An unmet actin requirement explains the mitotic inhibition of clathrin-mediated endocytosis
Figures
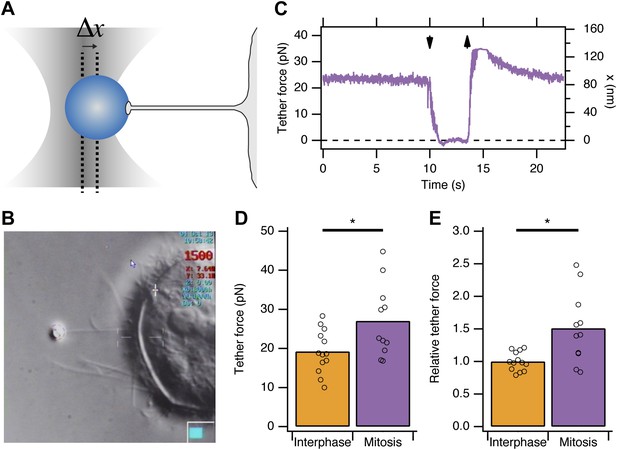
Membrane tension in mitotic cells is elevated.
(A) Schematic diagram to show the principle of using tether force to measure membrane tension. Displacement of the bead from the trap centre (Δx) can be converted to a tether force measurement (pN) as the stiffness of the trap is known. This tether force is proportional to the membrane tension. (B) Still video image of a membrane tension measurement in a mitotic cell. Note the thin tether is formed by moving the cell away from the bead held in the trap. (C) A typical trace showing bead displacement from the trap centre and the tether force. The cell was moved towards the bead (down arrow) to eliminate tension and give a zero value. The cell is then moved away (up arrow) and tether force is re-established. (D) Summary plot of tether force measurements from HeLa cells (dots) in interphase (orange) or at metaphase (purple). The mean is shown by a bar. (E) Plot of tether force relative to the average interphase measurement from the same experiment. Ncell = 13 or 11, Nexp = 5. *p<0.05.
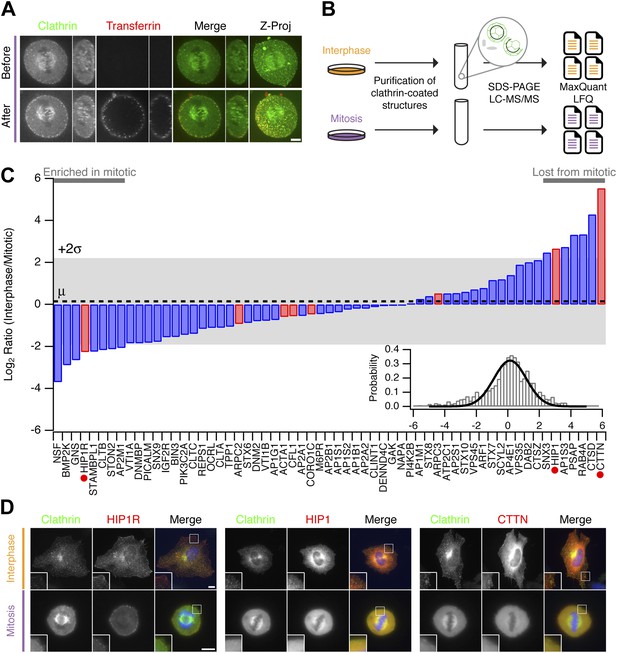
Comparative proteomics of fractions containing clathrin-coated membranes purified from interphase and mitotic HeLa cells.
