Intact protein folding in the glutathione-depleted endoplasmic reticulum implicates alternative protein thiol reductants
Figures
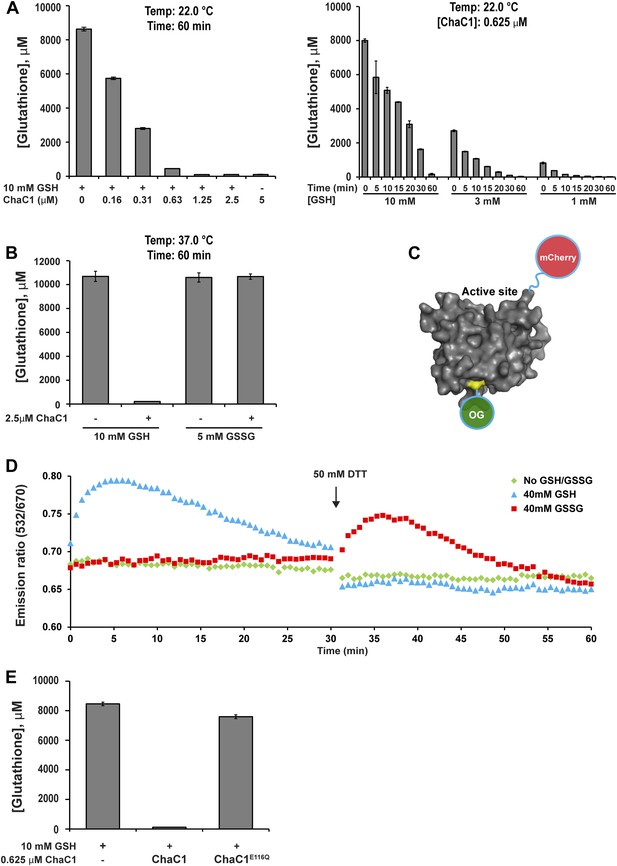
ChaC1 efficiently and selectively degrades reduced glutathione.
(A) A bar-graph representation of residual glutathione levels following incubation of 10 mM glutathione with the indicated concentrations of bacterially expressed mouse ChaC1. Varying concentrations of enzymes were assayed at a single time point (left panel) and varying initial concentrations of glutathione were assayed at different time points (right panel). (B) Comparison of the ability of ChaC1 to eliminate reduced (GSH) and oxidized glutathione (GSSG). Note that ChaC1 effectively eliminated reduced glutathione, but had no effect on oxidized glutathione. (C) Cartoon of the fluorescent resonance energy transfer (FRET) probe, OG-ChaC1-Cherry, used to detect substrate binding to ChaC1. Shown is a model of murine ChaC1 (UniProt Q8R3J5) residues 31–204, created by Phyre2 (Kelley and Sternberg, 2009) based on the crystal structure of γ-glutamyl cyclotransferase (PDB 2RBH). The side chain of Cys 92, which has been modified with the Oregon Green (OG) donor, is highlighted, as is the C-terminus of the protein, site of the fused mCherry fluorescent acceptor. (D) Time-resolved FRET signal (expressed as the ratio of the emission signal at 532 nm and 670 nm upon excitation at 480 nm) of the OG-ChaC1-Cherry probe [2.5 µM] following exposure to 10 mM reduced (GSH) or oxidized glutathione(GSSG). Where indicated, the sample was injected with dithiotreitol (DTT) to reduce the GSSG and convert it to a substrate for ChaC1. The biphasic change in FRET signal upon exposure to GSH is consistent with binding followed by breakdown of GSH by the probe, which retains its enzymatic activity. (E) Comparison of glutathione elimination by purified bacterially expressed wild-type and E116Q mutant ChaC1 in vitro.
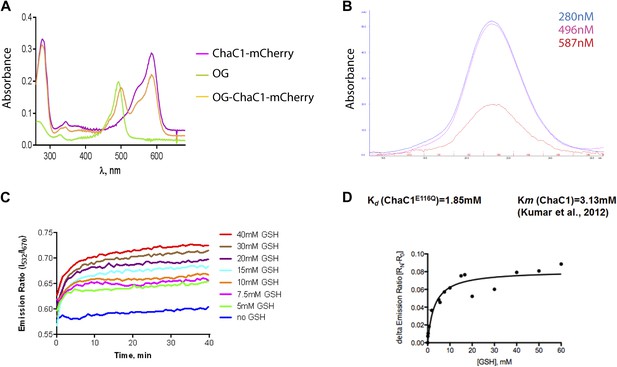
Analysis of the substrate binding properties of ChaC1.
