Domain–domain interactions determine the gating, permeation, pharmacology, and subunit modulation of the IKs ion channel
Figures
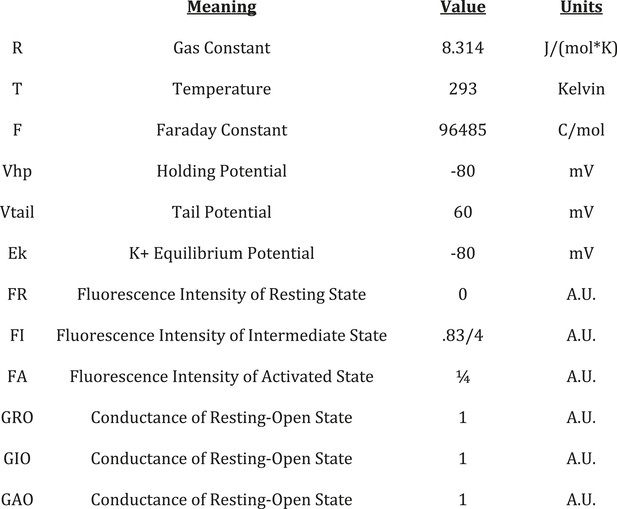
KCNE1 suppresses the intermediate-open state of KCNQ1.
(A–H) Fluorescence (green) and current (black) signals from Xenopus oocytes injected with cRNA encoding pseudo-WT (C214A/G219C/C331A) KCNQ1 alone (KCNQ1, A–D) or coinjected with cRNAs encoding pseudo-WT KCNQ1 and KCNE1 (KCNQ1+KCNE1, E–H). The cells were labeled with Alexa 488 C5-maleimide. (A and E) GV and FV relationships (solid) with the main and high voltage FV components plotted (dotted lines). (B and F) normalized fluorescence and current responses to a 60 mV pulse shown with fits (thin grey lines) to a single- or bi-exponential function. Averaged fast (C and G) and slow (D and H) tau values of fluorescence and current responses to various voltage pulses. (I) Intermediate- (E1-R2, top) and activated- (E1-R4, bottom) state homology models of KCNQ1 after 100 ns of MD simulation. Side view of one VSD (left) and bottom view of the pore (right). (J) Currents from the cells expressing E160R/R231E (E1R/R2E, top) or E160R/R237E (E1R/R4E, bottom) both alone (−KCNE1, middle) or with KCNE1 (+KCNE1, right).
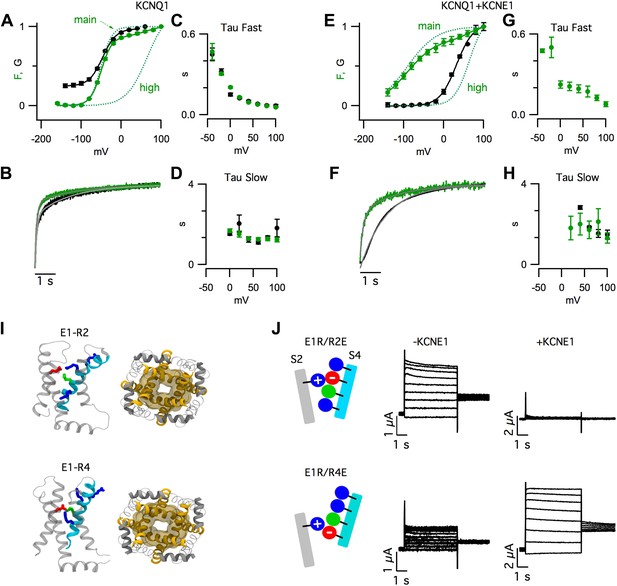
Improved resolution of Fhigh.