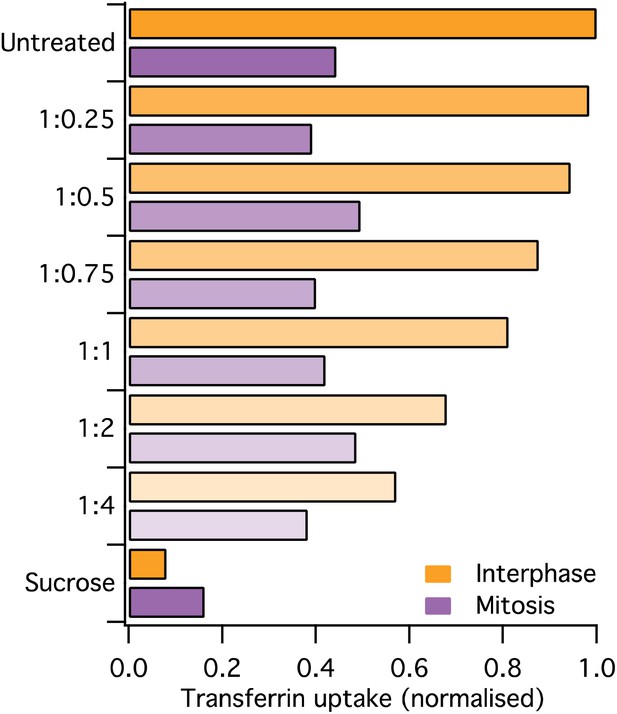
(A) Confocal micrographs of a HeLa cell expressing GFP-tagged clathrin light chain a (clathrin, green). The same cell is shown before and after application of transferrin-Alexa568 (Tf, red). A single XY section is shown together with a YZ view through the cell centre (right). Far right, 3D projection of the whole confocal series. (B) Schematic diagram of the purification and proteomic analysis of clathrin-coated structures purified from cells in interphase or metaphase. (C) Bar chart to show the comparative interphase/mitotic abundance for CCV proteins from the search list. The interphase or mitotic LFQ value for each protein derived from four separate experiments were compared. Red bars show proteins related to the actin cytoskeleton. Red circles indicate proteins verified in D. Inset: histogram to show the frequency of abundances for all proteins in the analysis. A single Gaussian function was fitted to the data, the mean and variance of which is shown in the main bar chart. Dotted line shows the average comparative abundance and the shaded area represents ± 2 standard deviations. (D) Representative fluorescence micrographs to show the colocalisation of GFP-tagged clathrin light chain a (clathrin, green) in interphase or mitosis, with HIP1R-tDimer-RFP, tdTomato-HIP1 or mCherry-cortactin (red). Scale bar, 10 µm.
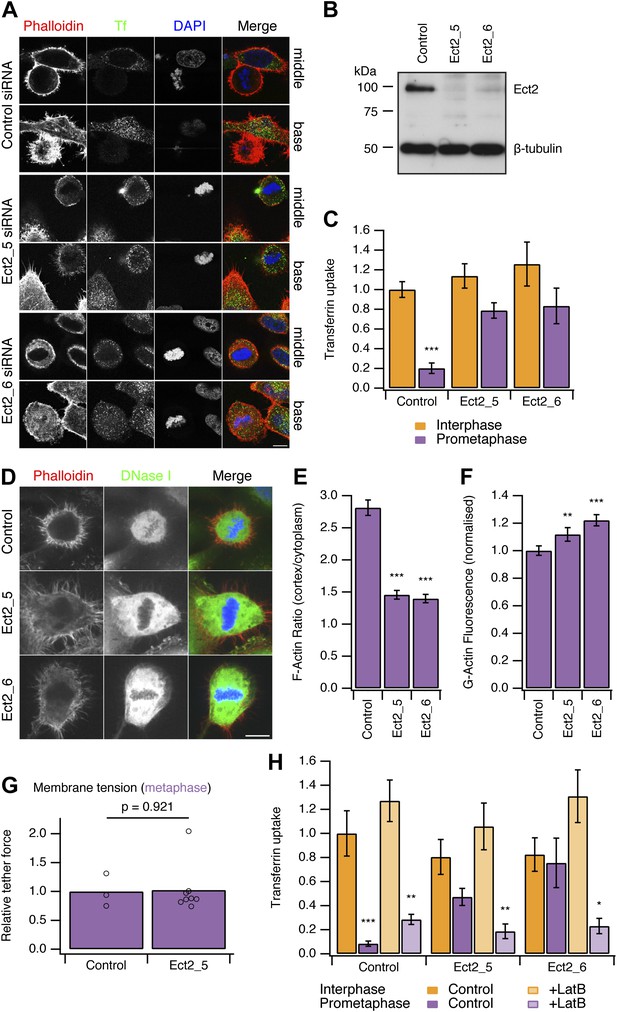
Restoring CME in mitotic cells by depletion of Ect2.