(A) Absorbance spectrum of ChaC1-mCherry, Oregon Green (OG), and Oregon green-labeled ChaC1-mCherry. (B) Absorbance profile of size-exclusion chromatogram of Oregon green-labeled ChaC1 mCherry. Note the coincidence of the absorption peak for protein (280 nm), Oregon Green (496 nm) and mCherry (587 nm) (C) FRET signal of an enzymatically inactive OG-ChaC1E116Q-Cherry probe upon exposure to varying concentrations of reduced glutathione. Note the mono-phasic change in FRET signal, consistent with inability of the mutant enzyme to break down glutathione and the contrast with wild-type OG-ChaC1-mCherry (Figure 1D). (D) Graph of the relationship between the steady-state FRET signal of the OG-ChaC1E116Q-Cherry probe and the concentration of reduced glutathione (GSH).
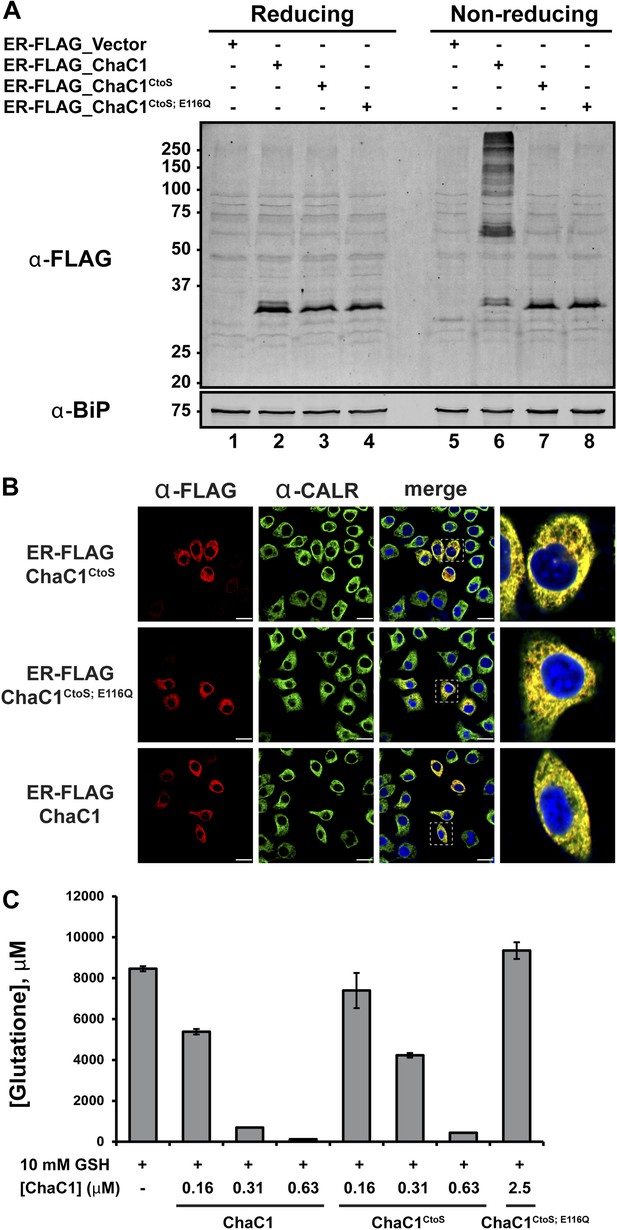
Cysteine-free ChaC1 is an active enzyme that can be targeted to the endoplasmic-reticulum.
