A single pair of neurons links sleep to memory consolidation in Drosophila melanogaster
Figures
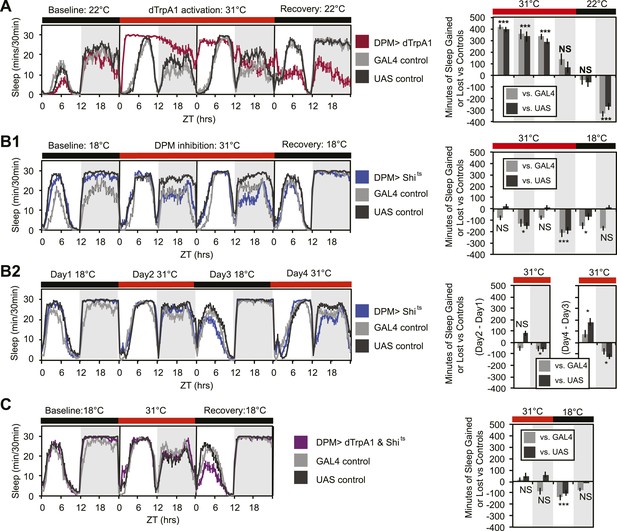
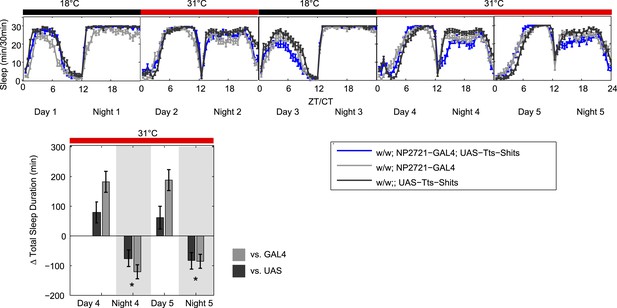
DPM activity and synaptic release are sleep promoting in a cell-autonomous manner.
(A) Flies exhibit large gains in sleep when DPM neurons are activated with dTrpA1 at 31°C (w-; NP2721-GAL4/ UAS-dTrpA1-II). Compensatory sleep loss is apparent during recovery following 2 days of dTrpA1 activation. (B1) Flies show small but significant sleep loss when DPM synaptic release is inhibited with Shits after shift to 31°C (w-; NP2721-GAL4; 20xUAS-IVS-Syn21-Shits). Continuing sleep loss is apparent during the first 12 hr of recovery following Shits inhibition of DPM synaptic release. (B2) Sleep loss can be seen over multiple cycles of temperature shift when DPM synaptic release is inhibited with Shits (w-; NP2721-GAL4; 20xUAS-IVS-Syn21-Shits). For quantification in B2, day 1 was used as a baseline to calculate day 2 sleep changes and day 3 was used as a baseline to calculate day 4 sleep changes. (C) Sleep gains resulting from dTrpA1 activation are fully blocked when DPM synaptic release is inhibited with Shits at 31°C (w-; NP2721-GAL4/ UAS-dTrpA1-II; 20xUAS-Tts-Shits). Left plots show sleep in 30-min bins during a baseline day (22°C for dTrpA1 alone, 18°C for Shits or combined UAS experiments), followed by 1–2 days of DPM hyperactivation or inhibition (31°C) and 1 day of recovery (22°C for dTrpA1 alone, 18°C for Shits or combined UAS experiments). Right plots show a quantification of the 30-min data in 12-hr bins across 1 or 2 days of heating and 1 day of recovery. Sleep change is quantified as the minutes of sleep gained or lost by the experimental genotype in comparison to either the UAS or GAL4 control genotypes during heating and recovery periods. Grey shading indicates the dark period/night, red bars indicate increased temperature. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001, and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Calculation of sleep gain or loss and statistics are described in the ‘Materials and methods’ section.
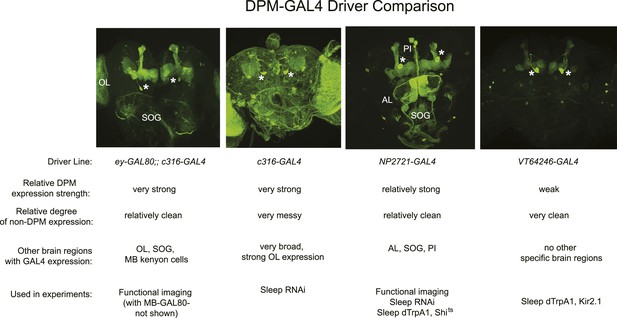
Comparison of DPM-expressing GAL4 lines used in experiments.
