An essential and NSF independent role for α-SNAP in store-operated calcium entry
Figures
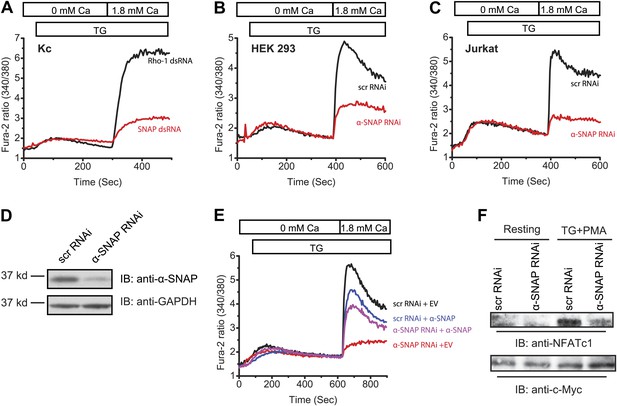
α-SNAP depletion inhibits SOCE and NFAT activation.
(A) Average Fura-2 ratios of Drosophila Kc cells treated with dsRNA targeting SNAP (red) or Rho-1 (black) for 3 days, and stimulated with 1 μM TG to measure SOCE using flexstation. (n > 3). (B and C) Average Fura-2 ratios of α-SNAP (red) or scramble (scr) (black) RNAi treated HEK 293 cells (B) or Jurkat T cells (C) stimulated with 1 μM TG to measure SOCE using flexstation. (n > 20) (D) A representative Western Blot for α-SNAP. WCLs of α-SNAP and scr RNAi treated HEK 293 cells were subjected to western blot analysis for each experiment using α-SNAP monoclonal antibody followed by anti-mouse secondary antibody. (E) Reconstitution of α-SNAP deficient cells with α-SNAP. Average Fura-2 ratios of RNAi treated HEK 293 cells stimulated with 1 μM TG to measure SOCE using flexstation, analyzed 3–4 days post RNAi transduction and 24 hr post-transfection with α-SNAP. (Black) cells transduced with scr RNAi and transfected with empty vector; (Red) cells transduced with α-SNAP RNAi and transfected with empty vector; (Blue) cells transduced with scr RNAi, transfected with α-SNAP; and (Purple) cells transduced with α-SNAP RNAi, transfected with α-SNAP. (n = 3) (F) Western blot for nuclear NFATc1 in RNAi treated Jurkat T cells. Nuclear extracts were prepared from RNAi treated Jurkat T cells, unstimulated, or stimulated with 1 μM TG and 10 ng/ml PMA and subjected to western blot using anti-NFATc1 antibody. c-Myc was used as a loading control. (n = 3)
Alignment of Drosophila and human SNAP amino acid sequences.
Purple (basic), Blue (acidic), Red (hydrophobic), Green (polar).
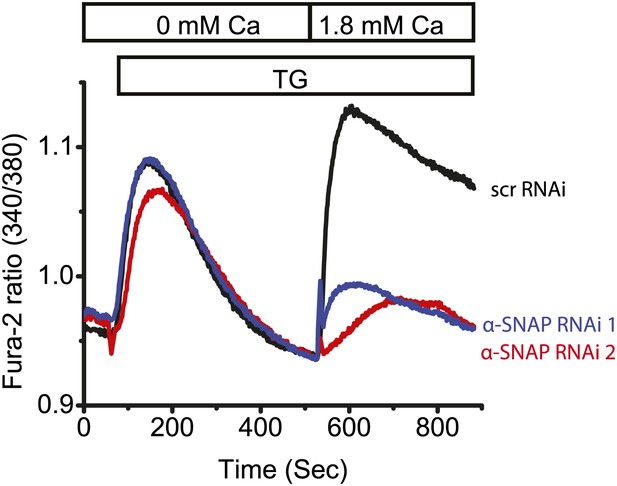
α-SNAP depletion using two different RNAi sequences in U2OS cells.
Average single cell Fura-2 ratios of U2OS cells treated with two different α-SNAP RNAi or scr RNAi and stimulated with 1 μM TG (n = 2).
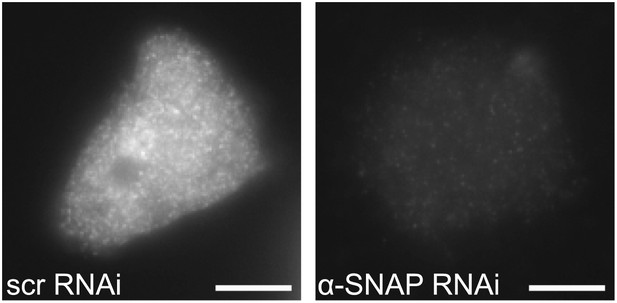
Immuno-staining for α-SNAP in α-SNAP depleted and control cells.
