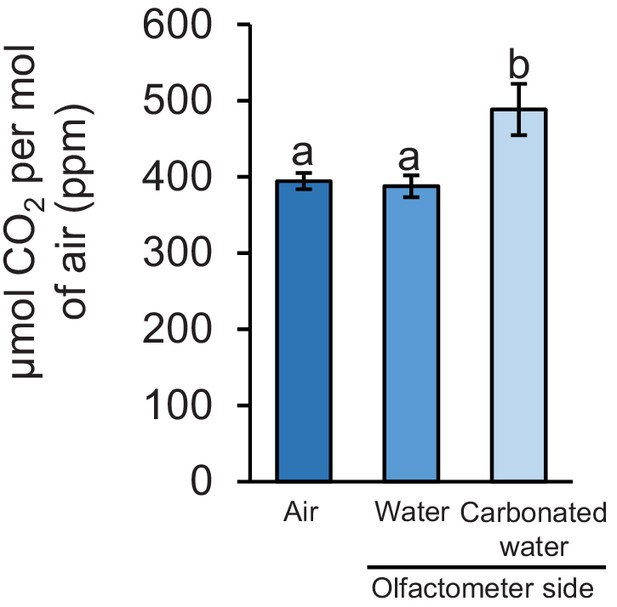
Carbon dioxide levels at different sampling points.
Mean (± SEM) CO2 levels in each olfactometer side and in the air (n = 3–4). Different letters indicate significant differences in CO2 levels (p < 0.05 by one-way ANOVA with Holm’s multiple-comparisons test).
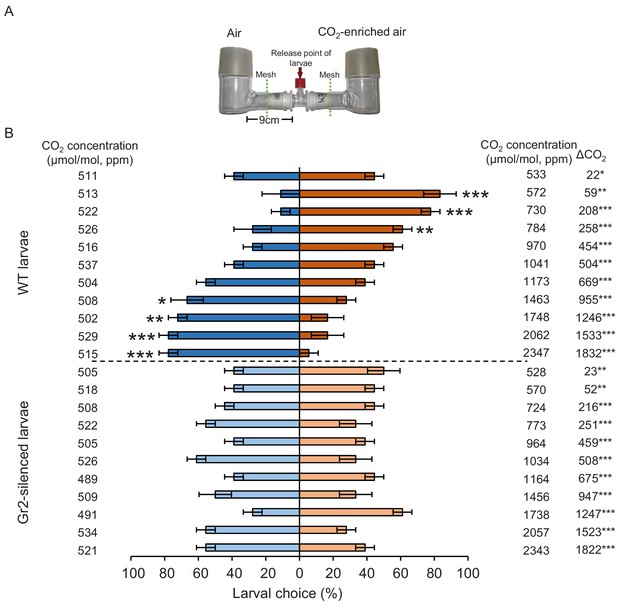
DvvGr2 is required for dose-dependent western corn rootworm (WCR) responses to CO2.
(A) Two-arm olfactometer used to test the attractive and repellent effects of CO2 on WCR larvae. (B) Mean ( ± SEM) proportion of WCR larvae observed in each arm of the olfactometers. Larvae were considered to have made a choice when they were found at a distance of 1 cm or less from the wire mesh, indicated by dashed green lines. Three olfactometers with six larvae each were assayed (n = 3). Asterisks indicate statistically significant differences between larval choices (*p < 0.05; **p < 0.01; ***p < 0.001 by generalized linear model [GLM] followed by FDR-corrected post hoc tests). Mean CO2 concentrations in each olfactometer side and the difference between them (∆CO2) are indicated. Asterisks indicate significant differences in the CO2 levels of each olfactometer arm (*p < 0.05; **p < 0.01; *** p < 0.001 by GLM followed by FDR-corrected post hoc tests). For detailed data on CO2 levels, refer to Figure 4—figure supplement 1. For details regarding the statistical results, refer to Supplementary file 1. Raw data are available in Figure 4—source data 1.
-
Figure 4—source data 1
Raw data for Figure 4.