Top: representative maximum z-projection images through the central brain of flies from each GAL4 line with the respective promoter-GAL4 driving expression of mCD8-GFP. Asterisks denote DPM cell bodies and brain regions with ectopic GAL4 expression are labeled. Bottom: Relative DPM expression strength, relative degree of non-DPM expression, other brain regions with GAL4 expression and a list of experiments in which each GAL4 line was used. Ectopic expression amongst the three GAL4 lines appears to be non-overlapping, suggesting DPMs are the only common locus of expression in these lines. Although addition of ey-GAL80 greatly reduces background expression of the c316-GAL4 insertion, these flies were less healthy than other lines and thus were only used for functional imaging, but not generally for behavior experiments. Addition of the MB-GAL80 transgene was also necessary in the ey-GAL80;; c316-GAL4 line to suppress a low level of GAL4 expression in MB Kenyon cells (the brain shown above does not contain MB-GAL80). Abbreviations: OL-optic lobe, SOG-subesophageal ganglion, AL-antennal lobe, PI- pars intercerebralis.
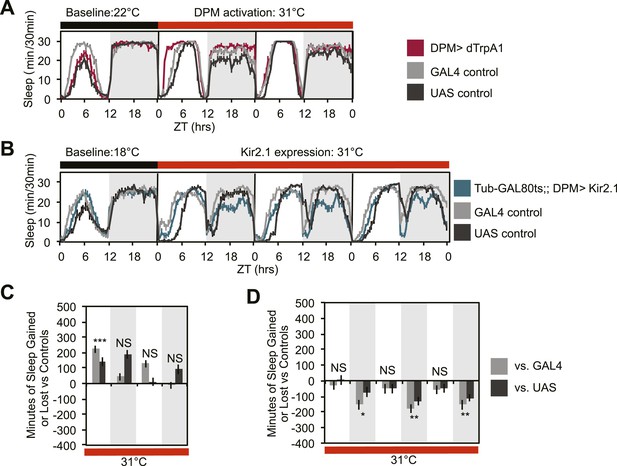
DPM activity regulated by a different GAL4 insertion is also sleep-promoting.
(A and C) Flies exhibit gains in sleep when DPM neurons are activated with dTrpA1 at 31°C using the weak, but very clean GAL4 driver, VT64246-GAL4, to drive dTrpA1 expression (w-; UAS-dTrpA1-II; VT64246-GAL4). (B and D) Flies show nighttime sleep loss when DPM depolarization is suppressed following the conditional expression at 31°C of the hyperpolarizing potassium channel, Kir2.1, driven by the weak, but very clean VT64246-GAL4 driver (w-, Tub-GAL80ts; UAS-Kir2.1; VT64246-GAL4). Top plots (A and B): show sleep in 30-min bins during a baseline day (22°C for dTrpA1, 18°C for Tub-GAL80ts; UAS-Kir2.1), followed by 2–3 days of DPM hyperactivation or repression (31°C). Bottom plots (C and D): show a quantification of the 30-min data (C is a quantification of data in A and D is a quantification of data in B) in 12-hr bins across 2 or 3 days of heating. Sleep change is quantified as the minutes of sleep gained or lost by the experimental genotype in comparison to either the UAS or GAL4 control genotypes during heating. Grey shading indicates the dark period/night, red bars indicate increased temperature. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001 and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Calculation of sleep gain or loss and statistics are described in the ‘Materials and methods’ section.
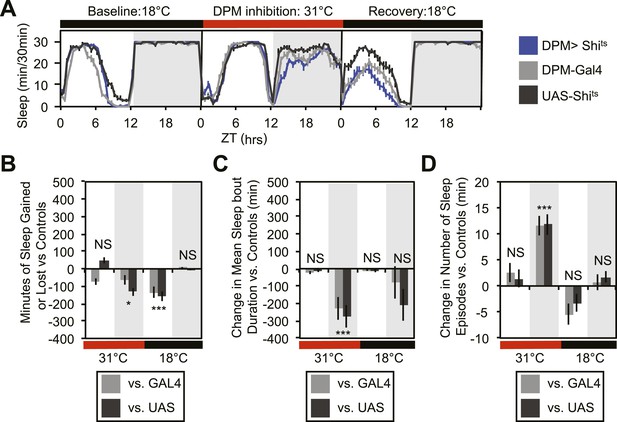
Vesicle release from DPMs promotes consolidated nighttime sleep.
(A and B) Flies show nighttime sleep loss when DPM synaptic release is inhibited with Shits at 31°C (w-, ey3.5-GAL80; MB-GAL80; c316-GAL4/ 20xUAS-Tts-Shits). Continuing sleep loss is apparent during the first 12 hr of recovery following release of Shits inhibition. (C and D) Flies exhibit highly fragmented nighttime sleep during the first night of DPM inhibition at 31°C . This impairment of sleep quality is characterized by a dramatic decrease in mean nighttime sleep bout duration (C) and an increase in the number of nighttime sleep bouts (D). Top (A): shows sleep in 30-min bins during a baseline day (18°C), followed by 1 day of DPM repression (31°C) and 1 day of recovery (18°C). Bottom plots (B, C, and D): show a quantification of 30-min data in 12 hr-day/night bins. Sleep change, as well as changes in mean sleep episode duration and number of sleep episodes, is quantified as the minutes of sleep or number of sleep episodes gained or lost by the experimental genotype in comparison to either the UAS or GAL4 control genotypes during heating. Grey shading indicates the dark period/night, red bars indicate increased temperature. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001 and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Calculation of sleep gain or loss and statistics are described in the ‘Materials and methods’ section.