(A) Representative confocal micrographs to show transferrin uptake (Tf, green), actin organisation (phalloidin, red) and DNA (blue) in control or Ect2-depleted cells. For each condition, a section though the middle or base of the mitotic cell with a neighbouring interphase cell is shown. Cells were transfected with control siRNA or one of two Ect2 siRNAs (Ect2_5 or Ect2_6). Scale bars, 10 µm. (B) Representative western blot to show the amount of Ect2 remaining after RNAi. Blots were probed for Ect2 and β-tubulin as a loading control. (C) Bar chart to show normalised transferrin uptake in interphase and mitotic (prometaphase) cells as quantified by confocal microscopy. Ncell = 5–8. (D) Representative confocal micrographs to show the distribution of F-actin (Phalloidin, red), G-actin (DNase I, green) and DNA (blue) in mitotic HeLa cells. The cells were transfected with control (GL2) or Ect2 (Ect2_5 or Ect2_6) siRNA. Scale bar, 10 µm. (E and F) Bar charts to summarise the quantification of F-actin ratio at the cell cortex vs the cytoplasm (E) or the amount of G-actin in the cytoplasm (F). Ncell = 40, Nexp = 3. (G) Summary of membrane tension measurements on mitotic HeLa cells transfected with control (GL2) or Ect2 (Ect2_5) siRNA. Relative tether force is shown for individual cells (dots) and the bar indicates the mean. (H) Bar chart to show normalised transferrin uptake in interphase and mitotic (prometaphase) cells. Note the inhibition by latrunculin B (LatB, 1 µM 30 min) of restored CME in mitotic cells depleted of Ect2. Ncell = 7–14. All bars show mean ± SEM, *p<0.05; **p<0.01, ***p<0.001. One-way ANOVA with Tukey’s post-hoc test, comparison to control RNAi, interphase.
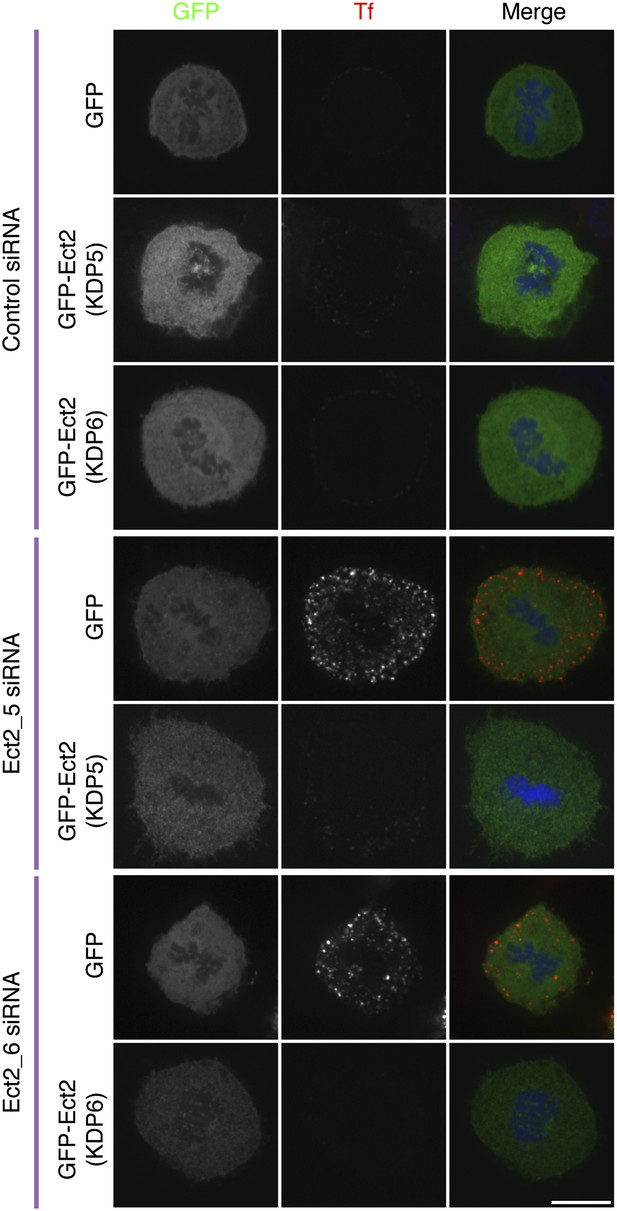
Ect2 RNAi rescue experiment to show that restoration of CME in Ect2-depleted cells is due to loss of Ect2.
Representative fluorescence micrographs of a transferrin uptake experiment in mitotic HeLa cells. The cells expressed GFP or siRNA-resistant forms of GFP-Ect2 and were treated with control (GL2) or Ect2 (Ect2_5 or Ect2_6) siRNAs as indicated. Transferrin uptake is shown in red, the GFP construct is shown in green and DNA in blue. Scale bar, 10 µm.
