RecA filament sliding on DNA facilitates homology search
Figures
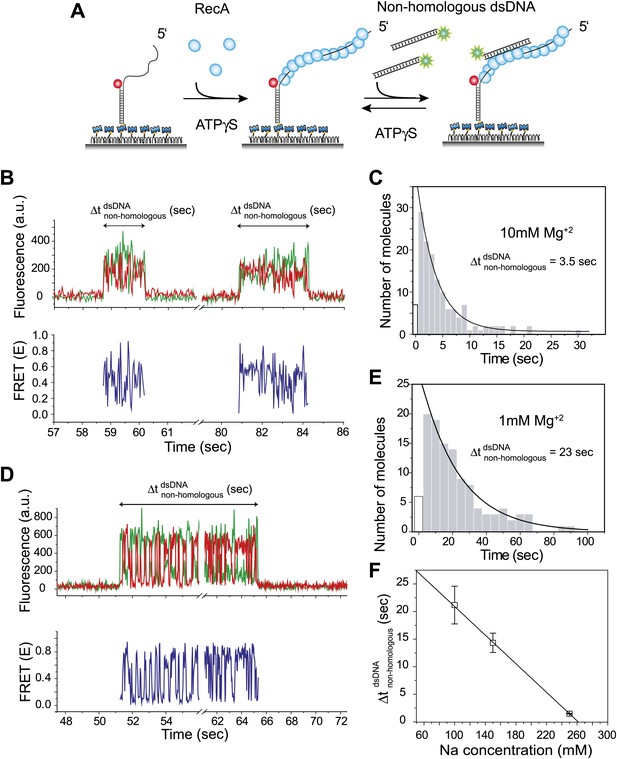
Dynamic interactions between RecA filament and non-homologous dsDNA.
(A) A schematic of the single molecule FRET based assay to detect interactions between RecA filament and non-homologous dsDNA. After RecA filament formation on ssDNA (Lfilament = 39 nt) labeled with an acceptor (red), a non-homologous dsDNA (LdsDNA = 39 bp) labeled with a donor was added. DNA docking results in appearance of donor (green) signal with FRET reporting on the changes in distance. (B) Single molecule time traces showing donor (green) and acceptor (red) intensities exhibits rapid FRET fluctuations with multiple binding and dissociation events within a single time trace (top panel). Corresponding FRET time traces (blue) are shown in the bottom panel (C) Histogram of the duration of the bound state for non-homologous dsDNA () and a single exponential decay fit. (D) Same as in (B), except that the Mg+2 concentration in solution was 1 mM. (E) Same as in (C), with 1 mM Mg+2 in solution. (F) Plot of Na+ concentration vs dwell time of dsDNA interaction with a RecA filament. Mg+2 concentrations in all cases was maintained at 1 mM. Error bars are standard errors of the mean obtained from single exponential decay fitting of dwell times. Linear fitting was used as a guide.
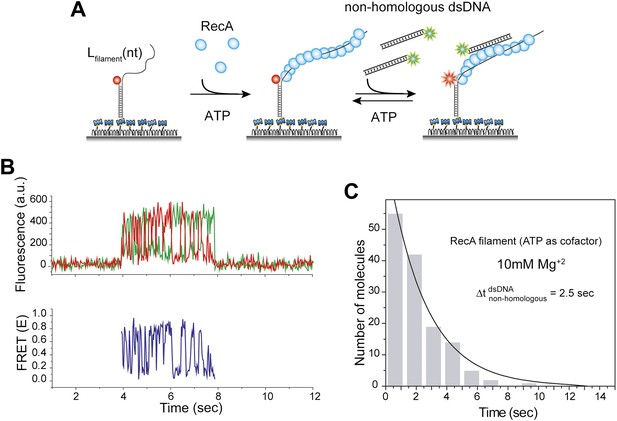
Non-homologous DNA interactions with the RecA filament are independent of ATP hydrolysis.
(A) A ssDNA (Lfilament = 39 nt) with an acceptor fluorophore (Cy5) at the junction was immobilized. After RecA filament formation in the presence of ATP as a co-factor, non-homologous dsDNA with a donor fluorophore (Cy3) was added along with free RecA protein (1 μM) and ATP (1 mM) to prevent filament dissociation. The Mg+2 concentration in solution was maintained at 10 mM. (B) Single molecule time trace showing donor (green) and acceptor (red) intensities (top panel) and the corresponding FRET values (bottom panel). (C) Histogram of the dwell time of DNA docking to the RecA filament and a single exponential decay fit.
