Sex-specific triacylglycerides are widely conserved in Drosophila and mediate mating behavior
Figures
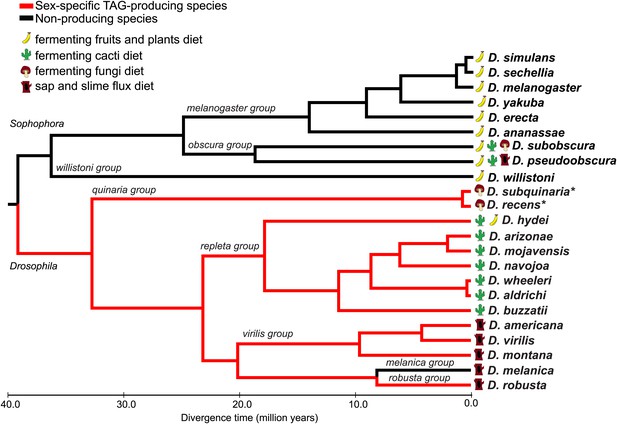
Male-specific TAG expression is broadly conserved across the Drosophila subgenus and not found in species from Sophophora.
The primary diets of each species are indicated, based on the previous studies. Branches for TAG-producing species are colored in red. Branch lengths are proportional to evolutionary time. *Evidence for TAG-expression is based on Curtis et al., 2013.
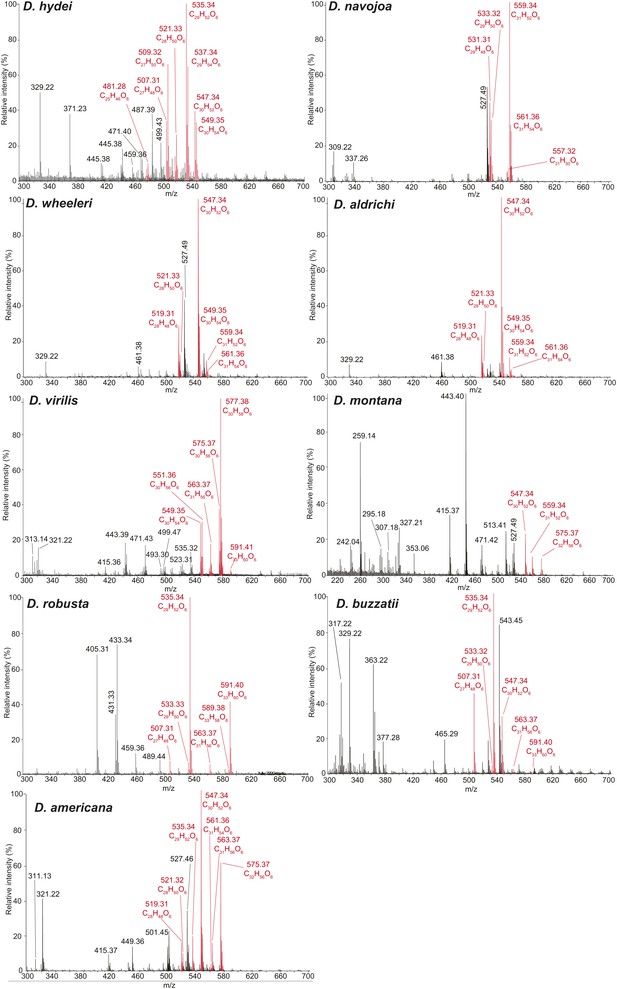
Representative UV-LDI spectra from distantly related drosophilids in the Drosophila subgenus.
Signals corresponding to sex-specific TAGs (in red) are identified based on the exact mass measurements, predicted elemental composition, and number of double bonds. Labeled signals correspond to potassiated molecules [M + K]+. Unlabeled peaks correspond to sodiated molecules [M + Na]+.
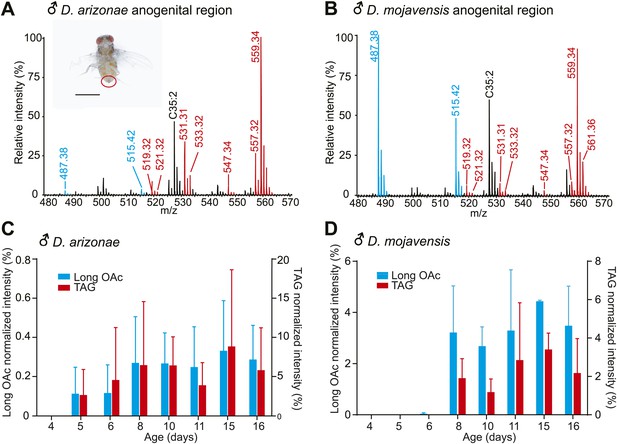
Pheromone profiles and age-related increase in sex-specific TAGs.
