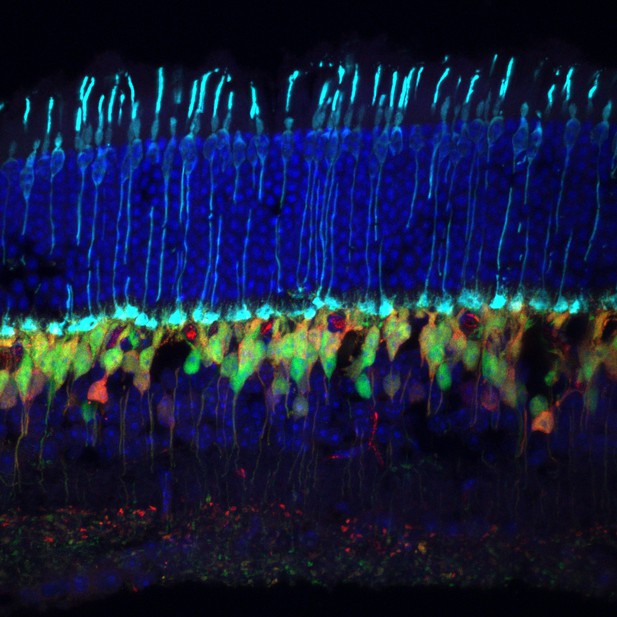
Image of the healthy mouse retina, with cones shown in cyan, bipolar cells shown in red and green, and cell nuclei are labelled dark blue. Image credit: Miranda Scalabrino.
Our sense of sight depends on the retina, a thin layer of cells at the back of each eye. Its job is to detect light using cells called photoreceptors, then send that information to the rest of the brain. The retina has two kinds of photoreceptors: rods (active in dim light) and cones (which detect colour and work in bright light). We rely heavily on cone cells for vision in our daily lives.
Retinitis pigmentosa is a progressive eye disease affecting photoreceptors. In the early stages of this disease, rods gradually die off. Next, cone cells start to die, inevitably resulting in blindness. There is currently no cure, although some experimental treatments are being developed that aim to prevent rod death or replace missing rod cells.
However, it is unclear if these therapies will be effective, because we do not fully understand how rod death affects cone cells – for example, whether or not it damages the cones irreversibly. Scalabrino et al. therefore set out to track how the signals that cones send to the brain changed over time during progression of the disease using genetically altered mice that reproduced the symptoms of retinitis pigmentosa.
In these mice, rod cells die off over several months, followed by complete loss of cones a few months later. Initial microscopy experiments looking at the shape and appearance of the cone cells revealed that the cones started looking abnormal long before all the rods died. Next, to determine if these unhealthy cones had stopped working, Scalabrino et al. measured the activity of the mice’s retinal ganglion cells (RGCs) in bright light – in other words, when cones are normally active.
RGCs transmit signals from photoreceptors to the brain, like a ‘telephone line’ between our brains and eyes. Applying a technique called information theory – which was originally used to determine how efficiently signals travel down telephone lines – to these experiments revealed that the RGCs still sent high-quality visual information from the cones to the brain. This is was surprising because the cones appeared to be dying and were surrounded by dead rods.
This study sheds new light on the biological processes underpinning a devastating eye disease. The results suggest that treatments to restore vision could work even if given after a patient’s cones start looking unhealthy, giving hope for the development of new therapies.