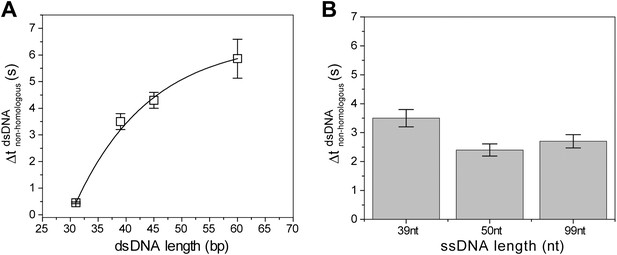
Non-homologous dsDNA interaction with RecA filament: dependence on filament length, Lfilament (nt) and dsDNA length, LdsDNA (bp).
(A) Off-time of non-homologous dsDNA binding vs ssDNA length. RecA filament formation was initiated on ssDNA of different lengths followed by addition of non-homologous dsDNA (LdsDNA = 39 bp). Error bars are standard errors obtained from single exponential decay fitting. (B) Off time of non-homologous dsDNA vs dsDNA length. RecA filament formation was initiated on ssDNA (Lfilament = 39 nt) followed by addition of non-homologous dsDNA (LdsDNA = 31, 39, 45 and 60 bp). Error bars are standard errors obtained from single exponential decay fitting. Dwell time for LdsDNA = 60 bp represents the mean lifetime of all binding events. Exponential fit was used as a guide.
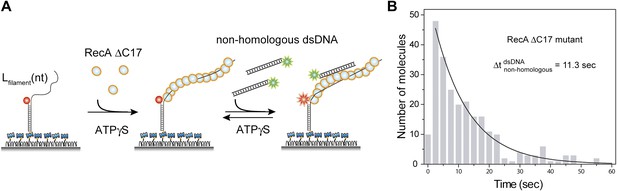
Deletion of acidic residues enhances RecA affinity for non-homologous dsDNA.
(A) A ssDNA (Lfilament = 39 nt) with an acceptor fluorophore (Cy5) at the junction was immobilized. After filament formation using a C terminal deletion mutant of RecA in the presence of ATPγS as a co-factor, non-homologous dsDNA with a donor fluorophore (Cy3) was added. (B) Histogram of the dwell time of DNA docking to the RecA filament and a single exponential decay fit.
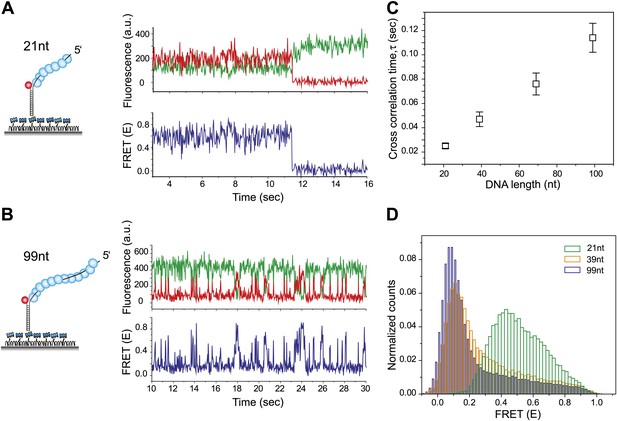
RecA filament slides along dsDNA.
(A) Single molecule traces showing donor (green) and acceptor (red) intensities (top panel) upon docking of non-homologous dsDNA (LdsDNA = 39 bp) to a RecA filament assembled on a ssDNA overhang, Lfilament = 21 nt. Corresponding FRET time traces (blue) are shown in the bottom panel. (B) Same as (A), except that the RecA filament is assembled on a ssDNA, Lfilament = 99 nt. (C) Average cross correlation time, vs Lfilament. Error bars are standard errors of the mean determined from three independent datasets. (D) FRET efficiency (E) histograms of single molecule traces for individual docking events of non-homologous dsDNA to RecA filaments assembled on ssDNA, Lfilament = 21 nt, 39 nt and 99 nt.