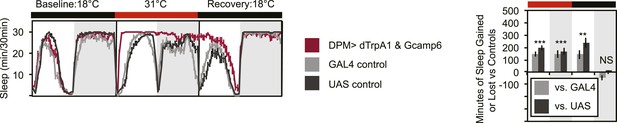
An additional UAS transgene does not prevent dTrpA1-induced sleep gains.
Flies exhibit large gains in sleep when DPM neurons are activated with dTrpA1 at 31°C in the presence of an additional GAL4 target (w-; NP2721-GAL4/ UAS-dTrpA1-II; 20xUAS-IVS-GCaMP6m). Left plot: shows sleep in 30-min bins during a baseline day (18°C), followed by 1 day of DPM hyperactivation (31°C) and 1 day of recovery (18°C). Right plot: shows a quantification of the 30-min data in 12-hr bins across 1 day of heating and 1 day of recovery. Sleep change is quantified as the minutes of sleep gained or lost by the experimental genotype in comparison to either the UAS or GAL4 control genotypes during heating and recovery periods. Grey shading indicates the dark period/night, red bars indicate increased temperature. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001 and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Calculation of sleep gain or loss and statistics are described in the ‘Materials and methods’ section.
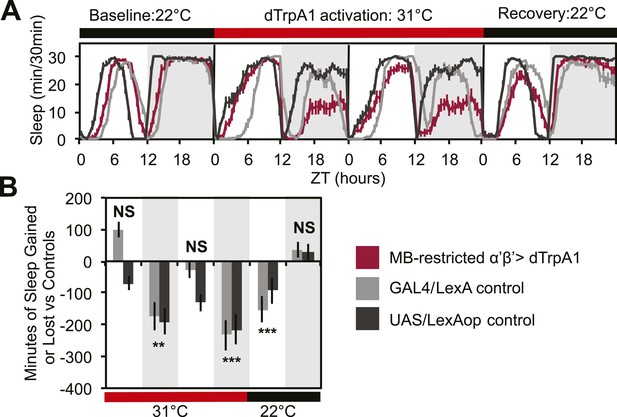
MB α’β’ neuron activity promotes wakefulness.
The α’β’ c305a-GAL4 driver line was crossed to UAS-dTrpA1-II (with c305a expression restricted to the MB) to determine effects on sleep of α’β’ activation. (A) shows sleep in 30-min bins during a baseline day (22°C), followed by 2 days of DPM hyperactivation (31°C) and 1 day of recovery (22°C). (B) shows minutes of sleep gained or lost by the experimental genotype in comparison to either the UAS or GAL4 control genotypes during heating and recovery periods. MB-restricted genotypes are: 1). UAS-dTrpA1, ptub>GAL80>/c305a-GAL4; MB-LexA/LexAop-Flp, 2). c305a-GAL4; MB-LexA (GAL4/LexA control), and 3). UAS-dTrpA1, ptub>GAL80>; LexAop-Flp (UAS/LexAOP control). Grey shading indicates the dark period/night, red bars indicate increased temperature. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001 and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Calculation of sleep gain or loss and statistics are described in the ‘Materials and methods’ section.
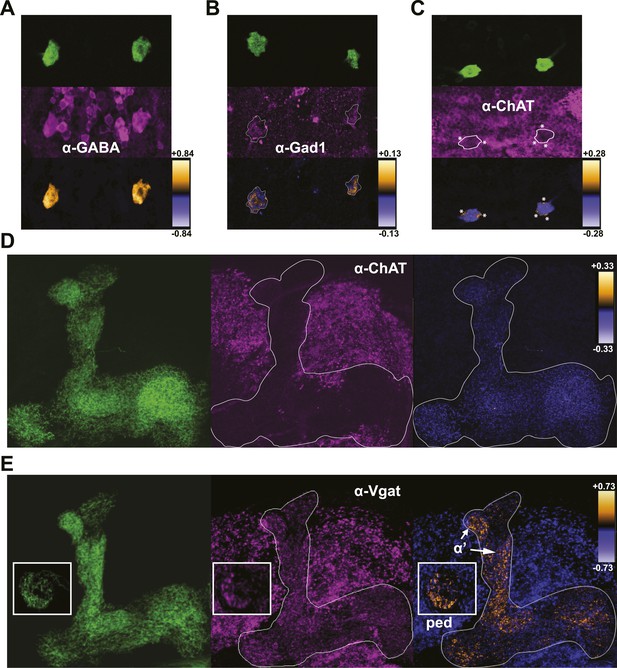
DPMs are GABAergic, but not cholinergic.
