Release of human cytomegalovirus from latency by a KAP1/TRIM28 phosphorylation switch
Figures
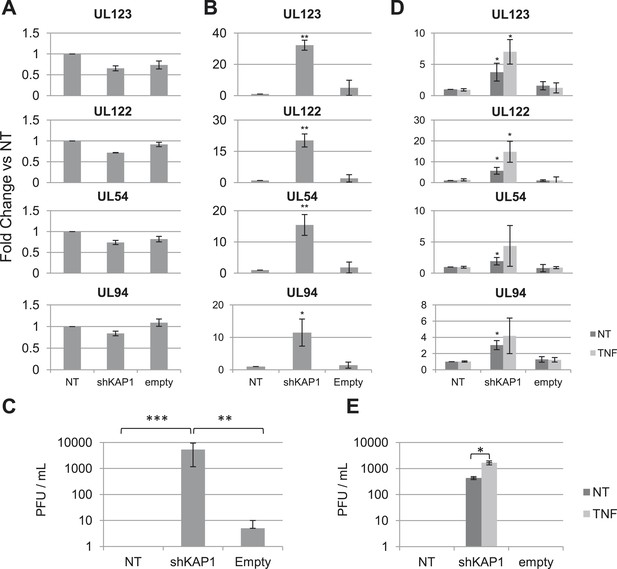
KAP1 is required for HCMV latency.
RT-qPCR analysis of indicated HCMV genes expression in MRC-5 (A) or cord blood CD34+ (B, C, D, E) cells infected with the TB40-E strain 3 days after (A, B, C) or 7 days prior to (D, E) being transduced or not (NT) with lentivectors expressing (shKAP1) or not (empty) a small hairpin RNA against Kap1, sorting CD34+ cells for GFP and CD34 expression. RT-qPCRs were performed 7 days after HCMV infection (A and B) or 7 days after lentiviral transduction (D and E). Results are presented as average of fold change expression vs NT after GAPDH and β-2M normalization (n = 4, *p < 0.05, **p < 0.01, error bars as s.d.). (C and E) HCMV production in CD34+ cells was quantified by plaque assay on MRC-5 cells. Results are presented as average of PFU/ml (n = 3, *p < 0.05, **p < 0.01, ***p < 0.001, error bars as s.d.). See Figure 1—figure supplements 3, 4 for all individual experiments.
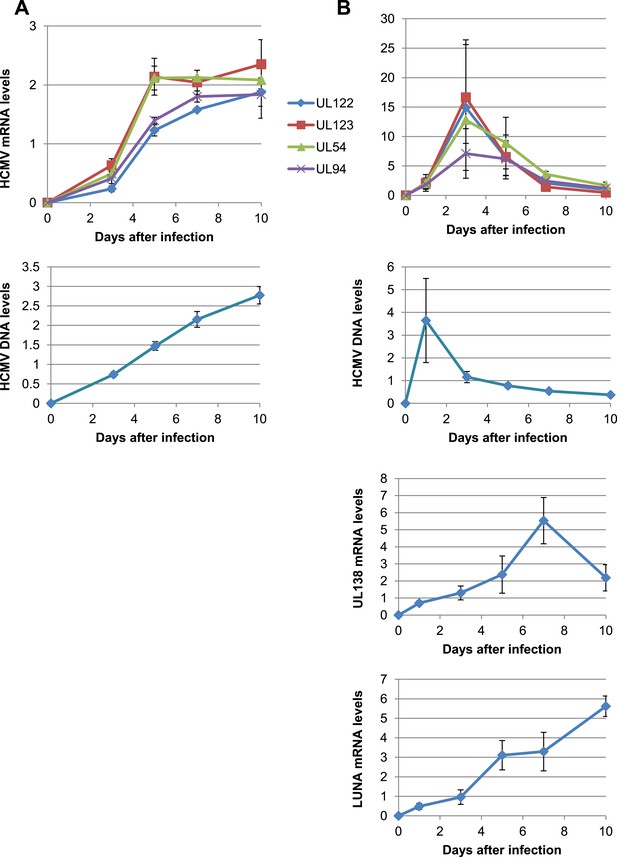
KAP1 is necessary for both establishment and maintenance of HCMV latency in HSC.
