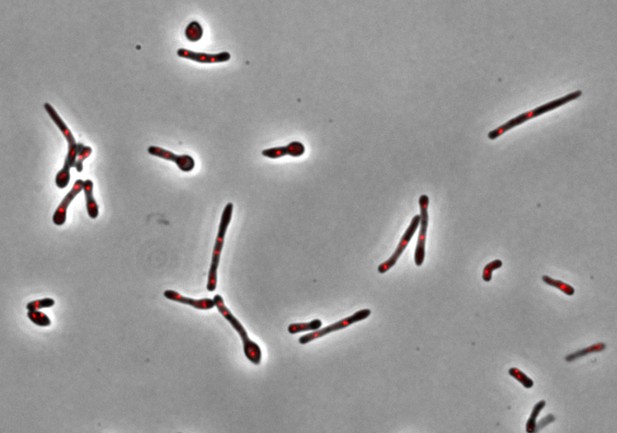
Mycobacterium smegmatis mutants in which expression of the essential gene, carA, is blocked by CRISPR interference. Image credit: Timothy de Wet (CC BY 4.0)
Caused by the microorganism Mycobacterium tuberculosis, tuberculosis kills more people around the world than any other infectious disease. M. tuberculosis is also becoming increasingly resistant to treatments, which are particularly difficult for patients to complete. The M. tuberculosis genome carries about four thousand genes, with several hundred being vital for survival. Finding new ways to fight tuberculosis relies on understanding the exact role of these essential genes, but they are difficult to study in living bacteria.
To investigate this question, de Wet et al. used the related, fast-dividing bacterial species called M. smegmatis as a model. Microscopic imaging was combined with CRISPR-interference – a method that temporarily disrupts expression of a specific gene – to examine how blocking an essential gene would affect the shape of the living microorganism.
Experiments were conducted on a collection of 270 mutants, capturing single-cell data for hundreds of thousands of live bacteria. To analyze the data, a computational pipeline was built, which automatically clustered similar-shaped bacteria. These groups, or ‘phenoprints’, brought together genes of known and unknown roles; this indicated that these genes participate in similar biological networks – and, if unknown, hinted at their function.
Finally, targeting essential genes with CRISPR-interference often yielded the same shape changes as blocking their encoded proteins with antibiotics. This suggests that phenoprints could be useful to understand the mode of action of potential new tuberculosis treatments. When applied to M. tuberculosis and other deadly bacteria, the approach developed by de Wet et al. might speed up drug development.




