Precardiac deletion of Numb and Numblike reveals renewal of cardiac progenitors
Figures
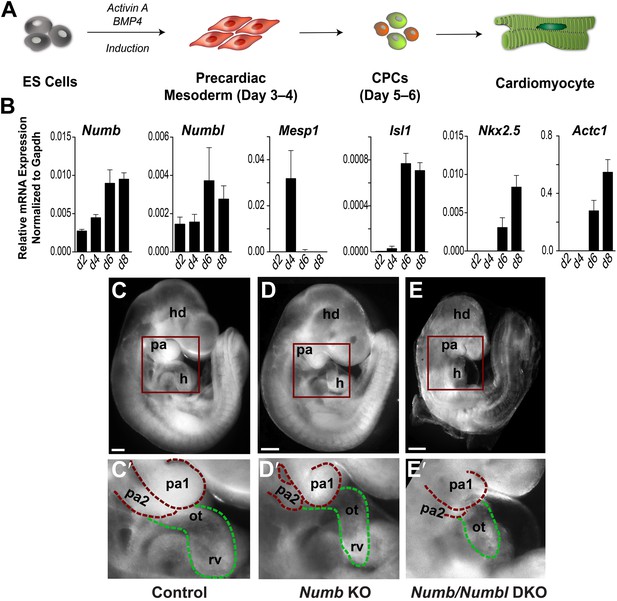
Numb and Numbl are required for PA2 and heart development.
(A) Schema of cardiac differentiation in ES cell system. (B) Expression profiles of genes indicated during cardiac differentiation of ES cells. Gene expression was analyzed by qPCR. Data are mean ± SD; n = 4; d, day. (C–E) Lateral views of control (C), Mesp1Cre; Numbflox/flox (Numb KO, D), Mesp1Cre; Numbflox/flox; Numbl–/– (Numb/Numbl DKO, E) embryos. (C′–E′) Enlargement of boxed areas in (C–E), showing normal, hypoplastic or atrophic PA2 and heart in control (C′), Numb KO (D′) or Numb/Numbl DKO (E′) embryos, respectively. Pharyngeal arches (red) and outflow tract/right ventricle (green) are outlined in dashes. Scale bars, 150 μm. hd, head; pa, pharyngeal arch; h, heart; ot, outflow tract; ra, right atrium; rv, right ventricle.
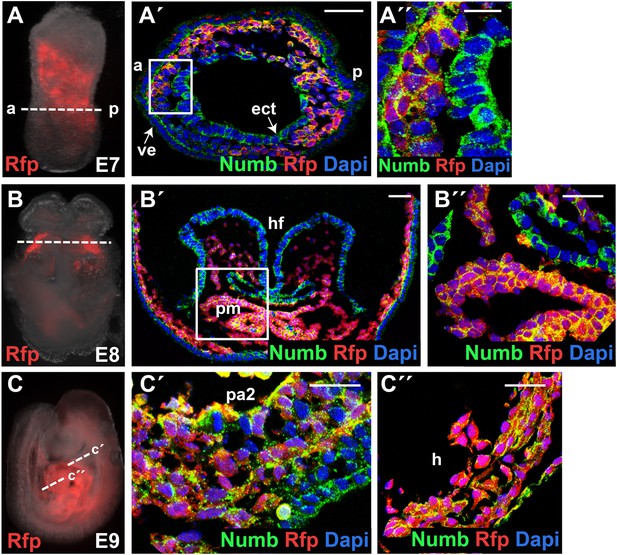
Numb is ubiquitously expressed in developing embryos.
(A–C) Mesp1 lineage-traced embryos (Mesp1Cre; Ai9) at E7 (A), E8 (B), or E9 (C). RFP marks Mesp1+ cells and their progeny. (A′, A″, B′, B″, C′, C″) Confocal images of corresponding embryos immunostained with Numb (green) or RFP (red) antibody, showing ubiquitous expression of Numb protein. Section planes are indicated by dotted lines in (A), (B), (C). Boxed areas in (A′) and (B′) are shown in higher magnification in (A″) and (B″), respectively. Second pharyngeal arch and heart sections are shown in (C′) and (C″), respectively. Dapi (blue) was used to counterstain the nuclei. Scale bars, 10 mm (A″), 25 mm (B″, C′, C″), 50 mm (A′ and B′), and 100 mm (C″). a, anterior; p, posterior; ect, ectoderm; ve, viceral endoderm; hf, head fold; pm, precardiac mesoderm; pa2, second pharyngeal arch; h, heart.