HCMV kinetics analysis performed on MRC-5 fibroblasts (A) or CD34+ HSC (B). HCMV mRNA and DNA expression were quantified at day 1-3-5-7 and 10 post-infection by RT-qPCR.
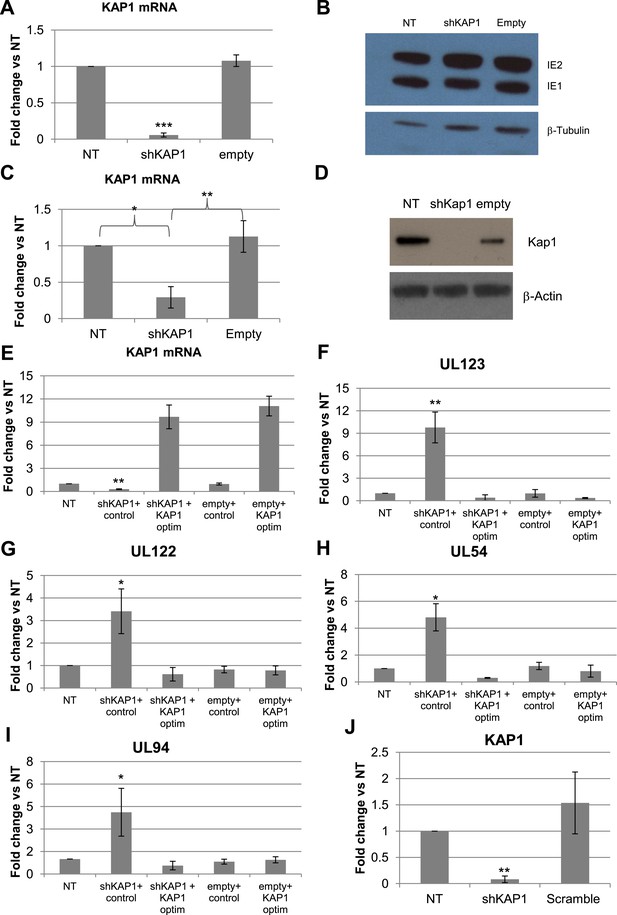
KAP1 is necessary for both establishment and maintenance of HCMV latency in HSC.
KAP1 knockdown and re-complementation efficiency were tested by RT-qPCR (A, C, E, J) and Western-blot analysis (D). MRC-5 fibroblasts (A and B) or CD34+ cells (C–J) were transduced or not (NT) with lentiviral vectors expressing (shKAP1) or not (empty, scramble) an shRNA against Kap1, and co-transduced with a vector expressing (KAP1-optim) or not (control) an shRNA-resistant Kap1 allele, 3 days before (A–H) or 7 days after (J) infection with the TB40-E HCMV strain. Indicated HCMV transcripts were quantified by RT-qPCR for CD34+ cells (F–I) and by western blot analysis for MRC-5 (B). All results are presented as average of fold change expression vs NT after GAPDH and β-2M normalization for mRNA and GAPDH and Albumin for DNA (n = 4, *p < 0.05, **p < 0.01, error bars as s.d.). Western-Blots are representative of three independent experiments.
KAP1 is necessary for establishment of HCMV latency in HSC.