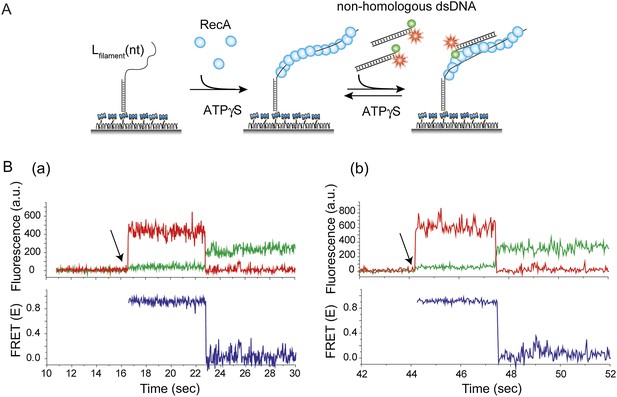
Thermal breathing of DNA ends does not contribute to the observed fluctuations in FRET.
(A) A ssDNA (Lfilament = 39 nt) with an no fluorophore was immobilized. After RecA filament formation in the presence of ATPγS as a co-factor, non-homologous dsDNA labeled with both a donor (Cy3) and acceptor (Cy5) fluorophores was added. The labeling scheme was such that the appearance of high FRET indicates the intact nature of the dsDNA. (B) (a) and (b) Single molecule time trace showing donor (green) and acceptor (red) intensities (top panel) and the corresponding FRET values (bottom panel). Arrows represent the time at which non-homologous dsDNA docking occurs. FRET remains high (approximately 0.9) until photobleaching of the acceptor.
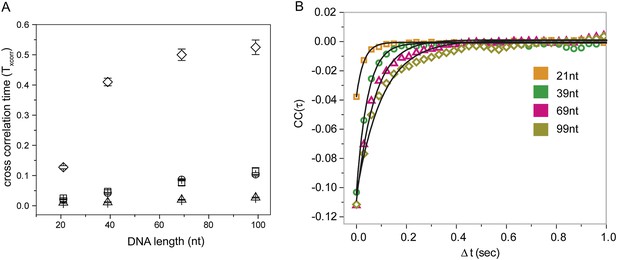
Cross correlation and corresponding Monte Carlo simulation of RecA filament sliding.
(A) Cross correlation curves and corresponding single exponential fits for the data as a function of R ecA filament length of the non-homologous dsDNA interaction with RecA filament changes as a function of filament length. (B) Monte Carlo simulations of RecA filament sliding were carried out as described in the ‘Materials and methods’ (□ experimental data same as Figure 2C), simulation with diffusion coefficient 0.9 × 10−3 μm2/s (○), simulation with diffusion coefficient 9 × 10−3 μm2/s (Δ) and simulation with diffusion coefficient 0.09 × 10−3 μm2/s (◊).
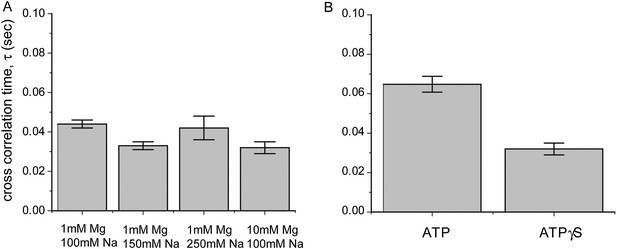
Dependence of sliding rate on solution conditions.
(A) Cross correlation time vs changes in sodium and magnesium concentrations. (B) Cross correlation time vs change in co-factor. The concentration of ATP and ATPγS in solution is 1 mM. Free RecA (1 μM) was maintained in solution for experiments involving the use of ATP.
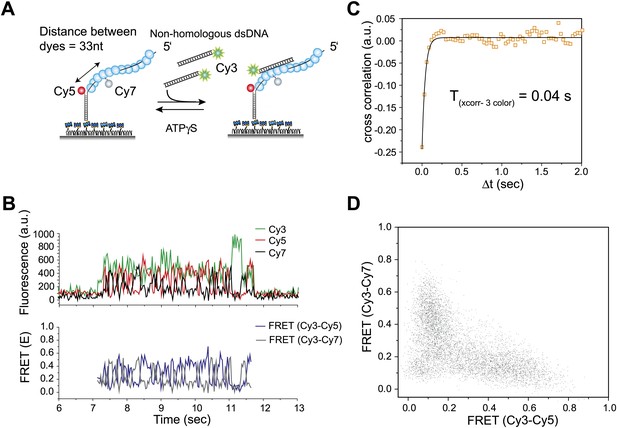
Three color FRET observations support RecA filament sliding.
