Bacillus velezensis HBXN2020 alleviates Salmonella Typhimurium infection in mice by improving intestinal barrier integrity and reducing inflammation
eLife Assessment
In this useful study, Wang and colleagues investigate the potential probiotic effects of Bacillus velezensis in a murine model. They provide convincing evidence that B. velezensis limits the growth of Salmonella typhimurium in lab culture and in mice, together with beneficial effects on the microbiota. The overall presentation of the manuscript has improved and the work will be of interest to infectious disease researchers.
https://doi.org/10.7554/eLife.93423.4.sa0Useful: Findings that have focused importance and scope
- Landmark
- Fundamental
- Important
- Valuable
- Useful
Convincing: Appropriate and validated methodology in line with current state-of-the-art
- Exceptional
- Compelling
- Convincing
- Solid
- Incomplete
- Inadequate
During the peer-review process the editor and reviewers write an eLife Assessment that summarises the significance of the findings reported in the article (on a scale ranging from landmark to useful) and the strength of the evidence (on a scale ranging from exceptional to inadequate). Learn more about eLife Assessments
Abstract
Bacillus velezensis is a species of Bacillus that has been widely investigated because of its broad-spectrum antimicrobial activity. However, most studies on B. velezensis have focused on the biocontrol of plant diseases, with few reports on antagonizing Salmonella Typhimurium infections. In this investigation, it was discovered that B. velezensis HBXN2020, which was isolated from healthy black pigs, possessed strong anti-stress and broad-spectrum antibacterial activity. Importantly, B. velezensis HBXN2020 did not cause any adverse side effects in mice when administered at various doses (1×107, 1×108, and 1×109 CFU) for 14 days. Supplementing B. velezensis HBXN2020 spores, either as a curative or preventive measure, dramatically reduced the levels of S. Typhimurium ATCC14028 in the mice’s feces, ileum, cecum, and colon, as well as the disease activity index (DAI), in a model of infection caused by this pathogen in mice. Additionally, supplementing B. velezensis HBXN2020 spores significantly regulated cytokine levels (Tnfa, Il1b, Il6, and Il10) and maintained the expression of tight junction proteins and mucin protein. Most importantly, adding B. velezensis HBXN2020 spores to the colonic microbiota improved its stability and increased the amount of beneficial bacteria (Lactobacillus and Akkermansia). All together, B. velezensis HBXN2020 can improve intestinal microbiota stability and barrier integrity and reduce inflammation to help treat infection by S. Typhimurium.
Introduction
Salmonella Typhimurium (S. Typhimurium) is a major foodborne zoonotic pathogen that can cause diarrhea and colitis in humans and animals (Fàbrega and Vila, 2013; Yang et al., 2021). According to reports, Salmonella infections are common in both industrialized and developing nations, posing a serious risk to public health and resulting in significant financial losses (Katiyo et al., 2019; Mohan et al., 2019). At present, antibiotics remain one of the most effective treatment strategies for Salmonella infections. However, numerous studies have reported that the prolonged use or misuse of antibiotics can lead to environmental pollution, an increase in multi-drug-resistant bacteria, as well as gastrointestinal microbiota dysbiosis (Ferri et al., 2017; Paulson and Zaoutis, 2015; Reyman et al., 2022). In order to tackle Salmonella infections, a hunt for novel antimicrobial drugs is now underway.
More research has recently demonstrated the critical role gut microbiota plays in reducing intestinal inflammation (Cristofori et al., 2021) and mending the intestinal barrier (Ohland and Macnaughton, 2010; Wang et al., 2020). The use of probiotics is a popular approach to modulating intestinal microbiota nowadays. Among all probiotics, Bacillus is one of the most popular probiotic species because of its ability to form endospores that survive gastric transit, storage, and delivery conditions (Gu et al., 2015). Furthermore, Bacillus species can generate extracellular enzymes and antimicrobial metabolites that inhibit enteric pathogens, which lowers the risk of infection and enhances nutrient utilization (Abdhul et al., 2015; Ouattara et al., 2017).
Recent studies have found that Bacillus velezensis (B. velezensis) has the enormous potential to produce a variety of metabolites with broad-spectrum antibacterial activity (Ye et al., 2018). Meanwhile, previous studies have reported that B. velezensis exhibits varying degrees of beneficial effects on plants, livestock, poultry and fish. For instance, the combined use of B. velezensis SQR9 mutant and FZB42 improved root colonization and the production of secondary metabolites that are beneficial to cucumber (Shao et al., 2022). B. velezensis isolated from yaks was shown to enhance growth performance and ameliorate blood parameters related to inflammation and immunity in mice (Li et al., 2019). The dietary B. velezensis supplementation can regulate the innate immune response in the intestine of crucian carp and reduce the degree of intestinal inflammation damage induced by Aeromonas veronii (Zhang et al., 2019). However, studies on B. velezensis in the prevention of S. Typhimurium infection are rarely reported; instead, the majority of investigations on B. velezensis focused on the biocontrol of fungal infections in plants. In this study, a strain of B. velezensis HBXN2020 with broad-spectrum antibacterial activity against Salmonella was selected from a vast number of Bacillus strains. Next, we evaluated its safety in mice by supplementing B. velezensis HBXN2020 and then explored the protective effects in STm-infected mice. Notably, B. velezensis HBXN2020 alleviated colon tissue damage caused by STm infection, as indicated by markers such as STm loads, Tnfa and Tjp1 levels. Moreover, supplementing B. velezensis HBXN2020 also increased the abundance of beneficial bacteria, specifically Lactobacillus and Akkermansia, within the colon microbiota. As a result, this research supports the creation of probiotic-based microbial products as a substitute method of preventing Salmonella infections.
Results
Isolation of Bacillus
Four strains of Bacillus with antibacterial activity were screened from a large amount of presumptive Bacillus isolates, based on spot-on-plate tests analysis (Figure 1—figure supplement 1), and the four strains clearly displayed the properties of bacteria in the genus Bacillus such as colonies were crateriform, Gram-positive, rod shape, and endospore-forming ability. Next, a comparative analysis of the antibacterial spectrum of four strains of Bacillus found that one strain exhibited excellent antibacterial activity against common pathogens (Supplementary file 1). Therefore, this strain was selected in this study for further research, which was designated as the HBXN2020.
Growth curve and in vitro resistance against environmental assaults
The growth of HBXN2020 was assessed in flat-bottomed 100-well microtiter plates by measuring the values of OD600 every hour using an automatic growth curve analyzer. After measurement, we found that HBXN2020 entered the logarithmic growth phase after 2 hr, and reached a plateau after 10 hr of culture (Figure 1A, Figure 1—source data 1).
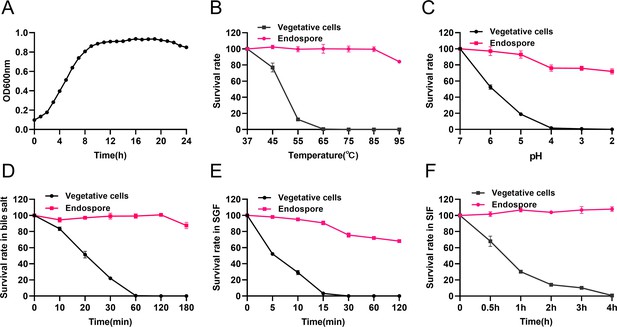
Growth curve of B. velezensis HBXN2020 and its in vitro resistance against environmental assaults.
(A) Growth curves of B. velezensis HBXN2020 cultured in Luria-Bertani (LB) medium at 37°C, detection of OD600 values at 1 hr intervals in microplate reader. (B) Survival rate of endospore and vegetative cells of B. velezensis HBXN2020 after 30 min at different temperatures (37°C, 45°C, 55°C, 65°C, 75°C, 85°C, or 95°C). Equal amounts of endospore and vegetative cells of HBXN2020 were exposed to the following: (C) acid solution (pH 2–7), (D) 0.3% bile salts, (E) simulated gastric fluid (SGF) (pH 1.2) supplemented with pepsin, and (F) simulated intestinal fluid (SIF) (pH 6.8) containing trypsin at 37°C. At predetermined time points, 100 μL was taken from each sample, and 10-fold serially diluted with sterile PBS (pH 7.2), and then spread on LB agar plates and cultured at 37°C for 12 hr before bacterial counting. Each group was repeated three times (n=3).
-
Figure 1—source data 1
Raw data values for Figure 1A–F.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig1-data1-v1.xlsx
Next, we evaluated the effect of physical, chemical, and biological sterilization conditions such as high temperature, strong acidity, and enzyme preparation on the survival of HBXN2020. The survival rate of HBXN2020 showed a decreasing trend with increasing temperature (Figure 1B, Figure 1—source data 1), but this decrease was not obvious. In a strong acid environment (pH 2.0), HBXN2020 maintained a high survival rate (Figure 1C, Figure 1—source data 1), suggesting that HBXN2020 spores can survive under extreme conditions. Based on these results, we hypothesized that HBXN2020 spores might also exhibit improved survival in gastrointestinal tract environments. Therefore, we further evaluated the survival rate of HBXN2020 in bile salt (0.3%, vol/vol), simulated gastric fluid (pepsin 1 mg/mL, pH 1.2), and simulated intestinal fluid (trypsin 1 mg/mL, pH 6.8). As shown in Figure 1D–F (Figure 1D–F, Figure 1—source data 1), HBXN2020 spores demonstrated significantly improved tolerance to these simulated gastrointestinal tract environments.
Antibiotic susceptibility of HBXN2020 and bacteriostasis effect in vitro
In order to assess HBXN2020’s drug resistance, 19 antibiotics that are frequently used in clinical settings were chosen in this study. The results indicated that only polymyxin B had a relatively small inhibition zone diameter (less than 15 mm). Ampicillin, meropenem, minocycline, ofloxacin, and trimethoprim had the strongest inhibition on HBXN2020, with an inhibition zone diameter exceeding 30 mm (Figure 2A, Figure 2—source data 1). This indicated that HBXN2020 was extremely sensitive to β-lactams, tetracyclines, and quinolone drugs. We next utilized an in vitro antibacterial assay to evaluate the antibacterial activity of HBXN2020. The results showed that HBXN2020 had a similar inhibitory effect on standard and wild strains of E. coli, Salmonella, S. aureus, and Clostridium perfringens (C. perfringens), as well as wild strains of Streptococcus suis (S. suis), Pasteurella multocida (P. multocida), and Actinobacillus pleuropneumoniae (A. pleuropneumoniae). Except for the wild strains of S. suis and A. pleuropneumoniae, the diameter of the inhibition zone of other strains was above 15 mm (Figure 2B, Figure 2—figure supplement 1, and Figure 2—source data 1), while the size of the inhibition zone of S. suis and A. pleuropneumoniae was also more than 12 mm.