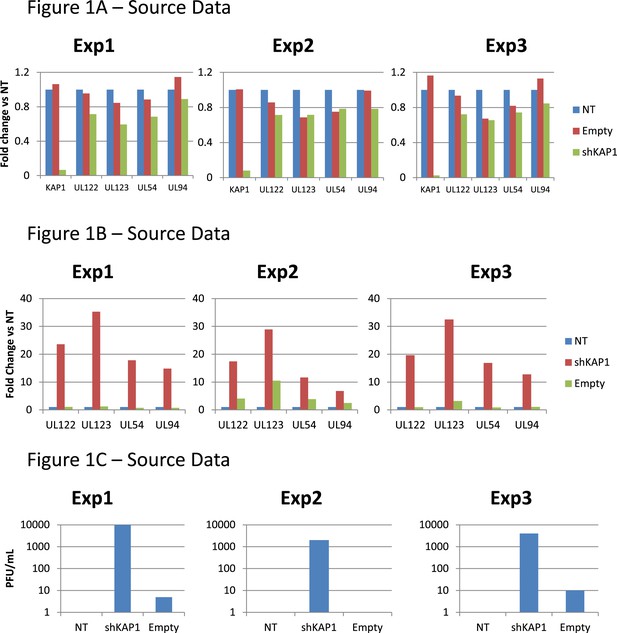
Figure 1A,B,C source datas.
KAP1 is necessary for maintenance of HCMV latency in HSC.
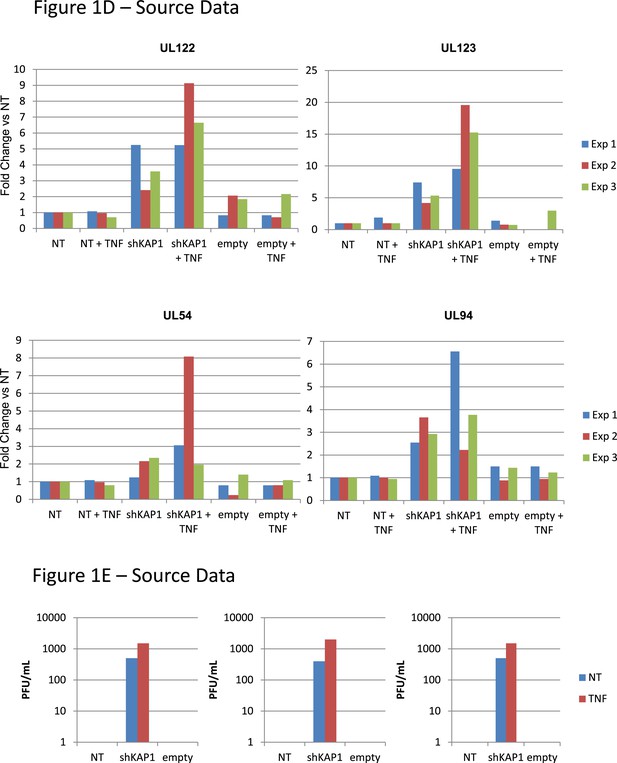
Figure 1D,E source datas.
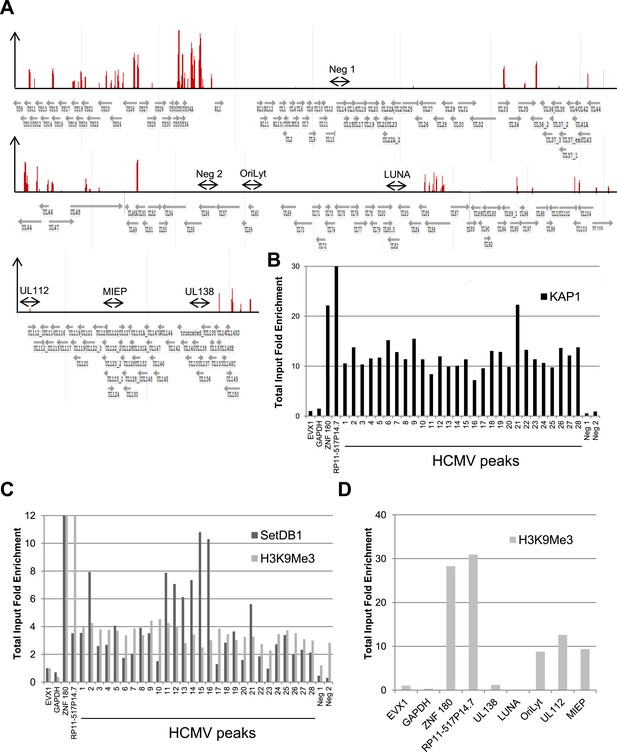
KAP1, SetDB1 and H3K9Me3 are enriched on the HCMV genome in latently infected HSC.