(A) Immunoblot of FLAG M1-tagged ER-localized wild type (WT), cysteine-free (CtoS), and compound CtoS; E116Q enzymatically dead ChaC1 in N-ethyl maleimide-blocked lysates of transfected HeLa cells. Lanes 1–4 are from a reducing and lanes 5–8 are from a non-reducing SDS-PAGE. Note the presence of high-molecular weight disulfide linked FLAG-tagged ChaC1 in cells transfected with the WT ER-targeted protein that is absent from those transfected with the cysteine-free, CtoS mutants. (B) Fluorescent photomicrographs of HeLa cells transfected with the indicated expression plasmids and immunostained for the FLAG tag (marking ChaC1) and calreticulin (CALR) as an ER marker. The merge panels show an overlap of the FLAG, CALR, and Hoechst 33,258 signal (to reveal the nuclei) at 630X with a close-up view in the right-most panel. (C) A bar-graph representation of residual glutathione levels following 60-min incubation of 10 mM glutathione with the indicated concentrations of bacterially expressed wild-type ChaC1, its cysteine free derivative, ChaC1CtoS, and its inactive mutant ChaC1CtoS;E116Q.
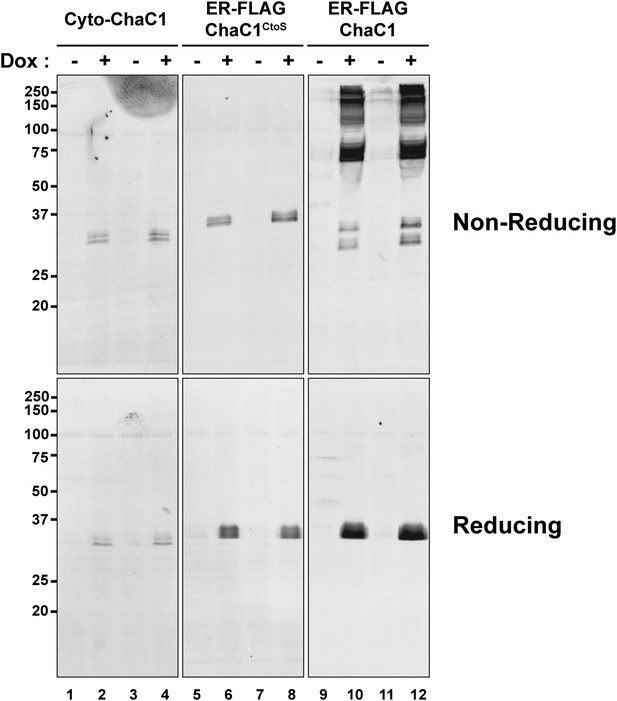
Replacement of cysteines with serines circumvents aberrant disulfide bond formation in ER-localized ChaC1.
Immunblot of FLAG-tagged authentic, cytosolic mouse ChaC1, ER-localized ChaC1 and ER-localized cysteine-free ChaC1CtoS in lysates of stably-transfected Flp-In T-REx HEK 293T cells expressing the proteins under the control of a tetracycline-inducible promoter. Shown are samples blocked with N-ethyl maleimide at the time of lysis and resolved by reducing and non-reducing SDS-PAGE. Where indicated the cells had been exposed to doxycycline to induce expression of the heterologous protein.
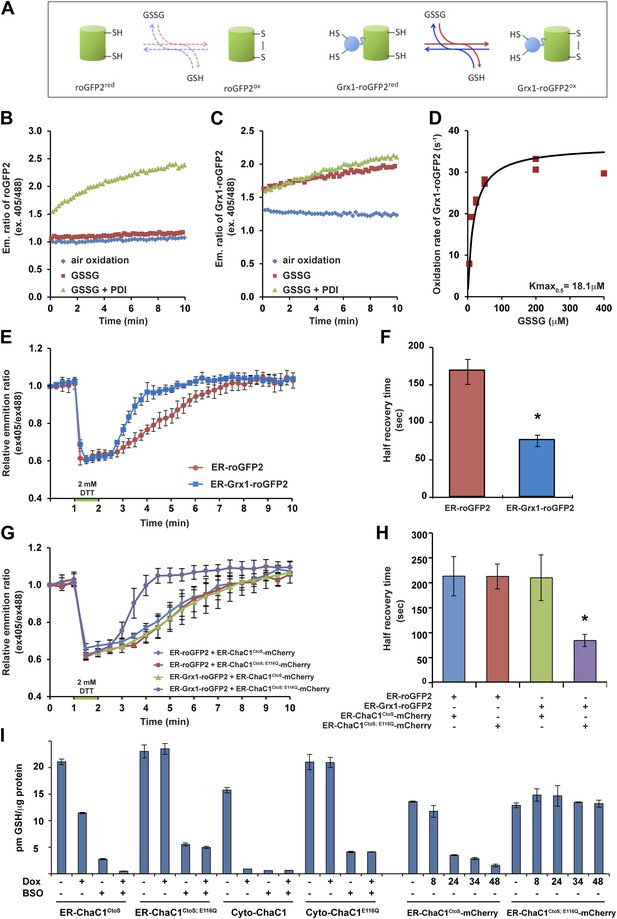
ER-targeted ChaC1 purges the organelle of its glutathione content.