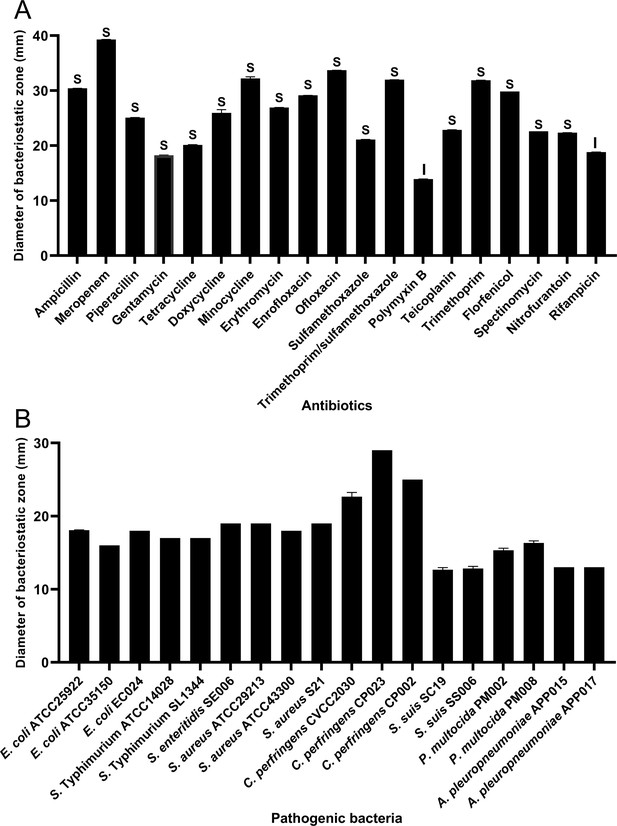
Antibiotic susceptibility of B. velezensis HBXN2020 and bacteriostasis assay in vitro.
(A) The diameter of the antibacterial zone indicates the extent of sensitivity to antibiotics. (B) The diameter of the antibacterial zone indicates the extent of inhibition against pathogenic bacteria. The diameter of the antibacterial zone was measured with vernier caliper. Each group was repeated three times (n=3). R, resistant; I, moderately sensitive; S, sensitive.
-
Figure 2—source data 1
Raw data values for Figure 2A and B.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig2-data1-v1.xlsx
Genomic characteristics
HBXN2020’s complete genome was sequenced using the Illumina HiSeq and PacBio platforms. The results showed that HBXN2020 has a circular chromosome of 3,929,792 bp (Figure 3A) with a GC content of 46.5%, including 3744 protein-coding genes (coding sequence [CDS]), 86 tRNA genes, and 27 rRNA genes (Supplementary file 2). Furthermore, the HBXN2020 genome also contains 13 different clusters of secondary metabolic synthesis genes, such as Fengycin (genomic position: 1,865,856) and Difficidin (genomic position: 2,270,091) (Supplementary file 2).
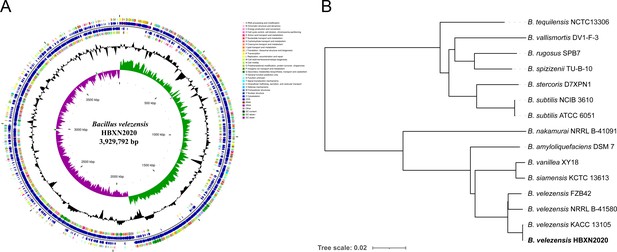
Genomic characteristics and phylogenetic relationships of B. velezensis HBXN2020.
(A) The whole-genome map of B. velezensis HBXN2020 with its genomic features. The map consists of six circles. From the inner circle to the outer circle: (1) GC-skew, (2) GC content, (3) reverse protein-coding genes, different colors represent different COG functional classifications, (4) genes transcribed in reverse direction, (5) genes transcribed in forward direction, (6) forward protein-coding genes, different colors represent different COG functional classifications. (B) The whole-genome phylogenetic tree was constructed based on genome-wide data from 14 Bacillus strains. B. velezensis HBXN2020 are indicated in bold.
A phylogenetic tree based on genome-wide data from all 14 Bacillus strains demonstrated that the HBXN2020 belongs to the B. velezensis species (Figure 3B). To further understand the classification status of HBXN2020, the online tool JSpeciesWS was used to calculate the average nucleotide identity (ANI) based on the BLAST (ANIb) method. As shown in Figure 3—figure supplement 1, HBXN2020 was found to be a member of the B. velezensis species due to the high percentage of ANIb (more than 97%).
Safety evaluation of B. velezensis HBXN2020
We assessed the in vivo effects of B. velezensis HBXN2020 in a mouse model in order to ascertain its safety. After gavage with B. velezensis HBXN2020 spores for 2 weeks, we observed no significant difference in the body weight of each group of mice (Figure 4A, Figure 4—source data 1). The gene expression levels of Tnfa, Il1b, Il6, and Il10 of colon from all groups of mice exhibited no remarkable changes (Figure 4B, Figure 4—source data 1). However, in the colon, mRNA levels of the barrier proteins Tjp1 and Ocln were trending toward an increase compared with the control group (Figure 4C, Figure 4—source data 1). Additionally, blood routine tests and serum biochemistry tests were performed for mice in the control group and H-HBXN2020 group on the 14th day after oral administration of B. velezensis HBXN2020 spore multiple times. As shown in Figure 4D, Figure 4—source data 1, the blood parameters of mice after B. velezensis HBXN2020 spores treatment, including red blood cells (RBC), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), hemoglobin (HGB), white blood cells (WBC), platelets (PLT), hematocrit (HCT), and mean corpuscular hemoglobin concentration (MCHC), were consistent with those of healthy mice (Figure 4—figure supplement 1, Figure 4—figure supplement 1—source data 1). The serum biochemical parameters, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin (ALB), total bilirubin (TBIL), serum creatinine (CREA), and blood urea nitrogen (BUN), were also within normal limits (Figure 4E, Figure 4—source data 1). The corresponding histological analysis of colon tissue from mice receiving low, medium, and high doses of B. velezensis HBXN2020 spores (L-HBXN2020, M-HBXN2020, and H-HBXN2020 groups, respectively) is presented in Figure 4F. The colon tissue sections of mice in the test groups showed no significant differences compared to the control group. Additionally, there were no observable differences in the major organ tissues (heart, liver, spleen, lung, and kidney) of mice treated with the high dose of B. velezensis HBXN2020 spores compared to healthy mice (Figure 4—figure supplement 2). The results of this trial show that B. velezensis HBXN2020 is safe to use and has no negative side effects in mice.
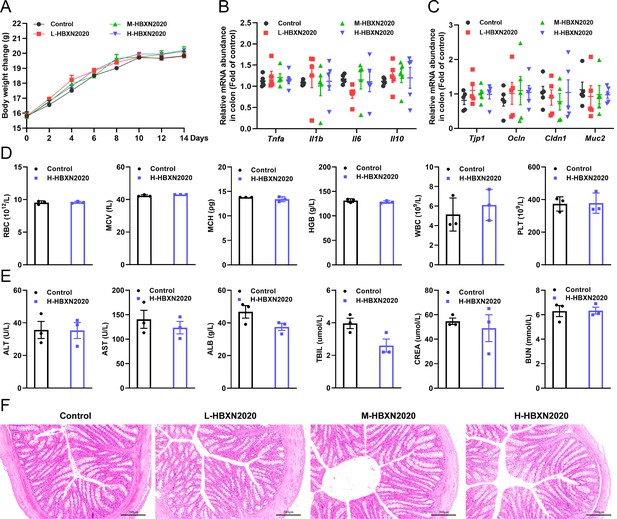
In vivo safety evaluation of B. velezensis HBXN2020 in a mouse model.
(A) Body weights changes of mice during gavage with B. velezensis HBXN2020 spores. Mice were treated with sterile PBS (control group) or low-dose (L-HBXN2020 group), medium dose (M-HBXN2020 group), and high-dose (H-HBXN2020 group) of B. velezensis HBXN2020 spores. Weighing and gavage were performed once every 2 days during the experimental period (15 days). Data were shown as mean values ± SEM (n=5). (B) The mRNA levels of inflammatory cytokines in the colon of mice measured by RT-qPCR. Data were shown as mean values ± SEM (n=5). (C) The mRNA levels of barrier protein Tjp1, Ocln, Cldn1, and Muc2 in the colon of mice measured by RT-qPCR. Data were shown as mean values ± SEM (n=5). (D) Major blood routine parameters and (E) serum biochemical parameters of mice in the control group and H-HBXN2020 group. Data were shown as mean values ± SEM (n=3). (F) Hematoxylin and eosin (H&E) stained colon sections in the different groups. Scale bar: 200 μm.
-
Figure 4—source data 1
Raw data values for Figure 4A–E.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig4-data1-v1.xlsx
Oral administration of B. velezensis HBXN2020 spores alleviated infection by S. Typhimurium
Testing both solid and liquid co-culture methods to see if B. velezensis HBXN2020 might suppress S. Typhimurium ATCC14028 (STm), the findings indicated that B. velezensis HBXN2020 decreased the development of STm in a dose-dependent manner (Figure 5A, Figure 5—figure supplement 1, Figure 5—source data 1, and Figure 5—figure supplement 1—source data 1). Next, the therapeutic potential of B. velezensis HBXN2020 was evaluated in an STm-infected mouse model. At days 1, 3, and 5 after STm infection, mice in the STm+HBXN2020 group and HBXN2020 group were orally administered B. velezensis HBXN2020 spores by gavage, STm+CIP group were given oral ciprofloxacin, while the control group and STm+PBS group were orally treated with sterile PBS (Figure 5B). Also, the number of excreted STm in feces was counted daily at the designated time points. As shown in Figure 5C, Figure 5—source data 1, there was a significant and continuous reduction in the number of STm in feces following treatment with B. velezensis HBXN2020 spores or ciprofloxacin, while the number of B. velezensis HBXN2020 viable bacteria in feces is also gradually decreasing (Figure 5—figure supplement 2, Figure 5—figure supplement 2—source data 1). The number of STm in the mice feces of STm+HBXN2020 group and STm+CIP group exhibited a reduction ranging from 0.12 to 1.18 logs and 0.76–1.81 logs, respectively, compared with the STm+PBS group, over the period from day 2 to day 7 post-treatment. Moreover, treatment with B. velezensis HBXN2020 spore or ciprofloxacin resulted in a 1.06, 1.69, 1.14 logs and 1.70, 2.15, 1.48 logs reduction in the number of STm in the cecum, colon, and ileum, respectively (Figure 5D and E, Figure 5—figure supplement 2, Figure 5—source data 1, and Figure 5—figure supplement 2—source data 1). The weight loss (p<0.05) and disease activity index (DAI) scores (p<0.01) were significantly reduced in mice after B. velezensis HBXN2020 spore treatment or ciprofloxacin treatment compared to that in the PBS treatment group (Figure 5F and G, Figure 5—source data 1). In addition, we also measured the colon length of the mice and found that the STm+PBS group had a significantly shorter colon length (p<0.001) than the control group and HBXN2020 group. However, compared to the STm+PBS group, the STm+HBXN2020 group (p<0.05) and STm+CIP group exhibited a longer colon (Figure 5H and I, Figure 5—source data 1).
