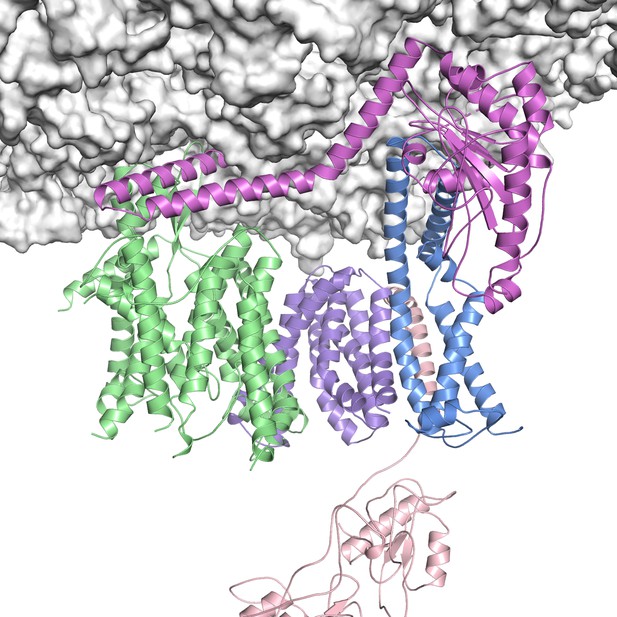
Cryo-EM structure of the TMCO1 translocon bound to the ribosome. Image credit: Robert Keenan and Phil McGilvray (CC BY 4.0)
Cell membranes are structures that separate the interior of the cell from its environment and determine the cell’s shape and the structure of its internal compartments. Nearly 25% of human genes encode transmembrane proteins that span the entire membrane from one side to the other, helping the membrane perform its roles.
Transmembrane proteins are synthesized by ribosomes – protein-making machines – that are on the surface of a cell compartment called the endoplasmic reticulum. As the new protein is made by the ribosome, it enters the endoplasmic reticulum membrane where it folds into the correct shape. This process is best understood for proteins that span the membrane once. Despite decades of work, however, much less is known about how multi-pass proteins that span the membrane multiple times are made.
A study from 2017 showed that a protein called TMCO1 is related to a group of proteins involved in making membrane proteins. TMCO1 has been linked to glaucoma, and mutations in it cause cerebrofaciothoracic dysplasia, a human disease characterized by severe intellectual disability, distinctive facial features, and bone abnormalities. McGilvray, Anghel et al. – including several of the researchers involved in the 2017 study – wanted to determine what TMCO1 does in the cell and begin to understand its role in human disease.
McGilvray, Anghel et al. discovered that TMCO1, together with other proteins, is part of a new ‘translocon’ – a group of proteins that transports proteins into the endoplasmic reticulum membrane. Using a combination of biochemical, genetic and structural techniques, McGilvray, Anghel et al. showed that the translocon interacts with ribosomes that are synthesizing multi-pass proteins. The experiments revealed that the translocon is required for the production of a multi-pass protein called EAAT1, and it provides multiple ways for proteins to be inserted into and folded within the membrane.
The findings of McGilvray, Anghel et al. reveal a previously unknown cellular machinery which may be involved in the production of hundreds of human multi-pass proteins. This work provides a framework for understanding how these proteins are correctly made in the membrane. Additionally, it suggests that human diseases caused by mutations in TMCO1 result from a defect in the production of multi-pass membrane proteins.