- https://cdn.elifesciences.org/articles/65575/elife-65575-fig4-data1-v1.xlsx
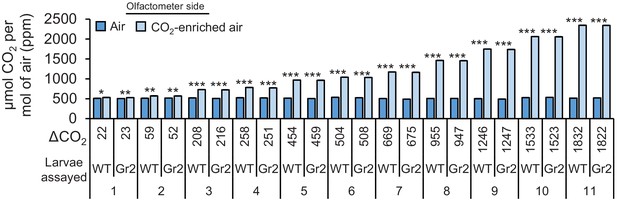
Carbon dioxide levels at different sampling points.
Mean (± SEM) CO2 levels in olfactometer sides that were treated with air or with CO2-enriched synthetic air (n = 3). Asterisks indicate significant differences in CO2 levels (*p < 0.05; **p < 0.01; ***p < 0.001 by generalized linear model (GLM) followed by FDR-corrected post hoc tests). For p-values of GLMs, refer to Supplementary file 1. Gr2: DvvGr2-silenced larvae.
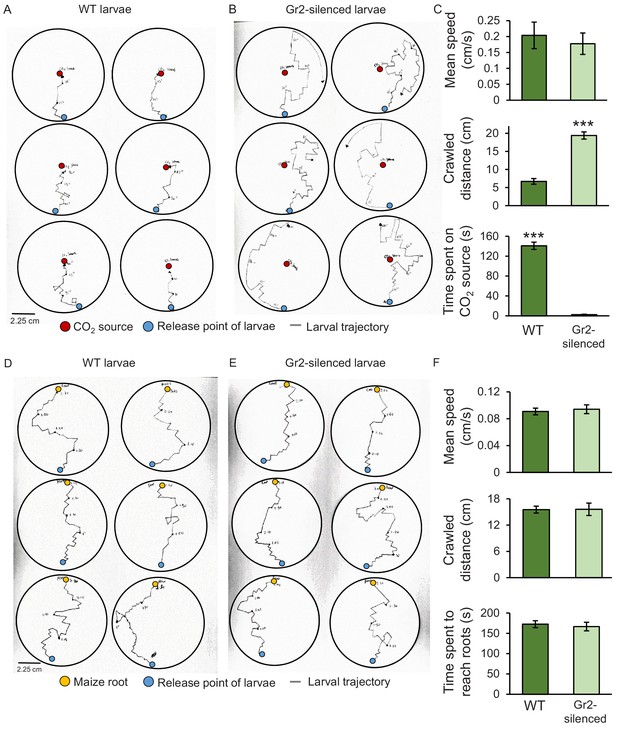
Silencing the carbon dioxide group 2 receptor (DvvGr2) impairs western corn rootworm (WCR) responses to CO2 without affecting larval motility or search behaviour.
(A, B) Trajectories of individual wild type (WT) (A) and DvvGr2-silenced (B) WCR larvae in Petri plates with a CO2 source. The blue circles represent larval release points. The red circles represent CO2 sources consisting of a fine needle that releases CO2 at 581 ppm, resulting in CO2 concentrations 60 ppm above ambient CO2 levels at the release point. (C) Mean (± SEM) speed and distance covered during the movement phase, and time spent at the CO2 source during the first 3 min of the experiment. (D, E) Trajectories followed by WT (D) and by DvvGr2-silenced (E) WCR larvae on Petri plates containing maize seedling roots. The blue circles represent larval release points. The yellow circles represent maize seedling roots. (F) Mean (± SEM) speed and distance covered during the movement phase, and time necessary to reach the maize root during the first 3 min of the experiment. For both experiments, six Petri plates with one larva each were assayed (n = 6). Asterisks indicate statistically significant differences between mobility parameters of WT and DvvGr2-silenced larvae (***p < 0.001 by Student’s t test). For details regarding the statistical results, refer to Supplementary file 1. Raw data are available in Figure 5—source data 1.
-
Figure 5—source data 1
Raw data for Figure 5.