(A) Cartoon contrasting the slow coupling of roGFP2 with the glutathione redox buffer (dashed lines) and the rapid coupling of Grx1-roGFP2 with the glutathione redox buffer (after Gutscher et al., 2008). (B) Trace of time-dependent changes in the ratio of reduced to oxidized roGFP2 detected optically as the ratio between the emission signal (at 535 nm) upon excitation at 405 nm vs 488 nm (ex 405/488) following introduction of the fully reduced probe into the indicated solutions of oxidized glutathione (GSSG) or PDI and GSSG. (C) Similar trace of reduced Grx1-roGFP2. Note the indifference of roGFP2 and the marked responsiveness of Grx1-roGFP to oxidized glutathione. (D) Graph of the initial velocity of Grx1-roGFP2 oxidation as a function of GSSG concentration, fitted to Linweaver–Burk plot. Half-maximal velocity is observed at 18 µM GSSG. (E) Trace of time-dependent changes in the ratio of oxidized and reduced roGFP2 and Grx1-roGFP2 probes expressed in the ER of HeLa cells following a brief (1 min) reductive pulse with dithiothreitol (DTT, 2 mM) followed by a washout. (F) Bar diagram of the half-time to recovery of oxidized roGFP2 and Grx1-roGFP2 following the reductive DTT pulse. Shown are means ±SD (N = 4, *p<0.01). (G) Trace of time-dependent changes in the ratio of oxidized to reduced probes expressed in the ER of HeLa cells alongside active or inactive ChaC1 (tagged at its C-terminus with mCherry to allow visualization of cells co-expressing the redox probes and the glutathione-depleting enzyme) following a brief reductive pulse with dithiothreitol and a washout. Note that the expression of active ChaC1 in the ER eliminates the kinetic advantage of Grx1-roGFP2 over roGFP2 in re-oxidation during the recovery from a DTT reductive pulse. (H) Bar diagram of the half-time to recovery of oxidized roGFP2 and Grx1-roGFP2 following the reductive DTT pulse in cells co-expressing active or inactive CHaC1. Shown are means ±SEM (N = 20, *p<0.01). (I) Bar diagram of cellular glutathione levels following 36 hr of doxycycline (DOX) induction of cytosolic and ER localized active and inactive ChaC1 in the absence and presence of concomitant exposure to buthionine-sulfoxide (BSO, 50 µM). Also shown is a time course of total cellular glutathione following induction of active and inactive mCherry-KDEL-tagged ChaC1.
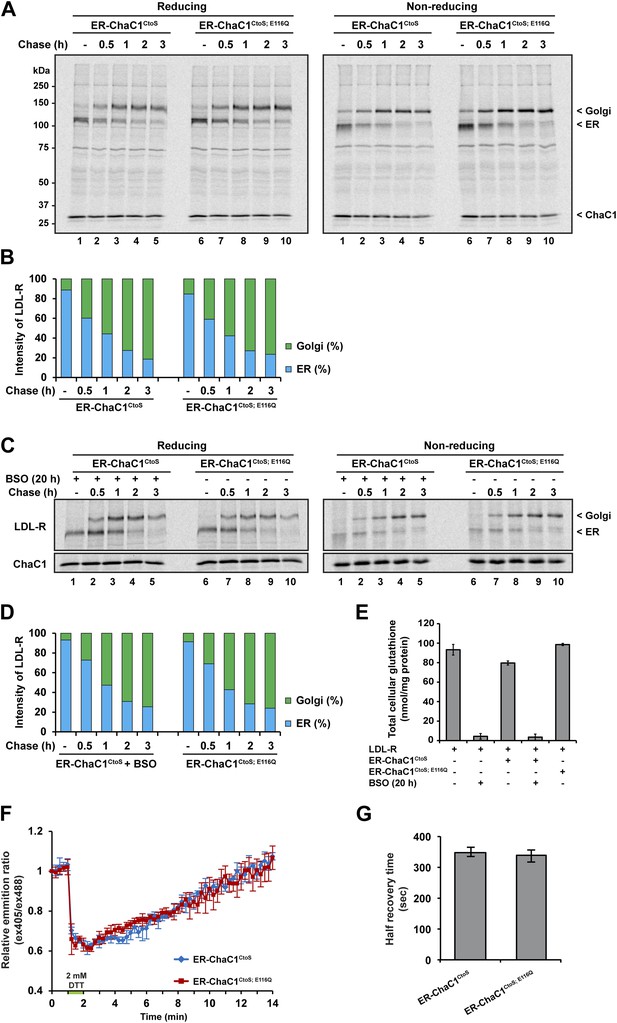
Maturation of the LDL-R receptor is unaffected by depletion of ER glutathione.