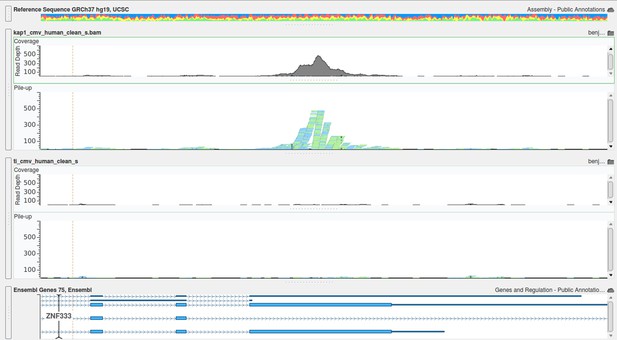
(A) KAP1 binding sites on HCMV genome in TB40-E-infected CD34+ cells, mapped by ChIP-Seq performed at day 7 post-infection. Results are presented as hit point upper 120 on the reference sequence GenBank EF999921.1, indicating all KAP1 peaks (1–28). (B and C) ChIP-PCR with anti-KAP1 (B), anti-SetDB1 and anti-H3K9Me3 (C) antibodies were performed on ChIP-Seq-mapped KAP1 peaks (1–28) plus two KAP1-negative HCMV regions (Neg 1 and 2), using EVX-1 and GAPDH as negative cellular gene controls, and ZNF180 and RP11-517P14.7 as positive controls. (D) H3K9me3-specific ChIP-PCR of two latency genes (UL138, LUNA) and three regions active during lytic replication (OriLyt, UL112 and MIEP), with the same controls as in (C). Results are presented as total input fold enrichment (EVX-1 normalized).
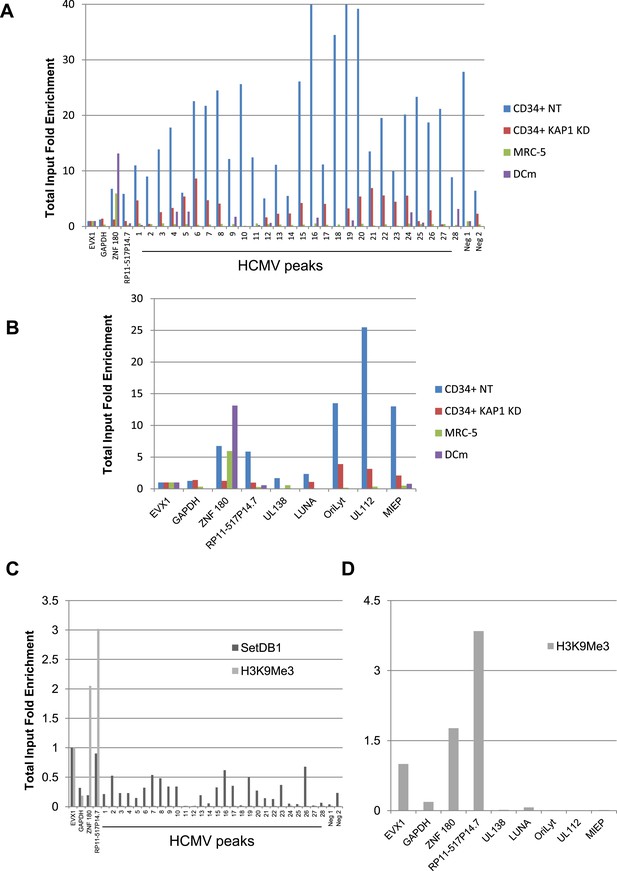
KAP1-dependent recruitment of HP1α and SETDB1 with H3K9 trimethylation of HCMV genome in latently infected HSC.