- https://cdn.elifesciences.org/articles/65575/elife-65575-fig5-data1-v1.xlsx
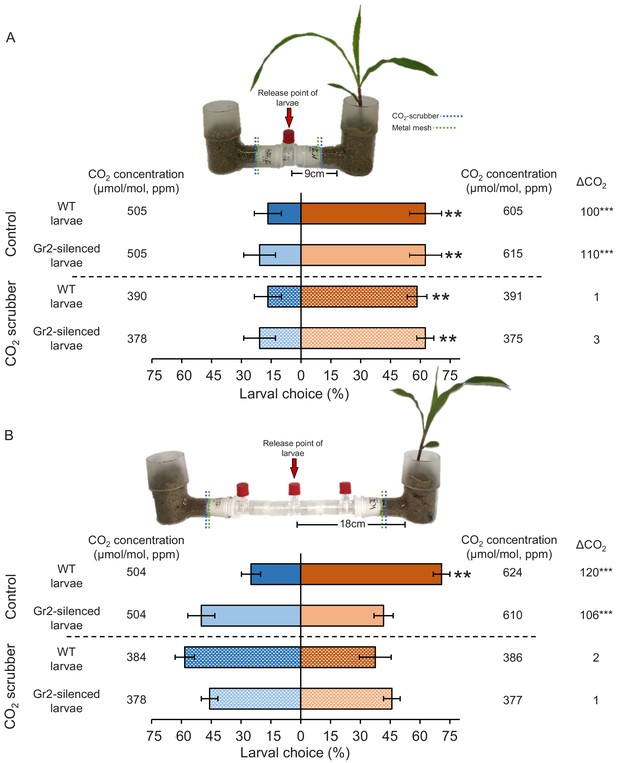
Plant-associated CO2 mediates host location by western corn rootworm (WCR) larvae in a distance-specific manner.
(A, B) Mean (± SEM) proportion (%) of WCR larvae observed on each side of the olfactometers. Larva were considered to have made a choice when they were found at a distance of 1 cm or less from the wire mesh, indicated by dashed green lines. Control olfactometers allowed for plant-associated CO2 to diffuse into the central glass tubes, while CO2 scrubber olfactometers were outfitted with soda lime to suppress CO2 diffusion while allowing for the diffusion of other volatiles. Mean CO2 concentrations in each olfactometer side and the difference between them (∆CO2) are given. Asterisks indicate significant differences in the CO2 levels of each olfactometer arm (***p < 0.001 by generalized linear model [GLM] followed by FDR-corrected post hoc tests). For detailed data on CO2 levels and other volatiles, refer to Figure 6—figure supplements 1 and 2. Four olfactometers with six larvae each were assayed using wild type (WT) or DvvGr2-silenced larvae (n = 4). Asterisks indicate statistically significant differences between treatments (**p < 0.01 by GLM followed by FDR-corrected post hoc tests). For details regarding the statistical results, refer to Supplementary file 1. Raw data are available in Figure 6—source data 1.
-
Figure 6—source data 1
Raw data for Figure 6.
- https://cdn.elifesciences.org/articles/65575/elife-65575-fig6-data1-v1.xlsx
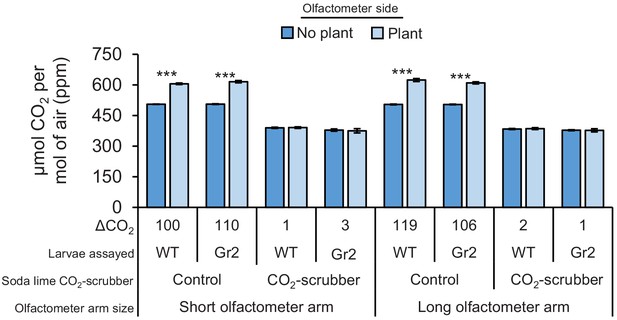
Carbon dioxide levels at different sampling points.
Mean (± SEM) CO2 levels in olfactometer sides with or without plants (n = 4). Asterisks indicate significant differences in CO2 levels (***p < 0.001 by generalized linear model [GLM] followed by FDR-corrected post hoc tests). For p-values of GLMs, refer to Supplementary file 1. Gr2: DvvGr2-silenced larvae.
