Cellular hallmarks reveal restricted aerobic metabolism at thermal limits
Figures
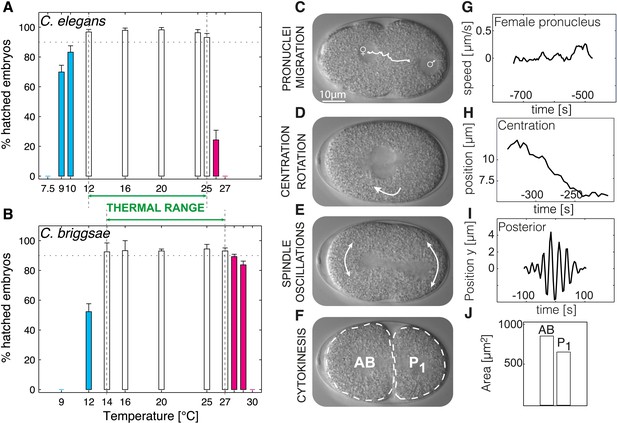
Defining the thermal range and quantifications.
(A and B) Progeny tests were performed on acclimated C. elegans worms from 7.5°C to 27°C and C. briggsae worms from 9°C to 30°C. Dotted line highlights 90% embryonic viability. Temperatures below 20°C exhibiting less than 90% viability are shown in cyan, temperatures above 20°C exhibiting less than 90% viability in magenta. Between panels A and B, we show the thermal range of each species. Error bars show SEM. (C–F) Stills from a time-lapse temperature-controlled DIC microscopy recording of a first-cell stage embryo at the indicated stages (G–J) Examples of feature quantification at the different cellular stages (24°C): female pronucleus speed (G), pronuclei position during centration-rotation (H), spindle pole oscillations (I), as well as areas of the AB (anterior) and P1 (posterior) daughter cells (J). See ‘Materials and methods’ for details on the quantifications. Figure 1—figure supplement 1 shows the temperature control setup. Figure 1—source data 1 lists all the quantified features and their thermal response within and beyond the thermal range.
-
Figure 1—source data 1
Quantified features.
List of features that were quantified and their thermal responses within and beyond the thermal range for C. elegans (N2). Within the thermal range, features were categorized as ‘temperature-dependent’ if the Pearson correlation p-value was below 0.0014 = 0.05/35 (see ‘Materials and methods’ for Bonferroni correction; ‘temperature-independent’ is shown underlined). Beyond the thermal limit, we performed an F-test to determine if the thermal response of the feature was changing compared to within the thermal range (see ‘Materials and methods’; we indicated a change in thermal response when the F-test p-value was below 0.0014, highlighted in bold). Abbreviations: PC: pseudo-cleavage, PM: pronuclear meeting, ME: mitotic entry, T: temperature, C/R: centration-rotation, MT: microtubules. The following features were also quantified but displayed no consistent thermal response both within and beyond the thermal range and hence were not included in the table: anterior-most position at the end of C/R, number of anterior and posterior oscillations, spindle position at the onset of oscillations.
- https://doi.org/10.7554/eLife.04810.004
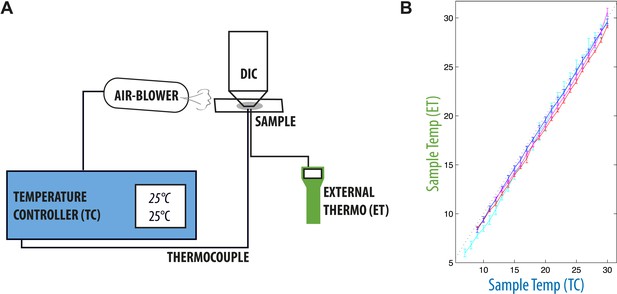
Temperature-control setup.
(A) The temperature within the sample was measured using a thin thermocouple of type K (see ‘Materials and methods’), connected to a temperature controller. This temperature feedback was used to control the temperature of the air blown on the sample and objective, so that sample and set temperatures always matched. (B) We verified that the temperature controller was well calibrated over the range of temperatures of interest by checking the temperature on the sample with yet another thermocouple connected to a separate external thermometer at the same time. We report the measured temperature on the sample by the temperature controller and external thermometer. The measured imprecision was of the order of 0.2°C at all temperatures.