(A) A schematic of the single molecule three color FRET assay to measure RecA filament sliding. ssDNA (Lfilament = 99 nt) labeled with two acceptor fluorophores (Cy5-red and Cy7-black) with a separation of 33 nt between the fluorophores, was immobilized on the surface. Upon docking of non-homologous donor (Cy3) labeled dsDNA to the pre-formed RecA filament formation, sliding predicts anticorrelated emissions between the two acceptors. (B) Single molecule time traces of Cy3 (green), Cy5 (red) and Cy7 (black) intensities (top panel). Corresponding FRET time traces of FRET between Cy3 and Cy5 (ECy3-Cy5-blue) and FRET between Cy3 and Cy7 (ECy3-Cy7-grey). (C) Normalized cross correlation plot of ECy3-Cy5 and ECy3-Cy7 averaged over 30 molecules and a single exponential fit of the data is overlaid (black). (D). Scatter plot of ECy3-Cy5 and ECy3-Cy7 for 30 molecules showing unique high FRET regions along both axes.
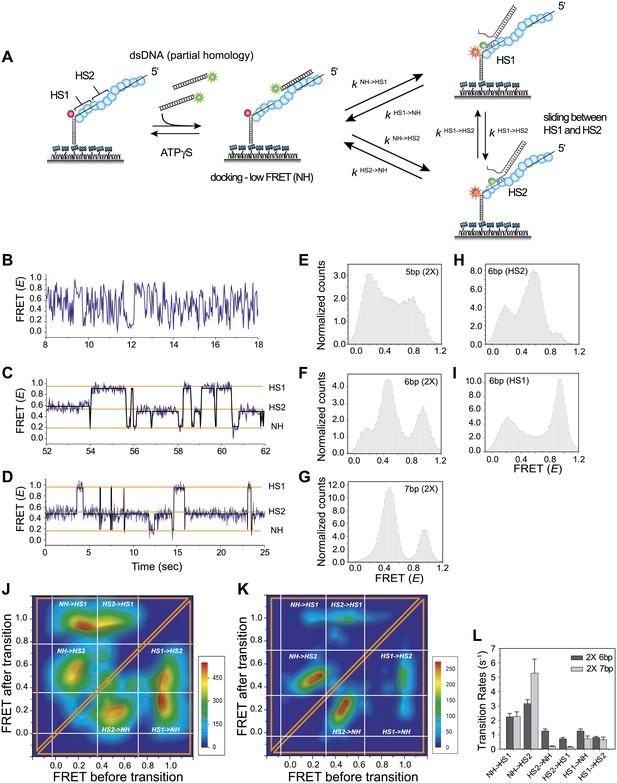
Homology recognition during sliding.
(A) A schematic of the single molecule FRET based assay to detect homology recognition between RecA filament and dsDNA. After RecA filament formation on ssDNA (Lfilament = 50 nt) labeled with an acceptor (red), a dsDNA (LdsDNA = 39 bp) labeled with a donor was added. Recognition of homology site 1 (HS1) or homology site 2 (HS2) results in the appearance distinct FRET states whose values depend on their relative distances from the acceptor. Docking at a location along the RecA filament outside a FRET sensitive regime results in low FRET (NH). (B) Single molecule time traces showing FRET for an immobilized ssDNA with two identical 5 nt homology sequences at HS1 and HS2 in a poly T sequence background. (C) Same as previous, with two identical 6 nt homology sequences at HS1 and HS2 in a poly T sequence background exhibits transitions between distinct FRET states. Idealized time trajectory obtained from HMM analysis is overlaid (black). (D) Same as previous, with two identical 7 nt homology sequences at HS1 and HS2 in a poly T sequence background. Idealized time trajectory obtained from HMM analysis is overlaid (black). (E) Normalized histograms of single molecule time traces exhibiting FRET for an immobilized ssDNA with two identical 5 nt homology sequences (HS1 and HS2). (F) Same as previous, with two identical 6 nt homology sequences (HS1 and HS2). (G) Same as previous, with two identical 7 nt homology sequences (HS1 and HS2). (H) Same as previous, with a single 6 nt homology site (HS2) at a position distal to the acceptor resulting in the appearance of a distinct mid FRET state. (I) Same as previous, with a single 6 nt homology site (HS1) in close proximity to the acceptor resulting in the appearance of a distinct high FRET state. (J) Transition density plot (TDP) of all FRET transitions for immobilized ssDNA with two 6 nt repeat sequences (HS1 and HS2) from 236 molecules exhibiting 13,020 transitions. (K) Transition density plot (TDP) of all FRET transitions for immobilized ssDNA with two 7 nt repeat sequences (HS1 and HS2) from 191 molecules exhibiting 3,819 transitions. (L) Forward and reverse transition rates between HS1, HS2 and NH states for Lh = 6 nt and 7 nt. Error bars denote standard errors of the mean for three measurements.