(A) Autoradiograph of metabolically labeled LDL receptor (LDL-R) immunopurified from HeLa cells co-expressing the FLAG-tagged LDL-R and ER-localized enzymatically active (ER-ChaC1CtoS) or inactive ChaC1 (ER-ChaC1CtoS;E116Q) and resolved on a reducing SDS-PAGE. Cells were lysed at the end of a 30-min labeling pulse (lanes 1 and 6) or after an additional chase period (indicated). The mobility of the ER and Golgi forms of the LDL-R on reducing (left) and non-reducing (right) SDS-PAGE is indicated, as is the labeled ChaC1, which is also recovered in this anti-FLAG immunoprecipitation. (B) Graphic presentation of the conversion of the ER to Golgi form of the LDL-R from ‘A’ above. Shown is a representative experiment reproduce three times with similar outcome. (C) Autoradiograph of an experiment similar in layout to that depicted in ‘A’ above. Where indicated, the cells were exposed to the glutathione synthesis inhibitor buthionine-sulfoxide (BSO, 100 µM, 20 hr) before the pulse-chase labeling. (D) Graphic presentation of the conversion of the ER to Golgi form of the LDL-R from ‘C’ above. Shown is a representative experiment reproduce three times with similar outcome. (E) Bar graph of total cellular glutathione in cells manipulated as in the experiment described in ‘C’ (shown is the mean ±SD, n = 3, *p<0.05). (F) Trace of time-dependent changes in the ratio of oxidized and reduced roGFP2 expressed in the ER of mouse embryonic fibroblasts with genetic lesions compromising disulfide bond formation (Ero1am/m; Ero1bm/m; Prdx4m/y) following a brief reductive pulse with dithiothreitol followed by a washout. The cells co-expressed ER-localized enzymatically active (ER-ChaC1CtoS) or inactive ChaC1 (ER-ChaC1CtoS;E116Q) marked at its C-terminus with an mCherry fluorescent probe. (G) Bar diagram of the half-time to recovery of oxidized roGFP2 following the reductive DTT pulse in ‘F’ above. Note that expression of active ChaC1 in the ER did not affect the rate of recovery of the sentinel disulfide in ER-localized roGFP2.
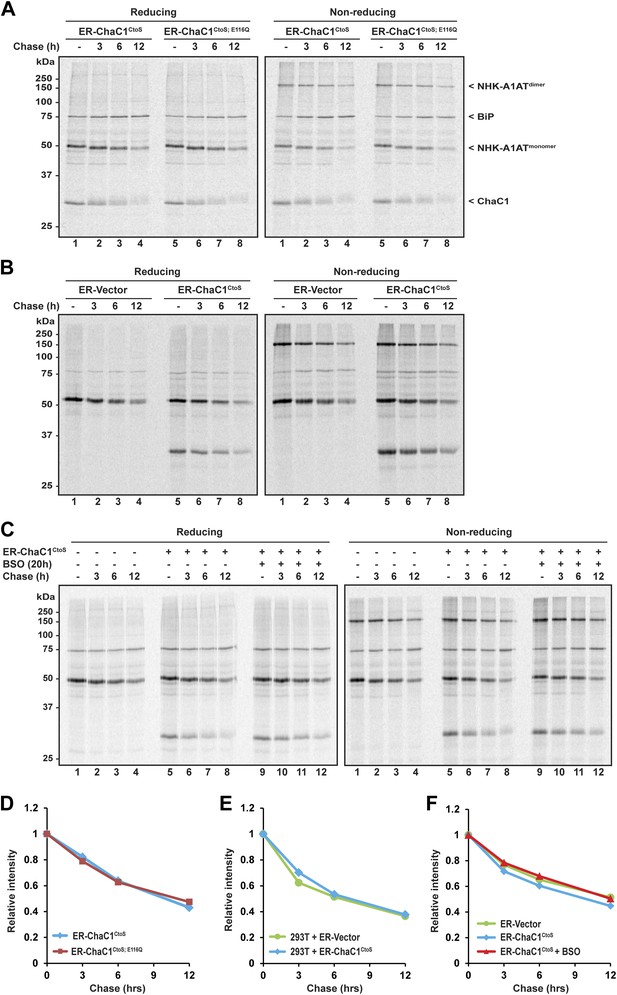
Clearance of misfolded null Hong Kong mutant alpha 1 anti-trypsin (NHK-A1AT) is unaffected by depletion of ER glutathione.