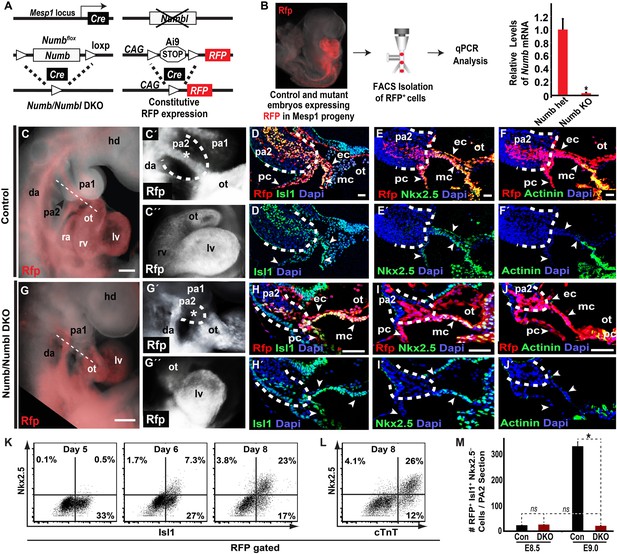
Deletion of Numb and Numbl depletes Mesp1+ progenitor-derived Isl1+ Nkx2.5– cells in PA2.
(A) Generation of Numb/Numbl DKO embryos expressing RFP in Mesp1 progeny (Mesp1Cre; Numbflox/flox; Numbl−/−; Ai9). (B) Relative mRNA levels of Numb in RFP+ cells isolated from control and Numb/Numbl DKO embryos. Data are mean ± SD; n = 9. (C, C′, G, G′) Lateral views of Mesp1+ cell-traced control (C and C′) or Numb/Numbl DKO (G and G′) embryos analyzed at E9.0. RFP marks Mesp1 progeny. Control embryos show continuous RFP expression from second pharyngeal arch (PA2, outlined) to heart (asterisk, C′), but the arch (outlined) is severely underdeveloped in Numb/Numbl DKO embryos without noticeable RFP expression (asterisk, G′). (C″ and G″) Frontal views of control (C″) or Numb/Numbl DKO (G″) heart. (D–F′), (H–J′), Representative confocal images of transverse sections through PA2 and outflow tract (OT) of control (D–F′) and Numb/Numbl DKO (H–J′) embryos. Cutting planes are shown in (C) and (G). Internal boundaries of PA2 are outlined in dashes. Dapi (blue) was used to counterstain the nuclei. (K and L) Representative plots of intracellular staining of RFP-gated cells. Isl1 and Nkx2.5 staining during day 5–8 (K) and cardiac troponin T (cTnT) and Nkx2.5 staining at day 8 (L). (M) Average number of RFP+ Isl1+ Mef2c−Nkx2.5− cells in PA2 section (12-micron) of indicated embryos and stages. Data are mean ± SD; n = 5; *p<0.05; ns, not significant. p values were determined using the paired Student t test. Scale bars, 25 μm (D–F and H–J), 150 μm (C and G). hd, head; pa, pharyngeal arch; ot, outflow tract; da, dorsal aorta; fe, foregut endoderm; ra, right atrium; rv, right ventricle; lv, left ventricle; ec, endocardial layer; mc, myocardial layer; pc, pericardial layer.
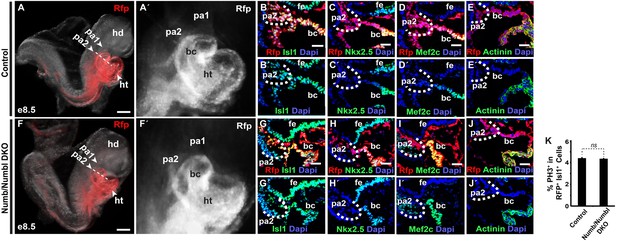
Numb/Numbl DKO embryos are grossly normal at E8.5.