Epifluorescence images of α-SNAP and scr RNAi treated HEK 293 cells stained using α-SNAP monoclonal antibody followed by anti-mouse IgG secondary antibody. Scale bar 10 μm.
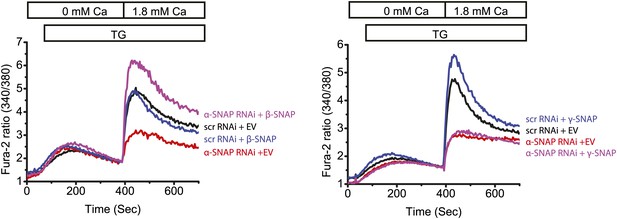
Reconstitution of α-SNAP deficient cells with β- or γ-SNAP.
Average Fura-2 ratios of RNAi treated HEK 293 cells stimulated with TG to measure SOCE (using flexstation), analyzed 3–4 days post RNAi transduction and 24 hr post-transfection with β- or γ-SNAP. (Black) cells transduced with scr RNAi and transfected with empty vector; (Red) cells transduced with α-SNAP RNAi and, transfected with empty vector; (Blue) scr RNAi, transfected with β-SNAP (Left Panel) or γ-SNAP (Right Panel); and (Purple) α-SNAP RNAi, transfected with β-SNAP (Left Panel) or γ-SNAP (Right Panel). (n = 2)
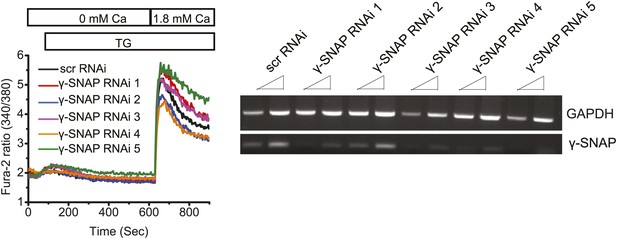
RNAi mediated depletion of γ-SNAP and measurement of SOCE.
(Left Panel) Average Fura-2 ratios of HEK 293 cells transduced with scr RNAi or five different RNAi targeting γ-SNAP, and stimulated with TG to measure SOCE (using flexstation). (Right Panel) Semi-quantitative PCR on total RNA to assess the level of γ-SNAP mRNA depletion compared to GAPDH in RNAi treated cells.
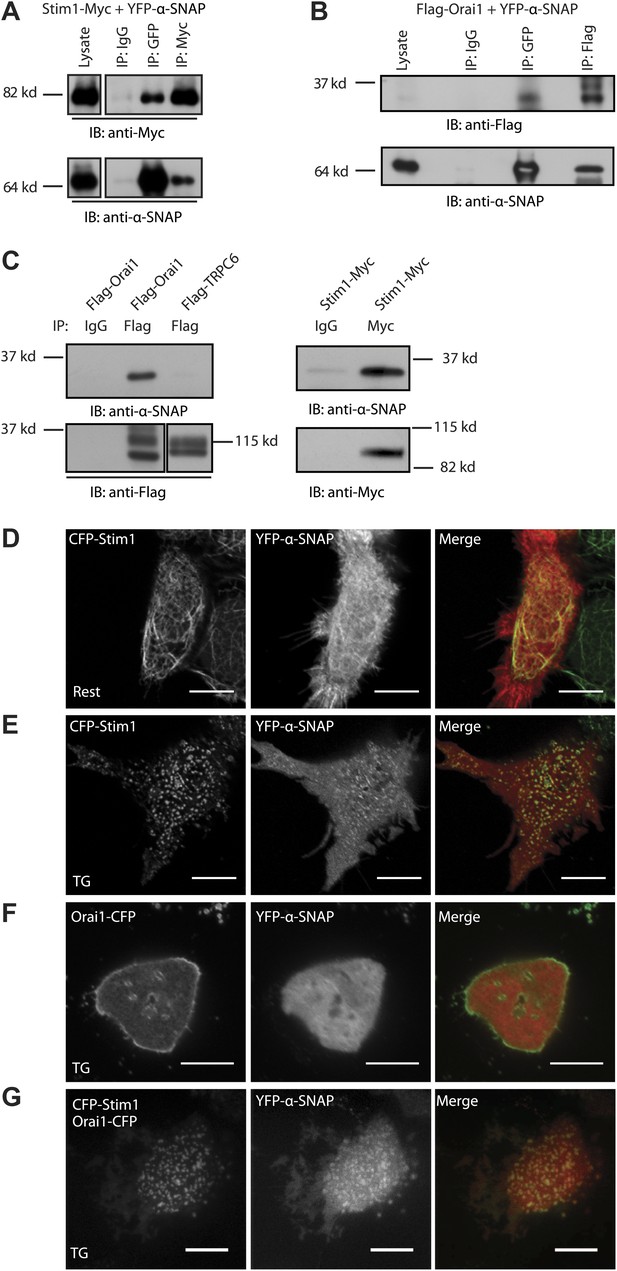
α-SNAP directly binds Stim1 and Orai1 and co-localizes with CRAC channel clusters.