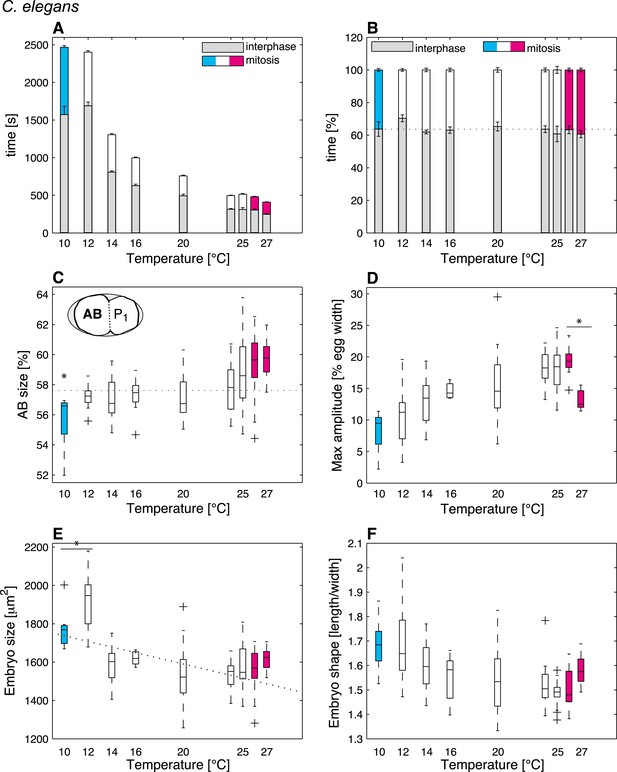
C. elegans thermal responses.
(A) Cell cycle duration as a function of temperature (error bars show SEM). (B) Relative cell cycle duration as a function of temperature (error bars show SEM). (C) Relative size of the AB blastomere as a function of temperature. Dotted line represents the average relative size within the thermal range (57.4%). (D) Maximum amplitude of posterior pole oscillations during anaphase. (E) Embryo size as a function of temperature. Dotted line shows a linear regression of the data within the thermal range (white boxes). (F) Embryo shape, measured as the ratio of embryo length over embryo width, as a function of temperature. See main text for p-values. Color code (for the whole figure): white bars show data within the thermal range. Colored bars show data below (cyan) and above (magenta) the thermal limit. Boxplots show median as well as 25th and 75th percentiles. Whiskers extend to the most extreme points not considered outliers (i.e., within 99.3% coverage). Note that the variance of cellular features does not increase beyond the thermal limits as compared to within the thermal range. Figure 2—figure supplement 1 depicts embryo size and shape at various temperatures.
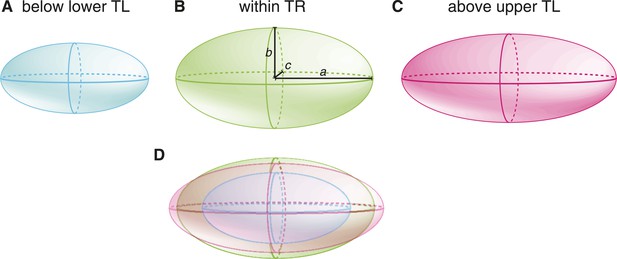
Embryo size and shape at various temperatures, exaggerating the actual differences for visualization purposes.
We show a cartoon embryo below the lower thermal limit (A), which has the same shape as the embryo within the thermal range (B), that is, a/b is equal in both cases. Above the upper thermal limit (C), the embryo elongates (i.e., a/b increased). In (D), we show an overlay of the embryos in (A–C).
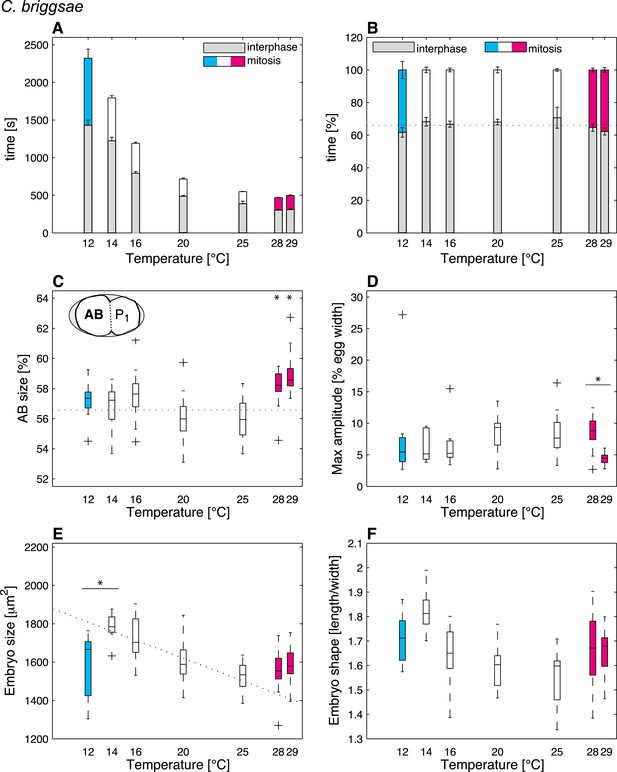
Thermal responses in C. briggsae.
See legend of Figure 2 and main text for p-values.