(A, A′, F, F′) Lateral views of Mesp1+ cell-traced control (A and A′) or Numb/Numbl DKO (F and F′) embryos analyzed at E8.5. RFP marks Mesp1 progeny. (B–E′ and G–J′) Representative confocal images of transverse sections through primordial PA2 and BC of control (B–E′) and Numb/Numbl DKO (G–J′) embryos (Cutting planes are shown in A and F) with indicated markers. Internal boundaries of PA2 are outlined in dashes. Dapi (blue) was used to counterstain the nuclei. (K) Percentage of PH3+ cells in RFP+ Isl1+ cells in control and Numb/Numbl DKO embryos at E8.5. Data are mean + SD; n = 4; *p<0.05; ns, not significant. p values were determined using the paired Student t test. Scale bars, 15 mm (B–E and G–J), 200 mm (A and F). hd, head; ht, heart tube; pa, pharyngeal arch; bc, bulbus cordis; fe, foregut endoderm.
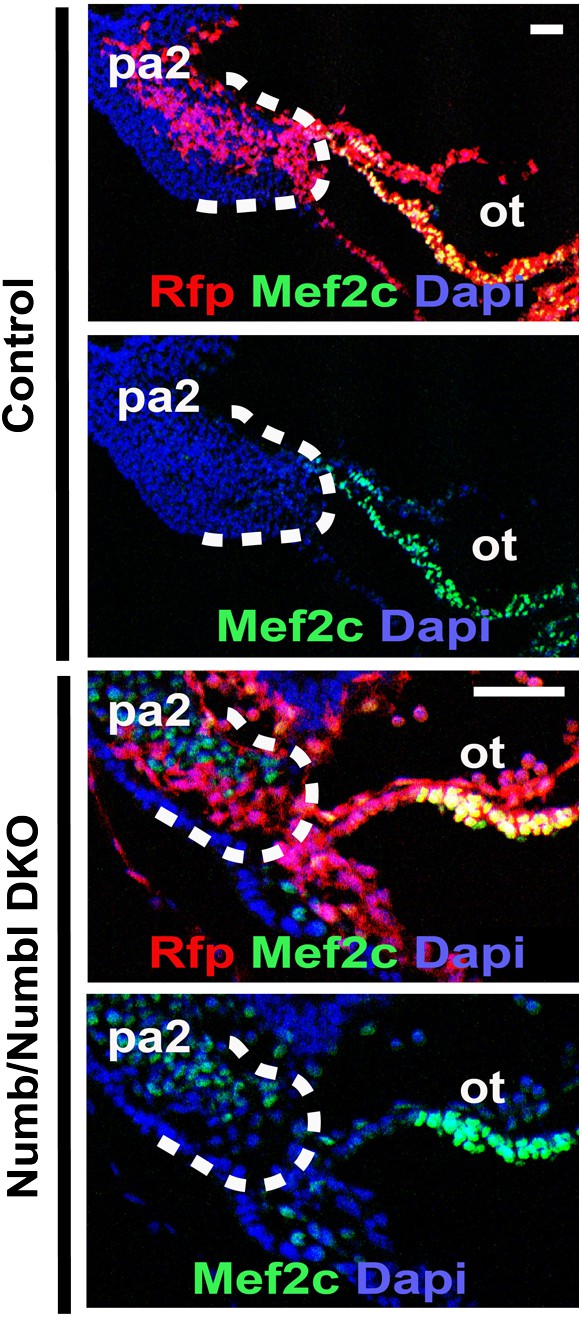
Mef2c expression in PA2 and OT of control and Numb/Numbl DKO embryos.
Scale bars, 25 mm. pa2, second pharyngeal arch; ot, outflow tract.