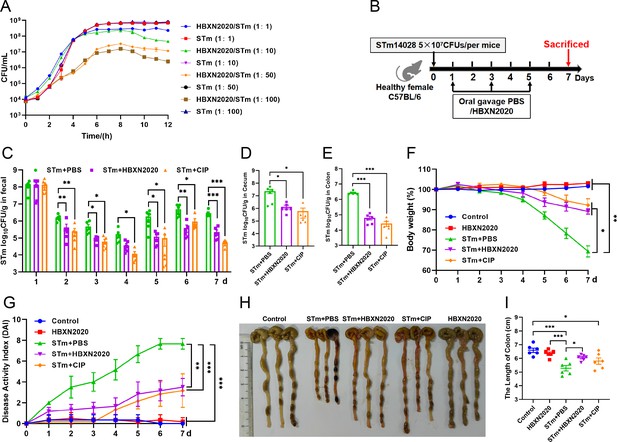
Oral B. velezensis HBXN2020 spores alleviated infection by S. Typhimurium.
(A) In vitro bacterial competition between STm and B. velezensis HBXN2020. STm were co-incubated with B. velezensis HBXN2020 at various ratios at 37°C with shaking. The growth of STm was reflected by bacterial counting per hour. (B) Experimental design for treatment in this study. Orally administrated with either sterile PBS, B. velezensis HBXN2020 spores or ciprofloxacin by gavage at days 1, 3, and 5 after STm (5×107 CFU/mouse) infection, respectively. All mice were euthanized at day 7 after STm infection. (C) Bacterial count of STm in mouse feces. Fecal samples were collected per day after STm infection and resuspended in sterile PBS (0.1 g of fecal resuspended in 1 mL of sterile PBS). One hundred microliters of each sample performed a serial of 10-fold dilutions and spread on selective agar plates (50 µg/mL kanamycin) and incubated at 37°C for 12 hr before bacterial counting. The bacterial loads of STm in (D) cecum and (E) colon. The cecum and colon were harvested and then homogenized. Data were shown as mean values ± SEM (n=6). Statistical significance was evaluated using Student’s t-test (*, p<0.05, **, p<0.01, and ***, p<0.001). (F) Daily body weight changes and (G) daily disease activity index (DAI) scores of mice with different treatment groups. Data were shown as mean values ± SEM (n=6). Statistical significance was evaluated using one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test (*, p<0.05, **, p<0.01, and ***, p<0.001). (H) Colonic tissue images. (I) The length of the colon from per group (n=6). Statistical significance was evaluated using one-way ANOVA with Tukey’s multiple comparisons test (*, p<0.05, **, p<0.01, and ***, p<0.001).
-
Figure 5—source data 1
Raw data values for Figure 5A, C, D, E, F, G, I.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig5-data1-v1.xlsx
-
Figure 5—source data 2
PDF file containing original colonic tissue image for Figure 5H.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig5-data2-v1.zip
-
Figure 5—source data 3
Original files for colonic tissue image displayed in Figure 5H.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig5-data3-v1.zip
Histological analysis further showed that in the control group and HBXN2020 group, the colon epithelial cells and crypt structure were intact with neat villi, while STm+PBS group showed significant histological damage, including erosion or loss of the intestinal epithelium, crypt destruction, and inflammatory cell infiltration in the colonic tissues (Figure 6A). Compared with the STm+PBS group, B. velezensis HBXN2020 and ciprofloxacin protected the mucosal architecture and the loss of intestinal epithelial cells, and reduced inflammatory cell infiltration (Figure 6A).
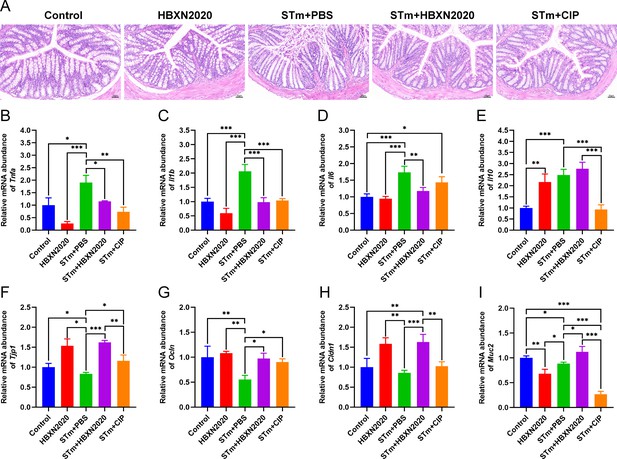
Oral B. velezensis HBXN2020 spores attenuated colonic damage and inflammatory reaction.
(A) Hematoxylin and eosin (H&E) stained colon tissue sections. Scale bar: 50 μm. The mRNA levels of (B) Tnfa, (C) Il1b, (D) Il6, and (E) Il10 were detected by RT-qPCR. Data were shown as mean values ± SEM (n=5). The mRNA levels of (F) Tjp1, (G) Ocln, (H) Cldn1, and (I) Muc2 in colon tissue were detected by RT-qPCR. Data were shown as mean values ± SEM (n=5). Statistical significance was evaluated using one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test (*, p<0.05, **, p<0.01, and ***, p<0.001).
-
Figure 6—source data 1
Raw data values for Figure 6B–I.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig6-data1-v1.xlsx
To further assess the impact of B. velezensis HBXN2020 on intestinal inflammatory response, the mRNA levels of inflammatory cytokines in the colonic tissues were measured. As shown in Figure 6B–D; Figure 6—source data 1, the mRNA levels of Tnfa (p<0.05), Il1b (p<0.001), Il6 (p<0.001), and Il10 (p<0.001) in the colon tissue were significantly increased in the STm+PBS group compared to the control group. B. velezensis HBXN2020 spore treatment significantly decreased the mRNA levels of Tnfa (p<0.05), Il1b (p<0.001), Il6 (p<0.01), and increased the Il10 level, compared with the STm+PBS group. In contrast, ciprofloxacin treatment failed to significantly decrease the mRNA levels of Il6 in the colon of STm-infected mice (Figure 6D, Figure 6—source data 1). Similarly, ciprofloxacin treatment drastically dampened the mRNA levels of Il10 in the colon of STm-infected mice (Figure 6E, Figure 6—source data 1).
To assess the influence of B. velezensis HBXN2020 on the intestinal barrier integrity, the mRNA levels of tight junction proteins in the colonic tissues were measured. As presented in Figure 6, compared with control group, the mRNA levels of tight junction protein in the STm+PBS group was significantly reduced, including Tjp1 (p<0.05), Ocln (p<0.01), Cldn1, and Muc2 (p<0.05) (Figure 6F–I, Figure 6—source data 1). Moreover, the levels of tight junction protein transcription were remarkably elevated in the STm+HBXN2020, STm+CIP, and HBXN2020 group, including Tjp1 (p<0.001, p<0.05, and p<0.05, respectively), Ocln (p<0.05, p<0.05, and p<0.01, respectively), and Cldn1 (p<0.001, p=0.0857, and p<0.01, respectively), except for Cldn1 (p=0.0857) in group STm+CIP, compared with that of the STm+PBS group (Figure 6F–I, Figure 6—source data 1). In addition, we also detected the transcription level of Muc2 in colon tissue, which mainly exists in and around goblet cells and is an important component of the mucus layer. The transcription levels of Muc2 were dramatically increased in the STm+HBXN2020 group compared with the STm+PBS group (Figure 6I, Figure 6—source data 1). Conversely, the mRNA levels of Muc2 were significantly decreased in the HBXN2020 group and STm+CIP group (Figure 6I, Figure 6—source data 1).
Next, we further explore the impact of B. velezensis HBXN2020 on the intestinal microbiota composition of STm-treated mice by 16S rRNA gene high-throughout sequencing. As shown in Figure 7, alpha diversity analysis revealed that both the richness and diversity (calculated in Sobs, Chao, Shannon, and Simpson indexes) were lower in STm+PBS group than control group, HBXN2020 group, STm+HBXN2020 group, and STm+CIP group (Figure 7A–D, Figure 7—source data 1). Although an increasing trend of alpha diversity was observed in STm+CIP group, but without statistical difference was achieved as compared with other groups. Principal components analysis (PCA) based on Bray-Curtis distance showed that a separation in the gut microbiota structure among control and STm+PBS group. When B. velezensis HBXN2020 spores were supplemented, the gut microbiota composition of the STm+HBXN2020 group, HBXN2020 group, and the control group were closer together (R2= 0.1616, p = 0.006, Figure 7E).
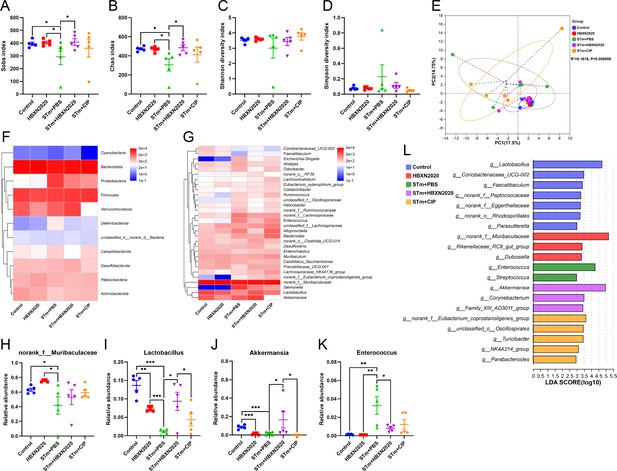
Oral B. velezensis HBXN2020 spores regulated the composition of intestinal microbiota.
The alpha diversity of the gut microbiota, determined by the (A) Sobs, (B) Chao, (C) Shannon, and (D) Simpson index. Data were shown as mean values ± SEM (n=5). (E) The principal components analysis (PCA) plot showed the β diversity of the gut microbiota based on Bray-Curtis distance at the operational taxonomic unit (OTU) level. Heatmap of the community composition of colonic microbiota at the phylum (top 15 phyla) (F) and genus (top 30 genera) (G) level. Relative abundance of selected taxa (H) norank_f_Muribaculaceae, (I) Lactobacillus, (J) Akkermansia, and (K) Enterococcus. Data were shown as mean values ± SEM (n=5). (L) Analysis of differences in the microbial communities by LEfSe (linear discriminant analysis [LDA] effect size) (LDA score>2) among different groups. Significance was evaluated by one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test (*, p<0.05, **, p<0.01, and ***, p<0.001).