(A–C) Top: VT64246-GAL4 was used to drive expression of membrane-localized mCD8-GFP in DPM cell bodies, which was visualized with an anti-GFP antibody. Middle: brains were stained using antibodies against (A) GABA (N = 11/11 cell bodies with positive staining), (B) Gad1 (N = 12/13 cell bodies with positive staining), and (C) ChAT (N = 11/11 cell bodies had no staining). Although a number of neighboring ChAT-positive cell bodies cross over the periphery of the DPM cell bodies resulting in very localized correlation between channels (* in image), the DPMs do not show a general colocalization with anti-ChAT. Bottom: ICA was used to visualize the relative colocalization between DPM>GFP and transmitter staining in pairs of DPMs. (D–E) Left: VT64246-GAL4 was used to drive expression of a presynaptic marker, BRP-short-GFP, in DPM projections to the MB. Middle: brains were stained with antibodies against (D) ChAT (N = 16/16 MB lobe sets with negative staining) and (E) VGAT, with insets showing the MB peduncles (N = 10/10 MB lobe sets with positive staining). Right: ICA was used to build false color maps of relative colocalization between DPM>Brps-GFP and transmitter staining in DPM projections. For ICA, orange indicates colocalization/correlation of pixel intensities between channels (PDM>0) and purple indicates a lack of colocalization/anticorrelation of pixel intensities between channels (PDM<0) relative to the scale shown for each image (see ‘Materials and methods’ for further details). ‘α’’ indicates the MB α’ lobe and ‘ped’ indicates peduncles, shown in the inset.
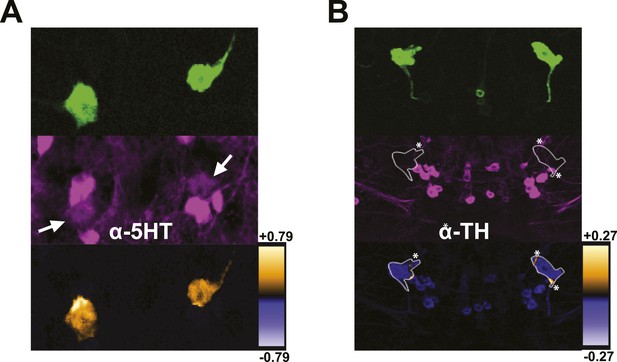
DPMs are serotonergic, but not dopaminergic.
(A–B) Top: VT64246-GAL4 was used to drive expression of membrane-localized mCD8-GFP in DPM cell bodies, which was visualized with an anti-GFP antibody. Middle: Brains were stained with antibodies against (A) 5-HT (N = 6/6 cell bodies with positive staining) and (B) TH (N = 6/6 cell bodies with no staining). Although some TH-positive neurites from other neurons cross over the location of the DPM cell bodies resulting in very localized correlation between channels (* in image), the DPMs do not show a general colocalization with anti-TH. Bottom: ICA was used to visualize the relative colocalization between DPM > GFP and transmitter staining in pairs of DPMs. For ICA, orange indicates colocalization/correlation of pixel intensities between channels and purple indicates a lack of colocalization/anticorrelation of pixel intensities between channels relative to the scale shown for each image (see ‘Materials and methods’ for further details).
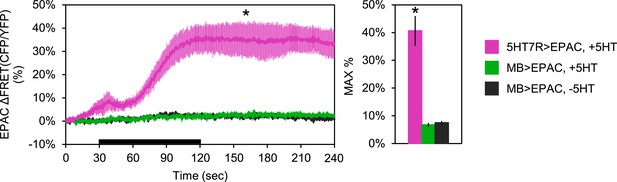
MB neurons do not express stimulatory serotonin receptors.
Mean EPAC response traces of w-; 5HT7-GAL4/+; UAS-EPAC/+ (pink, N = 4), or w-; lexAop-EPAC/+; MB247-lexA/+ (green, N = 10) to 90 s perfusion of 1 mM serotonin (5HT) or vehicle (black, N = 10). Black bar denotes the time of perfusion of 5HT or vehicle. For MB247-lexA, ROIs were taken from both the horizontal and vertical lobes of the MBs. For 5HT7-GAL4, ROIs were taken from the ellipsoid body ring. p = 0.002 for Mann–Whitney U test. Histogram summarizes mean maximum percentage change in EPAC fluorescence, values are 40.6 + 5.3% (pink), 6.7 + 0.6% (green), 7.4 + 0.6% (black).
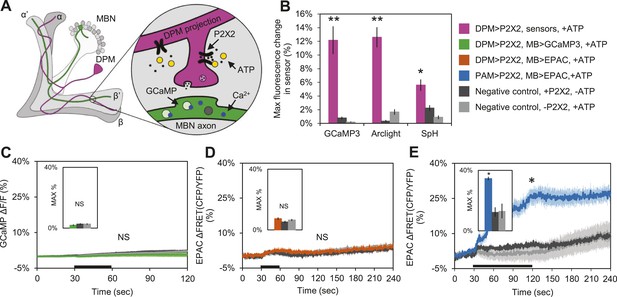
DPM activation has no excitatory effect on the MBs.