(A) Autoradiograph of metabolically labeled NHK-A1AT immunopurified from HeLa cells co-expressing C-terminally FLAG-tagged NHK-A1AT and ER-localized enzymatically active (ER-ChaC1CtoS) or inactive ChaC1 (ER-ChaC1CtoS;E116Q) and resolved on a reducing or non-reducing SDS-PAGE. Cells were lysed at the end of a 30-min labeling pulse (lanes 1 and 5) or after an additional chase period (indicated). The mobility of the labeled NHK-A1AT, and the ER-chaperone BiP that co-purifies with it, are indicated, as is the labeled ChaC1, which is also recovered in this anti-FLAG immunoprecipitate. (B) Autoradiograph of samples recovered from 293T cells in an experimental design as in ‘A’. (C) Autoradiograph of samples recovered from HeLa cells in an experimental design as in ‘A’. Where indicated, cells were exposed to the glutathione synthesis inhibitor buthionine-sulfoxide (BSO, 100 µM, 20 hr) before the pulse-chase labeling. (D–F) Plot of time-dependent change in NHK-A1AT (monomer) signal from the reducing gels A–C above. Shown are representative experiments reproduced twice with similar outcome.
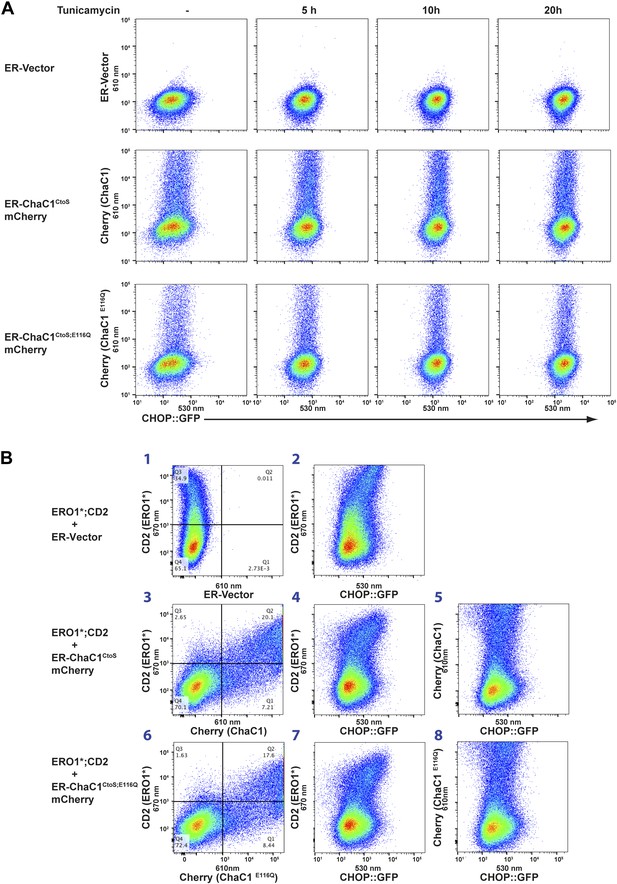
Activity of the unfolded protein response (UPR) is unaffected by depletion of ER glutathione.
