CAPS-1 promotes fusion competence of stationary dense-core vesicles in presynaptic terminals of mammalian neurons
Figures
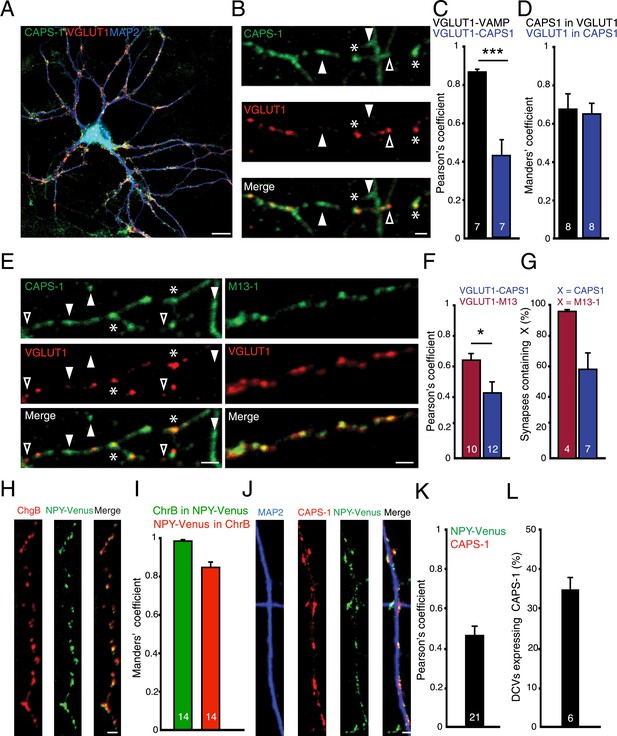
CAPS-1 clusters are present at synaptic and extra-synaptic sites and partly co-localize with DCVs.
(A) Example image of a hippocampal neuron (DIV 14) stained for endogenous CAPS-1 (green), dendrite marker MAP2 (blue) and synapse marker VGLUT1 (red). Scale bar 10 µm. (B) Zoom of a neurite stained for CAPS-1 and VGLUT1. CAPS-1 rich domains not overlapping with VGLUT1 (filled arrowhead), VGLUT1 punctum not enriched for CAPS-1 (open arrowhead), VGLUT1 puncta overlapping with a CAPS-1 rich domain (stars). Scale bar 2 µm. (C) Co-localization of CAPS-1 with VGLUT1 in the entire neuron quantified by Pearson's correlation. Co-localization of VAMP2 with VGLUT1 was used as a positive control (VGLUT1-VAMP2: 0.8±0.02, n=7 neurons; VGLUT1-CAPS-1: 0.4±0.05, n=7 neurons, ***p<0.0001). (D) Mander's coefficients for the proportion of CAPS-1 immuno-reactivity in VGLUT1 positive locations: 0.67±0.08, n=8 neurons or proportion of VGLUT1 immunoreactivity in CAPS-1 positive locations: 0.64±0.05, n=8 neurons. (E) Example images of neurites from hippocampal neurons (DIV 14) stained for endogenous CAPS-1 (green, left panel) and VGLUT1 (red) or for Munc13-1 (M13-1, green, right panel) and VGLUT1 (red). CAPS-1 domains not overlapping with VGLUT1 (filled arrowheads), VGLUT1 puncta not enriched for CAPS-1 (open arrowheads). Synapses (VGLUT1 puncta) overlapping with CAPS-1 rich domains (stars). Scale bar 5 µm. (F) CAPS-1 co-localization with VGLUT1 in the entire neuron is lower compared to co-localization of Munc13-1 and VGLUT1. Pearson's correlation M13-1-VGLUT1: 0.6±0.04, n=10; CAPS-1-VGLUT1: 0.4±0.08, n=12, *p<0.05. (G) Percentage of VGLUT1 labeled synapses expressing CAPS-1 is lower than VGLUT1 labeled synapses expressing (VGLUT1/CAPS-1: 58.5±10.9%, n=7 neurons, number of synapses = 239; VGLUT1/M13-1: 96.2±0.7%, n=4 neurons, number of synapses = 350). (H) Example images of neurites from hippocampal neurons (DIV 14) infected with lentivirus encoding NPY-Venus (green) and stained for chromogranin B (ChgB, red). Scale bar 2 µm. (I) Mander's coefficients for the proportion of endogenous ChrB immuno-reactivity in NPY-Venus puncta: 0.97±0.02, n=14 neurons or proportion of NPY-Venus immunoreactivity in ChrB puncta: 0.84±0.01, n=14 neurons. (J) Example images of neurites from hippocampal neurons (DIV 14) infected with lentivirus encoding NPY-Venus and stained for CAPS-1 (red) and MAP2 (blue). Scale bar 2 µm. (K) Quantification of co-localization of CAPS-1 and NPY-Venus in the entire neuron. Pearson's coefficient: 0.45±0.05; n=21 neurons. (L) Percentage of NPY-Venus labeled DCVs co-localizing with CAPS-1: 34.71±3.03%, n=6, number of DCVs = 414.