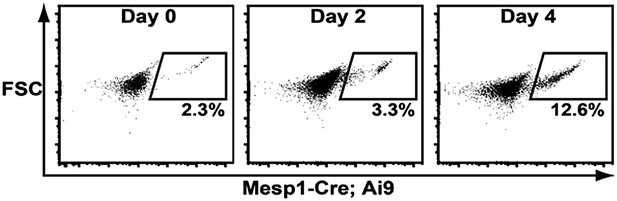
Histogram of RFP + cell induction from Mesp-Cre; Ai9 ES Cells.
RFP+ cells are induced from day 2–4.
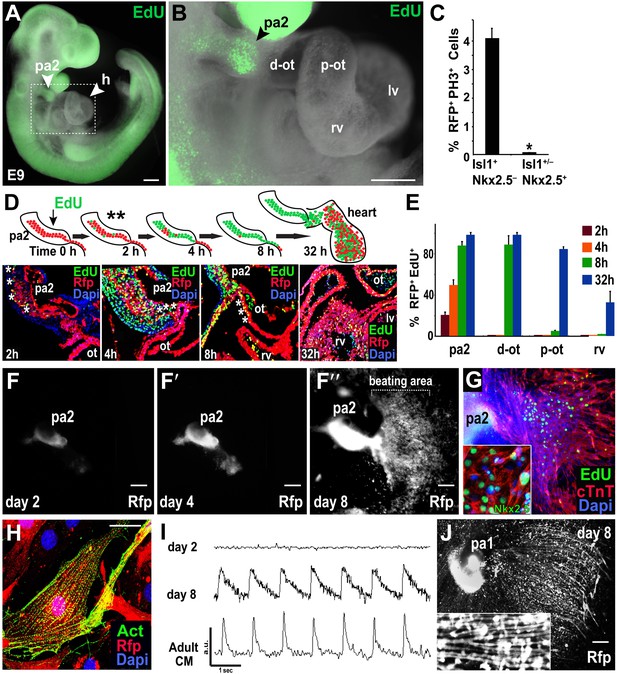
Mesp1+ progenitor-derived Isl1+ Nkx2.5– cells expand in PA2 and differentiate into Nkx2.5+ heart cells after leaving PA2.
(A) Whole-mount view of EdU (green)-treated embryo at E9.0. (B) Enlargement of the boxed area in (A), showing enrichment or lack of EdU+ cells in the PA2 or heart region, respectively. (C) Percentage of RFP+ PH3+ cells in Isl1+ Mef2c−Nkx2.5− cells and Isl1+/− Mef2c+ Nkx2.5+ cells. Data are mean ± SD; n = 4. (D) EdU pulse experiment. Top, EdU experiment schema. PA2s were also dissected out for ex vivo culture after 2 hr (**). Bottom, progeny of EdU+ cells at 2, 4, 8, and 32 hr after single pulse of EdU injection at E9.0 demonstrating that Mesp1 progeny in PA2 proliferate and migrate to form the OT/RV. (E) Quantification of RFP+ EdU+ cell progeny shown in (D). Data are mean ± SD; n = 3. (F–F″), Cultured PA2 explants in 2D culture form a sheet of beating cardiomyocytes (see Video 1). (G) EdU-pulsed PA2-derived sheet of beating cells stained with cTnT, EdU, or Nkx2.5 (inset). (H) Confocal image of RFP+ cells migrated from PA2 stained with cardiac α-actinin (Act). (I) Intracellular Ca2+ transients from day 2 and day 8 PA2 explants and adult cardiomyocyte (CM). a.u., arbitrary unit. (J) Cultured PA1 explants form myotube-like cells. Inset shows a magnified view. *p<0.05. D, day; Heart, E10.5 embryonic heart; ACTC1, actin, alpha, cardiac muscle 1. Dapi (blue) was used to counterstain the nuclei. p values were determined using the paired Student t-test. Scale bars, 10 μm (H), 150 μm (A and B), 250 μm (F–F″ and J). pa, pharyngeal arch; h, heart; d-ot, distal outflow tract; p-ot, proximal outflow tract; rv, right ventricle; lv, left ventricle.