(A and B) Co-immunoprecipitation of α-SNAP with Stim1 and Orai1. WCLs of store-depleted HEK 293 cells expressing Stim1-Myc and YFP-α-SNAP (A) or Flag-Orai1 and YFP-α-SNAP (B) were subjected to immunoprecipitation using anti-Myc, anti-Flag, or anti-GFP antibodies and Western Blot as indicated (n = 10). (C) In vitro binding of recombinant α-SNAP to full length Stim1 and Orai1. Flag-Orai1, Flag-TRPC6, and Stim1-Myc immunoprecipitates were incubated with purified α-SNAP protein (100 nM). Post-incubation, beads were washed, boiled, and subjected to western blot analysis using anti-α-SNAP antibody. (n > 10). (D, E and F) Representative confocal images of resting HEK 293 cells co-expressing YFP-α-SNAP and CFP-Stim1 (D) or store-depleted cells showing significant co-localization (E). Store-depleted HEK 293 cells co-expressing YFP-α-SNAP and Orai1-CFP (F). Scale bar 10 μm. (n = 3) (G) TIRF images of live, store-depleted HEK 293 cells, co-expressing CFP-Stim1, Orai1-CFP, and YFP-α-SNAP. Scale bar 10 μm. (n = 3)
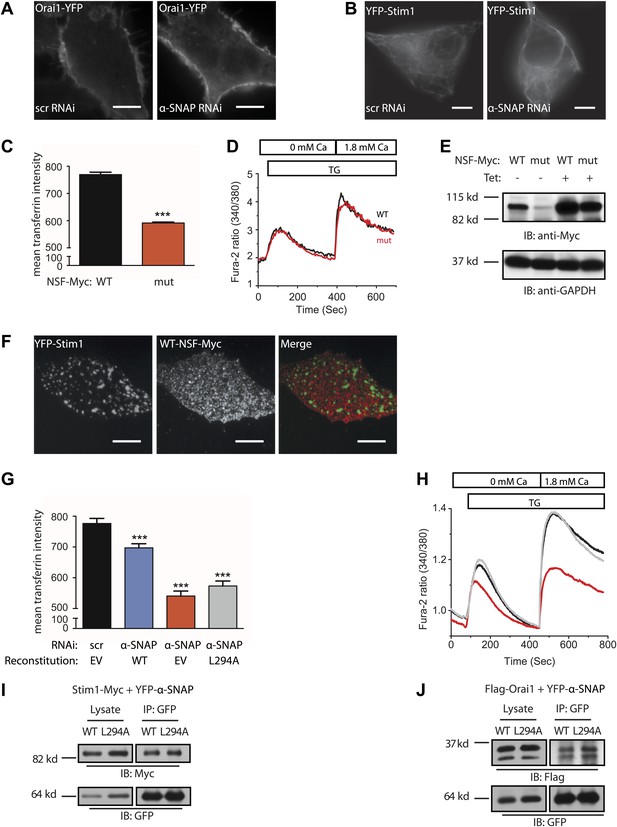
Regulation of SOCE by α-SNAP is NSF independent.