(A and B) UV-LDI MS allows spatially resolved detection of high molecular weight lipids directly from intact insects, with minimal damage to the cuticle. Representative mass spectra from the anogenital region (inset) of D. arizonae and D. mojavensis males show signals corresponding to triglycerides (TAGs, red) and long chain alkadienyl acetates (long OAcs, blue). The hydrocarbon C35:2 (number of carbons: number of double bonds) is found on cuticles of males and females. Labeled signals correspond to potassiated molecules [M + K]+. Scale bar: 1 mm. (C and D) Relative intensity of TAGs and long OAcs on male D. arizonae and D. mojavensis, respectively. TAGs and long OAcs increase with age, with trace quantities first appearing at 4 day old. The signal intensity for all detected TAGs or long OAcs was normalized to the signal intensity of C35:2.
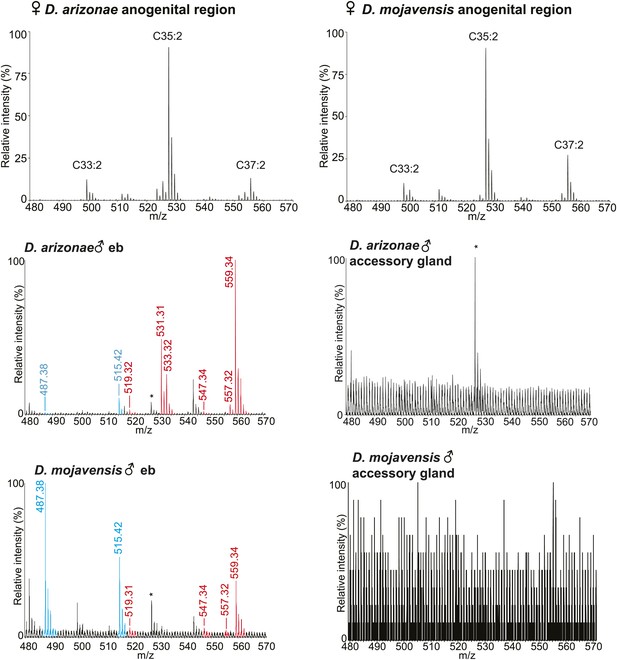
UV-LDI MS profiles from the anogenital region of virgin females and dissected ejaculatory bulb (eb) and accessory glands.
*: Background peak from fly wing matrix.
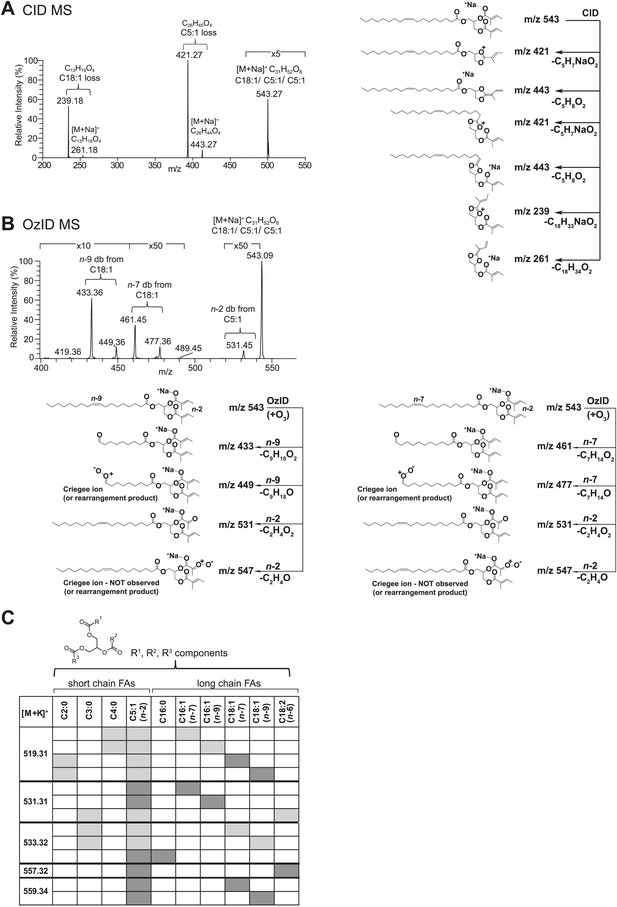
Structural elucidation of sex-specific TAGs.