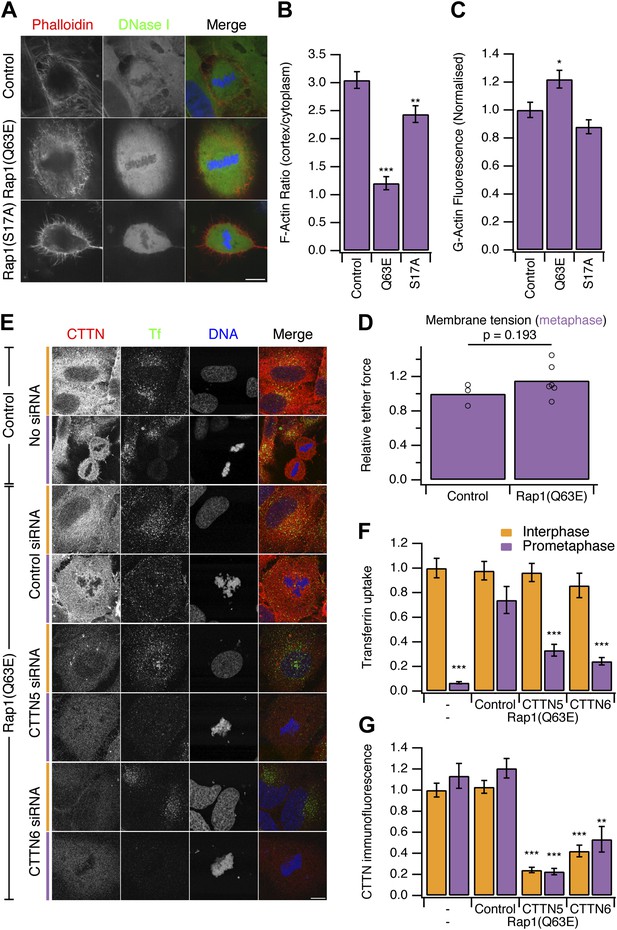
Restoring CME in mitotic cells by expression of Rap1(Q63E).
(A) Representative confocal micrographs to show the distribution of F-actin (Phalloidin, red), G-actin (DNase I, green), and DNA (blue) in mitotic HeLa cells. The cells either expressed no protein, the constitutively active Rap1(Q63E) or the inactive Rap1(S17A). Scale bar, 10 µm. (B and C) Bar charts to summarise the quantification of F-actin ratio at the cell cortex vs the cytoplasm (B) or the amount of G-actin in the cytoplasm (C). Ncell = 18. (D) Summary of membrane tension measurements on mitotic HeLa cells expressing Rap1(Q63E) compared to control. Relative tether force is shown for individual cells (dots) and the bar indicates the mean. (E) Representative confocal micrographs of interphase (orange) or mitotic (purple) HeLa cells to show transferrin uptake (Tf, green), cortactin immunofluorescence (CTTN, red) and DNA (blue). The cells were transfected as indicated to express Rap1(Q63E) and with control or either one of two CTTN siRNAs (CTTN_5 or CTTN_6). Scale bar, 10 µm. (F and G) Bar charts to show normalised transferrin uptake (F) or CTTN immunofluorescence (G) for each condition as indicated. Ncell = 10–29. All bar charts in the figure show mean ± SEM. All bars show mean ± SEM, *p<0.05; **p<0.01, ***p<0.001. One-way ANOVA with Tukey’s post-hoc test, comparison to control (B and C) or normal HeLa, interphase (F and G).
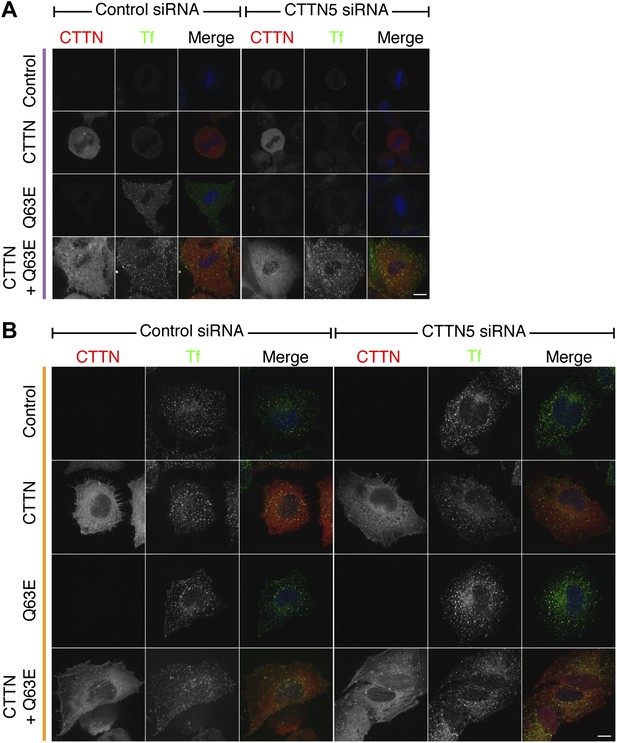
Cortactin RNAi prevents the restoration of CME by Rap1(Q63E) expression, and this effect is due to loss of cortactin.