(A and B) Resting localization of Orai1 and Stim1 in control and α-SNAP depleted cells. Epifluorescence images of α-SNAP and scr RNAi treated, resting HEK 293 cells stably expressing Orai1-YFP (A) or YFP-Stim1 (B). Scale bar 10 μm. (n > 5) (C) Quantification of intracellular transferrin-alexa 555 fluorescence. U2OS T-REx cells stably transfected with inducible WT-NSF (black) or E329Q-NSF (mut) (red) were induced with doxycycline for 8 hr. Post induction, cells were incubated with transferrin-alexa 555, washed, and fixed. Alexa 555 fluorescence was quantified in cells using ImageJ. (n = 2 with ∼80–100 cells scored per group) ***p<0.001 (D) SOCE in live and adherent U2OS cells expressing WT-NSF (black) vs E329Q-NSF (mut) (red). Fura-2 ratios averaged from cells induced with doxycycline for 16–20 hr and stimulated with TG. (n = 3) (E) Western blot on WCLs of WT- or E329Q-NSF expressing adherent U2OS cells analyzed in panel D. (F) Localization of WT-NSF with respect to Stim1 clusters in store-depleted cells. TIRF images of Myc-tagged WT-NSF expressing U2OS T-REx cells transiently transfected with YFP-Stim1 and stimulated with TG prior to imaging. Scale bar 10 μm. (G) Quantification of intracellular transferrin-alexa 555 fluorescence in RNAi treated HEK 293 cells. Cells transduced with scr RNAi and reconstituted with YFP alone (EV) (black bars) or transduced with α-SNAP RNAi and reconstituted either with YFP-alone (EV) (red), with YFP-tagged WT α-SNAP (blue) or with YFP-tagged α-SNAP-L294A mutant (gray). Post RNAi transduction and reconstitution, cells were incubated with transferrin-alexa 555, washed, and fixed. Alexa 555 fluorescence was quantified in YFP positive cells using ImageJ. (n = 2, with each experiment scoring ∼100 cells) ***p<0.001 (H) Average single cell Fura-2 ratios of scr RNAi treated (black) or α-SNAP RNAi treated HEK 293 cells reconstituted with empty vector (red) or α-SNAP-L294A mutant (gray) showing SOCE in response to stimulation with TG. Cells were analyzed 3 days post RNAi transduction and 16 hr post-reconstitution. (n = 2) (I and J) Co-immunoprecipitation of Stim1 (I) and Orai1 (J) with WT α-SNAP or α-SNAP-L294A. WCLs of store-depleted HEK 293 cells co-expressing Stim1-Myc with YFP-α-SNAP or Stim1-Myc with YFP-α-SNAP- L294A (I) and Flag-Orai1 with YFP-α-SNAP or Flag-Orai1 with YFP-α-SNAP-L294A (J) were subjected to immunoprecipitation using anti-GFP antibodies and Western blot as indicated.
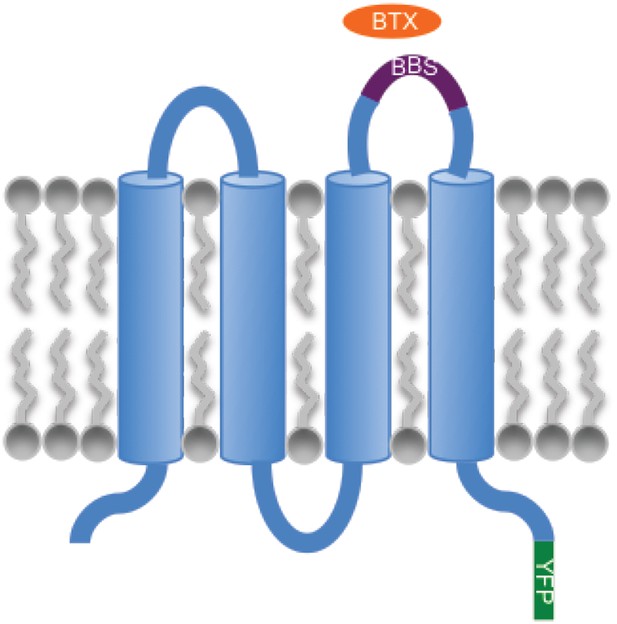
Strategy for quantifying Orai1 levels in the plasma membrane.
Schematic showing the site of insertion of α-bungarotoxin (BTX) binding site (BBS) in the second extracellular loop of Orai1 and the C-terminal YFP tag.