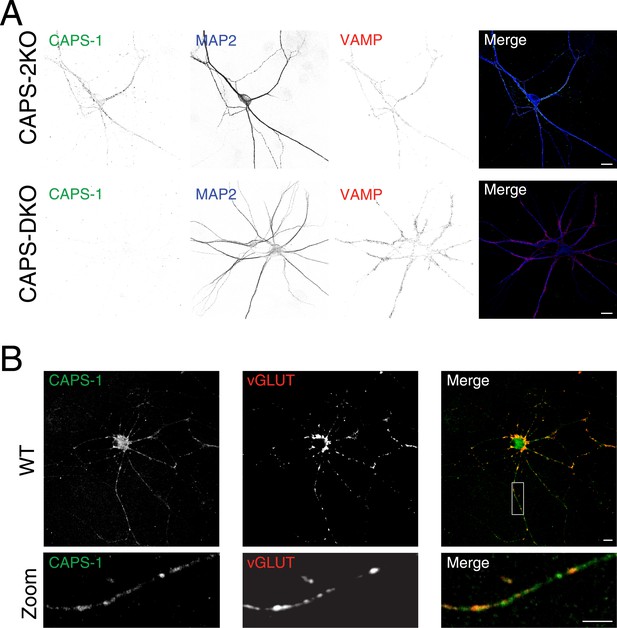
Specificity of CAPS-1 antibody.
(A) CAPS-1 immunoreactivity (green) and co-localization with synaptobrevin (VAMP, red) in CAPS-2 KO neurons stained for the dendritic marker MAP2 (blue) (top panel). Bottom panel: absence of immunoreactivity in CAPS DKO neurons. Scale bar 10 μm. (B) CAPS-1 immunoreactivity (green) and co-localization with VGLUT1 (red). Bottom panel: zoom indicated by the box in top panel. Scale bar 10 μm.
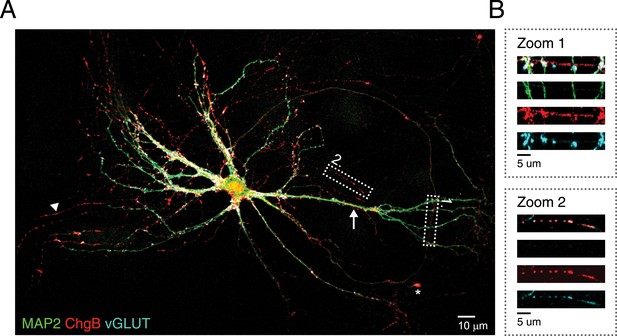
Endogenous DCV marker (chromogranin B) distribution in isolated single neurons.
(A) Confocal image of a WT hippocampal neuron (DIV 14) labeled with antibodies for the dendritic marker MAP2 (green), endogenous DCV cargo protein ChgB (red) and the synaptic marker VGLUT1 (blue). A punctate distribution of ChgB is seen in dendritic (arrow) and axonal regions (MAP2-negative; arrowheads) and accumulations of ChgB are found in growth cones (asterisks). (B) Higher magnification of boxed area 1 (zoom 1) shows ChgB puncta in axonal regions. Zoom 2 shows co-localization of ChgB and VGLUT1, indicating the presence of DCVs in synapses.
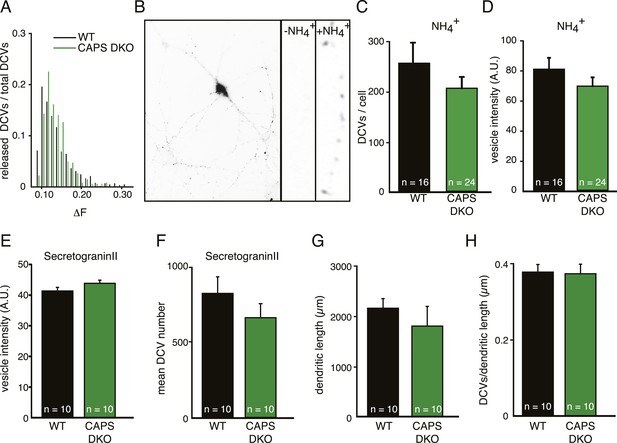
CAPS deletion does not influence DCV peptide loading or DCV biogenesis in hippocampal neurons.
(A) Frequency distribution of fluorescence intensity increase (ΔF) of individual DCV fusion events in WT and CAPS DKO neurons. No major differences are observed in ΔF of fusion events between WT and CAPS DKO neurons indicating similar Semaphluorin loading per DCV in WT and CAPS DKO neurons. The number of DCVs per bin is normalized to the total number of DCVs released. (B) Inverted wide-field image of a neuron expressing Semaphluorin upon NH4+ application to reveal all DCVs present in the cell. Zoom shows the effect of vesicle de-acidification upon NH4+ application (−NH4+ before application, +NH4+ during application). (C) Average number of DCV puncta per neuron quantified from the NH4+ response is similar in WT (n=16 neurons) and CAPS DKO (n=24) neurons. (D) Average intensity (in arbitrary units, AU) of single DCV puncta quantified from the NH4+ response in WT and CAPS DKO neurons is similar (WT n=16 neurons and 4435 puncta, CAPS DKO n=24 neurons and 4892 puncta). (E) Average intensity (in AU) of single DCV puncta in the field of view of confocal images is similar in non-transfected WT (n=10 neurons) and CAPS DKO (n=10) neurons stained for the endogenous DCV cargo secretogranin II and the dendritic marker MAP2. (F) Average number of DCV puncta per field of view. (G) Average dendritic length per field of view. (H) Number of DCV puncta per dendritic length.