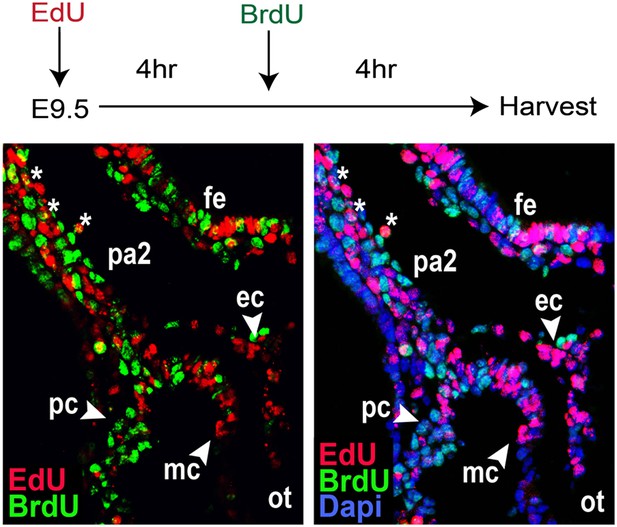
Dual Injection of EdU and BrdU.
EdU and BrdU were sequentially injected at E9.5 at a 4-hr interval. The resulting embryos were harvested after 4 hr (8 hr after EdU injection) and analyzed by immunostaining. EdU+ cells were mostly negative for BrdU, suggesting the purdurance of EdU is shorter than 4 hr. Some EdU+ BrdU+ cells (asterisks) were present in the PA2, implying their continued proliferation. ot, outflow tract; fe, foregut endoderm; ec, endocardial layer; mc, myocardial layer; pc, pericardial layer.
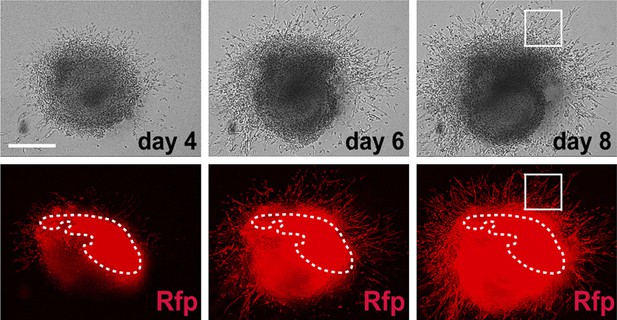
Time lapse images of 3-D matrigel PA2 culture.
These show growth and migration of Mesp1 progeny (RFP+) in the PA2 (Video 1). Boxed area is shown in Video 2.
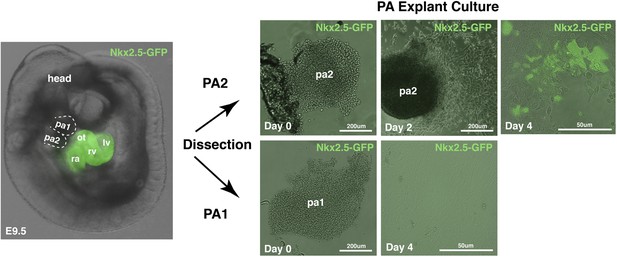
Explant culture of PA1 and PA2 dissected from Nkx2.5GFP embryo.
pa, pharyngeal arch; ot, outflow tract; rv, right ventricle; ra, right atrium; lv, left ventricle.
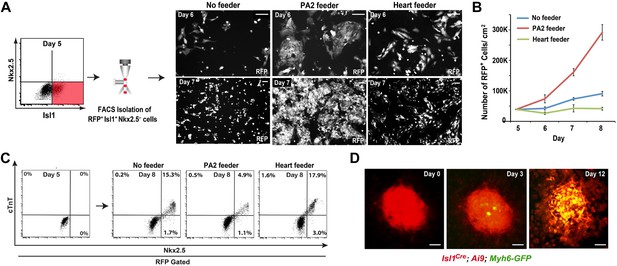
PA2 cells promote CPC expansion and suppress cardiac differentiation.