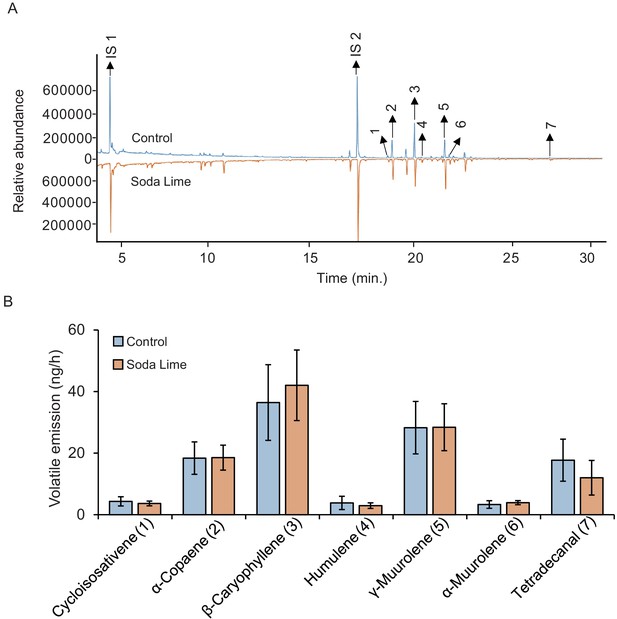
Soda lime does not influence the diffusion of plant volatiles other than CO2 into olfactometer arms.
(A) Representative chromatograms of volatiles from maize roots in the absence (control, blue chromatogram) or the presence of a soda lime layer that removes CO2 (soda lime, orange chromatogram). (B) Mean (± SEM) levels of the most abundant plant volatiles are shown (n = 5).
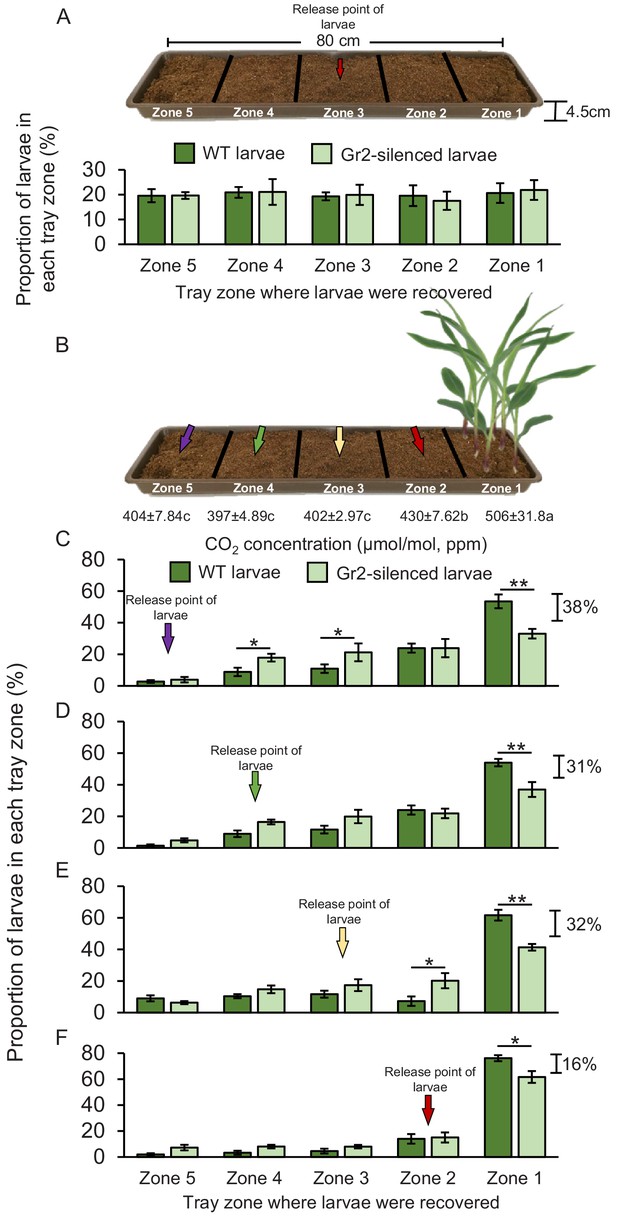
Root-associated CO2 is used by western corn rootworm (WCR) larvae for host location in a distance-specific manner.
