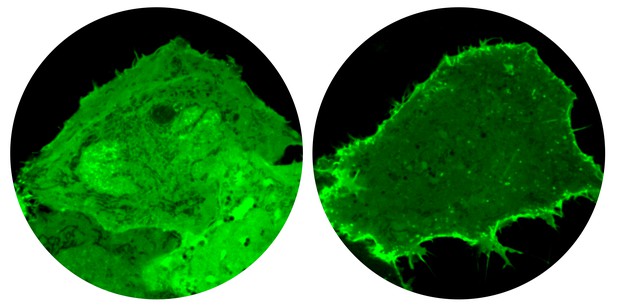
In human cells without GRAMD1 proteins (right), the pool of accessible cholesterol at the plasma membrane (green fluorescence) becomes greatly expanded. Image credit: Naito et al. (CC BY 4.0)
The human body contains trillions of cells. At the outer edge of each cell is the plasma membrane, which protects the cell from the external environment. This membrane is mostly made of fatty molecules known as lipids and about half of these lipids are specifically cholesterol. Human cells can either take up cholesterol that were obtained via the diet or produce it within a compartment of the cell called the endoplasmic reticulum.
Cells need to monitor the cholesterol levels in both the endoplasmic reticulum and the plasma membrane in order to regulate the uptake or production of this lipid. For example, if there is too much of cholesterol in the plasma membrane, then the cell transports some to the endoplasmic reticulum to tell it to shut down cholesterol production. However, how these different areas of the cell communicate with each other, and transport cholesterol, has remained unclear.
Naito et al. set out to look for key regulators of cholesterol transport and identified a group of endoplasmic reticulum proteins called GRAMD1 proteins. Cholesterol in the plasma membrane is either accessible or inaccessible, meaning it either can or cannot be moved back into the cell. The GRAMD1 proteins sense accessible cholesterol, and experiments with human cells grown in the laboratory showed that, specifically, the GRAMD1 proteins work together in a complex to sense accessible cholesterol at or near the plasma membrane. One particular part of the protein senses when the amount of accessible cholesterol reaches a certain level at the plasma membrane; when this threshold is reached, the complex flips a switch to start the transport of cholesterol to the endoplasmic reticulum and tell it to shut down cholesterol production.
This coupling of sensing and transporting lipids by one protein complex also helps maintain the right ratio of accessible and inaccessible cholesterol in the plasma membrane to prevent cells from activating unwanted cell-signaling events. Getting rid of the GRAMD1 proteins in cells, or removing sensing part of these proteins, leads to inefficient transport of cholesterol. A better understanding of how GRAMD1 proteins sense the accessibility of cholesterol could potentially help identify new approaches to control cholesterol transport inside cells. This may in turn eventually lead to new treatments that counteract the defects in cholesterol metabolism seen in some forms of neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease.




