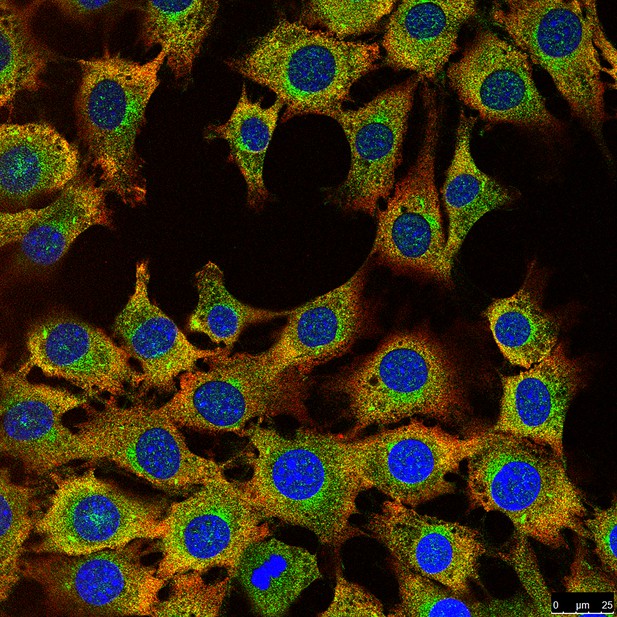
Microscopy image of fibroblast cells stained for DNA (blue), the PER2 protein (green) and phosphorylated PER2 (red). The PER2 protein can be detected in the nucleus alongside DNA, whereas phosphorylated PER2 can only be detected in the cytoplasm of the cells. Image credit: Andrea Brenna and Iwona Olejniczak (CC BY 4.0)
Anyone who has crossed multiple time zones on a long flight will be familiar with jet lag, and that feeling of wanting to sleep at lunchtime and eat in the middle of the night. Many bodily processes, including appetite and wakefulness, roughly follow a 24-hour cycle. These cycles are known as circadian rhythms, from the Latin ‘circa diem’ meaning about a day. An area of the brain called the suprachiasmatic nucleus (SCN) coordinates circadian rhythms. It acts as a master clock by generating a 24-hour signal for the rest of the body to follow. Jet lag occurs when this internal circadian rhythm becomes out of sync with the local day-night cycle.
Although jet lag can be uncomfortable, it tends to disappear over the course of a few days. This is because exposure to daylight in our new location resets the SCN master clock, enabling us to adapt to a new time zone. But evidence suggests that long-term disruption of circadian rhythms, for example as a result of shift work, may have lasting harmful effects. These include an increased risk of degenerative brain disorders such as Parkinson's disease and Alzheimer's disease.
Brenna et al. now identify a molecular mechanism that could explain this link. A key component of the SCN master clock is a protein called Period2 (PER2). Levels of PER2 rise and fall over each 24-hour period, helping the brain keep track of time. Brenna et al. show that PER2 interacts with CDK5, a protein that helps regulate brain development and that has been implicated in Parkinson's disease and Alzheimer's disease. Reducing CDK5 levels in mice shortened their circadian rhythms by several hours. It also altered the animals’ behavioral patterns over a 24-hour period. Deleting the gene for PER2 had a similar effect, suggesting that CDK5 helps regulate PER2.
Future studies should investigate the molecular links between CDK5, circadian rhythms and processes such as neurodegeneration. The results would provide clues to whether manipulating the circadian clock could help prevent or treat neurological disorders.