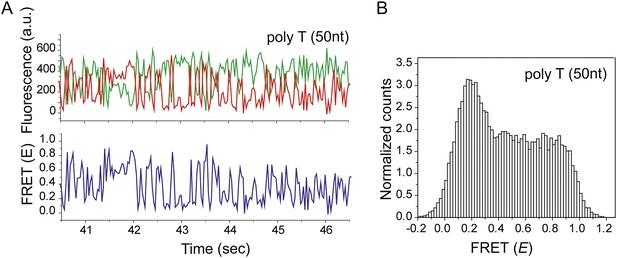
Non-homologous dsDNA interaction with a poly T (Lfilament = 50 nt) coated by RecA.
(A) A poly T ssDNA (Lfilament = 50 nt) with an acceptor fluorophore (Cy5) at the junction was immobilized. After RecA filament formation in the presence of ATPγS as a co-factor, non-homologous dsDNA with a donor fluorophore (Cy3) was added. Single molecule time trace showing donor (green) and acceptor (red) intensities (top panel) and the corresponding FRET values (bottom panel). (B) Histogram of the all single molecule time traces exhibiting FRET displays a broad distribution.
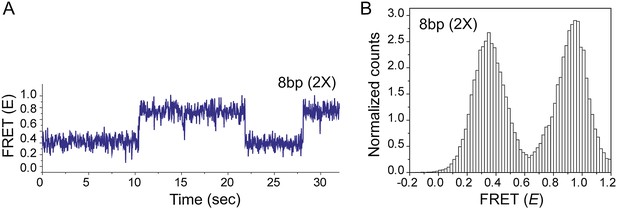
Effect of inserting two identical 8 bp homology sites (HS1 and HS2) in a poly T sequence background.
(A) Single molecule FRET time trace obtained following dsDNA docking to a RecA filament formed on a DNA strand containing two identical homology sites HS1 and HS2 with 8 nt homology at each site. Transitions between homology sites is rare due to stable binding of the target dsDNA to each homology site. (B) Histogram of single molecule time traces exhibiting FRET shows presence of distinct peaks which correspond to homology recognition and basepairing at HS1 and HS2.
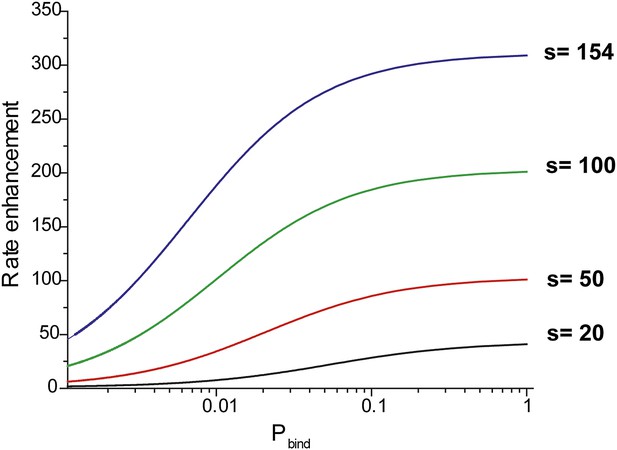
Effective rate enhancement of target search due to sliding.