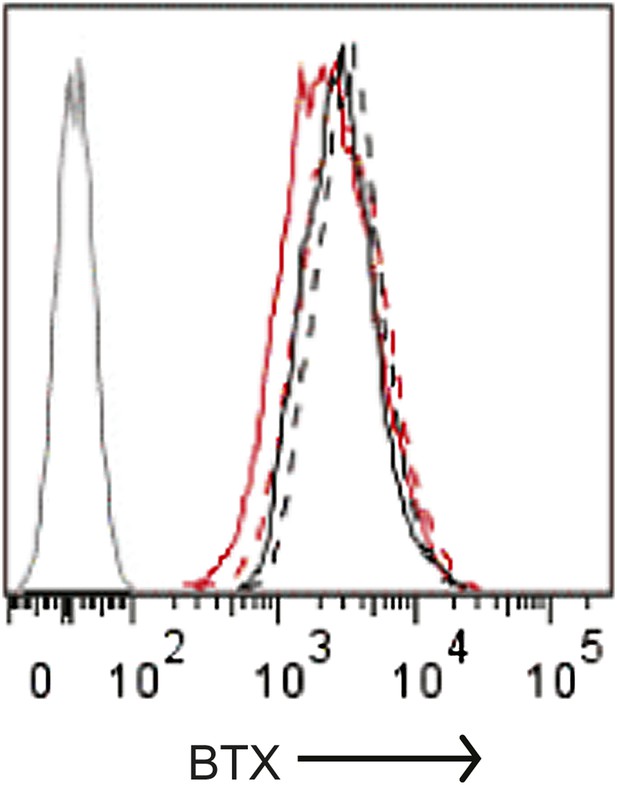
Quantification of cell surface Orai1 in α-SNAP depleted and control cells.
U2OS cells stably expressing Orai1-BBS-YFP were transduced with scr (black) or α-SNAP RNAi (red), stimulated with 0 (hashed line), or 1 μm TG (solid line), and incubated with α-bungarotoxin alexa 647 (BTX). Shown here is BTX binding on YFP-positive cells measured using FACS caliber. BTX binding to Orai1-YFP expressing cells was used as a control (gray line) (n > 3).
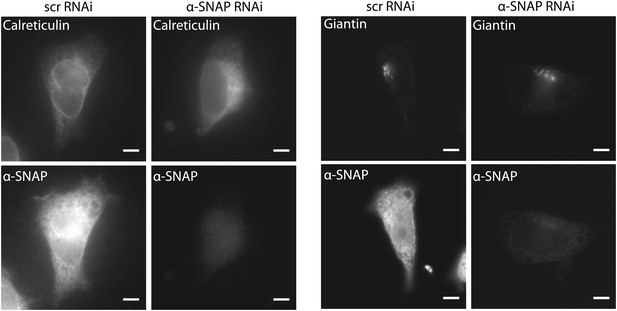
Morphology of ER and Golgi in control and α-SNAP depleted cells.
Epifluorescence images of α-SNAP or scr RNAi treated HEK 293 cells, stained with anti-calreticulin (Left Panel), or anti-giantin (Right Panel) primary antibodies followed by respective secondary antibodies. Scale bar 10 μm. (n = 3)
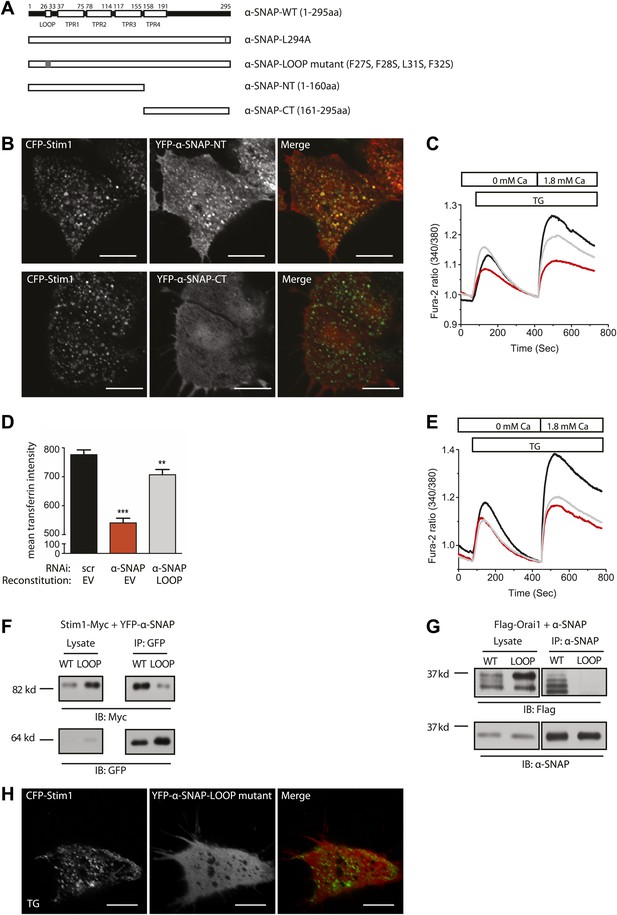
α-SNAP requires its hydrophobic loop for regulating SOCE.