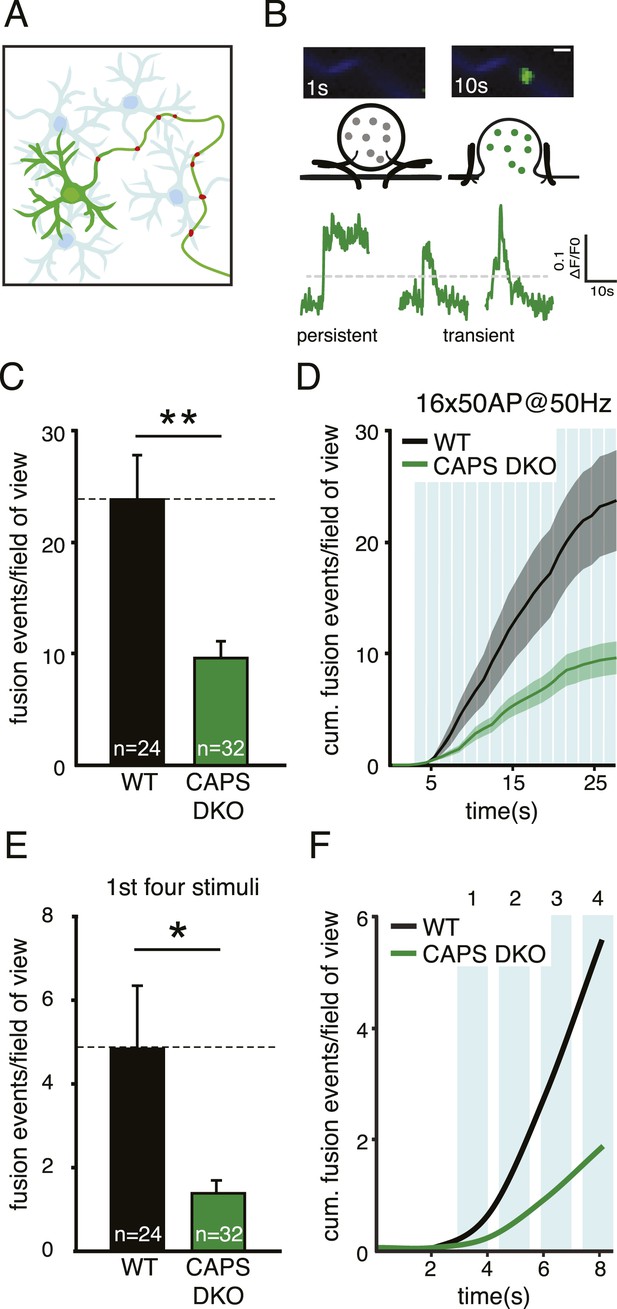
CAPS deletion reduces DCV fusion in hippocampal neurons.
(A) Schematic drawing of a neuronal mass culture used in these experiments in which one neuron expresses a fluorescently tagged morphology marker (green) and a DCV marker (red). Non-labeled neurons are indicated in light blue. (B) Top panel: two stills showing a typical example of a fusion event of a SemapHluorin labeled DCV with the corresponding schematic drawing. Fusion pore opening causes a sudden increase of fluorescence intensity corresponding to the increase in intravesicular pH. Bottom panel: example traces of SemapHluorin labeled DCV fusion events. Fluorescence increase of two standard deviations above initial fluorescence (grey dotted line) was scored as fusion event in C and E. After which, fluorescence may decrease (transient events) or remain high (persistent events). Figure 3—figure supplement 1 explains this behavior in detail. Scale bar 1 μm. (C) Average number of DCV fusion events per field of view during electrical stimulation with 16 bursts of 50 AP at 50 Hz with 0.5 s interval (WT: 23.7±4.5, n=24 neurons; CAPS DKO: 9.6±1.5, n=32 neurons, N=4, independent experiments, **p<0.01). (D) Cumulative frequency plot of DCV fusion events during stimulation (average vesicle fusion rate per cell during stimulation WT: 1.2±0.4 vesicles/s; CAPS-1/2 DKO: 0.15±0.1 vesicles/s). Blue bars represent stimulation of 16×50 AP at 50 Hz with 0.5 s interval. (E) Number of DCV fusion events per field of view during the first four bursts of the stimulation in C, (WT: 4.8±1.5, n=24; CAPS DKO: 1.4±0.3, n=32, *p<0.05). (F) Cumulative frequency plot of DCV fusion events during the first four bursts of the stimulation, showing that the initial release rate is slower in CAPS DKO neurons.
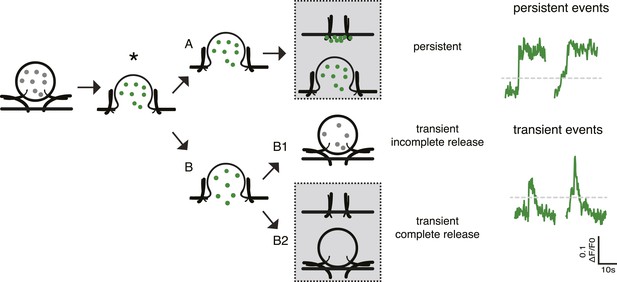
Different fusion events reported by SemapHluorin.