ChIP-PCR analysis of TB40-E-infected WT MRC-5 (A and B) KAP1-depleted CD34+ (A–D), WT CD34+ (A and B) or mDC (A and B) with antibodies against HP1α (A and B) SETDB1 (C) or H3K9me3 (C and D), as described in Figure 3.
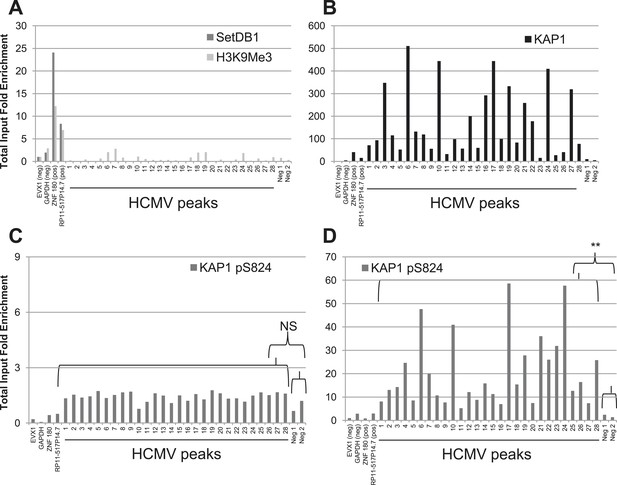
A KAP1 phosphorylation switch governs HCMV progression from latency to lytic replication.
ChIP-PCR of indicated HCMV genomic regions was performed with anti-SetDB1 and anti-H3K9Me3 (A), anti-KAP1 (B) or anti- S824 phosphoKAP1 antibodies (C and D) on material extracted from TB40-E-infected cells CD34+ cells (C) or mature dendritic cells derived therefrom (A, B, D), using same controls as in Figure 2. Results are presented as total input fold enrichment after EVX-1 normalization.
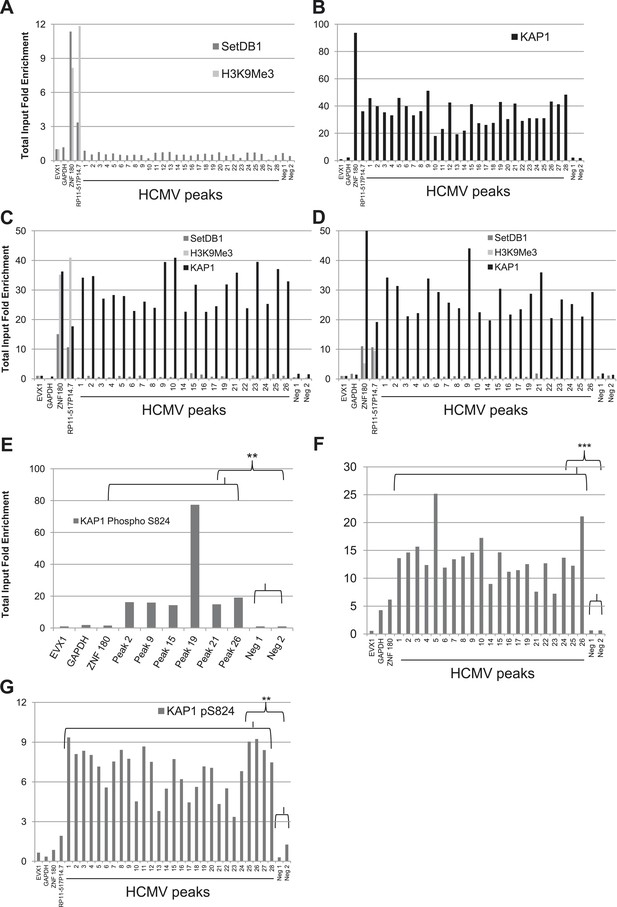
A KAP1 phosphorylation switch distinguishes HCMV latency and lytic replication.
ChIP-PCR with SETDB1-, H3K9Me3-, KAP1-, pS824KAP1-specific antibodies were performed on MRC-5 fibroblasts (A, B, F, G) or CD34+ HSC (C, D, E) infected with the HCMV TB40-E (A, B, G) or AD169 (C, D, E, F) strains, as described in Figure 3.