(A and B) VCF data from cells expressing pseudo-WT KCNQ1, labeled with Alexa 546 C5-maleimide (KCNQ1 [Alexa 546]). (C and D) VCF data from pseudo-WT KCNQ1+R67E/K69E/K70E KCNE1 labeled with Alexa 488 C5-maleimide (KCNQ1+RKK/EEE [Alexa488]). (A and C) Normalized current and fluorescence responses to a 60 mV pulse. (B and D) GV and FV relationships (solid) with the main and high voltage FV components plotted (dotted lines). Current signals (black), fluorescence signals (color, red = Alexa 546, green = Alexa 488). The RKK/EEE mutation in KCNE1 causes a leftward shift of Fmain, relative to that of WT KCNE1, which increases the separation between Fmain and Fhigh.
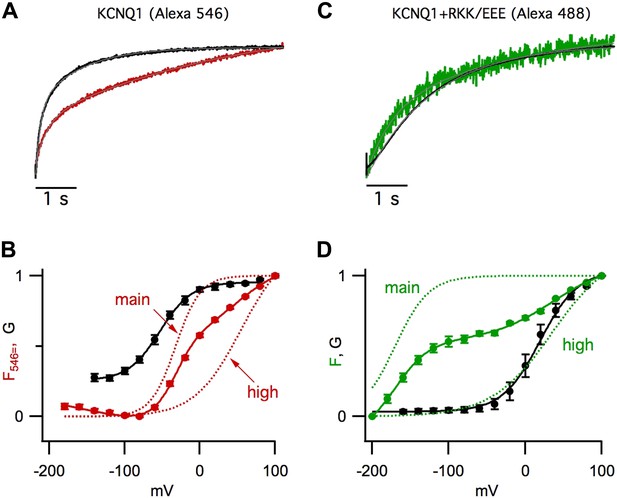
GV/FV relationships are maintained in channels the mutation R243Q in KCNQ1.
GV (solid black) and FV (solid green) relationships for R243Q/psWT KCNQ1 channels expressed alone (R243Q) (A) or with R67E/K69E/K70E KCNE1 (R243Q+RKK/EEE) (B). Green dotted lines show the main and high voltage FV components. The GV relationship of pseudo-WT channels (black dashed lines) is significantly different from that of R243Q; however, the relationship of the GV to different FV components are preserved.
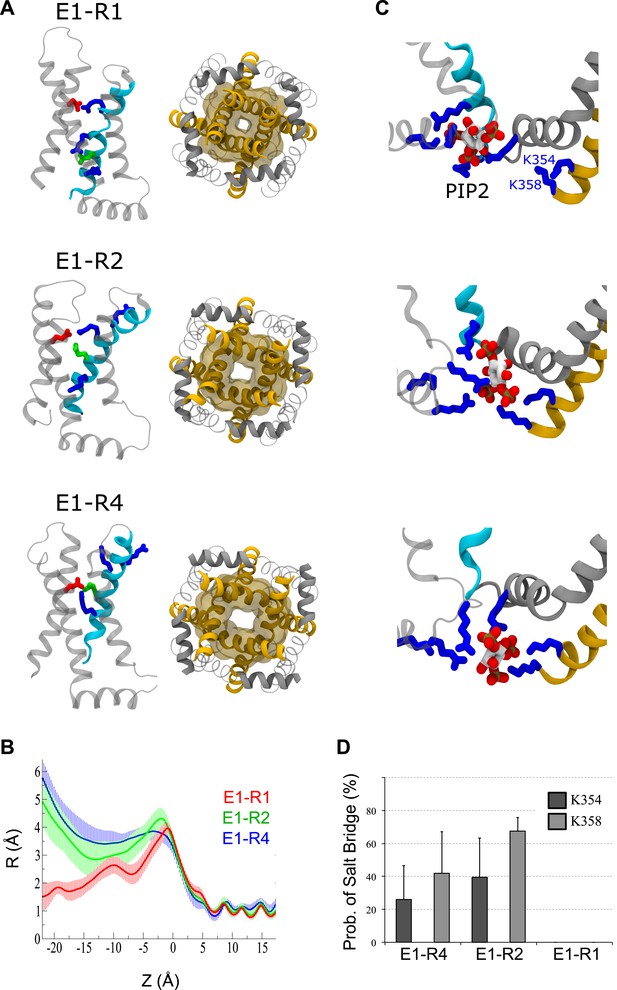
MD simulations predict that, unlike the resting-state, both the intermediate- and activated-states of the VSD stabilize pore opening through state-dependent protein and lipid interactions.