(A) Schematic showing domains of interest in human α-SNAP protein sequence and strategy for the generation of α-SNAP mutants. (B) Representative confocal images of store-depleted CFP-Stim1 expressing HEK 293 cells; transiently transfected with YFP-α-SNAP-NT (1–160 aa) or YFP-α-SNAP-CT (161–295 aa) Scale bar 10 μm (n = 3). (C) Average single cell Fura-2 ratios of scr (black) or α-SNAP RNAi treated HEK 293 cells, reconstituted either with empty vector (red) or with α-SNAP-NT (gray), showing SOCE in response to TG. Cells were analyzed three days post RNAi transduction and 16 hr post-reconstitution (n = 2). (D) Quantification of intracellular transferrin-alexa 555 fluorescence. HEK 293 cells were transduced with scr RNAi and reconstituted with YFP alone (black bars) or with α-SNAP RNAi and either reconstituted with YFP-alone (red) or with YFP-tagged α-SNAP-LOOP mutant (gray). Post RNAi transduction and reconstitution cells were incubated with transferrin-alexa 555, washed, and fixed. Intracellular alexa 555 fluorescence was quantified in YFP positive cells using ImageJ. (n = 2, with each experiment scoring ∼100 cells) **p<0.01, ***p<0.001 (E) Average single cell Fura-2 ratios of scr (black) or α-SNAP RNAi treated HEK 293 cells reconstituted either with empty vector (red) or with α-SNAP LOOP mutant (gray), showing SOCE in response to TG. Cells were analyzed three days post RNAi transduction and 16 hr post-reconstitution. (n = 2) (F and G) Co-immunoprecipitation of Stim1 (F) and Orai1 (G) with WT-α-SNAP or α-SNAP-LOOP mutant. WCLs of store-depleted HEK 293 cells expressing Stim1-Myc and YFP-α-SNAP or YFP-α-SNAP-LOOP mutant (F) and HEK 293 cells expressing Flag-Orai1 and α-SNAP or α-SNAP-LOOP mutant (G) were subjected to immunoprecipitation using anti-GFP or anti-α-SNAP antibody and western blot as shown. (n = 2) (H) Representative confocal images of store-depleted CFP-Stim1 expressing HEK 293 cells; transiently transfected with YFP-α-SNAP-LOOP mutant. Scale bar 10 μm. (n = 3)
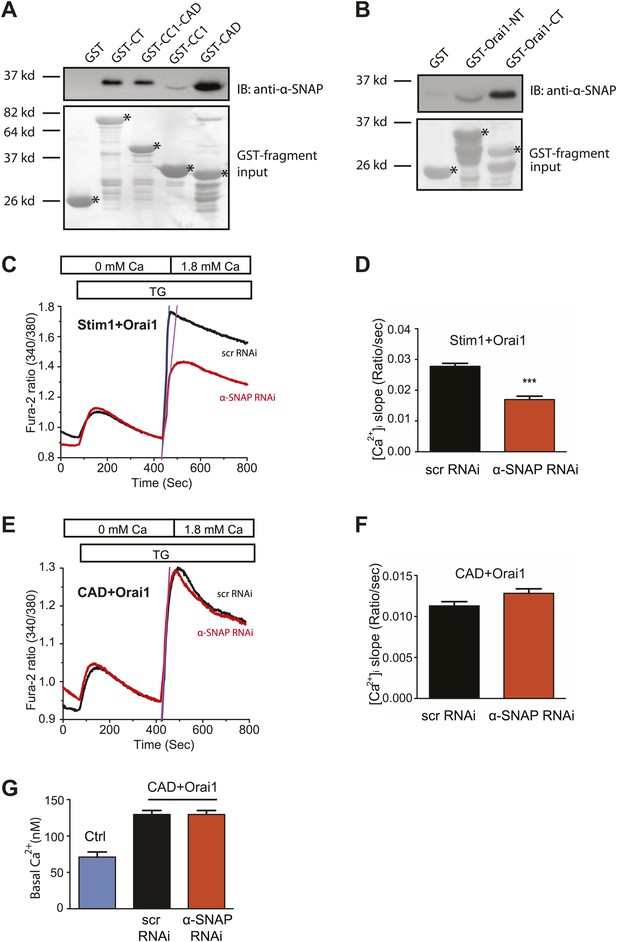
α-SNAP directly binds the CRAC activation domain of Stim1 and the C-terminal tail of Orai1.