(A) The low energy collision-induced dissociation (CID) mass spectrum of a TAG-related signal from crude D. arizonae extract ([M + Na]+ 543) shows fragments corresponding to losses of a 5 carbon fatty acid with a single double bond (C5:1) and an 18 carbon fatty acid with a single double bond (C18:1). Both sodiated (major peak) and protonated chain side losses are observed. The schematic rationalizes the product ions formed during CID of mass-selected [M + Na]+ of unsaturated lipids. (B) Ozone-induced dissociation (OzID) of a TAG-related signal (shown in A) indicates isomers with variant double bond positions. The fragments at m/z 461 and m/z 433 are aldehyde products consistent with double bonds (db) at positions n-7 and n-9, respectively. The fragment at m/z 531 confirms the n-2 double bond position found in the tiglic acyl component. The corresponding Criegee product ions (m/z 477 and m/z 449, respectively) are also observed. The schematic rationalizes the product ions formed during OzID of mass-selected [M + Na]+ of unsaturated lipids. Product ions are assigned as outlined by Thomas et al., 2008 and Brown et al., 2011. (C) CID and OzID MS analyses of the most abundant sex-specific TAGs reveal significant combinatorial complexity. A generic TAG molecule consisting of a glycerol backbone and 3 fatty acyl (FA) side chains, R1, R2, and R3, is shown. Each TAG species is comprised of 2 short chain and 1 long chain FA component. Shaded boxes indicate the composite side chains of each TAG species. The glycerol backbone positions for several TAGs are assigned based on the comparison with synthetic standards and ion product abundance patterns (dark gray boxes). Ambiguous backbone positions are in light gray.
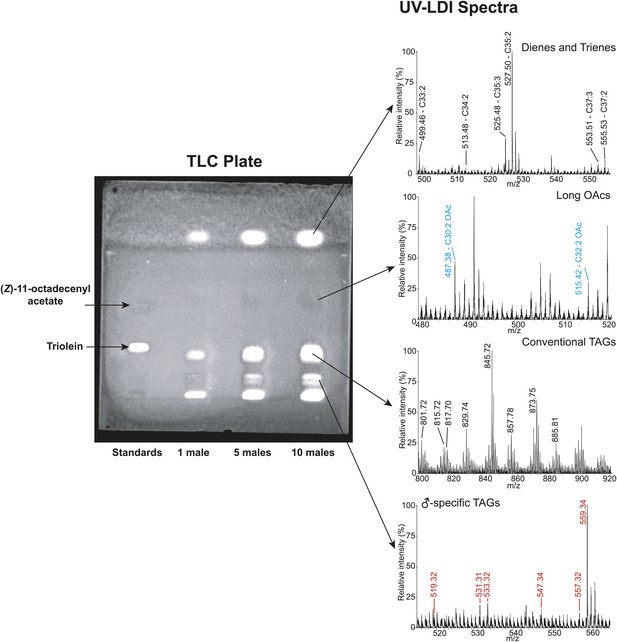
Thin layer chromatography (TLC) separation of D. arizonae male cuticular lipid extract.
Direct analysis of contents using fly-assisted laser desorption/ionization (FALDI) MS reveal signals corresponding to male-specific triacylglycerides (TAGs), long OAcs, conventional TAGs (from internal fat stores), and cuticular hydrocarbons.
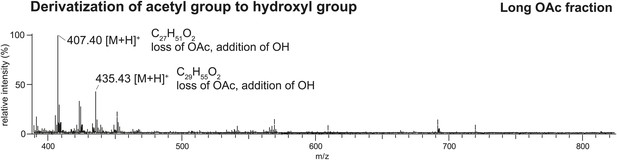
Direct analysis in real time (DART) MS spectrum of the TLC fraction containing long OAcs after derivatization by base hydrolysis confirms the presence of an acetyl functional group.
Based on the high mass accuracy predictions, the molecules [M + H]+ 449 and 477 (respectively, [M + K]+ 487 and 515 in Figure 2 UV-LDI spectra) are predicted to contain three double bonds. Derivatization of the putative acetyl functional group with base hydrolysis resulted in a mass shift (loss of m/z 42) consistent with the replacement of an acetyl with a hydroxyl group.
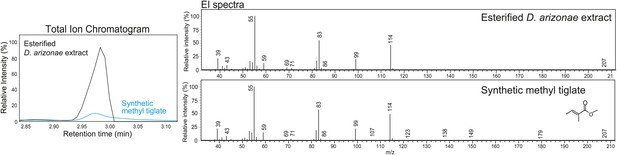
Structural elucidation of sex-specific TAGs using gas chromatography MS.
GCMS analysis of the male-specific TAG fraction following trans-esterification confirms that tiglic acid is one of the fatty acyl components. The retention time (at 2.98 min) and the electron ionization (EI) spectrum of synthetic methyl tiglate are identical to the analysis of a TAG fraction purified from crude extract.