-
Figure 7—source data 1
Raw data values for Figure 7A–D and H–K.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig7-data1-v1.xlsx
We analyzed the community composition of colonic microbiota at the phylum and genus level, and the results revealed that the composition of the gut microbiota changed markedly after STm-infected mice. At the phylum level, Bacteroidetes, Firmicutes, Verrucomicrobiota, and Proteobacteria were predominant phyla in the fecal microbiota (Figure 7F, Figure 7—figure supplement 1, and Figure 7—source data 1). At the genus level, infection with STm in the STm+PBS group dramatically reduced the relative abundance of norank_f_Muribaculaceae, Lactobacillus, and Akkermansia (Figure 7G and H–J, Figure 7—source data 1) and enhanced the abundance of Enterococcus, Salmonella, Bacteroides, Alloprevotella, and Escherichia-Shigella compared to the control group (Figure 7G and K, Figure 7—figure supplement 1, and Figure 7—source data 1). In contrast, the STm+HBXN2020 group and HBXN2020 group significantly improved the relative abundance of norank_f_Muribaculaceae (p=0.49, p<0.05) and Lactobacillus (p<0.05, p<0.001) compared with the STm+PBS group (Figure 7H and I, Figure 7—source data 1). However, ciprofloxacin treatment significantly decreased the relative abundance of Lactobacillus (p<0.05) and Akkermansia (p<0.05) compared with the STm+HBXN2020 group (Figure 7I and J, Figure 7—source data 1). Differentially abundant fecal bacterial taxa in STm-treated mice in response to B. velezensis HBXN2020 and ciprofloxacin were identified by linear discriminant analysis (LDA) effect size (LEfSe) analysis (Figure 7L). Additionally, we found that two bacterial genera including Enterococcus and Streptococcus were enriched in the STm+PBS group, while the other three taxa were enriched including Akkermansia, Family_XIII_AD3011_group, and Corynebacterium in the STm+HBXN2020 group, Parabacteroides, NK4A214_group, Turicibacter, unclassified_o__Oscillospirales, and norank_f__Eubacterium_coprostanoligenes_group in the STm+CIP group, and norank_f_Muribaculaceae, Rikenellaceae_RC9_gut_group and Dubosiella in the HBXN2020 group (Figure 7L).
Prophylactic B. velezensis HBXN2020 spores alleviated infection by S. Typhimurium
Based on the improved therapeutic efficacy of B. velezensis HBXN2020 in the treatment of STm-infected mice, we further explored its potential for disease prevention by evaluating pretreatment. Mice in the PBS+STm group, HBXN2020+STm group, and CIP+STm group were given the same amount of sterile PBS, B. velezensis HBXN2020 spores or ciprofloxacin by gavage 1 week in advance. As shown in Figure 8, compared with the PBS+STm group, oral administration of B. velezensis HBXN2020 spores or ciprofloxacin as a preventive measure remarkably alleviated infection by STm, including weight loss of mice (p<0.01) (Figure 8B, Figure 8—source data 1), a significant reduction in DAI (p<0.001) (the comprehensive score of weight loss, stool consistency, and blood in the feces, Figure 8C, Figure 8—source data 1), and the prevention of colon length shortening (Figure 8D and E, Figure 8—source data 1). Meanwhile, the number of B. velezensis HBXN2020 viable bacteria in feces is also gradually decreasing with time prolonging (Figure 8—figure supplement 1, Figure 8—figure supplement 1—source data 1). Furthermore, compared with the PBS+STm group, oral administration of B. velezensis HBXN2020 spores or ciprofloxacin not only reduced the number of STm in mouse feces (Figure 8F, Figure 8—source data 1) but also decreased STm colonization in the ileum, cecum, and colon (Figure 8G and H, Figure 8—figure supplement 1, Figure 8—source data 1, and Figure 8—figure supplement 1—source data 1).
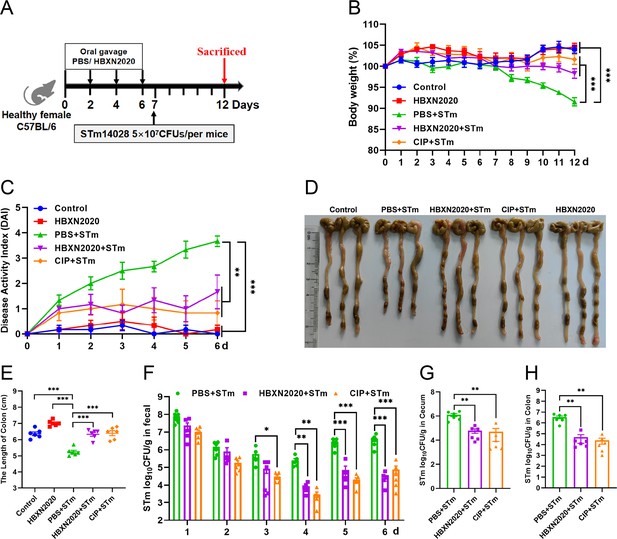
Prophylactic B. velezensis HBXN2020 spores attenuated the symptoms of S. Typhimurium-infected mouse.
(A) Experimental design for treatment in this study. At days 1, 3, 5, and 7, each mouse in the HBXN2020+STm group, CIP+STm group, and PBS+STm group were received 200 μL of B. velezensis HBXN2020 spores (1×108 CFU/mouse), ciprofloxacin or sterile PBS by gavage, respectively. Then, mice in PBS+STm group, HBXN2020+STm group, and CIP+STm group were orally inoculated with 200 μL (5×107 CFU/mouse) of STm on day 7, respectively. On day 12, all mice were euthanized. (B) Daily body weight changes and (C) daily disease activity index (DAI) scores of mice with different groups following STm treatment. Data were shown as mean values ± SEM (n=6). Statistical significance was evaluated using one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test (*, p<0.05, **, p<0.01, and ***, p<0.001). (D) Colonic tissue images. (E) The length of the colon from per group (n=6). (F) Bacterial count of STm in mouse feces. Fecal samples were collected every day after STm infection and resuspended in sterile PBS (0.1 g of fecal resuspended in 1 mL of sterile PBS) (n=6). One hundred microliters of each sample performed a serial of 10-fold dilutions and spread on selective agar plates (50 µg/mL kanamycin) and incubated at 37°C for 12 hr before bacterial counting. The bacterial loads of STm in (G) cecum and (H) colon (n=6). The cecum and colon were harvested and then homogenized. Statistical significance was evaluated using Student’s t-test (*, p<0.05, **, p<0.01, and ***, p<0.001).
-
Figure 8—source data 1
Raw numerical data for Figure 8B, C, and E–H.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig8-data1-v1.xlsx
-
Figure 8—source data 2
PDF file containing original colonic tissue image for Figure 8D.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig8-data2-v1.zip
-
Figure 8—source data 3
Original files for colonic tissue image displayed in Figure 8D.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig8-data3-v1.zip
Histological analysis further revealed that prophylactic B. velezensis HBXN2020 spore suppressed STm-induced loss of the intestinal epithelium, inflammatory cell infiltration, and colonic mucosa damage (Figure 9A), while prophylactic ciprofloxacin failed to inhibit the loss of colonic epithelial cells. We next measured the mRNA levels of inflammatory cytokines and tight junction proteins in colon tissue in the prophylactic animal experiment. As with the treatment test, prophylactic B. velezensis HBXN2020 spores largely attenuated the mRNA levels of Tnfa (p<0.001), Il1b (p<0.001), and Il6 (p<0.001), and significantly increased levels of Il10 (p<0.05), in the colon tissue of mice compared with the PBS+STm group (Figure 9B–E, Figure 9—source data 1). Similarly, prophylactic ciprofloxacin significantly reduced the mRNA levels of Tnfa (p<0.01) and Il1b (p<0.001), and drastically elevated levels of Il10 (p<0.001) in the colon tissue of mice compared with the PBS+STm group (Figure 9B, C, and E, Figure 9—source data 1). Also, prophylactic B. velezensis HBXN2020 spores significantly increased the transcription levels of Tjp1 (p<0.001), Ocln (p<0.001), Cldn1 (p<0.05), and Muc2 (p<0.001) compared with that of the PBS+STm group (Figure 9F–I, Figure 9—source data 1). Notably, the mRNA levels of Tjp1 (p<0.001), Ocln (p<0.05), and Muc2 (p<0.05) in the CIP+STm group were significantly lower than those in the HBXN2020+STm group. Overall, these results suggested that prophylactic B. velezensis HBXN2020 spores was capable of alleviating STm-induced colonic injury and inflammation.
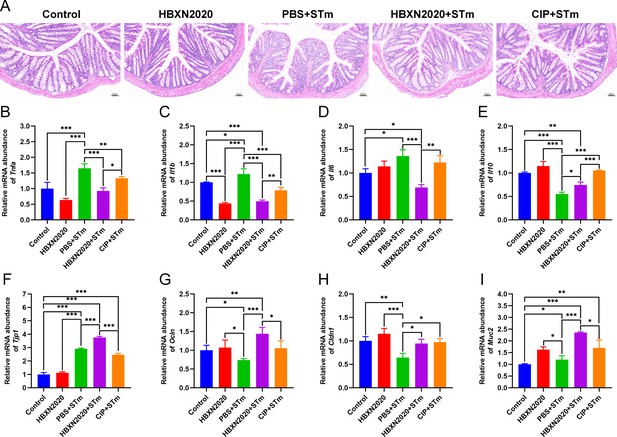
Prophylactic B. velezensis HBXN2020 spores attenuated colonic damage and inflammatory reaction.
(A) Hematoxylin and eosin (H&E) stained colon tissue sections. Scale bar: 50 μm. The mRNA levels of (B) Tnfa, (C) Il1b, (D) Il6, and (E) Il10 were detected by RT-qPCR. Data were shown as mean values ± SEM (n=5). The mRNA levels of (F) Tjp1, (G) Ocln, (H) Cldn1, and (I) Muc2 in colon tissue were detected by RT-qPCR. Data were shown as mean values ± SEM (n=5). Statistical significance was evaluated using one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test (*, p<0.05, **, p<0.01, and ***, p<0.001).
-
Figure 9—source data 1
Raw numerical data for Figure 9B–I.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig9-data1-v1.xlsx
Next, we examined the impact of prophylactic B. velezensis HBXN2020 on the intestinal microbiota composition of STm-treated mice. Sobs, Chao, and Shannon index were significantly increased in the HBXN2020, PBS+STm group, HBXN2020+STm group, and CIP+STm group compared with the control group (Figure 10A–C, Figure 10—source data 1), while the Simpson index (not statistically different) significantly decreased (Figure 10D, Figure 10—source data 1). PCA showed significant separation between control and PBS+STm groups (R2=0.3186, p=0.014, Figure 10E). When B. velezensis HBXN2020 spores or ciprofloxacin were supplemented, the community clustering was significantly similar to that of the control group rather than the PBS+STm group (Figure 10E). Meanwhile, the overall fecal bacterial composition of mice at the phylum and genus level in all groups was similar to that of the treatment experiment. The fecal microbiota was dominated by phyla Firmicutes, Bacteroidetes, Verrucomicrobiota, Patescibacteria, and Actinobacteria in all five groups (Figure 10—figure supplement 1). At the genus level, PBS+STm group exhibited lower relative abundances of Lactobacillus and Akkermansia (Figure 10G–I, Figure 10—source data 1), and higher relative abundances of Alistipes, Bacteroides, Escherichia-Shigella, Enterococcus, and Alloprevotella, compared with the control (Figure 10G and J–M, Figure 10—figure supplement 1, and Figure 10—source data 1). Moreover, compared with the PBS+STm group, following supplementation with B. velezensis HBXN2020 spores or ciprofloxacin, Lactobacillus significantly increased, and the gut microbiota was restored to a composition similar to the control group. It was noteworthy that six bacterial genera including Alistipes, Odoribacter, and norank_f__Oscillospiraceae were enriched in PBS+STm group, while other three taxa (e.g. Lactobacillus and Akkermansia) were enriched in the control group (Figure 10N). Moreover, we found that Candidatus_Arthromitus was enriched in the HBXN2020+STm group, Dubosiella was enriched in the CIP+STm group, and Faecalibaculum, Rikenellaceae_RC9_gut_group, and unclassified_f__Lactobacillaceae were enriched in the HBXN2020 group (Figure 10N). Above all, these results indicated that prophylactic B. velezensis HBXN2020 spores regulated the gut microbiota composition, leading to the potential to attenuate the STm-induced dysbiosis.