(A) Snapshots of one VSD (left, side view) or the pore domain (right, bottom view) following 100 ns of MD simulations. In the resting, intermediate or activated VSD, E160 (E1, red) forms a salt bridge with R228 (R1), R231 (R2) or R237 (R4), respectively. (B) Averaged (over several trajectories) pore radius vs position along the axis normal to the membrane (Z). (C) Snapshots of the PIP2 intrasubunit-binding site in the three states. In the resting/closed state, PIP2 interacts with positive residues of S4 (cyan). When the VSD is intermediate or activated, PIP2 shifts closer to S6 (yellow) and anchors its positive residues (K354 and K358). (D) Probability of salt bridges formation between positive residues of S6 (K354 and K358) and PIP2. The lipid interacts with S6 only when the VSD is intermediate or activated, not when it is resting. Error bars represent SD. K354 and K358 interactions are not statistically different for the E1-R2 and E1-R4 states. Figure 1—figure supplement 3B represents the averaged pore radius profiles along the axis normal to the membrane (Z). In the activated/open and intermediate states, the minimal radiuses of the pore at this level are 3.5 ± 0.4 Å and 2.8 ± 0.6 Å respectively. For comparison, in the Kv1.2 open state, the corresponding radius (pdb 3LUT [Chen et al., 2010]) is 4.2 Å, in the Kv1.2/2.1 paddle chimera open state (pdb 2 R9R [Long et al., 2007]) it is 4.2 Å also, and in the NavMS open state (pdb 3ZJZ [Loussouarn et al., 2003]) it is 2.3 Å. Therefore, the minimal pore radius at the intercellular gate level in the models of the Kv7.1 activated and intermediate states corresponds to the open pore. In the resting/closed state, this radius decreases to 1.5 ± 0.5 Å. This is similar to the closed states of KcsA (pdb 1K4C [Zhou et al., 2001]), NavAB (pdb 4EKW [Payandeh et al., 2012]) and NavAP (pdb 4DXW [Zhang et al., 2012]), where these values are 1.1, 1.2 and 0.9 Å respectively. The activated/open, intermediate and resting/closed states of Kv7.1 differ by their properties as evidenced from the reported experimental data. Taking advantage of our simulations, we attempted to investigate whether the interactions between PIP2 and positive residues of the Kv7.1 intrasubunit binding site are different. Indeed PIP2 interacts preferably with the VSD (S4) when the channel is resting/closed or with the pore (S6) when the channel is activated/open (Kasimova et al., 2014) (Figure 1—figure supplement 3C, top and bottom panels). In the intermediate state, the lipid forms salt bridges with both S4 (R243) and S6 (K354 and K358) simultaneously (Figure 1—figure supplement 3C, middle panel). Its equilibrium position is also between these in the activated/open and resting/closed states. Interestingly, the probability of interaction between PIP2 and S6 (K354 and K358) is rather high (Figure 1—figure supplement 3D). The average values are slightly higher for the intermediate than for the activated/open states: 40 and 26% for K354, 68 and 42% for K358 respectively. However, this difference is statistically insignificant due to the estimated error bars.