(A) In vitro binding of α-SNAP to cytosolic domains of Stim1. (Top panel) GST-tagged Stim1 fragments, expressed in E.Coli and immobilized on resin, were incubated with purified α-SNAP protein (10 nM). Post-incubation, beads were washed, boiled, and subjected to western blot analysis using anti-α-SNAP antibody. (Bottom panel) Ponceau S staining showing the input of GST-tagged fragments and their expected sizes. (n = 3) (B) In vitro binding of α-SNAP to cytosolic tails of Orai1. (Top panel) GST-tagged Orai1 cytosolic tails, expressed in E.Coli and immobilized on resin, were incubated with purified 10 nM α-SNAP protein as described above and subjected to western blot analysis using anti-α-SNAP antibody. (Bottom panel) Ponceau S staining showing GST-tagged fragment input and their expected sizes. (n = 3) (C and E) SOCE in RNAi treated HEK 293 cells stably expressing Orai1 and STIM1 (C) or Orai1 and CAD (E). Average, single cell Fura-2 ratios of cells transduced with α-SNAP (red) or scr (black) RNAi and stimulated with TG. (n = 3) (D and F) Measurement of the rate of increase in intracellular Ca2+. The rate of store-operated Ca2+ influx in (C) and (E) was estimated by measuring the maximal rate of initial rise in Fura-2 ratios after replenishing Ca2+ in the extracellular buffer. (p value***<0.001) (G) Basal intracellular Ca2+ concentration in unstimulated HEK 293 cells (Ctrl) or RNAi treated HEK 293 cells stably expressing Orai1 and CAD.
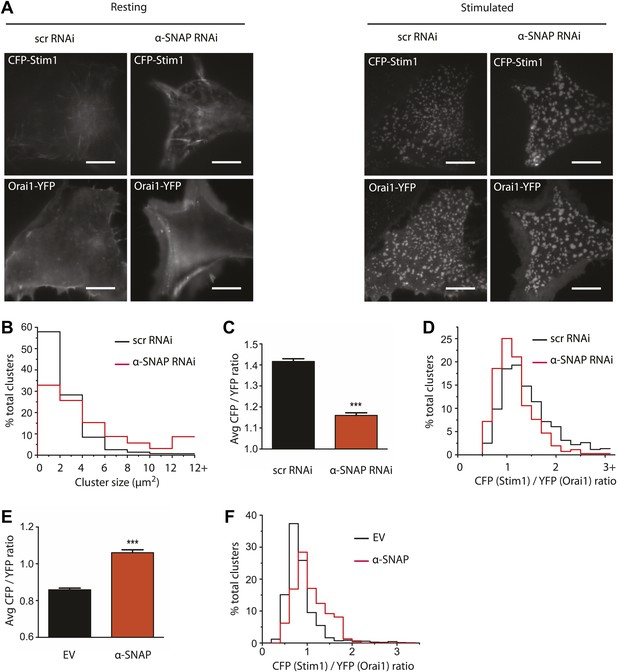
α-SNAP regulates the molecular composition of CRAC channel clusters in ER–PM junctions.
(A) Stim1 translocation and Orai1 clustering in control and α-SNAP deficient cells. TIRF images of resting and store-depleted, control, and α-SNAP deficient HEK 293 cells stably expressing CFP-Stim1 and Orai1-YFP. Scale bar 10 μm. (n > 5) (B) Quantification of Stim1-Orai1 cluster size in control and α-SNAP deficient cells. TIRF images were acquired as in (A) and boundaries for individual Stim1-Orai1 clusters were detected (as shown in Figure 6—figure supplement 1), pooled from nine cells and compared between control (black) and α-SNAP depleted cells (red). Histograms show the size comparison of ∼1000–1600 clusters. (C and D) Quantification of the relative intensities of CFP-Stim1 vs Orai1-YFP in CRAC channel clusters of RNAi treated cells. Stim1-Orai1 cluster boundaries were detected as described in (B) and average CFP vs YFP intensity of each cluster was calculated, pooled from nine cells and compared between control (black) and α-SNAP depleted cells (red). (C) Average CFP/YFP ratio of pooled clusters from RNAi treated cells and (D) CFP/YFP ratio of individual clusters from Figure 6C. (E and F) Independent quantification of relative intensities of CFP-Stim1 and Orai1-YFP in CRAC channel clusters over-expressing α-SNAP or empty vector. TIRF images were acquired from Orai1-Stim1 co-expressing HEK 293 cells transiently transfected with α-SNAP (red) or empty vector (black). Stim1-Orai1 cluster boundaries were detected and average intensity of CFP-Stim1 vs Orai1-YFP per cluster was calculated as described in Figure 6B–D above, pooled from six cells per group and compared. (E) Average CFP/YFP ratio of pooled clusters from α-SNAP (red) and empty vector (black) over-expressing cells. (F) CFP/YFP ratio of individual clusters from Figure 6E. (***p<0.001)
A representative Orai1-YFP image and its corresponding mask.