(A) Schematic of genetic set-up for functional imaging experiments. P2X2 receptors were expressed in the DPM neurons, and fluorescent sensors (GCaMP shown) were expressed in the MB neurons. (B) Bath-applied ATP is effective at activating DPMs expressing P2X2 receptors. Mean maximum percentage change in GCaMP3.0 (w-, eyeless-GAL80; UAS-GCaMP3.0/+; c316-GAL4/UAS-P2X2, N = 9 with UAS-P2X2 transgene, 8 without [9, 8], p < 0.001 by Mann–Whitney U test), Arclight (w-, eyeless-GAL80; UAS-Arclight/+; c316-GAL4/UAS-P2X2, N = [11,6], p < 0.001 by Mann–Whitney U test), and Synapto-pHluorin (w-, eyeless-GAL80; UAS-Synapto-pHluorin/+; c316-GAL4/UAS-P2X2, N = [11, 6], p = 0.002 for Mann–Whitney U test) fluorescence in horizontal DPM neuron projections in response to 30 s perfusions of 2.5 mM ATP. (C–E) Black bar denotes time of perfusion of 2.5 mM ATP or vehicle. Insets are histograms summarizing the mean maximum percentage change in fluorescence of the respective sensor. The eyeless-GAL80 and MB247-GAL80 transgenes were used to restrict GAL4 driven expression to DPMs. (C) Mean GCaMP3.0 response traces of w-, eyeless-GAL80; lexAop-GCaMP3.0/MB247-GAL80; c316-GAL4, MB247-lexA/UAS-P2X2 (green), or without the UAS-P2X2 transgene (grey), to 30 s perfusion of 2.5 mM ATP or vehicle (black). N = [10, 8], p > 0.05 for Kruskal–Wallis one-way ANOVA, histogram values are 1.9 + 0.5% (green), 2.6 + 0.5% (black), 2.7 + 0.5% (grey). (D) Mean EPAC response traces of w-, eyeless-GAL80; lexAop-EPAC/MB247-GAL80; c316-GAL4, MB247-lexA/UAS-P2X2 (orange), or without the UAS-P2X2 transgene (grey), to 30 s perfusion of 2.5 mM ATP or vehicle (black). N = [10, 8], p > 0.05 for Kruskal–Wallis one-way ANOVA, histogram values are 7.4 + 0.8% (orange), 5.3 + 0.8% (black), 6.6 + 0.7% (grey). (E) MBs respond to excitatory inputs. Mean EPAC response traces of w-; R58E02-lexA/lexAop-P2X2; MB247-GAL4/UAS-EPAC (blue), or without the lexAop-P2X2 transgene (grey), to 90 s perfusion of 2.5 mM ATP or vehicle (black). N = [9, 5], p < 0.001 for Mann–Whitney U test, histogram values are 34.6 + 3.1% (blue), 12.5 + 1.2% (black), 13.2 + 4.8% (grey). (C–E) Traces represent ROIs taken from horizontal sections of MB lobes. (C–D) ROIs were also taken from the vertical lobes and no change in fluorescence was seen (data not shown).
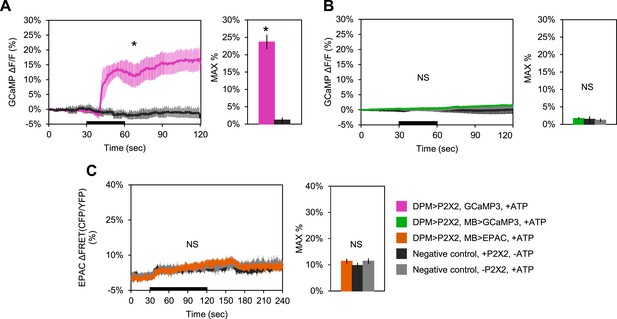
DPM activation has no excitatory effect on the MBs.
(A–C) Black bar denotes time of perfusion of 2.5 mM ATP or vehicle. Histograms summarize mean maximum percentage change in fluorescence of respective sensor. (A) Bath-applied ATP is effective at activating DPMs expressing P2X2 receptors. Mean GCaMP3.0 response traces of w-; UAS-GCaMP3.0/NP2721-GAL4; UAS-P2X2/MB247-lexA to 30 s perfusion of 2.5 mM ATP (pink) or vehicle (black). N = 6 for ATP responses, 3 for vehicle responses, p = 0.02 for Mann–Whitney U test, histogram values are 23.7 + 2.1% (pink), 1.2 + 0.7% (black). (B) Mean GCaMP3.0 response traces of w-; lexAop-GCaMP3.0/NP2721-GAL4; UAS-P2X2/MB247-lexA (green), or without UAS-P2X2 transgene (grey), to 30 s perfusion of 2.5 mM ATP or vehicle (black). N = 5 with UAS-P2X2 transgene, 5 without [5, 5], p > 0.05 for Kruskal–Wallis one-way ANOVA, histogram values are 1.7 + 0.3% (green), 1.5 + 0.8% (black), 1.2 + 0.5% (grey). (C) Mean EPAC response traces of w-; lexAop-EPAC/NP2721-GAL4; UAS-P2X2/MB247-lexA (orange), or without UAS-P2X2 transgene (grey), to 90 s perfusion of 2.5 mM ATP or vehicle (black). N = [8, 6], p > 0.05 for Kruskal–Wallis one-way ANOVA, histogram values are 11.4 + 0.9% (orange), 9.8 + 1.0% (black), 11.4 + 1.1% (grey). (B–C), ROIs were also taken from the vertical lobes and no change in fluorescence was seen (data not shown).