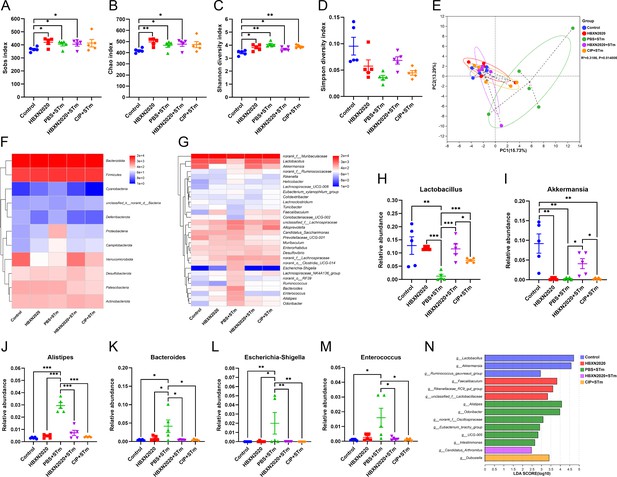
Prophylactic B. velezensis HBXN2020 spores regulated the composition of gut microbiota.
(A–D) Alpha diversity of the intestinal microbiota. Data were shown as mean values ± SEM (n=5). (E) The principal components analysis (PCA) plot showed the β diversity among different microbial community groups based on Bray-Curtis distance at the operational taxonomic unit (OTU) level. Heatmap of the community composition of colonic microbiota at the phylum (top 15 phyla) (F) and genus (top 30 genera) (G) level. Relative abundance of selected taxa (H) Lactobacillus, (I) Akkermansia, (J) Alistipes, (K) Bacteroides, (L) Escherichia-Shigella, and (M) Enterococcus. Data were shown as mean values ± SEM (n=5). (N) Analysis of differences in the microbial taxa by LEfSe (linear discriminant analysis [LDA] effect size) (LDA score>2) in different groups. Significance was evaluated by one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test (*, p<0.05, **, p<0.01, and ***, p<0.001).
-
Figure 10—source data 1
Raw numerical data for Figure 10A–D and H–M.
- https://cdn.elifesciences.org/articles/93423/elife-93423-fig10-data1-v1.xlsx
Discussion
S. Typhimurium is an important intestinal pathogen that can cause invasive intestinal diseases such as bacterial colitis (Herp et al., 2019; Schultz et al., 2017). Antibiotics are frequently used to treat colitis caused by bacteria; nevertheless, in recent years, their abuse has greatly increased bacterial resistance, leading to serious environmental pollution. A recent study showed that antibiotic resistance genes in probiotics could be transmitted to the intestinal microbiota, which may threaten human health (Crits-Christoph et al., 2022). It is crucial to consider the source of probiotics. Strong environmental adaptability and disease resistance make black pigs an outstanding household breed in China (Yang et al., 2022). In this study, B. velezensis HBXN2020 was isolated from the free-range feces of black piglets in a mountain village in Xianning City (Hubei, China) and exhibited excellent antibacterial activity. Otherwise, in vitro tolerance assays showed that B. velezensis HBXN2020 spores have good tolerance to high temperature, strong acids, bile salts, as well as simulated gastric and intestinal fluid, which was similar to previous research results (Du et al., 2022). Furthermore, studies for antibiotic susceptibility revealed that B. velezensis HBXN2020 is not resistant to antibiotics.
Probiotics are distinct from food or medications in that they can cause toxins or infections in the body when ingested, and they are living when swallowed (Sanders et al., 2010; Snydman, 2008). Therefore, assessing the biosafety of probiotics is crucial. In this study, the safety of B. velezensis HBXN2020 was evaluated by measuring the body weight, intestinal barrier proteins, and inflammatory cytokines of the experimented mice, as well as conducting routine blood and biochemical tests. The results showed that the expression levels of Tjp1 and Ocln in the colon of mice increased after treatment with different doses of B. velezensis HBXN2020 spores. It has been reported that Tjp1, Ocln, and Cldn1 are the three most important tight junction proteins in intercellular connections, which play a crucial role in maintaining the intestinal epithelial barrier (Bazzoni et al., 2000; Zihni et al., 2016). Thus, it is possible to improve intestinal barrier function by upregulating Tjp1 and Ocln expression levels.
Important aminotransferases in animals, AST and ALT, are regarded as critical metrics for assessing liver function damage (Gou et al., 2022; Ozer et al., 2008). Under normal circumstances, the levels of ALT and AST are in dynamic balance without notable changes. However, when the liver is damaged or becomes dysfunctional, the levels of aminotransferases (ALT and AST) increase significantly (Tang et al., 2012). Previous studies have shown that CCL4-induced liver injury strongly increases plasma ALT/AST levels (Singhal et al., 2018). Zhang et al. reported that the addition of Bacillus subtilis to chicken diets significantly decreased the serum levels of ALT and AST (Zhang et al., 2017). In this present study, the levels of ALT and AST in the probiotic-treated group were slightly lower than those in the control group, indicating that B. velezensis HBXN2020 had no negative effect on liver health in mice. The B. velezensis HBXN2020 treatment group’s blood indices were also found to be comparable to those of the control group, falling within the normal reference range, according to regular blood testing.
Mice were fed with B. velezensis HBXN2020 spores orally after being challenged with S. Typhimurium ATCC14028 in order to assess the spores’ potential to alleviate infection caused by S. Typhimurium. Our experimental results demonstrated that oral treatment with B. velezensis HBXN2020 spores could alleviate infection by S. Typhimurium, as evidenced by decreased weight loss, DAI, and histological damage, which is similar to prior studies (Cao et al., 2019; Wu et al., 2022). Previous research showed that macrophages are the first line of host defense against bacterial infection. They release proinflammatory cytokines, which are critical in initiating adaptive immune responses (Cheng et al., 2014; Dinarello, 2000). Nevertheless, proinflammatory cytokines have immunological properties that can be beneficial for the host to resist the invasion of bacteria and other microbes in the surrounding environment (Liu et al., 2021). For instance, pretreatment with recombinant murine Tnfa was shown to protect mice against lethal bacterial (E. coli) infection (Cross et al., 1989). PJ-34 exerted protective effects on intestinal epithelial cells against invasive Salmonella infection by upregulating Il6 expression through the ERK and NF-κB signaling pathways (Huang, 2009).
On the other hand, some research has demonstrated that over-activation of immune cells can result in tissue damage, organ failure, systemic or persistent inflammation, and autoimmune diseases (Karki et al., 2021; Kotas and Medzhitov, 2015). Il10 is a recognized anti-inflammatory mediator that plays a crucial role in maintaining intestinal microbe-immune homeostasis, regulating the release of inflammatory mediators, and inhibiting proinflammatory responses of innate and adaptive immunity (Maloy and Powrie, 2011; Ouyang et al., 2011; Saraiva et al., 2020). For instance, previous studies have shown that Clostridium butyrate can induce the production of Il10 in the intestine, thereby alleviating experimental colitis in mice (Hayashi et al., 2013). Similar results were observed in our study, where oral administration of B. velezensis HBXN2020 spores reduced the expression levels of Tnfa, Il1b, and Il6 while increasing the levels of Il10 in the colon of mice. Additionally, as demonstrated by the decreased histological damage and increased expression levels of intestinal barrier proteins, oral treatment of B. velezensis HBXN2020 spores reduced the functional damage of the intestinal barrier induced by S. Typhimurium infection.
Besides host itself, environmental factors such as diet and gut microbiota have been associated with the development of STm-infected. Gut microbiota constitutes a critical bridge between environmental factors and host health, where the beneficial bacteria such as Lactobacillus in the microbiota may exert repair and anti-inflammatory function (Li et al., 2022; Liu et al., 2022). Moreover, supplementation of probiotics has been shown to modulate intestinal microbiota and reduce the risk of STm-infected (Buddhasiri et al., 2021; Zhang et al., 2022). This is consistent with our research results between the B. velezensis HBXN2020 treatment group and STm-infected mice. Here, we found that oral B. velezensis HBXN2020 spores raised the colonic microbiota’s alpha diversity and modulated the composition of the intestinal microbiota, namely, enriching the relative abundance of Lactobacillus (known for enhancing the epithelial barrier by increasing mucus secretion and upregulating the expression of tight junction proteins such as Cldn1, Ocln, and Tjp1) (Kaur et al., 2021) and Akkermansia (known for modulating intestinal immune response by producing short-chain fatty acids and reducing the secretion of proinflammatory cytokines) (Cani et al., 2022), reducing the relative abundance of Enterococcus, Salmonella, and Bacteroides (harmful to intestinal homeostasis) (Wang et al., 2023). Notably, we also found that the relative abundance of Lactobacillus and Akkermansia in the ciprofloxacin treatment group was significantly lower than that in the B. velezensis HBXN2020 spores treatment group, indicating that excessive use of antibiotics may interfere with the recovery of beneficial bacteria in the gut. Collectively, infection with S. Typhimurium disrupts gut microbiota and the gut barrier, leading to intestinal inflammation, while oral B. velezensis HBXN2020 spores protects the colon and reduce infection by increasing the abundance of beneficial bacteria and barrier integrity and decreasing inflammation.