Representative confocal images of a rescue experiment to test for specificity of the cortactin RNAi phenotype. Cells either expressed no additional protein, mCherry-cortactin, Rap1(Q63E) or both proteins; and were either treated with control (GL2) or cortactin (CTTN_5) siRNA. The mCherry-cortactin construct was from mouse and was resistant to CTTN_5, which targets the human transcript. Transferrin uptake (Tf, green), mCherry-cortactin (if present–CTTN, red), and DNA (blue). Cells were transfected as indicated to express Rap1(Q63E) and with control or CTTN siRNA. Micrographs of mitotic (purple, A) or interphase (orange, B) HeLa cells are shown. Note that there is no restoration of mitotic CME in control RNAi cells expressing mCherry-cortactin. This indicates that mitotic shutdown of CME is not simply due to loss of cortactin. Scale bars, 10 µm.
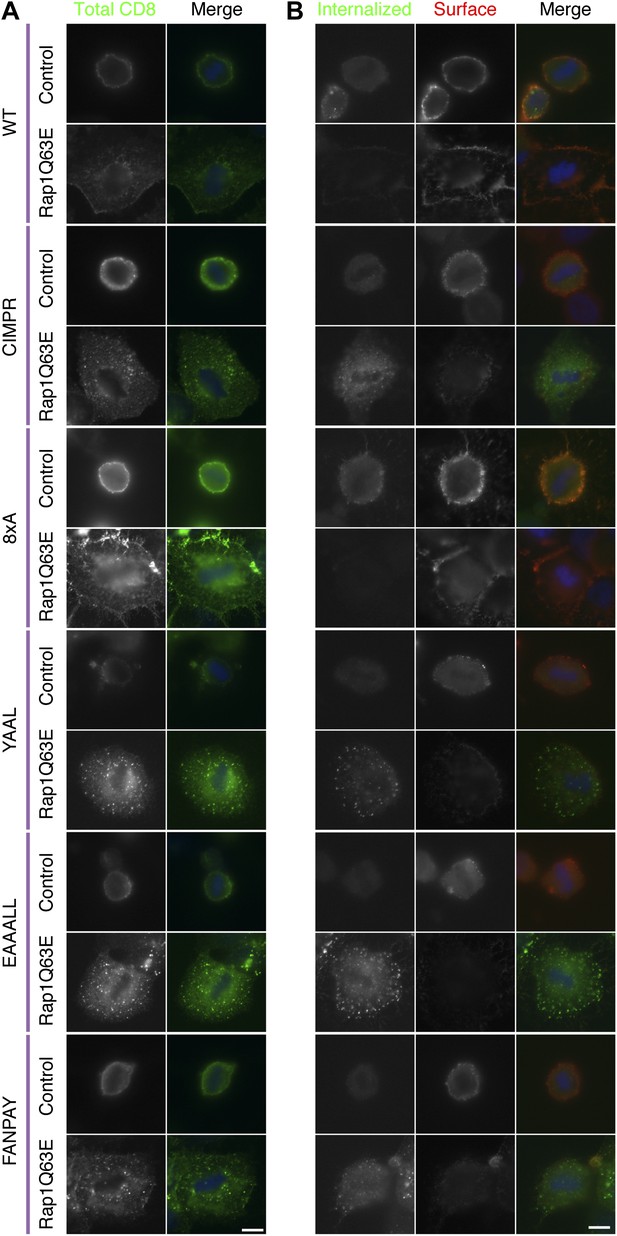
Internalisation of diverse cargo is restarted in mitotic Rap1(Q63E)-expressing cells.
(A) Representative confocal images of control and Rap1(Q63E)-expressing HeLa cells in mitosis that express the indicated CD8 chimera. The cells were fixed and stained for total CD8 to show the steady-state distribution of CD8 chimeras (green; DNA, blue). (B) Representative fluorescence micrographs of anti-CD8 antibody uptake experiments. Control and Rap1(Q63E)-expressing HeLa cells in mitosis are shown that express the indicated CD8 chimera. Uptake was performed as previously described (Fielding et al., 2012). Internalised CD8 is shown in green, and surface CD8 is shown in red, and DNA in blue. Scale bars, 10 µm.