(A) Mean (± SEM) proportion of wild type (WT) (dark green) or DvvGr2-silenced (light green) WCR larvae observed in the different tray zones 8 hr after releasing the larvae in the centre of soil-filled trays without plants. Three trays per larval type with 20 larvae each were assayed (n = 3). (B) Schematic representation (photomontage) of experimental set-up used to test distance-specific host location abilities of WCR larvae depicting mean (± SEM) CO2 levels detected in the soil gas phase of each tray zone (n = 3–4). Different letters indicate significant differences in CO2 levels (p < 0.05 by one-way ANOVA with Holm’s multiple-comparisons test). For detailed data on CO2 levels, refer to Figure 7—figure supplement 1. (C–F) Mean (± SEM) proportion of WT (dark green) or DvvGr2-silenced (light green) WCR larvae observed in the different tray zones 8 hr after releasing the larvae at distances of 64 cm (C), 48 cm (D), 32 cm (E), or 16 cm (F) from the plants. Six trays per larval type and distance combination with 20 larvae each were assayed (n = 6). Asterisks indicate statistically significant differences in the proportion of WT and DvvGr2-silenced larvae found in each tray zone (*p < 0.05; **p < 0.01 by generalized linear model followed by FDR-corrected post hoc tests). For details regarding the statistical results, refer to Supplementary file 1. Raw data are available in Figure 7—source data 1.
-
Figure 7—source data 1
Raw data for Figure 7.
- https://cdn.elifesciences.org/articles/65575/elife-65575-fig7-data1-v1.xlsx
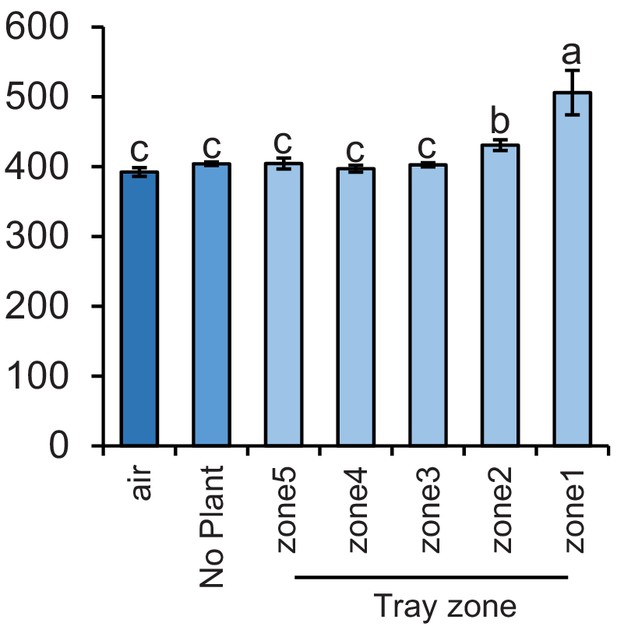
Carbon dioxide levels at different sampling points.
Mean (± SEM) CO2 levels detected in the soil gas phase of each tray zone or in the air (n = 3–4). Different letters indicate significant differences in CO2 levels (p < 0.05 by one-way ANOVA with Holm’s multiple-comparisons test).
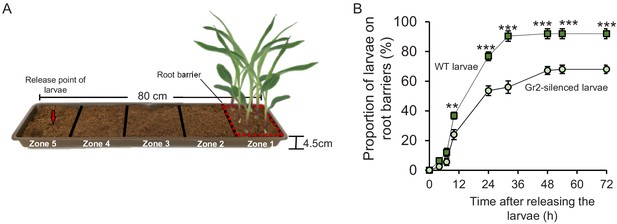
Root-associated CO2 is required for host location by western corn rootworm (WCR) larvae at long distances.
(A) Schematic representation (photomontage) of experimental set-up used. (B) Mean (± SEM) proportion of WCR larvae retrieved at close vicinity of root systems (i.e., on root barriers). Twenty-five larvae were released at 64 cm from the plants and five arenas were assayed (n = 5). Soil surrounding root barriers (indicated by dashed red lines) were carefully inspected, and the observed larvae were collected and counted regularly. Asterisks indicate statistically significant differences in the proportion of larvae retrieved at each time point (**p < 0.01; ***p < 0.001 by two-way repeated measures ANOVA with Holm’s multiple-comparisons test). For p-values of the different factors and their interactions, refer to Supplementary file 1.