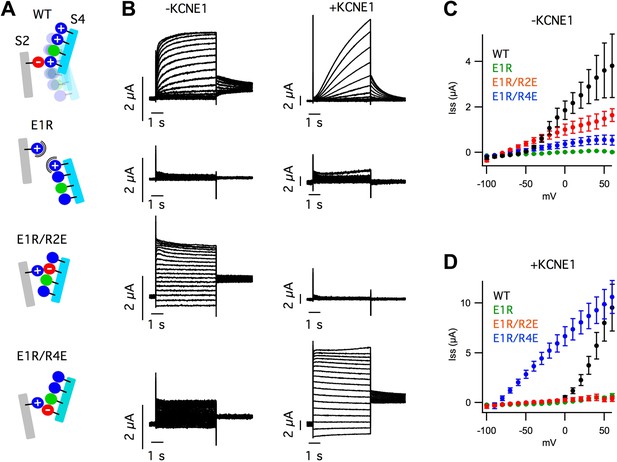
VSD mutations reveal that KCNE1 suppresses currents from intermediate-open states and increases those from activated-open states.
(A) Cartoons illustrating the mutational strategy used to arrest the VSD near the intermediate- and activated-states. The E160R (E1R) mutation was used to disrupt the native electrostatic interactions between the S2 and S4 segments that stabilize the VSD as it undergoes activation. E1R was paired with R231E (R2E) or R237E (R4E) to stabilize the putative intermediate- and activated-states, respectively. (B) Currents from oocytes expressing WT or mutant KCNQ1 subunits alone (−KCNE1, left) or with KCNE1 (+KCNE1, right) in response various voltage test pulses. (C and D) Averaged steady-state current–voltage relationships for cells expressing WT or mutant KCNQ1 subunits alone (C, −KCNE1) or with KCNE1 (D, +KCNE1).
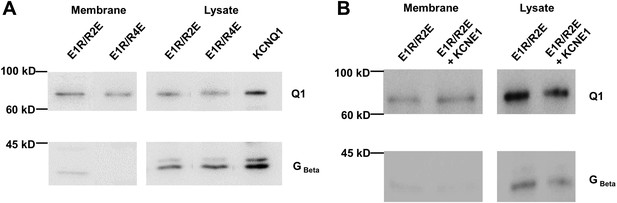
Surface membrane expression of E1R/R2E and E1R/R4E.
Biotinylation of intact oocytes allowed separation of membrane proteins from the cell lysate using streptavidin beads. The membrane fraction and the cell lysate were subjected to Western Blot using antibodies against KCNQ1 (Top) or Gβ, a soluble protein not found in the membrane. (A) E1R/R2E and E1R/R4E reached the cell membrane with similar efficiency. (B) KCNE1 did not decrease the expression of E1R/R2E to the membrane.
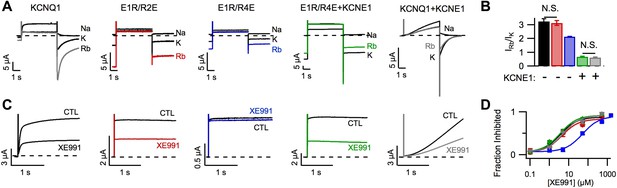
Permeation and pharmacological properties depend on VSD conformation.
Currents from cells expressing WT KCNQ1 alone (KCNQ1, black), E160R/R231E (E1R/R2E, red), E160R/R237E alone (E1R/R4E, blue), E160R/R237E+KCNE1 (E1R/R4E+KCNE1, green), or WT KCNQ1+KCNE1 (KCNQ1+KCNE1, grey). (A) Currents from a single cell in external solutions containing 100 mM of Na+, K+, or Rb+. The currents were elicited by first stepping the voltage to +60 mV for 5 s then to −60 mV for 3 s tails. (B) Averaged Rb+/K+ permeability ratios calculated by comparing the tail current amplitudes. (C). Currents before (CTL) and after (XE991) bath application of 5 μM XE991 in the external solution. (D) Fraction of original current inhibited after 2 s of depolarization vs concentration of XE991 applied shown with fits to the hill equation with a hill coefficient of 1. N.S. = not significant.