We further assessed pretreatment in order to learn more about the potential of B. velezensis HBXN2020 spores for disease prevention, given the increased efficacy of treating infection caused by S. Typhimurium. Similar to the therapeutic effect of oral B. velezensis HBXN2020, prophylactic B. velezensis HBXN2020 spores effectively alleviated the symptoms of STm-infected mice, namely, reduced the mRNA levels of proinflammatory cytokines and increased the anti-inflammatory cytokine level in the colon of STm-infected mice. Prophylactic B. velezensis HBXN2020 spores also increased the mRNA levels of gut barrier proteins in the colon of STm-infected mice. Furthermore, the microbiota composition was also dramatically modulated by prophylactic B. velezensis HBXN2020 spores in both healthy mice and STm-infected mice. Also, the community richness revealed by the alpha diversity index were not significantly impacted in STm-infected mice by prophylactic B. velezensis HBXN2020 spores. Interestingly, we found that prophylactic B. velezensis HBXN2020 spores significantly increased the community diversity in healthy mice, suggesting the potentially beneficial effects of B. velezensis HBXN2020 in gut microenvironment for animals in the future. Moreover, prophylactic B. velezensis HBXN2020 spores significantly boosted SCFA-producing bacteria including Lactobacillus and Akkermansia, which was consistent with the effects of therapeutic oral B. velezensis HBXN2020 spores. Meanwhile, harmful bacteria such as Alistipes, Bacteroides, Escherichia-Shigella, and Enterococcus were significantly enriched in STm-infected mice, reminding us that increased harmful bacteria might play a major role in promoting the development of STm-infected. In summary, our results demonstrated that B. velezensis HBXN2020-mediated microbial communities, especially Lactobacillus and Akkermansia, might play a crucial role in alleviating the development of STm-infected.
Conclusion
In conclusion, S. Typhimurium ATCC14028 infection can result in gut barrier disruption and intestinal inflammation and intestinal microbiota dysbiosis. Supplementing B. velezensis HBXN2020 spores can improve intestinal microbiota stability and gut barrier integrity and reduce inflammation to help prevent or treat infection by S. Typhimurium. Thus, our results indicate that supplementing B. velezensis HBXN2020 may represent a novel option for preventing Salmonella infections.
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Bacillus velezensis) | HBXN2020 | This paper | NCBI GenBank accession No: CP119399.1 | This strain is used in the entire text |
| Strain, strain background (Salmonella Typhimurium) | STm | ATCC | 14028 | |
| Strain, strain background (Escherichia coli) | 25922 | ATCC | 25922 | |
| Strain, strain background (E. coli) | 35150 | ATCC | 35150 | |
| Strain, strain background (E. coli) | EC024 | This paper | This strain is used in Figure 2B | |
| Strain, strain background (S. Typhimurium) | SL1344 | Gao et al., 2022 | ||
| Strain, strain background (S. Enteritidis) | SE006 | Gao et al., 2022 | ||
| Strain, strain background (Staphylococcus aureus) | 29213 | ATCC | 29213 | |
| Strain, strain background (S. aureus) | 43300 | ATCC | 43300 | |
| Strain, strain background (S. aureus) | S21 | This paper | This strain is used in Figure 2B | |
| Strain, strain background (Clostridium perfringens) | 2030 | CVCC | 2030 | |
| Strain, strain background (C. perfringens) | CP023 | This paper | This strain is used in Figure 2B | |
| Strain, strain background (C. perfringens) | CP002 | This paper | This strain is used in Figure 2B | |
| Strain, strain background (Streptococcus suis) | SC19 | Duan et al., 2023 | ||
| Strain, strain background (S. suis) | SS006 | This paper | This strain is used in Figure 2B | |
| Strain, strain background (Pasteurella multocida) | PM002 | This paper | This strain is used in Figure 2B | |
| Strain, strain background (P. multocida) | PM008 | This paper | This strain is used in Figure 2B | |
| Strain, strain background (Actinobacillus pleuropneumoniae) | APP015 | This paper | This strain is used in Figure 2B | |
| Strain, strain background (A. pleuropneumoniae) | APP017 | This paper | This strain is used in Figure 2B | |
| Sequence-based reagent | Tnfa-F | This paper | qPCR primers | CCACGCTCTTCTGTCTACTG |
| Sequence-based reagent | Tnfa-R | This paper | qPCR primers | ACTTGGTGGTTTGCTACGA |
| Sequence-based reagent | Il1b-F | This paper | qPCR primers | ACCTGTGTCTTTCCCGTGG |
| Sequence-based reagent | Il1b-R | This paper | qPCR primers | TCATCTCGGAGCCTGTAGTG |
| Sequence-based reagent | Il6-F | This paper | qPCR primers | GAGCCCACCAAGAACGATA |
| Sequence-based reagent | Il6-R | This paper | qPCR primers | TTGTCACCAGCATCAGTCC |
| Sequence-based reagent | Il10-F | This paper | qPCR primers | TGGACAACATACTGCTAACCG |
| Sequence-based reagent | Il10-R | This paper | qPCR primers | GGGCATCACTTCTACCAGGT |
| Sequence-based reagent | Tjp1-F | This paper | qPCR primers | CTGGTGAAGTCTCGGAAAAATG |
| Sequence-based reagent | Tjp1-R | This paper | qPCR primers | CATCTCTTGCTGCCAAACTATC |
| Sequence-based reagent | Ocln-F | This paper | qPCR primers | CAGGATGCCAATTACCATCAAG |
| Sequence-based reagent | Ocln-R | This paper | qPCR primers | GGGTTCACTCCCATTATGTACA |
| Sequence-based reagent | Cldn1-F | This paper | qPCR primers | AGATACAGTGCAAAGTCTTCGA |
| Sequence-based reagent | Cldn1-R | This paper | qPCR primers | CAGGATGCCAATTACCATCAAG |
| Sequence-based reagent | Muc2-F | This paper | qPCR primers | CGAGCACATCACCTACCACATCATC |
| Sequence-based reagent | Muc2-R | This paper | qPCR primers | TCCAGAATCCAGCCAGCCAGTC |
| Sequence-based reagent | β-actin-F | This paper | qPCR primers | GACCTCTATGCCAACACAGT |
| Sequence-based reagent | β-actin-R | This paper | qPCR primers | CACCAATCCACACAGAGTAC |
| Commercial assay or kit | HiScript III RT SuperMix | Vazyme Biotechnology Co., Ltd | #R323-01 | |
| Commercial assay or kit | qPCR SYBR Green Master Mix | Yeasen Biotechnology Co., Ltd | #11203ES08 | |
| Commercial assay or kit | E.Z-N.A Stool DNA Kit | Omega | #D4015-01 | |
| Chemical compound, drug | Kanamycin | Solarbio | #K8020 | |
| Chemical compound, drug | Ciprofloxacin | Solarbio | #C9710 | |
| Chemical compound, drug | Ampicillin | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C002 | 10 µg/tablet |
| Chemical compound, drug | Meropenem | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C102 | 10 µg/tablet |
| Chemical compound, drug | Piperacillin | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C005 | 10 µg/tablet |
| Chemical compound, drug | Gentamycin | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C017 | 10 µg/tablet |
| Chemical compound, drug | Tetracycline | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C021 | 30 µg/tablet |
| Chemical compound, drug | Doxycycline | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C032 | 30 µg/tablet |
| Chemical compound, drug | Minocycline | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C046 | 30 µg/tablet |
| Chemical compound, drug | Erythromycin | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C023 | 15 µg/tablet |
| Chemical compound, drug | Enrofloxacin | Shunyou Shanghai Biotechnology Co., Ltd | #CT0639B | 5 µg/tablet |
| Chemical compound, drug | Ofloxacin | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C044 | 5 µg/tablet |
| Chemical compound, drug | Sulfamethoxazole | Shunyou Shanghai Biotechnology Co., Ltd | #CT0051B | 25 µg/tablet |
| Chemical compound, drug | Trimethoprim-sulfamethoxazole | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C027 | 23.75 µg/tablet |
| Chemical compound, drug | Polymyxin B | Shunyou Shanghai Biotechnology Co., Ltd | #CT0044B | 300 U/tablet |
| Chemical compound, drug | Teicoplanin | Shunyou Shanghai Biotechnology Co., Ltd | #CT0647B | 30 µg/tablet |
| Chemical compound, drug | Trimethoprim | Shunyou Shanghai Biotechnology Co., Ltd | #CT0076B | 5 µg/tablet |
| Chemical compound, drug | Florfenicol | Shunyou Shanghai Biotechnology Co., Ltd | #CT1754B | 30 µg/tablet |
| Chemical compound, drug | Spectinomycin | Shunyou Shanghai Biotechnology Co., Ltd | #CT0046B | 10 µg/tablet |
| Chemical compound, drug | Nitrofurantoin | Shunyou Shanghai Biotechnology Co., Ltd | #CT0036B | 300 µg/tablet |
| Chemical compound, drug | Rifampicin | Hangzhou Binhe Microorganism Reagent Co., Ltd | #C013 | 5 µg/tablet |
Materials and methods
Sample collection and strain isolation
Request a detailed protocolFresh fecal samples were collected from the farms of various locations in southern China including Hubei, Anhui, Hunan, Jiangxi, Guizhou, and Guangxi. All samples were placed in sterile plastic bags, sealed and placed on ice, and immediately transported to the laboratory. Bacillus was isolated following a previously described method with slight modification (Unban et al., 2020). The obtained strains were kept in Luria-Bertani (LB) media containing 25% (vol/vol) glycerol and stored at −80°C, and prepared in LB medium or LB agar medium before use.
Antibacterial activity of Bacillus
Request a detailed protocolThe antibacterial activity of Bacillus isolates was evaluated by using the spot-on-plate method on LB agar plates supplemented with indicator bacteria (Salmonella, E. coli, S. aureus). Briefly, an overnight culture of indicator bacteria was dipped by sterile cotton swabs and spread on LB agar plates. Bacillus solution was spotted onto the double-layer agar plates and incubated at 37°C for 12 hr to measure the inhibition zone. A transparent zone of at least 1 mm around the spot was considered positive.
The antibacterial spectrum of Bacillus isolates was compared through the agar diffusion test, based on the results of the spot-on-plate test. In brief, an overnight culture of indicator strains was mixed with 10 mL of TSB soft agar (TSB broth containing 0.7% [wt/vol] agar) and poured into a sterile plate covered with LB agar and Oxford cups, 8 mm diameter wells were prepared in the TSB agar after removing the cups. The wells were filled with 100 μL of Bacillus solution. The plates were incubated at 37°C for 14 hr, and the inhibition zone was measured.
The indicator strains used in this study are listed in Supplementary file 3, and all the primers used in Supplementary file 3. C. perfringens were cultured in a fluid thioglycollate medium (FTG) (Hopebio, Qingdao, China) at 45°C. S. suis and P. multocida were cultured in TSB medium supplemented with 5% (vol/vol) sheep serum (Solarbio, Beijing, China) at 37°C, and A. pleuropneumoniae were cultured in TSB supplemented with 5% (vol/vol) sheep serum and 5 mM nicotinamide adenine dinucleotide at 37°C, while the other bacterial strains were cultured in LB medium (Solarbio, Beijing, China) at 37°C. In addition, the Difco sporulation medium was used for inducing the sporulation of Bacillus via the nutrient depletion method (Tang et al., 2017).
Growth curves of HBXN2020
Request a detailed protocolThe growth curve of HBXN2020 was recorded in flat-bottomed 100-well microtiter plates via detecting optical density at 600 nm (OD600) at 1 hr intervals using the automatic growth curve analyzer (Bioscreen, Helsinki, Finland).