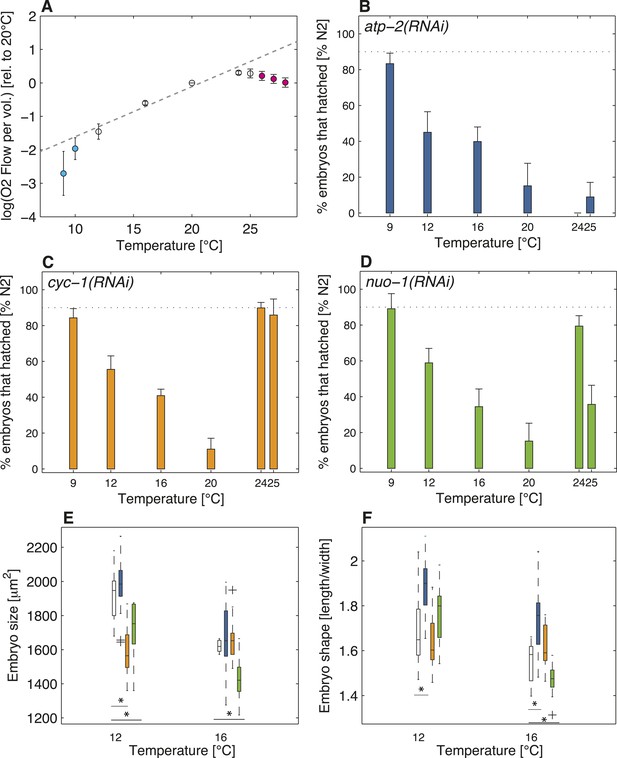
Restricted aerobic metabolism at the thermal limits.
(A) Oxygen consumption (y-axis displays the logarithm of O2 flow per volume) in embryos at different temperatures from 9°C to 28°C. Pooled data from two biological replicates, each with two technical replicates (see ‘Materials and methods’). Error bars represent the SEM. Note that respiration increases exponentially between 12°C and 24°C (white discs), as shown by the linear increase in log-scale (gray dashed line shows exponential fit between 12°C and 24°C). Note also that respiration decreases beyond both thermal limits (cyan and magenta discs, respectively), and no longer follows the exponential trend observed within the thermal range. (B) Color-code for panels (B–F): white (wild-type), blue (atp-2(RNAi)), orange (cyc-1(RNAi)), green (nuo-1(RNAi)). Progeny tests on atp-2(RNAi) embryos. (C) Same as B for cyc-1(RNAi). (D) Same as B for nuo-1(RNAi). (E) Embryo size as a function of temperature. (F) Embryo shape as a function of temperature. See main text for p-values. Figure 4—figure supplement 1 shows the RNAi feeding times as a function of temperature.
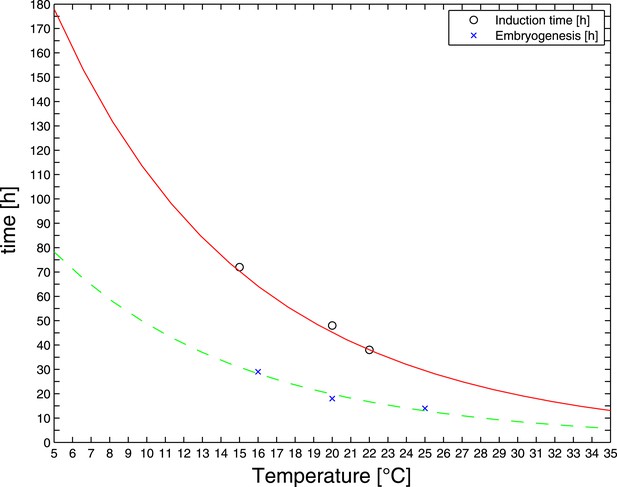
RNAi feeding time as a function of temperature.
Reported durations of embryogenesis (blue crosses) were fitted by an Arrhenius-like model (dashed green line) (Gillooly et al., 2002). The same activation energy was used to fit reported RNAi induction times (black circles and fitted red solid line). The latter fit was used to extrapolate RNAi feeding times at the temperatures of interest.
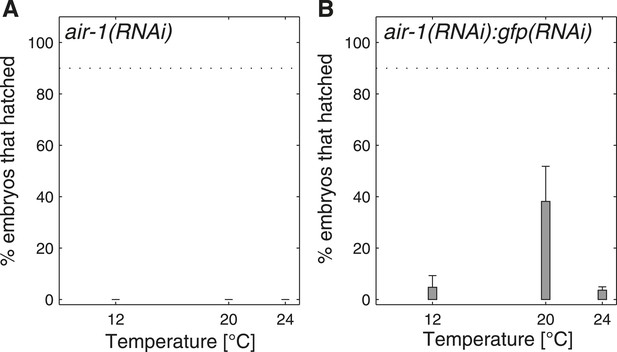
Progeny tests in air-1(RNAi).
(A) air-1(RNAi) is 100% embryonic lethal at 12°C, 20°C and 24°C, as anticipated (Schumacher et al., 1998; Hannak et al., 2001). (B) In order to titrate the phenotype, we performed double RNAi by mixing bacteria expressing dsRNA against air-1 with bacteria expressing dsRNA against gfp in a 1:3 ratio. We found that lethality is greater at 12°C and at 24°C than at 20°C, indicating that the results we uncovered when targeting mitochondrial respiratory chain components (Figure 4B–D) are not due to a general RNAi temperature-dependent response.
Additional files
-
Source code 1
In-house scripts to analyze embryos (Matlab).
- https://doi.org/10.7554/eLife.04810.012