(A) Two-dimensional plots (FACScans) of fluorescent intensity of individual CHO cells containing a stably-integrated UPR reporter, CHOP::GFP (Novoa et al., 2001), following transfection with plasmids encoding ER-localized, active (ER-ChaC1CtoS-mCherry-KDEL) or inactive ChaC1 (ER-ChaC1CtoS;E116Q-mCherry-KDEL). GFP fluorescent intensity, reporting on the activity of the UPR (X-axis), was detected at 530 ± 30 nm following excitation at 488 nm, whereas mCherry fluorescent intensity, reporting on the level of ChaC1-mCherry-KDEL in the ER (Y axis), was detected at 610 ± 20 nm following excitation at 561 nm. Where indicated the cells were exposed to the ER stress-inducing agent tunicamycin for the indicated period of time. (B) Three-color FACScans as in ‘A’ of CHOP::GFP cells co-transfected with expression plasmids for a hyperactive mutant of ERO1 (C104A, C133A; ERO1*) tagged by the cell-surface marker CD2 (decorated with an AF647-tagged antibody and detected by excitation at 640 nm and emission at 670 nm) with a wildtype or inactive mutants of ChaC1 fused to mCherry, as noted above. The axis of the scans are labeled with the cognate signals and panels numbered for ease of reference in the text.
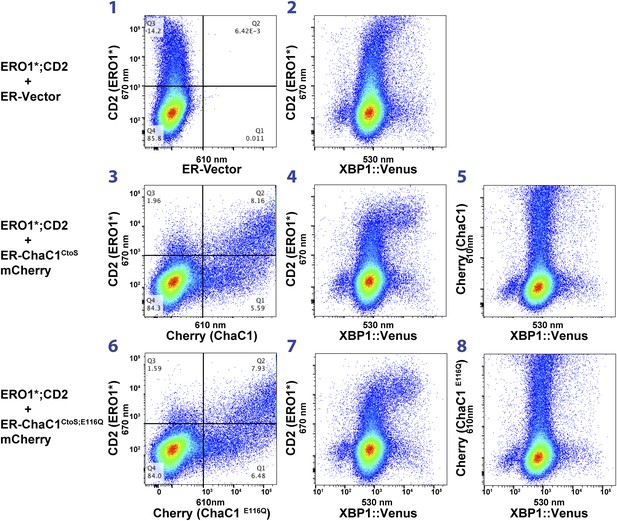
Activity of the unfolded protein response (UPR) as measured by an XBP1-splicing reporter is also unaffected by depletion of ER glutathione.
FACScans as in ‘6B’ of CHO cells stably transduced with an XBP1 splicing reporter linked to the expression of the fluorescent protein Venus (Iwawaki et al., 2004). Where indicated cells were co-transfected with expression plasmids for a hyperactive mutant of ERO1 (C104A, C133A; ERO1*) tagged by the cell-surface marker CD2 and wild-type or inactive mutants of ChaC1 fused to mCherry. The axis of the scans is labeled with the cognate signals.
Tables
List of the plasmids used in this study, their unique lab identifier, lab name, description, PMID of the relevant reference (if available), figure in which they first appear and cognate label in figure legend
| ID | Plasmid name | Description | Reference | First appearance | Label in figure |
|---|---|---|---|---|---|
| 15 | pFLAG-CMV1 | Mammalian expression bovine trypsinogen signal peptide-FLAGM1 fusion | PMID: 8024796 | 2A | ER-FLAG_vector |
| 242 | roGFP2_pRSETB | Bacterial expression of 6X His-tagged roGFP2 | PMID: 14722062 | 3A | roGFP2 |
| 836 | mChac1_1-224-H6-pET30a | Bacterial expression of mouse Chac1 C-terminal His-tagged | This paper | 1A | ChaC1 |
| 888 | pFLAG_mCherry_KDEL_CMV1 | ER localised FLAGM1-mCherry-KDEL in pFLAG-CMV1 | This paper | 6A | ER-vector |
| 915 | mChaC1_1-224_CtoS_pET30a | Bacterial expression mouse Chac1 CtoS (C92S, C169S, C190S, C212S) C-terminal His-tagged | This paper | 2C | ChaC1_CtoS |