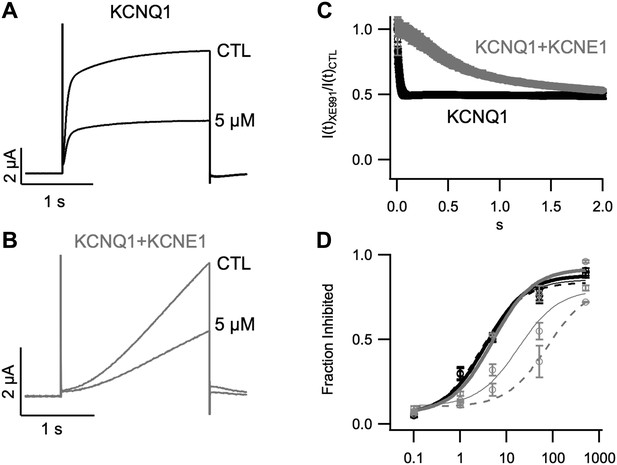
Inhibition of KCNQ1+KCNE1 channels by XE991 develops slowly over time.
Currents in response to a +60 mV test pulse from oocytes expressing KCNQ1 (A) or KCNQ1+KCNE1 (B), recorded in control (CTL) and 5 μM XE991 external solutions. (C) Time dependence of XE991 inhibition—the averaged ratio of the current in 5 μM XE991 to that in control solutions is plotted vs depolarization time. (D) Averaged fraction inhibited after 200 (dashed line), 500 (thin line) or 2000 (thick line) ms of depolarization is plotted vs concentration of XE991 for oocytes expressing WT KCNQ1 (black) or WT KCNQ1+KCNE1 (grey).
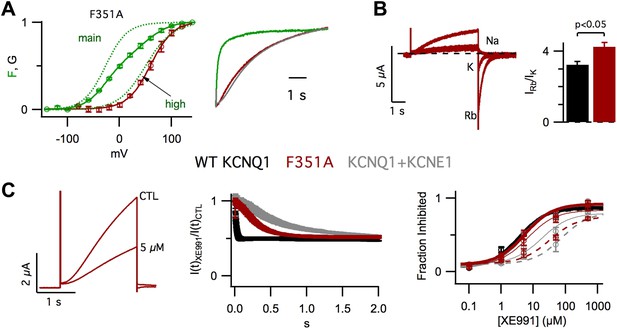
Altering VSD-pore coupling directly, by the mutation F351A, changes the gating permeation, and pharmacology of KCNQ1 channels.
(A) VCF recordings from oocytes expressing pseudo-WT/F351A (C214A/G219C/C331A/F351A), labeled with Alexa 488 C5-maleimide. Left–GV (red) and FV (solid green) relationships with the main and high voltage FV components plotted (dotted green lines). Right–normalized fluorescence (green) and current (red) responses to a 60 mV pulse, the current from a cell expressing pseudo-WT KCNQ1+KCNE1 is shown for comparison (grey). (B) Left–currents from a single oocyte expressing F351A in external solutions containing 100 mM of Na+, K+, or Rb+. Right–averaged Rb+/K+ permeability ratios for WT KCNQ1 (black) and F351A (red). (C) Left–currents from an oocyte expressing F351A in control and 5 μM XE991 external solutions. Middle–time dependence of XE991 inhibition—the averaged ratio of the current in 5 μM XE991 to that in control solutions is plotted vs depolarization time. Right–averaged fraction inhibited after 200 (dashed line), 500 (thin line) or 2000 (thick line) ms of depolarization is plotted vs concentration of XE991 for oocytes expressing WT KCNQ1 (black), F351A (red), or WT KCNQ1+KCNE1 (grey).