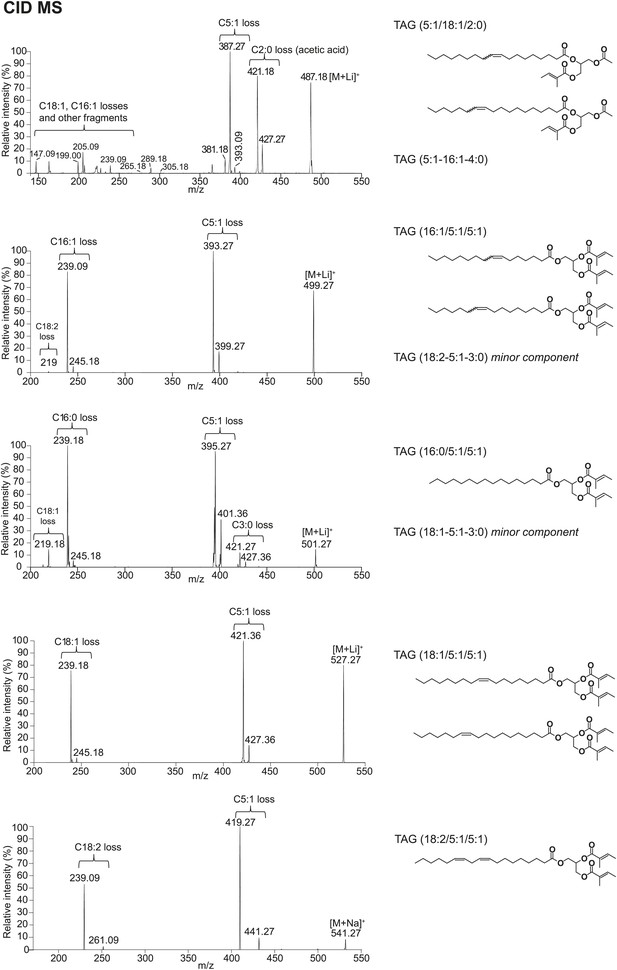
Structural elucidation of TAGs by CID MS reveals fatty acid (FA) components with 2, 3, 5, 16, or 18 carbons in length and 0–2 double bonds.
For more abundant TAG molecules, the FA substituent positions on the glycerol backbone are assigned based on the relative abundances of the product ions in the CID spectra, as described by Hsu and Turk (1999 and 2010). For minor TAG components, only the composite FA side chains are indicated. Substituent positions are ambiguous due to low-signal intensity.
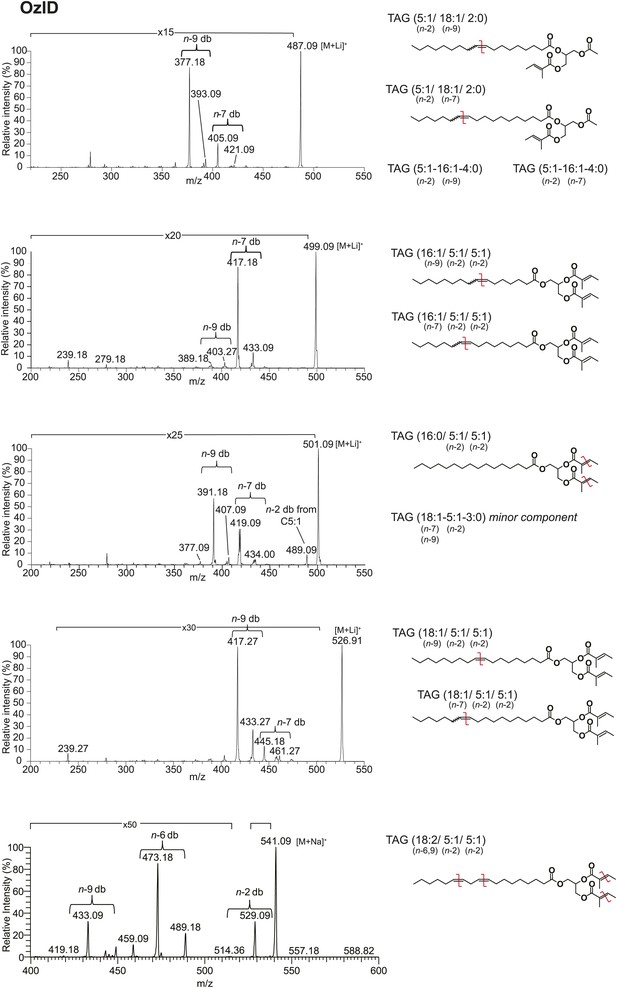
Analysis of TAGs by OzID reveals double bond positions of acyl side chains.
Fragments resulting from OzID analysis are aldehyde products with m/z consistent with double bonds at positions n-2, n-7, and n-9 for fatty acyl monoene constituents and at both n-6 and n-9 for a fatty acyl diene. n indicates the position of the terminal methyl group. The corresponding Criegee product ions (difference of m/z 16) were also found in each spectrum. Although the CID spectrum for the TAG species at [M + Li]+ 499 indicated a C18:2 FA component, the intensity of this signal was too low to allow for double bond placement by OzID. Red lines indicate putative cleavage points.