Cartoon depicting the different fusion events reported by SemapHluorin and analyzed in depth in de Wit et al., 2009. Electrical stimulation triggers calcium and SNARE protein-dependent membrane fusion. Opening of the fusion pore results in a sudden dequenching of vesicular SemapHluorin and increase in fluorescence (asterisk). This is scored as fusion event in Figure 3C,vE. Upon the sudden increase in fluorescence the signal either remains high (A: persistent event) or dims to baseline (B: transient event). Transient events represent incomplete release followed by vesicle retrieval and re-acidification (B1) or full release followed by cargo diffusion (B2). In B2 the vesicle may integrate into the plasma membrane or re-seal without SemapHluorin cargo (B2, grey box top and bottom panel, respectively). Persistent events reflect the continuous presence of cargo at the cell surface and may either represent stable deposits of SemapHluorin or vesicles with a permanently open fusion pore (A, grey box top and bottom panel, respectively). Persistent events are typical for cargo that interacts with the intra-vesicular matrix like Semaphorin. These events are very rare when using NPY as cargo. Our previous analysis of fusion events reported by SemapHluorin showed that 60% of fusion events are transient events of which half result in full release and 40% of events are persistent (de Wit et al., 2009). The typical example traces show persistent and transient fusion events. Both types of events were scored as fusion events in Figure 3C, E when fluorescence increased above two times the standard deviation of the initial fluorescence (grey dotted line).
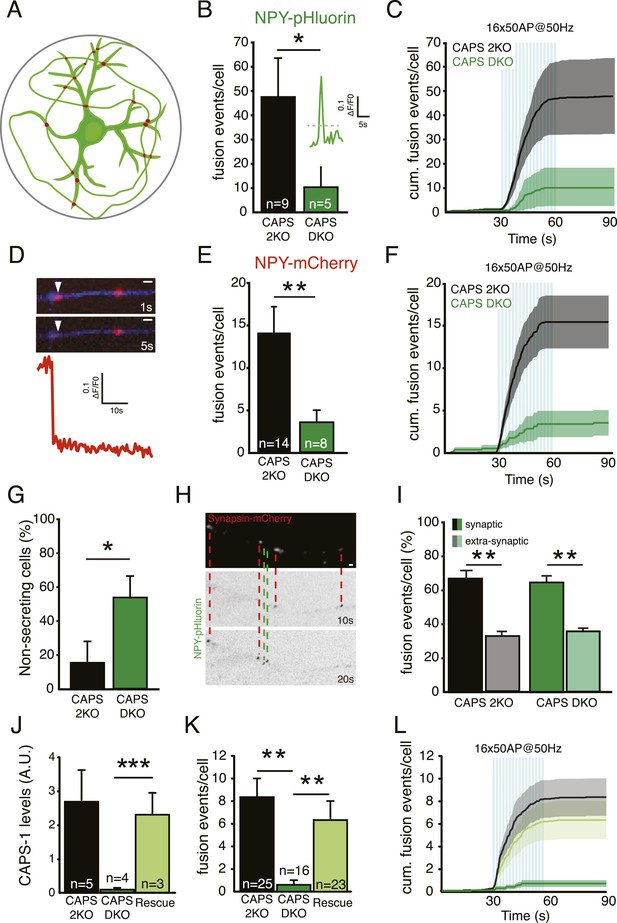
CAPS-1 deletion impairs DCV fusion in isolated neurons.
(A) Schematic drawing of a single isolated neuron grown on a micro island of astrocytes and expressing fluorescently tagged morphology marker (green) and a synapse marker (red) in addition to either NPY-pHluorin or NPY-mCherry (not shown). Average island diameter is 375 μm. (B) Average number of DCV fusion events per cell upon electrical stimulation of 16 bursts of 50 AP at 50 Hz using NPY-pHluorin as DCV marker (CAPS-2KO: 49.6±16.3, n=9; CAPS DKO: 10.6±7.9, n=5, number of independent experiments (N)=3, *p<0.05). Inset shows a typical example of a fusion event reported by NPY-pHluorin. Fluorescence increase of two standard deviations above initial fluorescence (grey dotted lines) was scored as fusion event in B. (C) Cumulative frequency plot of DCV fusion events in B, showing that DCV release is triggered by electrical stimulation paradigm (blue bars represent 16 bursts of 50 AP at 50 Hz, 16×50 AP at 50 Hz). (D) White arrowhead: Typical example of a fusion event with complete cargo release reported by the sudden and complete disappearance of NPY-mCherry fluorescence intensity. These fusion events were in measured in E and K. Time in seconds (s) after start of stimulation. Neurite marker is ECFP (blue). Scale bar 1 μm. (E) Average number of fusion events with complete cargo release per cell upon electrical stimulation using NPY-mCherry as DCV marker (CAPS-2KO: 14.6±3.3, n=14; CAPS DKO: 3.4±1.4, n=8, N=3, **p<0.01). (F) Cumulative frequency plot of DCV fusion events with complete cargo release in E, (blue bars represent 16 bursts of 50 AP at 50 Hz, 16×50 AP at 50 Hz). (G) Percentage of non-secreting cells is increased in CAPS DKO neurons (CAPS-2KO: 15±12.4%, n=26; CAPS DKO 53.6±12.6%, n=26, *p<0.05). Non-secreting cells were excluded from the analyses in B and E. (H) Example images showing NPY-pHluorin labeled DCVs fusing at synapsin-mCherry labeled synapses, indicated by the red dashed lines and DCV fusion events at extra-synaptic sites, indicated by the green dashed lines. Bar is 1 μm. (I) Percentage of synaptic and extra-synaptic DCV release events measured with NPY-pHluorin shows a similar distribution in CAPS DKO compared to CAPS-2KO (CAPS-2KO synaptic: 67.0±3.6, CAPS-2KO extra-synaptic: 0.32±1.4, n=20, **p<0.01; CAPS DKO synaptic: 64.7±2.3, CAPS DKO extrasynaptic: 35.6±0.9, n=28, **p<0.01). (J) CAPS-1 expression levels assessed by semi-quantitative immunofluorescence in CAPS-2KO, CAPS DKO, and CAPS DKO expressing CAPS-1 (Rescue) (CAPS-2KO: 2.8±0.7 AU, n=5; CAPS DKO: 0.1±0.0, n=4; Rescue: 2.4±0.7, n=3, ***p<0.0001). (K) Average number of fusion events leading to complete release per cell upon electrical stimulation with NPY-mCherry as DCV marker in CAPS-2KO, CAPS DKO and Rescue (CAPS-2KO: 8.5±1.7, n=25; CAPS DKO: 0.7±0.3, n=16; Rescue: 6.4±1.7, n=23, N=6). (L) Cumulative frequency plot of complete release DCV fusion events in K, (blue bars represent 16 bursts of 50 AP at 50 Hz, 16×50 AP at 50 Hz).