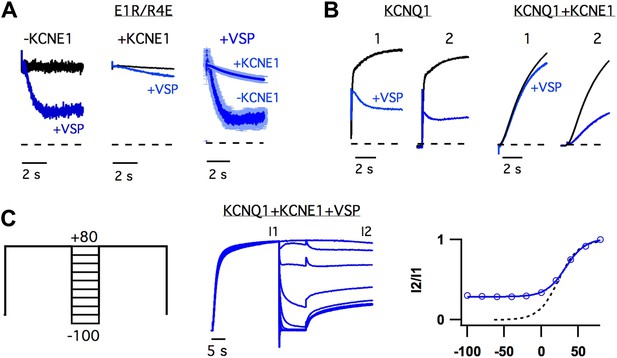
KCNE1 increases the apparent affinity of the activated-open state for PIP2.
(A) Responses of currents from oocytes expressing E1R/R4E alone (left) or E1R/R4E+KCNE1 (middle) to rapid depletion of PIP2 by CiVSP (VSP, blue). The membrane voltage was pulsed to +60 mV to activate CiVSP. Currents were normalized to the value 200 ms after depolarization for comparison with currents from oocytes not expressing CiVSP (black = channel subunits alone, blue = channel subunits + CiVSP). Right–averaged current responses for oocytes expressing E1R/R4E+CiVSP (−KCNE1) or E1R/R4E+KCNE1+CiVSP (+KCNE1). (B) CiVSP responses (VSP, blue) of currents from oocytes expressing WT KCNQ1 or WT KCNQ1+KCNE1. Two +80 mV depolarizing pulses were applied, spaced 30 s apart (note: time scale is broken). The currents were normalized to the value 200 ms into the first depolarizing pulse for comparison with currents from an oocyte expressing channel subunits alone (black). (C) Left–double pulse protocol in which two 25 s depolarizing pulses were applied. In between the two pulses the membrane potential was set to various voltages for 10 s. After each sweep, the membrane potential was held at −80 mV for 300 s to deactivate CiVSP and allow for PIP2 regeneration by the endogenous lipid kinases. Middle–currents from an oocyte expressing KCNQ1+KCNE1+CiVSP subjected to the voltage protocol shown and normalized to the value at the end of the first pulse (I1). Right–fraction of the first pulse current available on the second pulse (I2/I1) is plotted vs voltage of intervening 10 s (blue). The voltage-dependence of I2/I1 (solid blue line) is similar to that of the GV relationship for WT KCNQ1+KCNE1 (dotted black line) suggesting that the open probability during the 10 second interpulse determines the unbinding of PIP2.
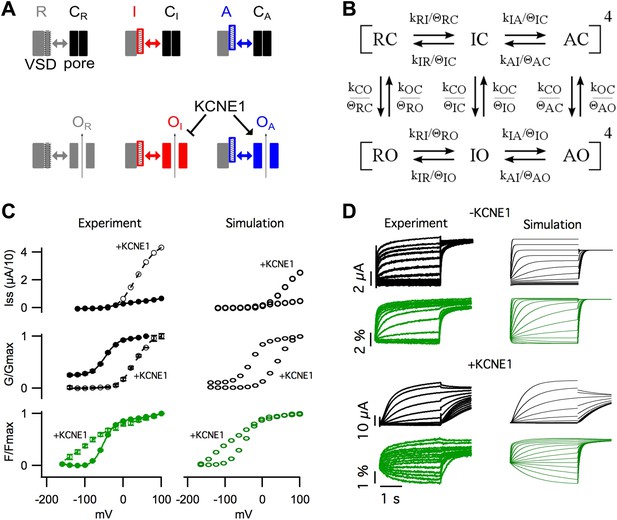
Modeling the effects of KCNE1.