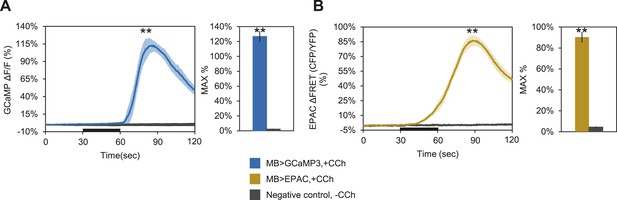
Bath-applied carbachol (CCh) evokes an excitatory response in MBs.
(A) MBs respond to CCh perfusion in the presence of TTX. Mean GCaMP3.0 response traces of w-; NP2721-GAL4/lexAop-P2X2; MB247-lexA/UAS-GCaMP3.0 to 30 s perfusion of 100 μM CCh (blue) or vehicle (black) in 1 μM TTX bath. N = 8, p < 0.001 for Mann–Whitney U test, histogram values are 126.7 + 7.5% (blue), 2.3 + 0.5% (black). (B) Mean EPAC response traces of w-; lexAop-EPAC/+; MB247-lexA/+ to 30 s perfusion of 100 μM CCh (yellow) or vehicle (black) in 1 μM TTX bath. N = 8, p < 0.001 for Mann–Whitney U test, histogram values are 90.3 + 5.0% (yellow), 4.4 + 0.6% (black). (A–B), traces represent ROIs taken from horizontal sections of MB lobes.
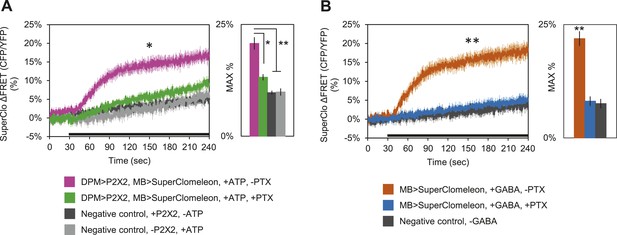
DPM activation has an inhibitory effect on the MBs.
(A) DPM activation evokes a chloride increase in MBs via that can be reduced by picrotoxin (PTX). Mean SuperClomeleon response traces of w-, eyeless-GAL80; lexAop-SuperClomeleon/MB247-GAL80; c316-GAL4, MB247-lexA/UAS-P2X2 to perfusion of 2.5 mM ATP alone (pink, N = 12) or in bath of 10 μΜ PTX (green, 10). Negative controls: mean response to ATP without UAS-P2X2 transgene (grey, 8), or vehicle (black, 12). p < 0.001 between pink and negative controls, p = 0.001 between pink and green, p < 0.01 between green and negative controls for Mann–Whitney U test. Histogram values are 20.8 + 1.4% (pink), 13.2 + 0.7% (green), 9.7 + 0.5% (black), 9.9 + 0.8% (grey). (B) Bath-application of GABA in the presence of TTX evokes a chloride increase in MBs that can be blocked by PTX. Mean SuperClomeleon response traces of w-; lexAop-SuperClomeleon/+; MB247-lexA/+ to perfusion of 1.5 mM GABA alone (orange, 8) or with 10 μΜ PTX (blue, 8), in 1 μM TTX bath. Negative control: Mean response to vehicle 1 μM TTX bath (black, 8). p < 0.001 for Mann–Whitney U test. Histogram values are 21.9 + 1.6% (orange), 8.1 + 1.1% (blue), 7.6 + 1.0% (black). (A–B) Black bar denotes time of perfusion. Histograms summarize the mean maximum percent change in fluorescence of SuperClomeleon. Traces represent ROIs taken from vertical sections of MB lobes. ROIs were also taken from the horizontal lobes and similar results were seen (data not shown).
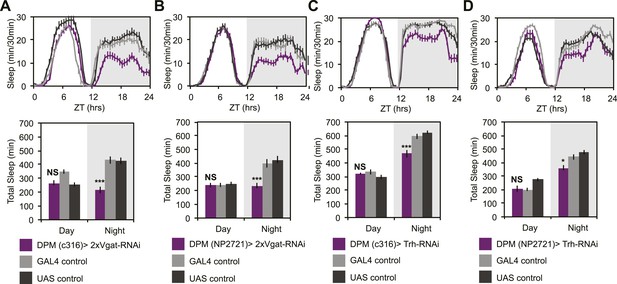
DPM GABA and 5HT promote nighttime sleep.