Rate enhancement due to sliding vs probability of binding (pbind) to a target site for different values of the sliding distance (s). The rate enhancement of target search due to sliding is given by the ratio of the rate of association of RecA filament with the target site in the presence of sliding to the rate of association with the target site in the absence of sliding (D = 0). In this hypothetical model, RecA filaments can bind non-specifically to DNA but do not slide.
pbind is the probability of binding with the target site in each nonspecific encounter and is dependent on the microscopic rate constant associated with the recognition of a target site. and are the association rates with and without sliding respectively.
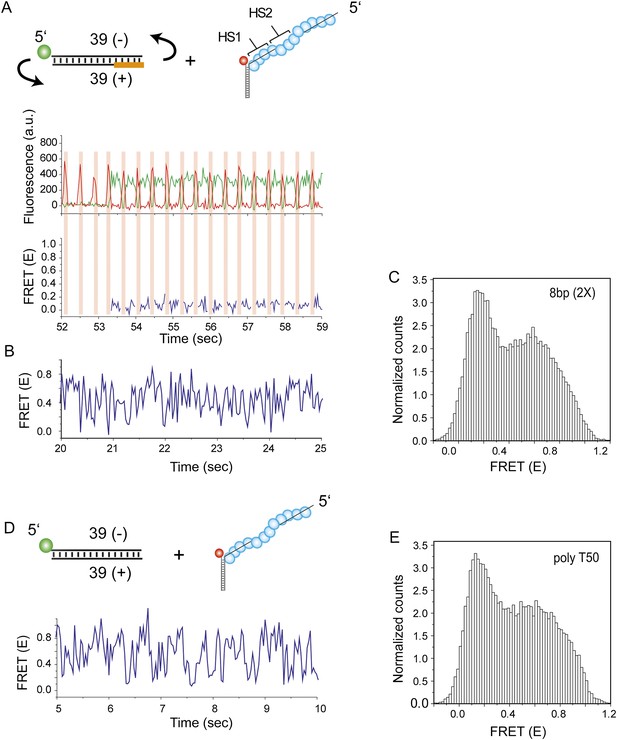
dsDNA flipping does not assist RecA mediated homology search.
To test if the dsDNA may flip its orientation relative to the RecA filament during homology search, we moved the donor fluorophore to the end opposite to the labeling position used for the experiments shown in Figure 4. Single molecule FRET time traces were obtained following dsDNA docking to a RecA filament formed on a DNA strand containing two identical homology sites with 8 nt homology at each site. (A) Orange highlight denotes the DNA segment which is complementary to HS1 and HS2. Homology recognition positions the unlabeled end of the dsDNA in close proximity to the acceptor dye resulting in stable low FRET. Time trace of fluorescence intensities (top panel) and corresponding FRET values (bottom panel) are shown. A pulse of red illumination (three frames, time resolution = 30 ms) with no green illumination was applied in order to verify the presence of a fluorescently active acceptor. Pulses of red illumination are indicated as red shaded portions in the single molecule time trace. (B) Binding in the opposite orientation results in lack of homology recognition leading to rapid FRET fluctuations which are not interrupted by stable low FRET states. If flipping was possible, homology recognition at the unlabeled end would result in the low FRET states interspersed with rapid FRET fluctuations. (C) Histogram of all single molecule time traces exhibiting FRET displays a broad distribution. (D) A poly T ssDNA (Lfilament = 50 nt) with an acceptor fluorophore (Cy5) at the junction was immobilized. Non-homologous dsDNA was labeled at the 5′end of the opposite strand. Single molecule FRET time exhibits rapid FRET fluctuations due to lack of homology recognition. (E) Histogram of single molecule time traces exhibiting FRET displays a broad distribution. The similarity in the time traces and FRET histograms shown in (B and C) and (D and E) suggest that the rapid FRET fluctuations with a broad distribution observed are due to dsDNA sliding on RecA filament without being perturbed by homology recognition events. Given that flipping does not occur, we conclude that a dsDNA that binds to a RecA filament in the wrong orientation cannot establish the correct orientation without full dissociation and rebinding.
Additional files
-
Supplementary file 1
DNA sequences used in measurements
- https://doi.org/10.7554/eLife.00067.017