(A) FACS-purification of RFP+ Isl1+ Nkx2.5− CPCs induced from ES cell-derived precardiac mesoderm and their culture with no, embryonic PA2, or embryonic heart feeders. Images show RFP+ cells at day 6 and day 7. Isl1Cre; Ai9 ES cells were used to purify the CPCs at day 5 of cardiac differentiation, when Nkx2.5 is not expressed in Isl1+ CPCs. (B) Quantification of numbers of RFP+ cells cultured with no, embryonic PA2, or embryonic heart feeders. Data are mean ± SD; n = 3. (C) FACS plot of RFP+ Isl1+ Nkx2.5− CPCs differentiating into Nkx2.5+/cTnT+ cells with no, embryonic PA2, or embryonic heart feeders, determined at day 8. (D) Time-lapse images of Isl1+ Nkx2.5− CPC colony showing cardiac differentiation after removal of PA2 cells, indicated by GFP expression driven by Myh6 promoter. Scale bars, 50 μm.
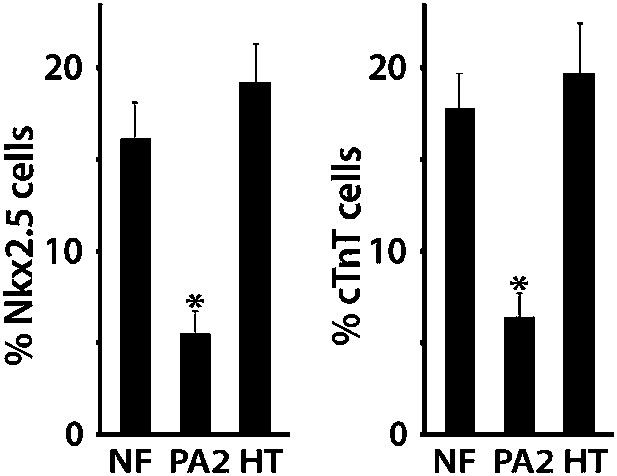
Percentages of Nkx2.5+ or cTnT + cells cultured with No, PA2, or embryonic heart feeders at day 8.
NF, no feeder; PA2, PA2 co-culture; HT, heart cell co-culture. Data are mean + SD; n = 3; *p<0.05. p values were determined using the paired Student t test.
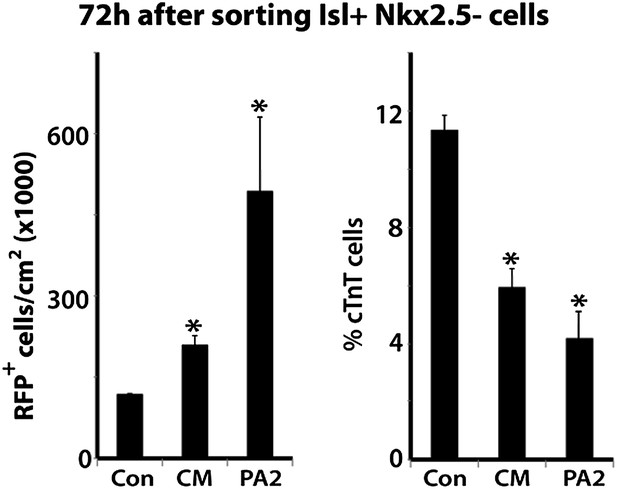
PA2 conditioned medium mimics PA2 co-culture.
Con, control; CM, PA2-conditioned medium; PA2, PA2 co-culture. Data are mean + SD; n = 3; *p<0.05. p values were determined using the paired Student t test.
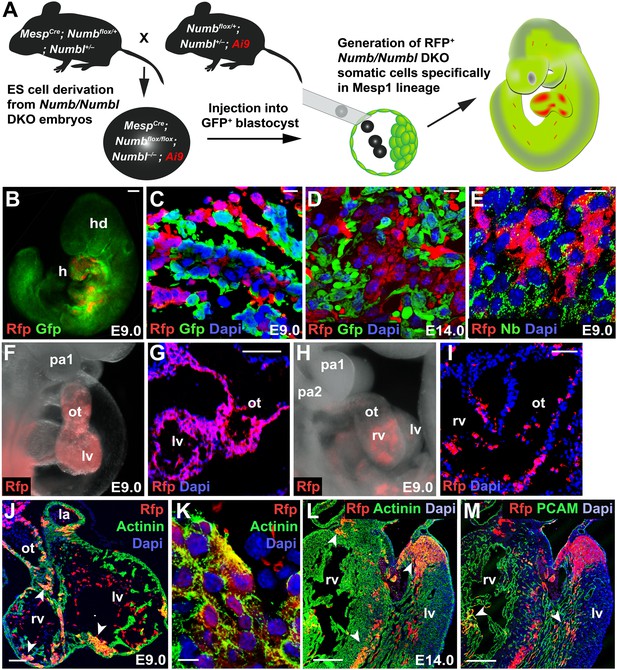
Generation of Mesp1 lineage-specific somatic cells lacking Numb and Numbl in vivo.