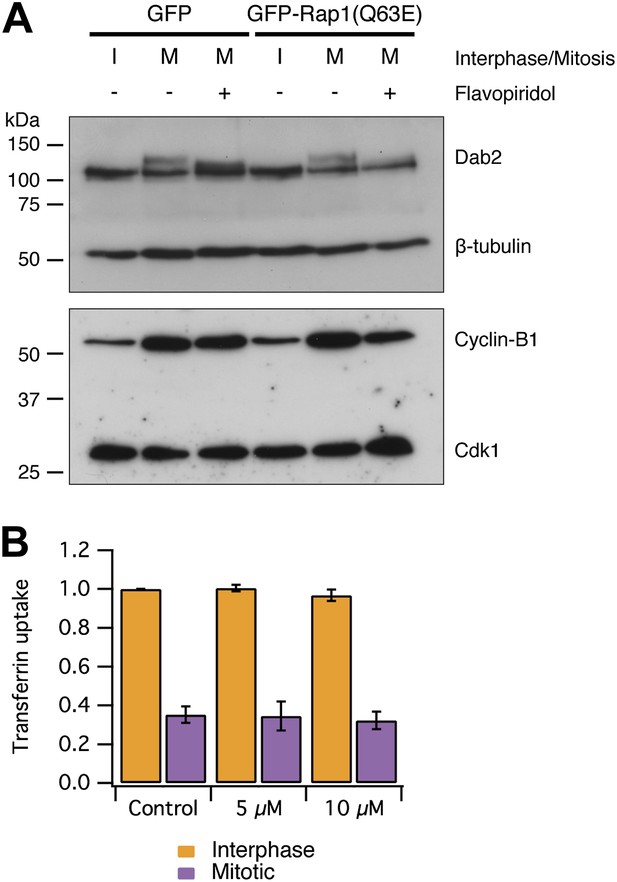
Mitotic phosphorylation does not explain mitotic shutdown of CME.
(A) Western blot to show Dab2 phosphorylation and inhibition by the Cdk1 inhibitor flavopiridol (5 µM, 10 min). Interphase or mitotic cell lysates prepared from cells expressing GFP or GFP-Rap(Q63E) to restart CME in mitotic cells. Blots were probed for Dab2 and β-tubulin as a loading control, or Cyclin-B1 and Cdk1. (B) Bar chart to show the average transferrin uptake in interphase (orange) or mitotic (purple) populations of cells by flow cytometry. The mean ± SEM of three separate experiments are shown.
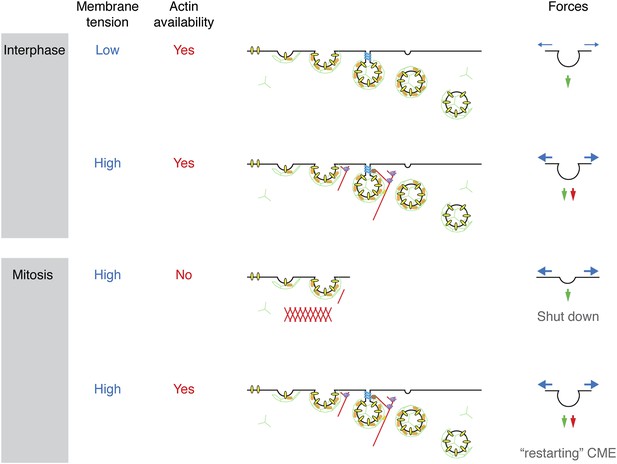
Model to show ‘two layer’ inhibition of CME during early mitosis.
In interphase, CME is continually active and can deploy actin to assist endocytosis if the membrane tension is increased. The forces of CME, actin and membrane tension are represented as green, red and blue arrows, respectively. In mitosis, the membrane tension is increased and actin is required to assist CME, but it is also unavailable due to the formation of the actin cortex. Increasing actin availability, either by Ect2-depletion or expression of Rap1(Q63E), can restore CME in mitotic cells. The membrane tension remains high, but actin is available to assist CME by providing additional force. If the actin cytoskeleton is considered to be a component of the endocytic machinery then its unavailability can be regarded as being due to a ‘moonlighting’ function in the formation of a rounded cortex (Royle, 2013).