In vitro resistance assay of HBXN2020
Request a detailed protocolHBXN2020 spores (100 μL) or vegetative cells (100 μL) were separately resuspended in 900 μL of LB medium supplemented with different pH values (2, 3, 4, 5, or 6), bile salts (0.85% NaCl, 0.3%), simulated gastric fluid (SGF, HCl, pH 1.2) containing 10 g/L of pepsin in 0.85% NaCl solution, or simulated intestinal fluid (SIF, NaOH, pH 6.8) containing 10 g/L of trypsin in 0.05 M KH2PO4 solution, and incubated at 37°C. A normal LB medium (pH 7.0) was used as the control. At predetermined time points, 100 μL was taken from each sample, serially diluted 10-fold with sterile PBS (pH 7.2), and then spread onto LB agar plates. The plates were incubated overnight in a constant temperature incubator at 37°C, and the bacterial colonies were counted. The survival rate was calculated using the following formula: Survival rate = (number of bacteria in the treatment group/number of bacteria in the control group)×100%.
One milliliter of HBXN2020 spores or vegetative cells were separately placed in water baths at different temperatures (37°C, 45°C, 55°C, 65°C, 75°C, 85°C, or 95°C) for 20 min, with a 37°C water bath used as the control. The survival rate was calculated as described above.
Antibiotic susceptibility assays
Request a detailed protocolAntimicrobial susceptibility testing was performed using the Kirby-Bauer disk diffusion method in accordance with the Clinical Laboratory Standards Institute (CLSI) guidelines (CLSI, 2018.). The antimicrobial agents tested were: Ampicillin, Meropenem, Piperacillin, Gentamycin, Tetracycline, Doxycycline, Minocycline, Erythromycin, Enrofloxacin, Ofloxacin, Sulfamethoxazole, Trimethoprim-sulfamethoxazole, Polymyxin B, Teicoplanin, Trimethoprim, Florfenicol, Spectinomycin, Nitrofurantoin, and Rifampicin. The diameter of the inhibition zone was measured using a vernier caliper.
Antimicrobial assays
Request a detailed protocolThe in vitro antagonistic activity of HBXN2020-CFS was tested using the agar well-diffusion method against 18 indicator strains (pathogens), which included 7 standard strains (E. coli ATCC 25922, E. coli ATCC 35150, S. Typhimurium ATCC 14028 (STm), S. Typhimurium SL1344, S. aureus ATCC 29213, S. aureus ATCC 43300, and C. perfringens CVCC 2030) and 11 clinical isolates (E. coli EC024, S. Enteritidis SE006, S. aureus S21, C. perfringens CP023, C. perfringens CP002, S. suis SC19, S. suis SS006, P. multocida PM002, P. multocida PM008, A. pleuropneumoniae APP015, and A. pleuropneumoniae APP017). All plates were cultured at 37°C for 16 hr before observing the inhibition zone, and the diameter of the inhibition zone was measured using a vernier caliper. The presence of a clear zone indicated antagonistic activity.
In vitro bacterial competition
Request a detailed protocolTo investigate whether HBXN2020 directly inhibited the growth of STm, we performed spot-on lawn and agar well-diffusion assays, as well as co-culture assays in liquid culture medium. In the spot-on lawn antimicrobial assays, we prepared double layers of agar by first pouring LB agar into the plate as the bottom layer. The top layer consisted of 10 mL TSB broth containing 0.7% agar with STm overnight culture. Then, 10 μL of HBXN2020 overnight culture and cell-free supernatant (CFS) were respectively spotted onto TSB agar and incubated at 37°C for 12 hr to measure the inhibition zone. A transparent zone of at least 1 mm around the spot was considered positive.
The antagonistic effect of HBXN2020-CFS against STm was determined using the agar well-diffusion assays. To collect HBXN2020-CFS, the culture was centrifuged at 9000×g for 15 min at 4°C, and the supernatant was filtered through a 0.22 μm membrane filter (Millipore, USA). The STm lawn medium was prepared by mixing 10 mL of TSB broth containing 0.7% agar with STm overnight culture and then poured into a sterile plate covered with LB agar and Oxford cups, 8 mm diameter wells were prepared in the TSB agar after removing the cups. The wells were filled with 100 μL of HBXN2020-CFS, LB medium, or ampicillin (100 µg/mL). The plates were incubated at 37°C for 14 hr, and the inhibition zone was measured.
The co-culture assay was conducted by incubating HBXN2020 and S. Typhimurium ATCC14028 (carry pET28a (+), kanamycin resistance) (Cao et al., 2019) separately overnight at 37°C and diluting them to 104 CFU/mL. Then, the two strains were mixed at different ratios (1:1, 1:10, 1:50, or 1:100) and co-cultured at 37°C with shaking (180 rpm). At predetermined time points, serial 10-fold dilutions were prepared for all samples and spread onto selective (kanamycin 50 µg/mL) LB agar plates and cultured at 37°C for 12 hr before bacterial counting. Viable colony counts ranged from 30 to 300 per plate.
Genome sequencing, annotation, and analysis
Request a detailed protocolThe complete genome of strain was sequenced using the Illumina HiSeq PE and PacBio RSII (SMRT) platforms (Shanghai Majorbio Bio-Pharm Technology Co., Ltd.). Briefly, the short reads from the Illumina HiSeq PE were assembled into contigs using SPAdenovo (http://soap.genomics.org.cn/). The long reads from the PacBio RSII and the Illumina contigs were then aligned using the miniasm and Racon tools in Unicycler (version 0.4.8, https://github.com/rrwick/Unicycler, copy archived at Wick, 2024) to generate long-read sequences. During the assembly process, sequence correction was performed using Pilon (version 1.22, https://github.com/broadinstitute/pilon/wiki/Standard-Output; https://github.com/broadinstitute/pilon copy archived at Walker, 2021). Lastly, a complete genome with seamless chromosomes was obtained.
The CDSs of the strain genome were predicted using Glimmer (version 3.02, http://ccb.jhu.edu/software/glimmer/index.shtml). The tRNA and rRNA genes were predicted using TRNAscan-SE (version 2.0, http://trna.ucsc.edu/software/) and Barrnap (version 0.8, https://github.com/tseemann/barrnap, copy archived at Seemann, 2019), respectively. The functional annotation of all CDSs was performed using various databases, including Swiss-Prot Database (https://web.expasy.org/docs/swiss-prot_guideline.html), Pfam Database (http://pfam.xfam.org/), EggNOG Database (http://eggnog.embl.de/), GO Database (http://www.geneontology.org/), and KEGG Database (http://www.genome.jp/kegg/). The circular map of the strain genome was generated using CGView (version 2, http://wishart.biology.ualberta.ca/cgview/) (Stothard and Wishart, 2005).
Comparative genomic analysis
Request a detailed protocolTo elucidate the phylogenetic relationships of the strain from a whole-genome perspective, a phylogenetic tree based on the whole genome was constructed using the Type (Strain) Genome Server (TYGS) online (https://ggdc.dsmz.de/) (Meier-Kolthoff et al., 2022). The ANI values of the strain genome to the reference strain genome were then calculated using the JSpeciesWS online service (Richter et al., 2016), and a heatmap was generated using TBtools (version 1.113) (Chen et al., 2020).
In vivo safety evaluation of B. velezensis HBXN2020
Request a detailed protocolFemale specific-pathogen-free (SPF) C57BL/6 mice aged 6–8 weeks were purchased from the Animal Experimental Center of Huazhong Agricultural University. All mice experiments were conducted in the standard SPF facility of Huazhong Agricultural University, with 12 hr of light and 12 hr of darkness at a temperature of 25°C and ad libitum access to food and water. The use of animals in this experiment was approved by the Animal Care and Ethics Committee of Huazhong Agricultural University (Ethics Approval Number: HZAUMO-2023-0089).
Safety assay of B. velezensis HBXN2020 referred to the method described by Zhou et al., 2022, with slight modifications. After a 7-day acclimation period (free access to water and food), mice were randomly divided into four treatment groups (n=5): low-dose B. velezensis HBXN2020 spores group (L-HBXN2020 group, 107 CFU/mouse), medium-dose B. velezensis HBXN2020 spores group (M-HBXN2020 group, 108 CFU/mouse), high-dose B. velezensis HBXN2020 spores group (H-HBXN2020 group, 109 CFU/mouse), and a control group. During the experimental period (15 days), mice were weighed and orally gavaged with their respective treatments once every 2 days. On day 15, all mice were euthanized, and blood, heart, liver, spleen, lung, kidney, ileum, cecum, and colon were collected. Blood samples were used for routine blood and biochemistry tests. Major organ tissues (heart, liver, spleen, lung, and kidney) and a 5 mm distal segment of the colon were used for histopathology. The remaining colonic tissues were rapidly frozen in liquid nitrogen and stored at −80°C for cytokine and tight junction protein expression analysis.
S. Typhimurium-infected mouse model
Request a detailed protocolAfter 7 days of acclimation (free access to water and food), mice were randomly divided into five treatment groups (n=6): control group, HBXN2020 group, STm+PBS group, STm+HBXN2020 group, and STm+CIP group. The S. Typhimurium-infected model was performed as previously described (Stecher et al., 2005), with slight modifications. On the first day of the experiment, all mice in the STm+PBS group, STm+HBXN2020 group, and STm+CIP group were orally inoculated with 200 μL (5×107 CFU/mouse) of STm. On days 1, 3, and 5 following STm infection, each mouse in the HBXN2020 group, STm+HBXN2020 group, and STm+CIP group received 200 μL (1×108 CFU/mouse) of B. velezensis HBXN2020 spores or ciprofloxacin (1 mg/mL) via gavage administration, respectively. In contrast, the control group and STm+PBS group received 200 μL of sterile PBS by oral gavage. Fecal samples were collected daily following STm infection and resuspended in sterile PBS. The number of STm in mice feces from both the STm+PBS, STm+HBXN2020, and STm+CIP groups was then determined by spreading a serial 10-fold dilution on selective LB agar plates containing 50 µg/mL kanamycin. Throughout the entire experiment, the body weight, stool consistency, and fecal occult blood of all mice were monitored daily. As shown in Supplementary file 3, DAI was calculated by the sum of the scores from three parameters (Praveschotinunt et al., 2019). On day 7 after STm infection, all mice were euthanized, their ileum, cecum, and colon were collected. The length of colon was measured, and a 5 mm distal segment of the colon was fixed in 4% paraformaldehyde for further histopathology. The remaining colon was then rapidly frozen in liquid nitrogen and stored at −80°C for cytokine and tight junction protein expression analysis. Then, the number of STm in the ileum, cecum, and colon was determined by spreading serial 10-fold dilutions on selective LB agar plates.