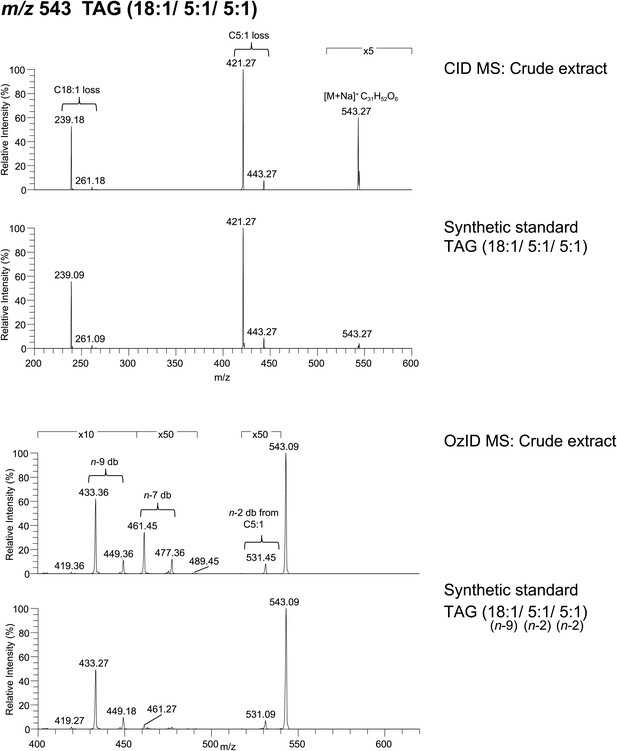
Spectra obtained from CID MS and OzID analyses of a synthetic TAG comprised of an oleic acid (cis-9-Octadecenoic acid) and tiglic acid side chains are consistent with the analysis of a TAG molecule with identical m/z found from crude extract.
The relative abundance of fragment ions resulting from fatty acyl side chain loss is consistent between natural and synthetic products and supports the backbone substituent positions. Following OzID analysis, the overall and relative abundance of aldehyde and Criegee ions to each other are similar between natural and synthetic products, indicating cis-double bond geometry. Difference in parent ion intensity between synthetic and natural products is likely due to isobaric interference from other minor components found in the crude extract.
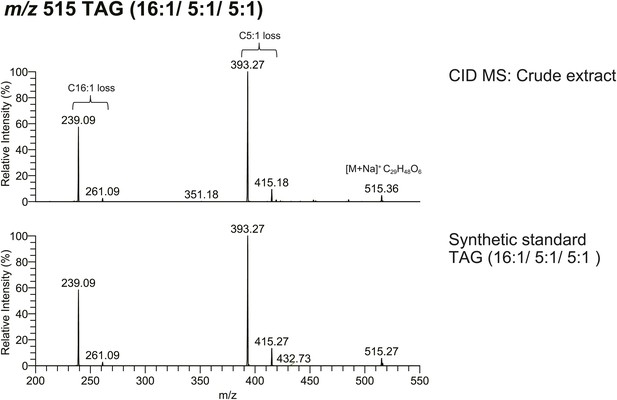
The spectrum obtained from CID MS analysis of a synthetic TAG (16:1/5:1/5:1) is consistent with the spectrum from a TAG molecule with identical m/z found from crude extract.
The relative abundance of fragment ions resulting from fatty acyl side chain loss is consistent between natural and synthetic products, supporting the backbone substituent positions.
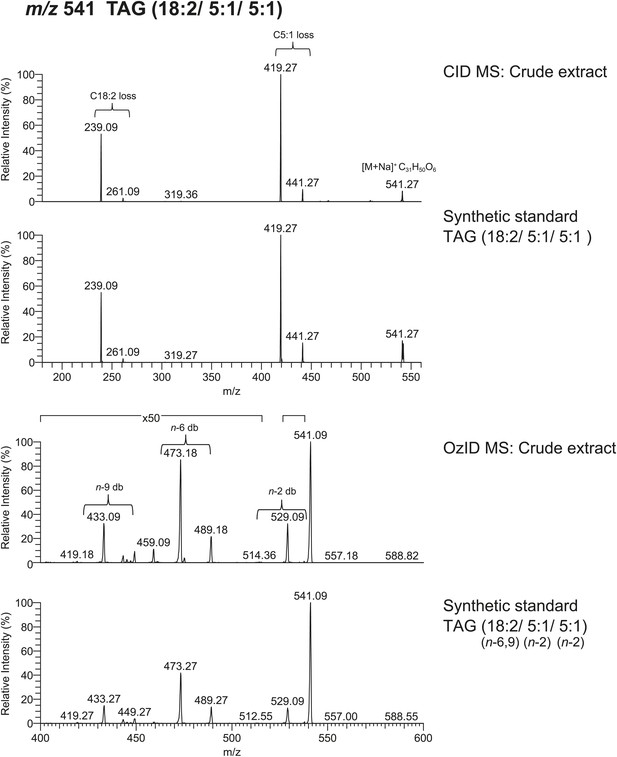
Spectra obtained from CID MS and OzID analyses of a synthetic TAG consisting of linoleic acid (cis, cis-9,12-Octadecadienoic acid) and tiglic acid side chains are consistent with spectra from analysis of a TAG molecule with identical m/z found from crude extract.
The relative abundance of fragment ions resulting from fatty acyl side chain loss is consistent between natural and synthetic products and supports the backbone substituent positions. Following OzID analysis, the overall and relative abundance of aldehyde and Criegee ions to each other are similar between natural and synthetic products, indicating cis and cis-double bond geometry.