| 932 | mChaC1_1-224_E116Q_pET30a | Bacterial expression of E116Q mutant mouse Chac1 C-terminal His-tagged | This paper | 1E | ChaC1_E116Q |
| 934 | mChaC1_3XFLAG_pCDNA5_FRT_TO | Mammalian expression of C-term FLAG-tagged mouse ChaC1 | This paper | 3H | Cyto-ChaC1 |
| 937 | mChaC1_CtoS_E116_pET30a | Bacterial expression of E116Q cysteine to serine mutant mouse Chac1 C-terminal His-tagged | This paper | 2C | ChaC1_CtoS_E116Q |
| 945 | mChaC1_E116Q_3XFLAG_pCDNA5_FRT_TO | Mammalian expression of C-term FLAG-tagged mouse ChaC1 E116Q mutant (Cyto-ChaC1_E116Q) | This paper | 3H | Cyto-ChaC1_E116Q |
| 950 | FLAGM1_mChaC1_CtoS_pCDNA5_FRT_TO | Mammalian expression ER-localised FLAG M1 tagged mouse CHAC1 CtoS (C92S, C169S, C190S, C212S) KDEL | This paper | 2A | ER-FLAG_ChaC1_CtoS |
| 951 | FLAGM1_mChaC1_CtoS_E116Q_pCDNA5_FRT_TO | Mammalian expression ER-localised FLAG M1 tagged mouse CHAC1 CtoS (C92S, C169S, C190S, C212S) E116Q KDEL | This paper | 2A | ER-FLAG_ChaC1_CtoS_E116Q |
| 974 | FLAGM1_mChaC1_WT_pCDNA5_FRT_TO | Mammalian expression ER-localised FLAG M1 tagged mouse CHAC1 KDEL | This paper | 2A | ER-FLAG_ChaC1 |
| 988 | FLAGM1_mChaC1_CtoS_mCherry_pCDNA5_FRT_TO | Mammalian expression ER-localised FLAG M1 tagged mouse CHAC1 mCherry-KDEL, CtoS (C92S, C169S, C190S, C212S) | This paper | 3F | ER-ChaC1_CtoS_mCherry |
| 993 | mChaC1_CtoS_92C_mCherry-pET30a | Bacterial expression of mouse ChaC1-mCherry fusion, C-terminal His-tagged, (C169S, C190S, C212S) | This paper | 1D | OG-ChaC1-Cherry probe |
| 1028 | FLAGM1_mChaC1_CtoS_E116Q_mCherry_pCDNA5 | Mammalian expression ER-localised FLAG M1 tagged mouse CHAC1 mCherry-KDEL, CtoS, E116Q | This paper | 3F | ER-ChaC1_CtoS_E116Q_mCherry |
| 1037 | mChaC1_CtoS_S92C_E116Q_mCherry-pET30a | Bacterial expression of mouse ChaC1-mCherry fusion, C-terminal His-tagged, (C169S, C190S, C212S), E116Q | This paper | S1C | OG-ChaC1_E116Q-Cherry |
| 1052 | ER_roGFP2_pCDNA3.1 | ER localized roGFP2 KDEL | This paper | 3D | ER-roGFP2 |
| 1063 | ER_Grx1_roGFP2_KDEL_pCDNA3.1 | ER localized Grx1 fused to roGFP2 KDEL | PMID:23424194 | 3D | ER-Grx1-roGFP2 |
| 1181 | hLDLR_3XFLAG_pCDNA5_FRT | Mammalian expression plasmid of human LDL receptor, cytosolic tail tagged with a 3X FLAG tag | PMID:12493918 | 4A | FLAG-tagged LDL-R |
| 1204 | A1AT_NHK_3XFLAG_pCDNA5_FRT_TO | Mammalian expression plasmid of null Hong-Kong mutant a1-antitrypsin C-terminally tagged with 3XFLAG | PMID:12736254 | 5A | FLAG-tagged NHK-A1AT |
| 1206 | Grx1_roGFP2_pET30a | Bacterial expression of Grx1-roGFP2 fusion protein | PMID: 18469822 | 3B | Grx1-roGFP2 |
| 1239 | pCAX-F-XBP1ΔDBD-venus | XBP1 mini-cDNA with Venus fused the post-IRE1 spliced open reading frame | PMID: 14702639 | 6C | XBP1-Venus |
| 1273 | hERO1A_C104A_C131A_pCDNA3-CD2 | Mammalian expression plasmid encoding human hyperactive ERO1L (ERO1a) and a co-expressed human CD2 FACS marker | PMID: 23027870 | 6B | ERO1* |