(A) Scheme for generation of RFP+ Numb/Numbl DKO cells in Mesp1 lineage. In this study, three independent sets of blastocyst injection were carried out and 16 chimeras were obtained from 132 embryos. (B–E) Chimeric embryos at E9.0 (B) generated with GFP+ host cells. Numb/Numbl DKO cells are shown in red. Sections were made transversely through cardiac region (C–E) and immunostained with RFP and GFP (C and D) or RFP and Numb (E) antibodies at corresponding stages. (F–I) Lateral views of chimera and sections, focused on PA and heart, showing contribution of RFP+ cells. Major contribution of RFP+ cells causes a phenotype similar to Numb/Numbl DKO embryos (F and G). (J–M) Confocal images of heart transverse sections at indicated stages. α-Actinin and PCAM are properly expressed in RFP+ cells (arrowheads). RFP+ area in (J) is enlarged in (K). Dapi (blue) was used to counterstain the nuclei. Scale bars, 10 μm (C, D, E, K), 100 μm (B, G, I, J), and 200 μm (L and M). a, anterior; p, posterior; hd, head; h, heart; pa, pharyngeal arch; ot, outflow tract; rv, right ventricle; lv, left ventricle; la, left atrium.
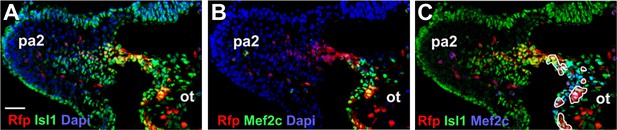
Numb/Numbl DKO cells are normally specified into OT cells.
(A–C) Confocal images of PA2 sections of E9.0 chimera, immunostained with RFP and Isl1 (A), RFP and Mef2c (B) or RFP, Isl1 and Mef2c (C) antibodies. RFP+ Isl1+ Mef2c+ cells are outlined in white (C). Dapi (blue) was used to counterstain the nuclei. Scale bars, 50 mm. ot, outflow tract; pa, pharyngeal arch.
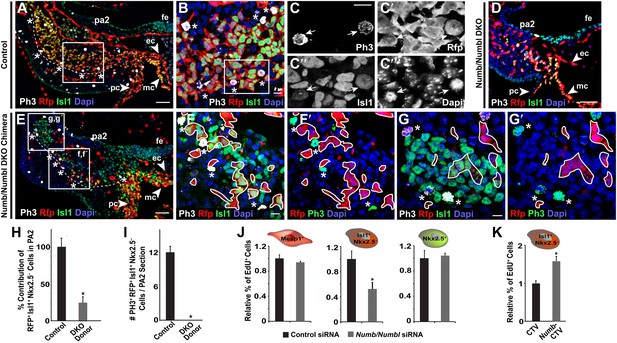
Numb and Numbl are required for proliferation of Mesp1+ progenitor-derived Isl1+ Nkx2.5– cells in PA2.