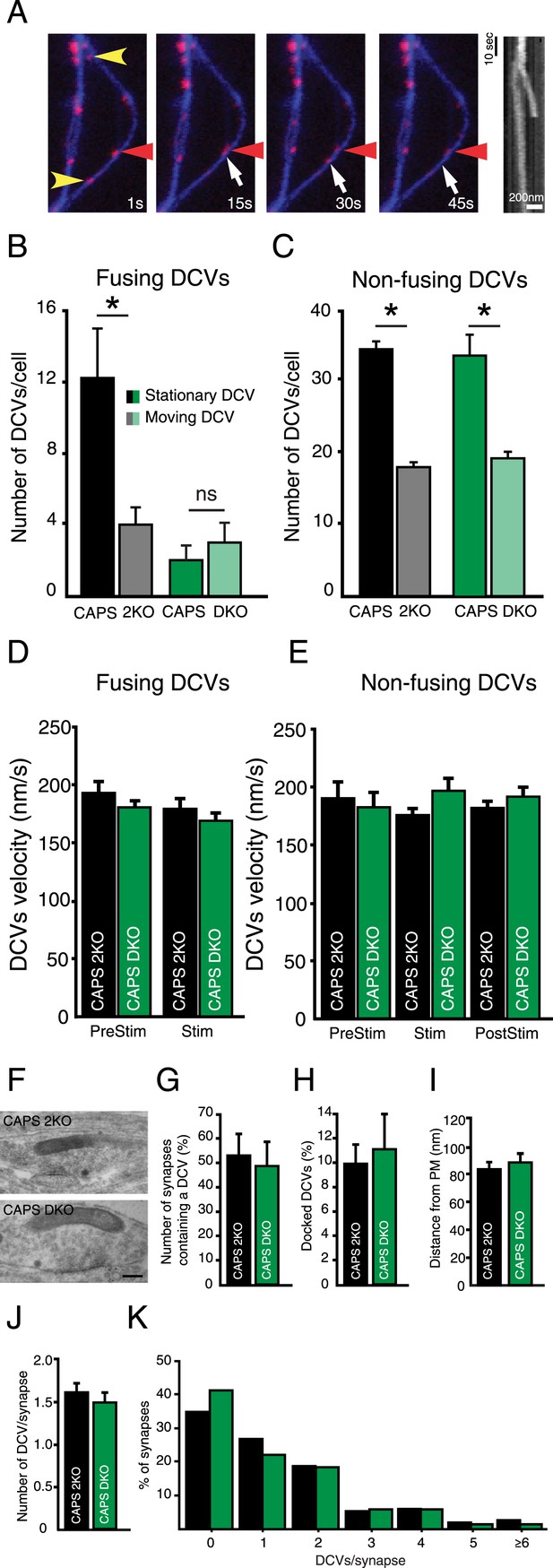
Deletion of CAPS-1 affects fusion of stationary DCVs.