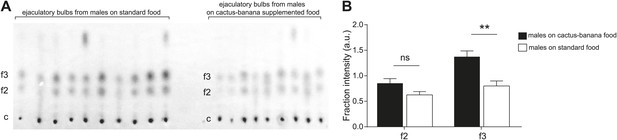
Diet changes the quantity but not composition of sex-specific TAGs.
(A) TAGs from individual ejaculatory bulbs of males raised on standard fly food (n = 10) or cactus-banana supplemented food (n = 9) were quantified using direct tissue thin layer chromatography. Each lane contains a single bulb. c: control band (point of origin) used for normalization; f2 and f3: fractions containing sex-specific TAGs. (B) The amount of TAGs in f3 from ejaculatory bulbs of males raised on standard food is significantly lower than supplemented food conditions (Student’s t-test, two-tailed, p=0.0016). TAGs found in f2 were not significantly different (p=0.062). Error bars indicate SEM. **: p<0.005; ns: not significant; a.u.: arbitrary units.
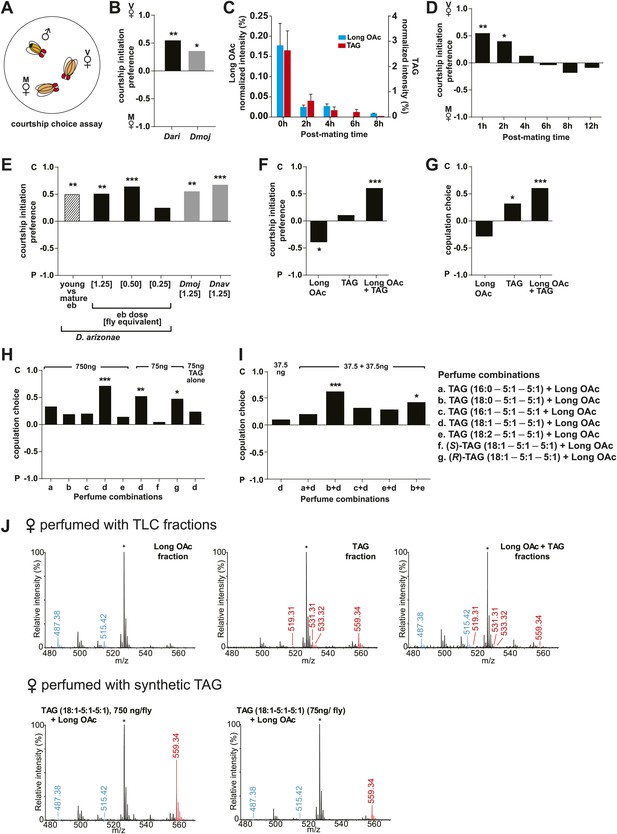
Sex-specific lipids suppress male mating behavior.
(A) To measure male courtship behavior, one male fly is placed with 2 females, one mated (M), and one virgin (V). (B) D. arizonae (Dari) and D. mojavensis (Dmoj) prefer to court virgin females over recently mated females (n = 20, Fisher’s exact test, p=0.00123; n = 31, p=0.0105). **: p<0.01; ns: not significant. A preference score of 1 indicates all males initiate courtship first with the virgin female; −1 indicates all males initiated courtship first with the mated female. (C) Levels of male-transferred TAGs and long OAcs on the female cuticle after first mating decreases by 2 hr post-mating. (D) Females are significantly less attractive for up to 2 hr after mating. By 4 hr, males do not exhibit significant preference for courting mated vs virgin females. **: Fisher’s exact test, p=0.00123; *: p=0.0256. (E) D. arizonae males are more reluctant to initiate courtship with females perfumed with the contents of [1.25] ejaculatory bulb (eb) (n = 27, Fisher’s exact test, p=0.000624) or [0.5] eb (n = 28, p<0.0001) but not [0.25] eb (n = 28, p=0.176). Extracts from immature male ebs were ineffective at inhibiting male courtship (n = 28, p=0.000389). D. mojavensis and D. navojoa (Dnav) males also avoided virgin females perfumed with eb contents (Dmoj: n = 21, p=0.00160; Dnav: n = 21, p<0.0002). **: p<0.002; ***: p<0.0001. C: solvent control; P: perfumed. (F) Suppression of D. arizonae courtship initiation is elicited only when TAGs and long OAcs are combined (n = 28, Fisher’s exact test, p<0.0001). TAGs alone are ineffective (n = 28, p=0.593). Long OAcs on their own could be attractive to males (n = 28, p=0.006). *: p<0.05; ***: p<0.0001. (G) D. arizonae copulation is suppressed in the presence of TAGs alone (n = 28; Fisher’s exact probability test, p=0.0287) or TAGs combined with long OAcs (n = 28; p<0.0001), but not long OAcs alone (n = 28; p=0.0543). *: p<0.05; ***: p<0.0001. A copulation choice score of 1 indicates all males copulated with solvent-perfumed females; −1 indicates all males copulated with TAG-perfumed females. (H) Perfuming with synthetic TAGs recapitulates copulation suppression. Oleic acid (C18:1)-containing TAGs produced significant effects at high and low doses (750 ng: n = 21, Fisher’s exact test, p<0.0001; 75 ng: n = 21, p=0.00167). Only the (R)-18:1 stereoisomer was bioactive (75 ng: n = 21, p=0.00480); the (S)-18:1 stereoisomer did not elicit a significant behavioral response (75 ng: n = 21, p=1). *: p<0.01; **: p<0.002; ***: p<0.0001. (I) Two combinations of TAGs produced synergistic effects on copulation suppression: oleic acid-TAG paired with stearic acid-TAG (n = 21, p=0.000139) and stearic acid-TAG paired with linoleic acid-TAG (n = 19, p=0.022). The oleic acid-containing TAG is not bioactive at a dose of 37.5 ng/fly (n = 20, Fisher’s exact test, p=0.751). *: p<0.05; ***: p<0.0005. (J) UV-LDI MS spectra of females perfumed with TLC fractions or synthesized TAGs. Signals for TAGs and long OAcs are indicated in red and blue, respectively. *: C35:2 Pentatriacontadiene reference peak, m/z 527.5 [M + K]+.