DPM expression of VGAT was reduced by combining two copies of UAS-VGAT-RNAi with each of two different DPM-GAL4 drivers, c316-GAL4 (A) and NP2721-GAL4 (B). Expression levels of TRH where reduced in DPMs by driving UAS-Trh-RNAi with each of two different DPM-GAL4 drivers, c316-GAL4 (C) and NP2721-GAL4 (D). Top: shows total sleep in 30-min bins averaged across 3 days. Bottom: shows the same data quantified in 12-hr day/night bins. In all cases, a decrease in VGAT or 5HT synthetic enzymes (TRH) in DPMs resulted in nighttime sleep loss, with no change in nighttime activity while awake, although increases in daytime activity while awake were often apparent. Grey shading indicates the dark period/night. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001 and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Statistics are described in the ‘Materials and methods’ section.
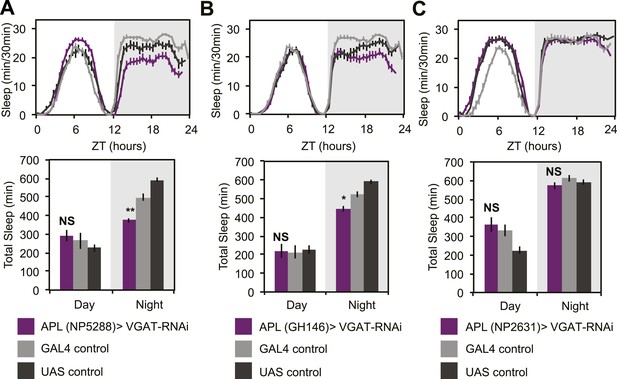
APL GABA can sometimes promote nighttime sleep.
APL expression levels of VGAT were reduced by combining two copies of UAS-VGAT-RNAi with each of three different APL-GAL4 drivers, NP5288-GAL4 (A), GH146-GAL4 (B) and NP2631-GAL4 (C). Top: shows total sleep in 30-min bins averaged across 3 days. Bottom: shows the same data quantified in 12-hr day/night bins. In some, but not all cases, APL>VGAT-RNAi resulted in modest decreases in nighttime sleep, which was often accompanied by concurrent increases in nighttime activity while awake (for NP5288: PGAL4<0.001, PUAS<0.05). Grey shading indicates the dark period/night. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001, and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Statistics are described in the ‘Materials and methods’ section.
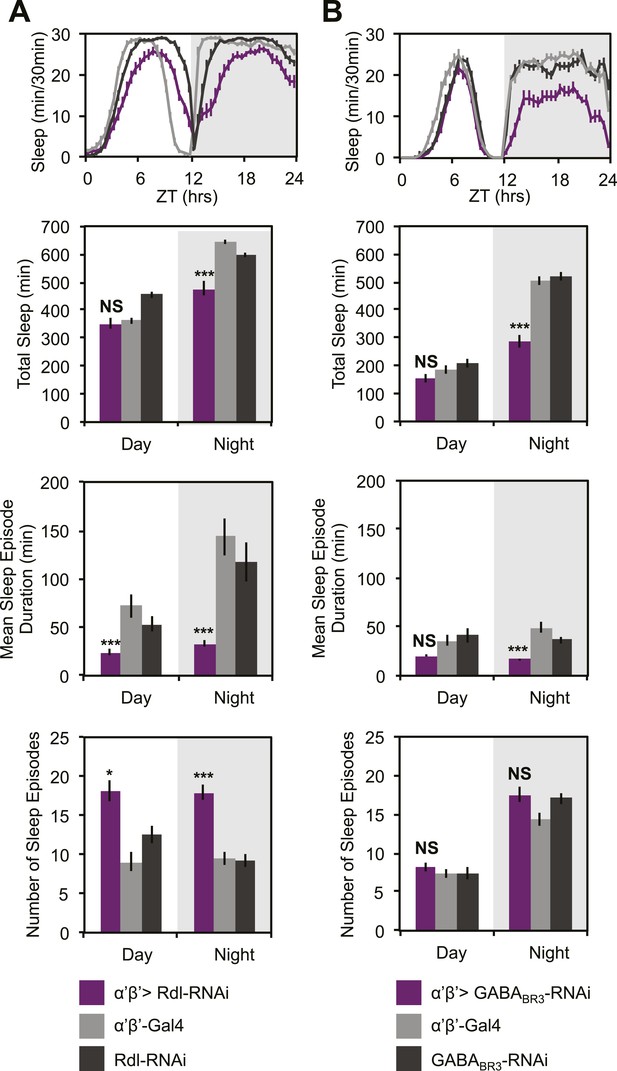
MB α’β’ GABA receptors promote nighttime sleep.
c305a-GAL4 was used to drive expression of Rdl-RNAi (A) or GABABR3-RNAi (B) in the α’β’ neurons. Top: shows total sleep in 30-min bins averaged across 3 days. Middle and bottom plots: show 3-day means of total sleep, mean sleep episode duration and number of sleep episodes quantified in 12-hr day/night bins. α’β’>Rdl-RNAi causes mild sleep loss and increases in nighttime sleep fragmentation, whereas α’β’>GABABR3-RNAi causes greater reductions in total sleep due to a decrease in the average sleep episode length. Grey shading indicates the dark period/night. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001 and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Statistics are described in the ‘Materials and methods’ section.
