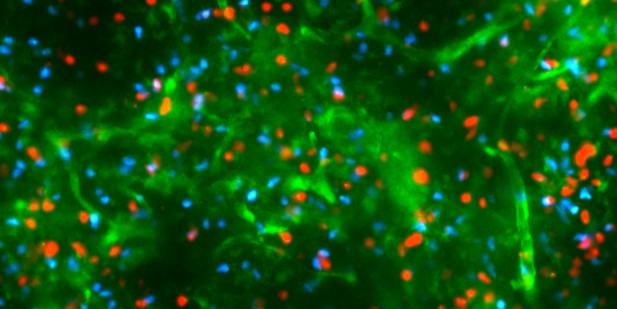
Immature T cells with (red) and without the CCR4 receptor (blue) migrating within the medulla of a mouse thymus (green). Image credit: Yu Li (CC BY 4.0).
Autoimmune diseases occur when immune cells mistakenly identify the body’s own tissues as ‘foreign’ and attack them. To reduce the risk of this happening, the body has multiple ways of removing self-reactive immune cells, including T cells. One such way, known as central tolerance, occurs in the thymus – the organ where T cells develop.
In the center of the thymus – the medulla – specialized cells display fragments of the majority of proteins expressed by healthy cells throughout the body. Developing T cells enter the medulla, where they scan these specialized cells to determine if they recognize the presented protein fragments. If an immature T cell recognizes and binds to these ‘self-antigens’ too strongly, it is either destroyed, or it develops into a regulatory cell, capable of actively suppressing T cell responses to that self-antigen. This ensures that T cells won’t attack healthy cells in the body that make those self-antigens, and therefore, it is important that T cells enter the medulla and carry out this scanning process efficiently.
T cells are recruited to the medulla from the outer region of the thymus by chemical signals called chemokines. These signals are recognized by chemokine receptors on T cells, which are expressed at different times during T cell development. Previous work has shown that one of these receptors, called CCR7, guides T cells to the medulla. Although it was thought that CCR7 was solely responsible for this migration, prior work suggests another receptor, CCR4, may also contribute to T cell migration into the medulla and central tolerance.
To determine whether CCR7 and CCR4 play the same or different roles in central tolerance, Li, Tipan et al. used a combination of experimental methods, including live imaging of the thymus, to study T cell development in mice. The experiments revealed that CCR4 is expressed first, and this receptor alone guides immature T cells into the medulla and ensures that they are the first to be checked for self-reactivity. In contrast, CCR7 is expressed by more mature developing T cells two to three days later, ensuring they also accumulate within the medulla and become tolerant to self-antigens. Both receptors are required for protection from autoimmunity, with results suggesting that CCR4 and CCR7 promote tolerance against different tissues.
Taken together, the findings provide new information about the distinct requirement for CCR4 and CCR7 in guiding immature T cells into the medulla and ensuring central tolerance to diverse tissues. One outstanding question is whether defects in T cells entering the medulla earlier or later alter tolerance to distinct self-antigens and lead to different autoimmune diseases. Future work will also investigate whether these observations hold true in humans, potentially leading to therapies for autoimmune diseases.