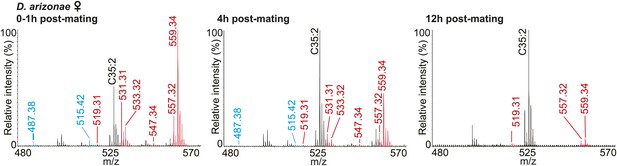
UV-LDI spectra of mated D. arizonae female anogenital regions reveal signals corresponding to male-specific TAGs (red) and long OAcs (blue) for up to 12 hr post-mating.
https://doi.org/10.7554/eLife.01751.018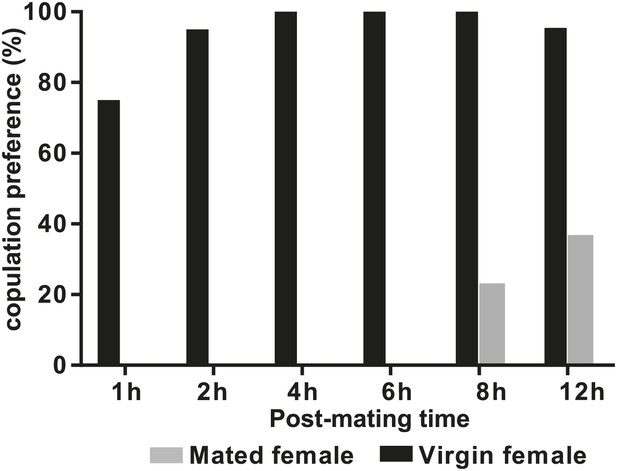
D. arizonae mated females are receptive to copulation starting at 8 hr after the first mating.
Copulation choice indicates the percentage of females that copulated with males from all trials within 30 min. In some cases, males copulated with both virgin and mated female targets within the same trial.
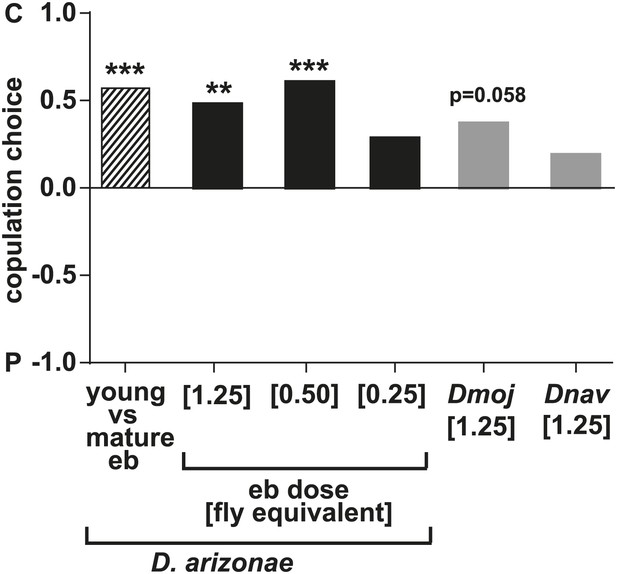
D. arizonae males avoided copulating with females perfumed with the contents of male ejaculatory bulbs (eb) at a concentration of [1.25] eb (n = 27, Fisher’s exact test, p=0.0003) or [0.5] eb (n = 28, Fisher’s exact test, p<0.0001) but not [0.25] eb (n = 28, p=0.176).