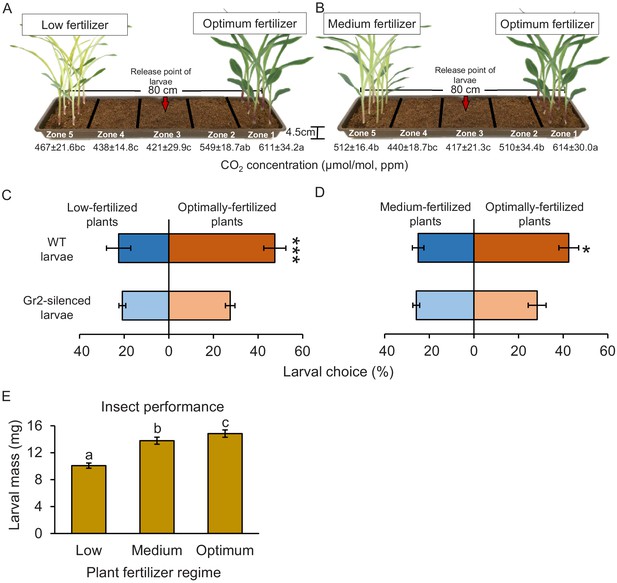
CO2 perception increases the location of more suitable host plants.
(A, B) Schematic representation (photomontage) of soil-filled trays used to evaluate location of differentially fertilized plants by western corn rootworm (WCR) larvae depicting mean (± SEM) CO2 levels detected in the soil gas phase of each tray zone (n = 10). Different letters indicate statistically significant differences in CO2 levels (p < 0.05 by one-way ANOVA with Holm’s multiple-comparisons test). For details regarding CO2 levels, refer to Figure 8—figure supplement 1. Mean (± SEM) proportion of WCR larvae recovered close to plants that received low (zone 5) or optimum (zone 1) fertilizer doses (C), or that were recovered close to plants that received medium (zone 5) or optimum (zone 1) fertilizer doses (D) 8 hr after releasing the larvae. Six trays with 20 larvae per tray were assayed (n = 6). Different letters indicate statistically significant differences in larval preferences (*p < 0.05; ***p < 0.001 by generalized linear model followed by FDR-corrected post hoc tests). (E) Mean (± SEM) weight of WCR larvae after 7 days feeding on plants fertilized with low, medium, or optimum fertilizer doses. Twenty solo cups with 4–7 larvae each were assayed (n = 20). Different letters indicate statistically significant differences in larval mass (p < 0.05 by one-way ANOVA followed by Holm’s multiple-comparisons tests). For details regarding the statistical results, refer to Supplementary file 1. Raw data are available in Figure 8—source data 1.
-
Figure 8—source data 1
Raw data for Figure 8.
- https://cdn.elifesciences.org/articles/65575/elife-65575-fig8-data1-v1.xlsx
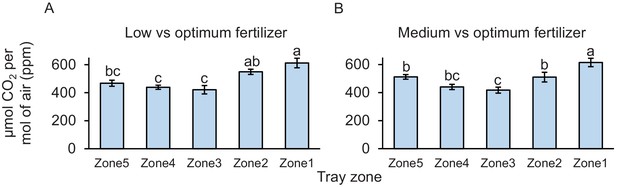
Carbon dioxide levels at different sampling points.
(A, B) Mean (± SEM) CO2 levels detected in the soil gas phase of each tray zone (n = 10). Different letters indicate statistically significant differences in CO2 levels (p < 0.05 by one-way ANOVA with Holm’s multiple-comparisons test).
Additional files
-
Supplementary file 1
Description and results of the statistical analysis methods used to analyse the data of this study.
- https://cdn.elifesciences.org/articles/65575/elife-65575-supp1-v1.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/65575/elife-65575-transrepform-v1.docx