(A) Stills from DCV fusion assay. Electrical stimulation starts at second 10. Yellow arrowheads indicate stationary vesicles that fuse, red arrowhead indicates a stationary vesicle that does not fuse and white arrow shows a moving DCV that fuses. Kymograph shows the trajectories of the stationary and moving vesicle over time. (B) Fusing DCVs classified as stationary or moving showing that CAPS deletion strongly affects fusion from stationary vesicles (CAPS-2KO stationary prior to fusion: 12.2±2.8, moving: 4±1.0, n=10, *p<0.05; CAPS DKO stationary: 2.3±1.3, moving: 4.0±1.7, n=3, ns, N=3). (C) Non-fusing DCVs classified as stationary or moving showing that CAPS deletion does not affect general trafficking behavior of non-fusing vesicles (CAPS-2KO stationary: 33.8±1.2, moving: 17.6±0.7, n=5, *p<0.05; CAPS DKO stationary: 33.7±3.0, moving: 19.3±2.4, n=3, *p<0.05). (D) Average velocity of DCVs classified as moving prior to fusion in B, before stimulation (PreStim = 30 s; CAPS-2KO: 193.6±9.9 nm/s, n=169 vesicles, CAPS DKO: 180.4±9.2 nm/s, n=18) and during electrical stimulation (Stim: from second 30 to the onset of fusion; CAPS-2KO = 180.9±6.6 nm/s, n=169; CAPS DKO: 169.2±7.5, n=18). (E) Average velocity of non-fusing DCVs in C, before stimulation (PreStim = 30 s; CAPS-2KO: 190.8±13.7 nm/s, n=168, CAPS DKO 186.8±13.5 nm/s, n=91), during stimulation (Stim = 24 s; CAPS-2KO: 175.3±6.7 nm/s, n=168; CAPS DKO 201.8±11.8 nm/s, n=91) and after stimulation (PostStim = 36 s CAPS-2KO: 182.2±6.2 nm/s, n=168, CAPS DKO 196.2±8.5 nm/s, n=91). (F) Typical examples of electron micrographs of neuronal DCVs in synapses. Scale bar 50 nm. (G) Number of synapses containing one or more DCVs (CAPS-2KO: 53.9±12.1%, n=198 synapses; CAPS DKO 47.6±5.4%, n=152 synapses, N=4). (H) Percentage of docked DCVs per synapse (CAPS-2KO: 10.3±1.7%, DCVs = 285, CAPS DKO: 11.1±3.7%, DCVs = 201). (I) Average distance of DCVs to the closest plasma membrane (CAPS-2KO: 81.2±4.8; CAPS DKO: 84.9±10.4). (J) Average number of DCVs per synapse (CAPS-2KO: 1.6±0.2; CAPS DKO: 1.5±0.2). (K) Frequency distribution of number of DCVs per synapse (% synapses, normalized to the total number of DCVs per group).
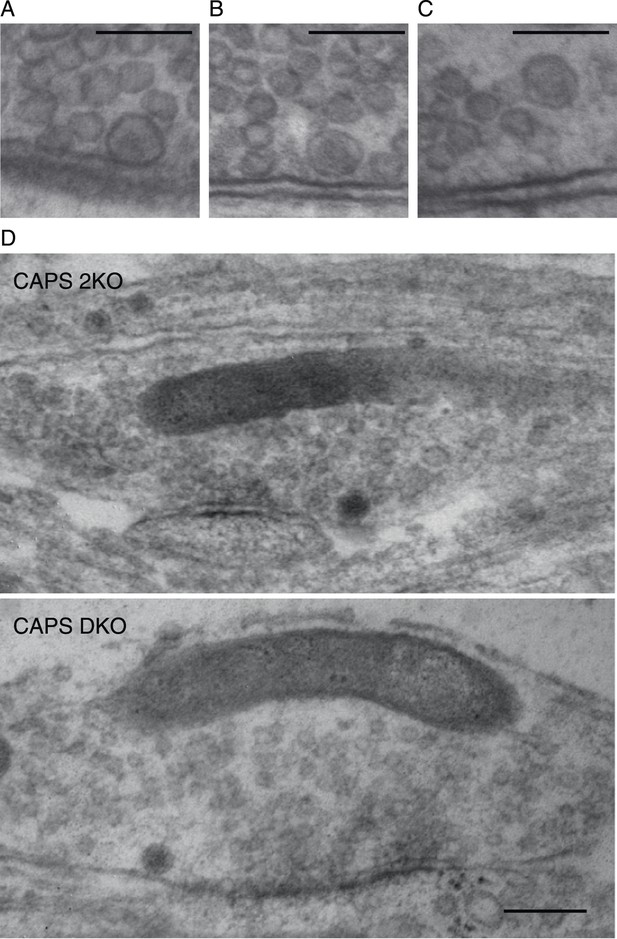
DCV docking definition and zooms of Figure 5F.
Zooms of three example DCVs showing the classification of docked versus undocked vesicles. We define a docked DCV as a vesicle containing a dense core that has no detectable distance between the vesicular membrane and the plasma membrane. The minimal distance between membranes we can visualize is 0.52 nm. (A) Electron micrograph showing an example of docked DCV. (B) Electron micrograph showing an example of DCV close but not docked to the plasma membrane. (C) Electron micrograph showing an example DCV far away to the plasma membrane. Scale bars 100 nm. (D) Enlarged high-resolution electron microscopy images of Figure 5F. Scale bar 50 nm.
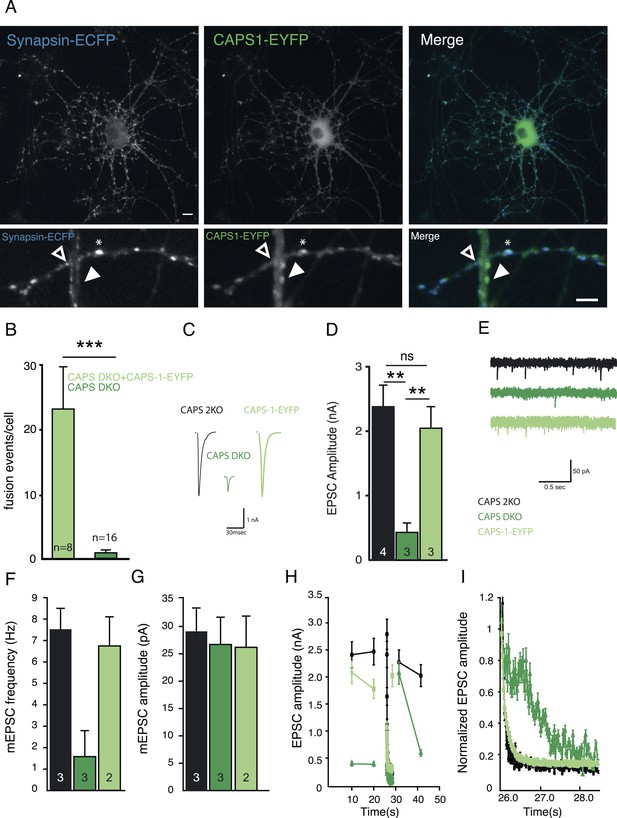
CAPS-1-EYFP fusion protein replaces CAPS-1 in DCV secretion and synaptic transmission.