(A) Cartoon illustrating the observed effects of KCNE1 on KCNQ1. The VSDs transit between resting (R, grey patterned), intermediate (I, red patterned), and activated (A, blue patterned) states. Each VSD conformation has unique interactions (double arrow) with the closed (C) and open (O) conformation of the pore. KCNE1 suppresses the intermediate-open (OI) state and modulates the activated-open (OA) states. (B) Kinetic model of KCNQ1 channel gating where the k parameters are the intrinsic transition rates of the VSD and the pore, and the θ parameters explicitly represent VSD-pore interactions. Fourth power notation ([ ]4) indicates that the model includes four VSDs. (C and D) Experimental data and model simulations with (+KCNE1) and without KCNE1. Steady-state current- (C, top), conductance- (C, middle), and fluorescence- (C, bottom) voltage relationships. (D) Current (black) and fluorescence (green) responses at various voltages.
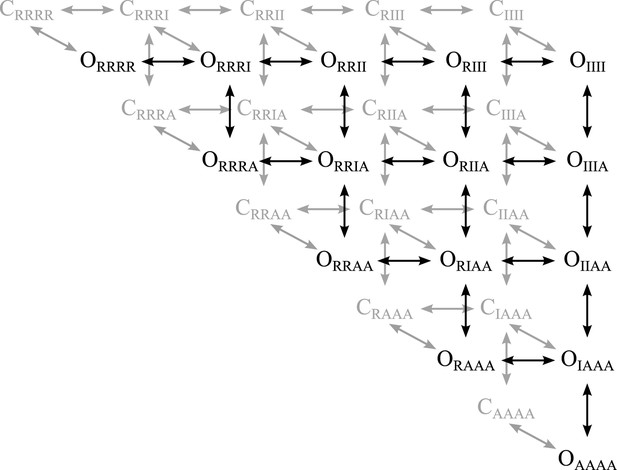
Scheme for voltage-depedendent gating.
The channel is modeled as a pore coupled to four voltage-sensing domains. VSD activation occurs in two transitions, resting (R) to intermediate (I) along the horizontal and intermediate to activated (A) along the vertical. The pore can be fully closed (C, back face, grey) or fully open (O, front face, black), with any combination of VSD-states. The state-notation indicates the conformation of the pore and the combination of the four VSDs (subscripted). For example CRRRR indicates the state where the pore is closed and all four VSDs are resting.
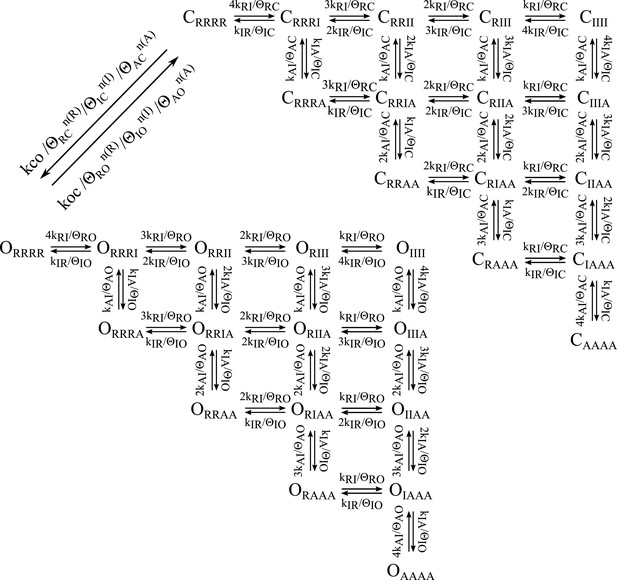
Balanced gating model showing transition rates.
As described in the text the k parameters represent the intrinsic rates of the two domains and the θ parameters represent the effect of VSD-pore interactions at different states. The closed to open transitions dependent on the intrinsic rate of the pore (kco) divided by the closed state interactions with the VSDs. The open to closed transitions depend on the koc divided by the open state interactions the VSDs. n(X) indiciated the number of VSDs in state X.

Desriptions, values and units for free parameters of gating model.
The values represent those used in the kinetic modeling of KCNQ1 gating. ΔKCNE1 column indicates the changes used to simulate the effects of KCNE1 on KCNQ1 channel gating.