TIRF image was acquired as described in Figure 6, and Orai1-YFP cluster boundaries were automatically detected using a localized thresholding method in Matlab. Shown here is a representative image and its corresponding binary cluster mask.
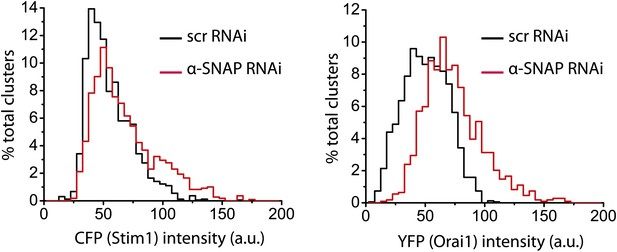
Quantification of CFP-Stim1 and Orai1-YFP intensities in Stim1-Orai1 clusters of RNAi treated cells.
Stim1–Orai1 cluster boundaries were detected as described in Figure 6—figure supplement 1 and Figure 6B. Average intensity of each CFP-Stim1 cluster (Left Panel) and Orai1-YFP cluster (Right Panel) was calculated, pooled from nine cells, and compared between control (black) and α-SNAP depleted cells (red).
α-SNAP depletion inhibits SOCE in Stim1-Orai1 over-expressing cells.
Average Fura-2 ratios of Orai1-Stim1 expressing HEK 293 cells transduced with α-SNAP RNAi (red) or scr RNAi (black) and stimulated with TG to measure SOCE. (n = 3)
α-SNAP co-expression augments SOCE in Stim1-Orai1 over-expressing cells.
Average Fura-2 ratios of Orai1-Stim1 expressing HEK 293 cells transiently transfected with α-SNAP (red) or empty vector as a control (black) and stimulated with TG to measure SOCE (n = 3).
Quantification of cell surface Orai1 in α-SNAP over-expressing cells.
Orai1-BBS-YFP expressing stable line was transiently transfected with α-SNAP (red) or empty vector (black). Subsequently, cells were stimulated with TG (solid lines) or left unstimulated (hashed lines) and incubated with BTX to estimate cell surface Orai1 as described in Figure 3—figure supplement 2. BTX binding to Orai1-YFP expressing cells was used as a control (gray line).
Quantification of Stim1-Orai1 cluster size in α-SNAP over-expressing cells.
CFP-Stim1 and Orai1-YFP expressing stable HEK 293 cells co-expressing α-SNAP (red) or empty vector control (black) from Figure 6E and 6F were quantified as in Figure 6B.
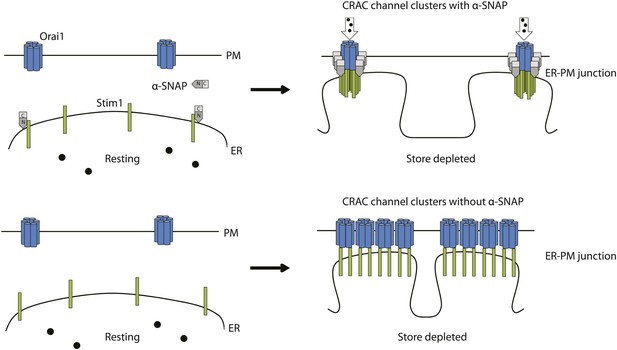
A hypothetical model of α-SNAP dependent re-arrangement of Stim1-Orai1 molecules within CRAC channel clusters in ER–PM junctions.
Calcium ions (black circles), PM localized Orai1 hexamers (light blue) ER localized Stim1 (green), α-SNAP (grey). (Top left panel) Resting localization of Orai1 hexamers and Stim1 in α-SNAP sufficient cells. (Top right panel) α-SNAP sufficient, store-depleted cells form functional CRAC channel clusters with relatively high Stim1/Orai1 ratio. (Bottom left panel) Resting localization of Orai1 hexamers and Stim1 in α-SNAP deficient cells is unaffected. (Bottom right panel) α-SNAP deficient, store-depleted cells form bigger non-functional Stim1–Orai1 clusters with low Stim1/Orai ratio.
Additional files
-
Supplementary file 1
dsRNA and shRNA sequences used in this study.
- https://doi.org/10.7554/eLife.00802.024