(A) Isolated single CAPS DKO neuron grown on glia micro-island expressing CAPS-1-EYFP (green) and synapsin-ECFP (blue). Top panels: maximum projection of the entire neuron, Scale bar 10 µm. Bottom panels: zooms of top panels showing synapses without detectable CAPS-1-EYFP (open arrowhead), CAPS-1 rich domain not overlapping with synapsin-ECFP (filled arrowhead) and a CAPS-1 rich synapse (star). Scale bar 5 µm. (B) Average number of DCV fusion events per cell (events/cell) upon electrical stimulation (16×50 AP at 50 Hz) using NPY-pHluorin as DCV marker in CAPS DKO neurons with (+CAPS-1-EYFP) and without (CAPS DKO) CAPS-1-EYFP (+CAPS-1-EYFP: 23.0±6.8, n=8, CAPS DKO: 0.7±0.3, n=16, ***p<0.0001, Mann–Whitney test). (C) Example traces of evoked EPSCs in CAPS-2KO (black), CAPS DKO (dark green) and CAPS-1-EYFP (light green) rescued CAPS DKO neurons. (D) Expression of CAPS-EYFP rescues EPSC amplitude in CAPS DKO neurons (CAPS-2KO: 2.4±0.3 nA, n=4; CAPS DKO: 0.48±0.15 nA, n=3; CAPS-1-EYFP: 2.0±0.4 nA, n=3, **p<0.01). (E) Example traces of spontaneous release (mEPSCs). (F) Mean mEPSC frequency. (CAPS-2KO: 7.5±1.1 Hz, n=3; CAPS DKO: 1.7±1.0 Hz, n=3; CAPS-1-EYFP: 6.9±1.3 Hz, n=2). (G) Mean mEPSC amplitude. (CAPS-2KO: 29.0±4.1 pA, n=3; CAPS DKO: 27.1±5.0 pA, n=3; CAPS-1-EYFP: 26.8±7.2 pA, n=2). (H) Changes in EPSC amplitude induced by 100-pulse train at 40 Hz during low frequency (0.1 Hz) stimulation. The interval between low- and high-frequency stimulation is 3 s (I) 100-pulses at 40 Hz induced rundown of normalized EPSC amplitude (zoom of H).
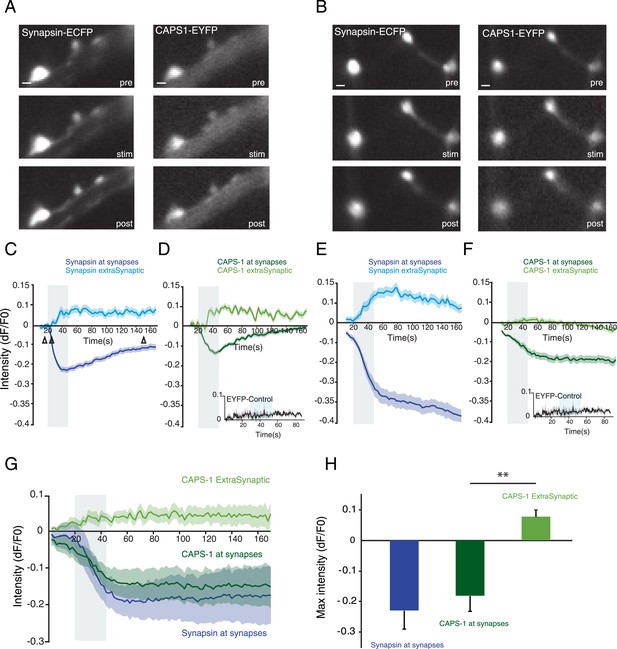
Localization of CAPS-1 at synapses is calcium dependent.
(A and B) Grey scale images of synapsin-ECFP (left) and CAPS-1-EYFP (right) labeled synapses showing the same region before (pre-15s) during (stim-22s) and after (post-150s) electrical stimulation (16×50 AP at 50 Hz). Synapsin-ECFP and CAPS-1-EYFP were imaged simultaneously at 0.5 Hz. (C and E) Traces of relative intensity changes (ΔF/F0) of synapsin-ECFP at synapses and extra-synaptic locations (79 synaptic and 31 extra-synaptic) showing increased extra-synaptic and decreased synaptic fluorescence upon stimulation (16×50 AP at 50 Hz). Open arrowheads in C indicate the pre- and post stimulations time points (same for D–F). (D and F) Example traces of relative intensity changes (ΔF/F0) of CAPS-1-EYFP at synapsin-ECFP labeled synapses from C. showing increased extra-synaptic and decreased synaptic fluorescence upon stimulation (16×50 AP at 50 Hz). Inset: synaptic fluorescence of membrane associated EYFP (EYFP control) as control. (G) Average relative intensity profiles of synapsin-ECFP, and CAPS-1-EYFP at synapses and CAPS-1-EYFP extra-synaptic, (395 synaptic regions, 155 extrasynaptic regions, n=5 cells). (H) Maximum relative intensity changes (max ΔF/F0) of synapsin-ECFP, and CAPS-1 at synapses and CAPS-1 at extra synaptic regions at t=160 s calculated from G (**p<0.01, n=5 cells each).