To investigate the prophylactic efficacy of B. velezensis HBXN2020 in ameliorating STm-infected, another independent experiment was conducted using 6-week-old female C57BL/6 mice (SPF). After a 7-day acclimation period, mice were randomly assigned to five groups (n=6): control group, HBXN2020 group, PBS +STm group, HBXN2020+STm group, and CIP+STm group. On days 1, 3, 5, and 7, each mouse in the HBXN2020 group, HBXN2020+STm group, and CIP+STm group received 200 μL (1×108 CFU/mouse) of B. velezensis HBXN2020 spores or ciprofloxacin (1 mg/mL) via gavage administration. Meanwhile, mice in the control group and PBS+STm group received 200 μL of sterile PBS by oral gavage. On day 7, all mice in the PBS+STm group, HBXN2020+STm group, and CIP+STm group were orally inoculated with 200 μL (5×107 CFU/mouse) of STm. Fecal samples were collected daily following STm infection from both groups and resuspended in sterile PBS. The number of STm in the feces of mice was then determined by spreading serial 10-fold dilutions on selective LB agar plates (50 µg/mL kanamycin). Throughout the entire experiment, the body weight and DAI scores of all mice were monitored daily. On day 12, all mice were euthanized, and the ileum, cecum, and colon were collected. The colon length was measured, and a 5 mm distal segment of the colon was fixed in 4% formalin for sectioning and staining. The remaining colon was stored at −80°C for future analysis. Lastly, the number of STm in the ileum, cecum, and colon was determined using a selective LB agar plate.
Determination of cytokines and tight junction protein expression in colon tissue
Request a detailed protocolTotal RNA was extracted from colon tissues using TRIpure reagent (Aidlab, China), and cDNA was obtained using HiScript III RT SuperMix for qPCR (+gDNA wiper) (Vazyme, China). RT-qPCR for each gene was performed in triplicate using qPCR SYBR Green Master Mix (Yeasen Biotechnology, Shanghai, China). The relative expression level of cytokine and tight junction protein genes was calculated using the 2−ΔΔCt method with β-actin and GAPDH as reference genes. The primer sequences used in the RT-qPCR test are listed in Supplementary file 3.
Histopathology analysis
Request a detailed protocolColon tissue samples (0.5 cm) were fixed in 4% paraformaldehyde for 24 hr, and the fixed tissues were then embedded in paraffin and sectioned. The sections were stained with hematoxylin and eosin (H&E) and observed and imaged using an optical microscope (Olympus Optical, Tokyo, Japan). The histopathological score included the degree of inflammatory infiltration, changes in crypt structures, and the presence or absence of ulceration and edema. The scoring criteria were determined as previously described (Wu et al., 2022).
16S rRNA gene sequencing and analysis
Request a detailed protocolAccording to the manufacturer’s instructions, colon microbial community genomes were extracted using E.Z-N.A Stool DNA Kit (Omega; D4015-01), and quality was detected by 1% agarose gel electrophoresis. The 16S rRNA V3-V4 variable region was amplified by PCR using universal primers 338F (5 '- ACTCCTACGGGGGGCAG-3') and 806R (5 '- GACTACHVGGGTWTCTAAT-3'). The PCR products were examined by electrophoresis on 2% agarose gels and then purified with the AxyPrep DNA gel extraction kit (Axygen Biosciences, USA). A sequencing library was constructed using the NEXTFLEX Rapid DNA-Seq Kit, and sequencing was performed using the Illumina MiSeq platform. Raw reads were quality evaluated and filtered by fastp (version 0.20.0) and merged using FLASH (version 1.2.7). The optimized sequences were clustered into operational taxonomic units (OTUs) based on 97% sequence similarity using UPARSE (version 7.1). The representative sequences of each OTU was classified by RDP classifier (version 2.2; confidence threshold value, 0.7). Alpha diversity was assessed using the ACE, Chao, Shannon, and Simpson indices. The β diversity analysis was performed using Bray-Curtis distances and visualized through PCA. LefSe was used to identify differential microbiota between groups.
Statistical analysis
Request a detailed protocolStatistical analysis was performed using GraphPad Prism 8.3.0 (GraphPad Software, San Diego, CA, USA) and Excel (Microsoft, Redmond, WA, USA). Data are presented as mean ± standard error of the mean (SEM). Differences between two groups were evaluated using two-tailed unpaired Student’s t-test, and all other comparisons were conducted using one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test. For all analyses, significance differences are denoted as: *, p<0.05, **, p<0.01, and ***, p<0.001.
Data availability
Whole genome sequencing data of Bacillus velezensis have been deposited in NCBI GenBank under accession code CP119399.1.16s rRNA sequencing data of mice have been deposited in NCBI under the accession code PRJNA1150571.
-
NCBI GenBankID CP119399.1. Bacillus velezensis strain HBXN2020 chromosome, complete genome.
-
NCBI BioProjectID PRJNA1150571. Effect of probiotic Bacillus on the gut microbiota of mice.
References
-
Bacteriocinogenic potential of a probiotic strain Bacillus coagulans [BDU3] from NgariInternational Journal of Biological Macromolecules 79:800–806.https://doi.org/10.1016/j.ijbiomac.2015.06.005
-
Interaction of junctional adhesion molecule with the tight junction components ZO-1, cingulin, and occludinThe Journal of Biological Chemistry 275:20520–20526.https://doi.org/10.1074/jbc.M905251199
-
Akkermansia muciniphila: paradigm for next-generation beneficial microorganismsNature Reviews. Gastroenterology & Hepatology 19:625–637.https://doi.org/10.1038/s41575-022-00631-9
-
Polyphenols from blueberries modulate inflammation cytokines in LPS-induced RAW264.7 macrophagesInternational Journal of Biological Macromolecules 69:382–387.https://doi.org/10.1016/j.ijbiomac.2014.05.071
-
BookPerformance Standards for Antimicrobial Disk Susceptibility Tests, M02Clinical and Laboratory Standards Institute.
-
Pretreatment with recombinant murine tumor necrosis factor alpha/cachectin and murine interleukin 1 alpha protects mice from lethal bacterial infectionThe Journal of Experimental Medicine 169:2021–2027.https://doi.org/10.1084/jem.169.6.2021
-
Salmonella enterica serovar Typhimurium skills to succeed in the host: virulence and regulationClinical Microbiology Reviews 26:308–341.https://doi.org/10.1128/CMR.00066-12
-
Antimicrobial resistance: A global emerging threat to public health systemsCritical Reviews in Food Science and Nutrition 57:2857–2876.https://doi.org/10.1080/10408398.2015.1077192
-
Potential probiotic attributes of a new strain of Bacillus coagulans CGMCC 9951 isolated from healthy piglet fecesWorld Journal of Microbiology & Biotechnology 31:851–863.https://doi.org/10.1007/s11274-015-1838-x
-
Epidemiology and outcomes of nontyphoidal Salmonella bacteremias from EnglandJournal of Clinical Microbiology 57:18.https://doi.org/10.1128/JCM.01189-18
-
Cytokines: from clinical significance to quantificationAdvanced Science 8:e2004433.https://doi.org/10.1002/advs.202004433
-
Probiotic bacteria and intestinal epithelial barrier functionAmerican Journal of Physiology. Gastrointestinal and Liver Physiology 298:G807–G819.https://doi.org/10.1152/ajpgi.00243.2009
-
Regulation and functions of the IL-10 family of cytokines in inflammation and diseaseAnnual Review of Immunology 29:71–109.https://doi.org/10.1146/annurev-immunol-031210-101312
-
Safety assessment of probiotics for human useGut Microbes 1:164–185.https://doi.org/10.4161/gmic.1.3.12127
-
Biology and therapeutic potential of interleukin-10The Journal of Experimental Medicine 217:e20190418.https://doi.org/10.1084/jem.20190418
-
Annulment of bacterial antagonism improves plant beneficial activity of a bacillus velezensis consortiumApplied and Environmental Microbiology 88:e0024022.https://doi.org/10.1128/aem.00240-22
-
Endothelial cell fitness dictates the source of regenerating liver vasculatureThe Journal of Experimental Medicine 215:2497–2508.https://doi.org/10.1084/jem.20180008
-
The safety of probioticsClinical Infectious Diseases 46 Suppl 2:S104–S111.https://doi.org/10.1086/523331
-
Quercetin prevents ethanol-induced dyslipidemia and mitochondrial oxidative damageFood and Chemical Toxicology 50:1194–1200.https://doi.org/10.1016/j.fct.2012.02.008
-
Combination of probiotics with different functions alleviate DSS-induced colitis by regulating intestinal microbiota, IL-10, and barrier functionApplied Microbiology and Biotechnology 104:335–349.https://doi.org/10.1007/s00253-019-10259-6
-
The interplay between herbal medicines and gut microbiota in metabolic diseasesFrontiers in Pharmacology 14:1105405.https://doi.org/10.3389/fphar.2023.1105405
-
Population genetic structure and selection signature analysis of beijing black pigFrontiers in Genetics 13:860669.https://doi.org/10.3389/fgene.2022.860669
-
Probiotic Bacillus subtilis LF11 protects intestinal epithelium against Salmonella infectionFrontiers in Cellular and Infection Microbiology 12:837886.https://doi.org/10.3389/fcimb.2022.837886
-
Tight junctions: from simple barriers to multifunctional molecular gatesNature Reviews. Molecular Cell Biology 17:564–580.https://doi.org/10.1038/nrm.2016.80
Article and author information
Author details
Funding
National Program on Key Research Project of China (2021YFD1800300)
- Ping Qian
Yingzi Tech Huazhong Agricultural University Intelligent Research Institute of Food Health (IRIFH202209)
- Ping Qian
Fundamental Research Funds for the Central Universities (2662016PY004)
- Ping Qian
National Program on Key Research Project of China (2022YFD1800800)
- Ping Qian
Yingzi Tech Huazhong Agricultural University Intelligent Research Institute of Food Health (IRIFH202301)
- Ping Qian
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
This work was supported by grants from the National Program on Key Research Project of China (2021YFD1800300, 2022YFD1800800), 'Yingzi Tech & Huazhong Agricultural University Intelligent Research Institute of Food Health' (No. IRIFH202209; IRIFH202301), and the Fundamental Research Funds for the Central Universities (2662016PY004).
RT-qPCR data were acquired at the National Key Laboratory of Agricultural Microbiology Core Facility.
Ethics
The use of all animals in this experiment was approved by the Animal Care and Ethics Committee of Huazhong Agricultural University (Ethics Approval Number: HZAUMO-2023-0089).
Version history
- Preprint posted:
- Sent for peer review:
- Reviewed Preprint version 1:
- Reviewed Preprint version 2:
- Reviewed Preprint version 3:
- Version of Record published:
Cite all versions
You can cite all versions using the DOI https://doi.org/10.7554/eLife.93423. This DOI represents all versions, and will always resolve to the latest one.
Copyright
© 2024, Wang et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 1,349
- views
-
- 195
- downloads
-
- 8
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 1
- citation for umbrella DOI https://doi.org/10.7554/eLife.93423
-
- 7
- citations for Version of Record https://doi.org/10.7554/eLife.93423.4