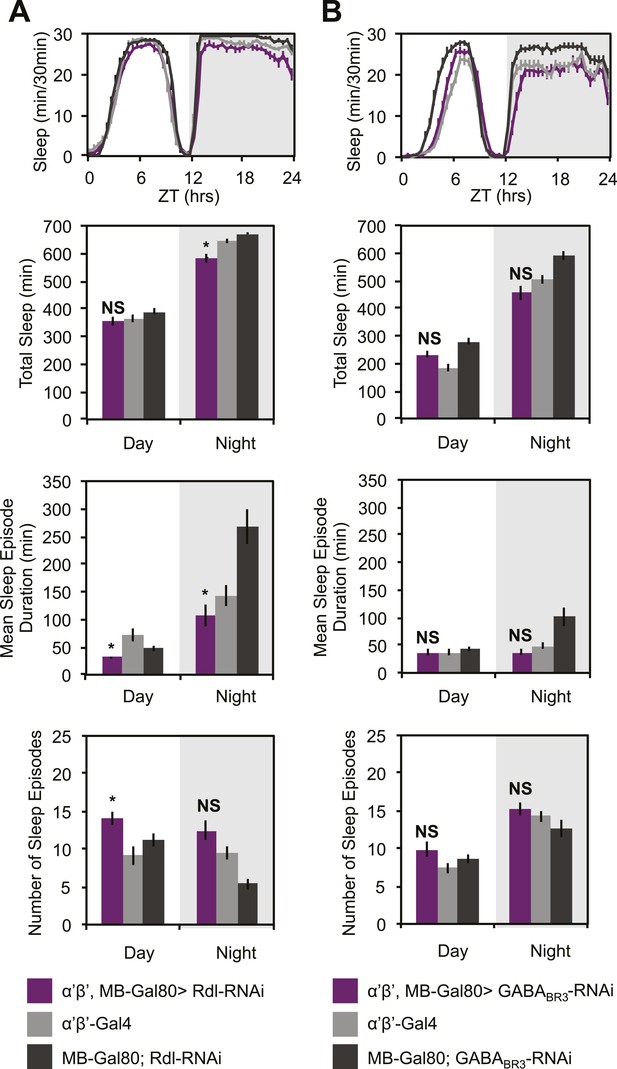
Sleep loss resulting from Rdl/GABABR3-RNAi is primarily due to MB α’β’ expression.
MB-GAL80 restricted c305a-GAL4 was used to drive Rdl-RNAi (A) or GABABR3-RNAi (B) in the c305a expression pattern outside of the MB. Top: shows total sleep in 30-min bins averaged across 3 days. Middle and bottom plots: show 3 day means of total sleep, mean sleep episode duration and number of sleep episodes quantified in 12-hr day/night bins. Non-MB c305a driven Rdl-RNAi results in a lesser degree of sleep loss and fragmentation and non-MB GABABR3-RNAi results in no changes in sleep or sleep structure, indicating these phenotypes are primarily due to MB α’β’ expression of c305a-GAL4. Grey shading indicates the dark period/night. All data are presented as mean ± SEM where * represents p < 0.05, **p < 0.001 and ***p < 0.0001 using the Mann-Whitney-Wilcoxon rank sum test. Statistics are described in the ‘Materials and methods’ section.
Videos
DPM > dTrpA1 activation induces sleep, but not locomotor impairment.
Groups of ten individual female flies with DPM(NP2721)> dTrpA1 (center), UAS-dTrpA1 (left) or DPM (NP2721)-GAL4 (right) were kept at 31°C for 2 hr before video recording. Flies with DPM driven dTrpA1 expression show normal locomotion when gently tapped (0:00:01), but quickly assume a stationary resting position after ∼30 s undisturbed (0:00:35), whereas control flies remain awake and continue to explore the environment. All flies were anesthetized with CO2, counted, and sorted into groups of ten 1 day prior to video recording and kept on food at 22°C prior to heating. Flies were heated at 31°C on food for 2 hr and flipped to empty vials just prior to recording.
Tables
Primer sequences
| Primers (5′-3′) | Sequence (Gateway sequences are in caps and vector sequences are lower case) |
|---|---|
| attB1-13xLexAOP forward | GGGGACAAGTTTGTACAAAAAAGCAGGCTATgcatgcctgcaggttactgtac |
| attB2-IVS reverse | GGGGACCACTTTGTACAAGAAAGCTGGGTAggccgcctgaagtaaaggataag |
| attB2r-Syn21-SuperClomeleon forward | GGGGACAGCTTTCTTGTACAAAGTGGAAAACTTAAAAAAAAAAATCAAAatggtgagcaagggcgagg, |
| attB3-SuperClomeleon reverse | GGGGACAACTTTGTATAATAAAGTTGCttaaagcttcttgtacagctcgtccatg |
Additional files
-
Source code 1
Custom software developed in MATLAB.
- https://doi.org/10.7554/eLife.03868.022