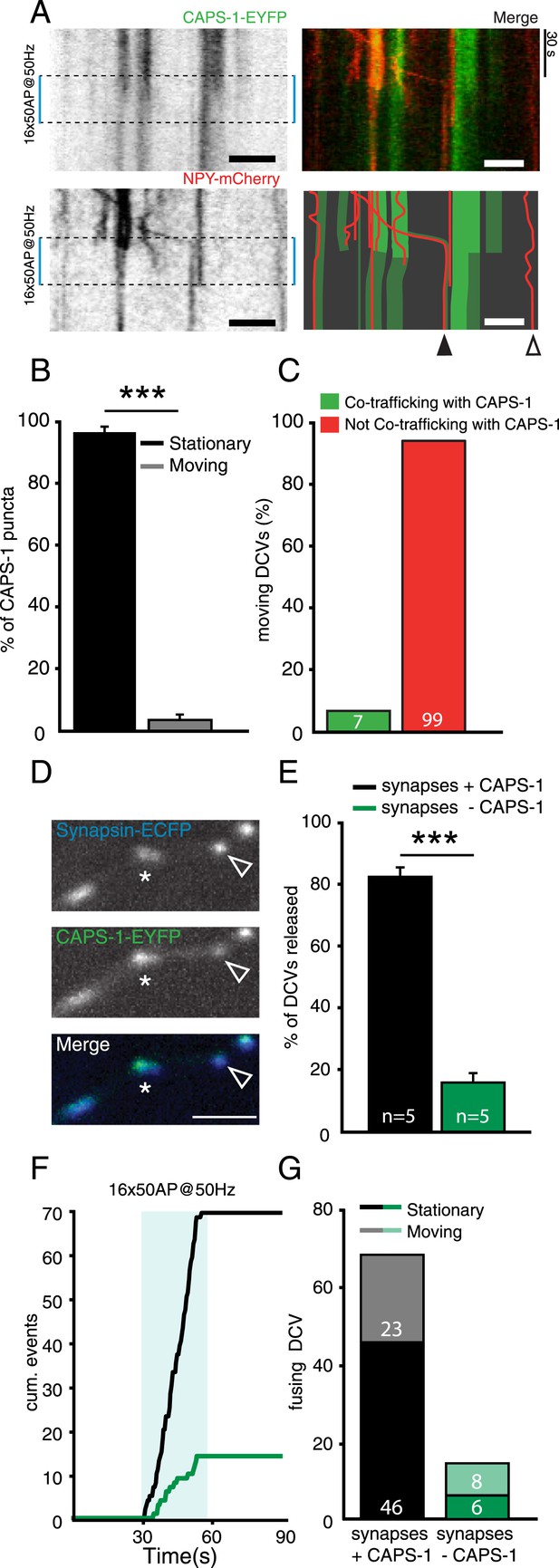
Presence of CAPS-1 increases DCV release probability at single synapses.
(A) Left top and bottom panels: kymographs of CAPS-1-EYFP and NPY-mCherry imaged simultaneously (acquisition frequency 2 Hz) for 90 s and stimulated at second 30 (dashed box, 16×50 AP at 50 Hz). Note the dispersion of the majority of CAPS-1-EYFP puncta upon stimulation and the disappearance of NPY-mCherry puncta upon stimulation. Top right panel shows the merge of the two channels. The bottom right panel shows a schematic drawing of the merged CAPS-1-EYFP and NPY-mCherry channels to aid in the interpretation of the kymographs. Open arrowhead indicates DCV not co-localizing with CAPS-1-EYFP. Filled arrowhead indicates a CAPS-1-EYFP punctum co-trafficking with a mobile DCV. Scale bar 5 µm. (B) Percentage of stationary and moving CAPS-1-EYFP puncta during image acquisition as described in (A). (Stationary: 96.6±1.7, moving: 3.3±1.7, number of cells = 9; number of kymographs per cell = 3, total number of puncta = 116). (C) Percentage of mobile DCVs co-trafficking with CAPS-1-EYFP (mobile DCVs not co-trafficking with CAPS-1-EYFP (green bar): 99 or 93.4%; mobile DCVs co-trafficking with CAPS-1-EYFP: 7 or 6.3%, total number of cells = 20, moving DCVs analyzed = 106). (D) Typical examples of synapsin-ECFP labeled synapses with high expression levels of CAPS-1EYFP (star) or low expression levels of CAPS-1-EYFP (open arrowhead). (E) Percentage of DCV release events occurring at synapses enriched for CAPS-1 (black bar) or depleted for CAPS-1 (green bar), (synapses + CAPS-1: 84.0±3.3%, synapses − CAPS-1: 16.0±3.3%, total DCVs released = 84, n=5, ***p<0.0001). (F) Cumulative frequency plot of the DCV release events in C. showing that release at CAPS-1 deficient synapses is reduced and delayed. Blue box represents 16×50 AP at 50 Hz stimulation. (G) Released DCVs categorized in stationary or moving before fusion (synapses + CAPS-1: stationary DCVs = 46, moving DCVs = 23, total DCVs released = 69; synapse − CAPS-1: stationary DCVs = 6 moving DCVs = 8, total DCVs released = 14).