(A–G′) Confocal images of PA2 sections of control (A–C‴), Numb/Numbl DKO (D), and chimeric (E–G′) embryos, immunostained with PH3, RFP, Isl1 antibodies. Asterisks indicate PH3+ RFP+ Isl1+ (triple positive) cells located in outer layers of RFP+ Isl1+ cell population (outlined, A and E). Boxed areas in (A), (B), and (E) are shown in higher magnification in (B), (C–C‴), and (F–G′), respectively. No PH3+ cells are found in RFP+ cells (asterisks, F–G′). RFP+ cells were outlined in white (F–G′). (H and I) Percentage of donor-derived Isl1+ Nkx2.5− cells in PA2 is shown in (H) and number of PH3+ RFP+ Isl1+ cells per 12-micron PA2 section is shown (I). Data are mean ± SD; n = 10; *p<0.05. (J and K) Relative percentage of EdU+ cells in ES cell-derived Mesp1+ progenitor, Isl1+ Nkx2.5− CPCs or Nkx2.5+ CPCs transfected with control or Numb/Numbl DKO siRNA (J) or Isl1+ Nkx2.5− CPCs transfected with control (CTV) or Numb overexpression construct (CTV-Numb) (K). Data are mean ± SD; n = 3; *p<0.05. The Mesp1+, Isl1+ Nkx2.5−, or Nkx2.5+ cells were FACS-purified from day 4 Mesp1Cre; Ai9, day 5 Isl1Cre; Ai9, or day 6 Nkx2.5GFP ES cells, respectively. Dapi (blue) was used to counterstain the nuclei. p values were determined using the paired Student t test. Scale bars, 10 μm (B, C, F, G). 50 μm (A, D, E). fe, foregut endoderm; pa, pharyngeal arch ec, endocardial layer; mc, myocardial layer; pc, pericardial layer.
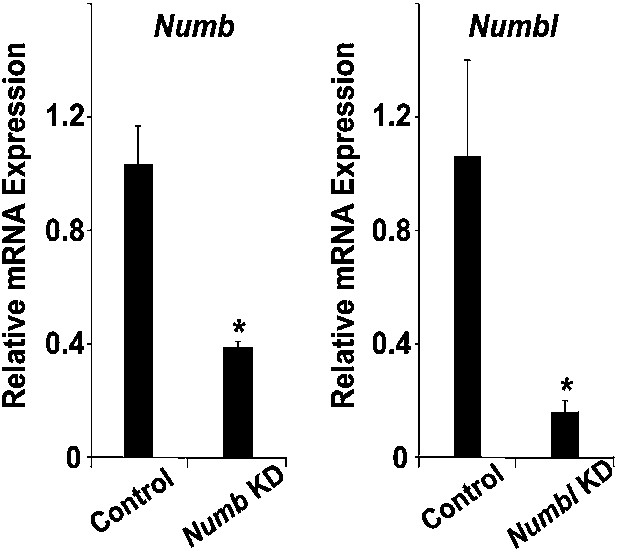
Knockdown efficiency of Numb siRNA and Numbl siRNA.
Relative mRNA expression levels of Numb and Numbl in CPCs after transfected with scrambled siRNA (Con), Numb siRNA (Numb KD), or Numbl siRNA (Numbl KD), determined by qPCR. Data are mean + SD; n = 3; *p<0.05. p values were determined using the paired Student t test.
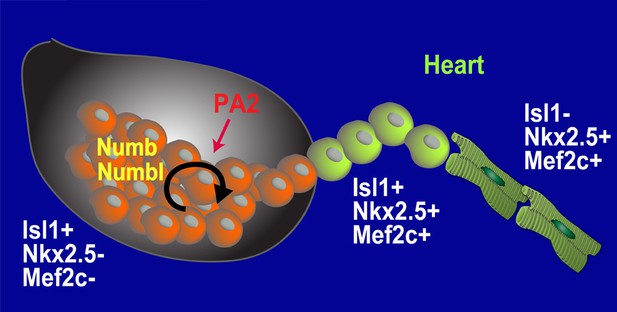
Model for Renewal and Niche of Mesp1+ progenitor-derived CPCs.
https://doi.org/10.7554/eLife.02164.023Videos
3D-cultured PA2 in Matrigel.
This video shows expansion and migration of Mesp1 progeny (RFP+) in ex-vivo cultured PA2.
High magnification of inset in Video 1.
This video shows RFP+ beating cardiac myocytes at a migrating edge of ex-vivo cultured PA2.
2D-cultured PA2.
This video shows a beating monolayer of cardiomyocytes derived from RFP+ Mesp1 Progeny in ex-vivo cultured PA2.