Eb extract from immature males was significantly less effective than extract from mature males (n = 28; *: Fisher's exact test, p<0.0001). D. mojavensis exhibited a tendency to avoid eb-perfumed females (n = 21, Fisher's exact test, p=0.0578). D. navojoa males courted but seldom copulated within the 1 hr observation time. Only 4 out of 21 males copulated, preferring the non-perfumed females. **: p<0.001; ***: p<0.0001. C: solvent control; P: perfumed.
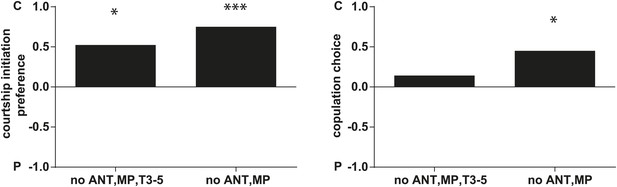
D. arizonae courtship and copulation preferences towards partly olfactory and gustatory perception-deficient virgin females that are perfumed with control or ejaculatory bulb extract.
Leaving gustatory perception intact and removing the antennae and maxiliary palps, there is no effect on male courtship or copulation preference towards control females. Males still showed a preference for control females (no ANT,MP; n = 20, p<0.0001; and p<0.01). Virgins without antennae, maxiliary palps, and having T3-5 segments of forelegs painted over with nail polish (T3-5), also did not affect male courtship choice (no ANT,MP,T3-5; Fisher's exact test, n = 21, p<0.01), suggesting that female behavior did not affect courtship initiation. However, these females are less likely to allow copulation (only 5 out of 21 copulated) (no ANT,MP,T3-5; n = 21, p=0.343). *: p<0.01; ***: p<0.0001. C: solvent control; P: perfumed.
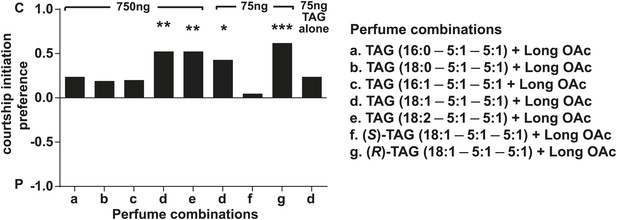
D. arizonae courtship preferences towards virgin females perfumed with synthetic TAGs together with extract-purified long OAcs.
Perfuming at the higher dose with the oleic acid (C18:1) or linoleic acid (C18:2)-containing. TAGs suppressed courtship initiation (n = 21, Fisher's exact test, p=0.00167; 750 ng: n = 21, p= 0.00158). Oleic acid-TAG was also effective at the 75 ng dose (n = 21, p=0.0126) paired with long OAcs, but not by itself. Only the (R)-18:1 stereoisomer was bioactive (75 ng: n = 21, p=0.000139); the (S)-18:1 stereoisomer did not elicit a significant behavioral response (75 ng: n = 21, p=1). *: p<0.02; **: p<0.002; ***: p<0.0002. C: solvent control; P: perfumed.
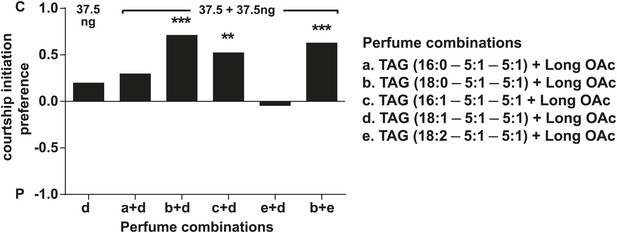
D. arizonae courtship preferences towards virgin females perfumed with different combinations of synthetic TAG together with extract-purified long OAcs.
Three combinations worked synergistically to inhibit courtship imitation: TAGs containing oleic acid (C18:1) paired with those containing stearic acid (C18:0; Fisher's exact test, n = 21, p<0.0001) or palmitoleic acid (C16:1; n = 21, p=0.00292) and TAGs containing stearic acid (C18:0) paired with those containing linoleic acid (C18:2; n = 19, p=0.00023). **: p<0.005; ***: p<0.0005. C: solvent control; P: perfumed.
Tables
Synthetic TAGs used in this study
| Calculated [M + K]+ | Fatty acyl components*,† | Long chain fatty acid |
|---|---|---|
| 531.31 | C16:1 (n-7)-C5:1-C5:1 | Palmitoleic acid |
| 533.32 | C16:0-C5:1-C5:1 | Palmitic acid |
| 557.32 | C18:2 (n-6)-C5:1-C5:1 | Linoleic acid |
| 559.34 | C18:1 (n-9)-C5:1-C5:1 | Oleic acid |
| 561.36 | C18:0-C5:1-C5:1 | Stearic acid |
| 559.34 | C18:1 (n-9)-C5:1-C5:1 (R isomer) | Oleic acid |
| 559.34 | C18:1 (n-9)-C5:1-C5:1 (S isomer) | Oleic acid |
-
*
Synthesized as racemic mixtures unless otherwise noted.
-
†
Notation indicates number of carbons followed by number of double bonds for each fatty acyl component; double bond position indicated in brackets.