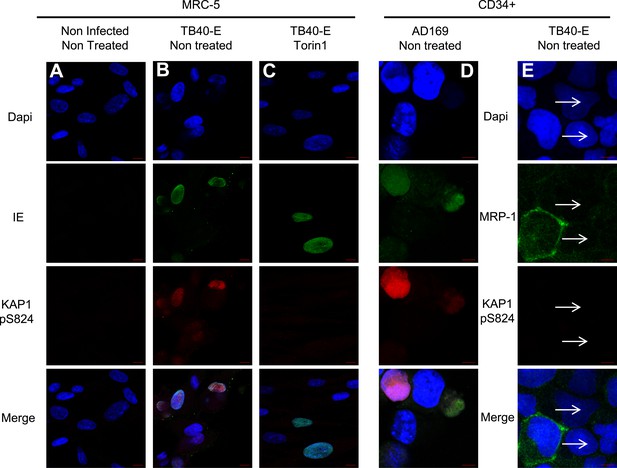
mTOR inhibition prevents HCMV-induced KAP1 phosphorylation.
MRC-5 (A, B, C) and CD34+ (D and E) cells, infected (AD169 or TB40-E) or not (Non Infected) with HCMV, and treated or not (Non Treated) with mTOR inhibitor (Torin1) were examined by immunofluorescence with anti-IE and anti-MRP1 antibodies (Alex-488, green), and anti-S824 phosphoKAP1 antibodies (Alexa 568, red). DNA was stained with Dapi (blue). White arrows represent latently infected cells (MRP-1 negative cells). Scale Bar in white represents 10 µm for MRC-5 and 5 µm for CD34+. All pictures are representative of slide overview from three independent experiments.
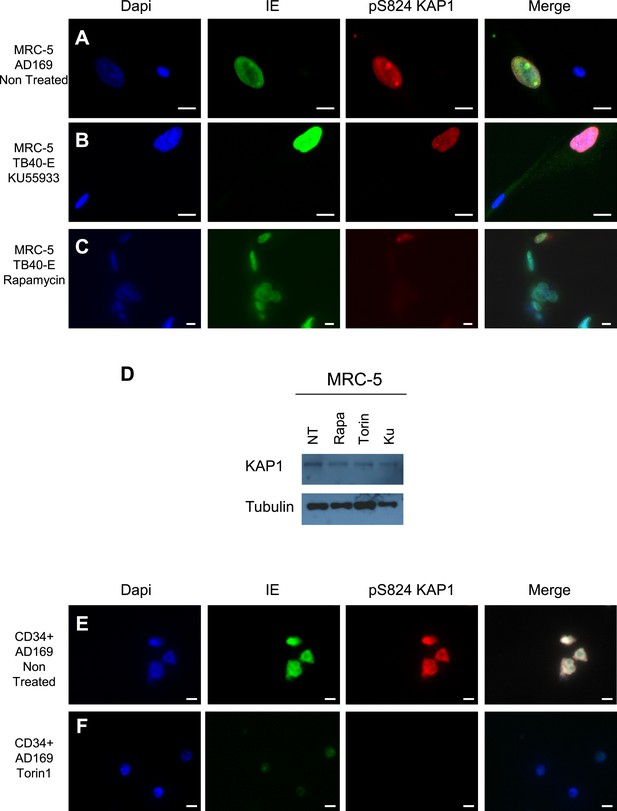
mTOR and HCMV-associated KAP1 phosphorylation.
(A–F) Confocal microscopy coupled to immunofluorescence was performed on MRC-5 (A–C) or CD34+ (E and F) HCMV infected (TB40-E, AD169) or not (Non Infected) and treated or not (Non Treated) with mTOR (Rapamycin, Torin1) or ATM (KU55933) inhibitors. Staining was performed with antibodies against IE (Alexa 488, green), and pS824KAP1 (Alexa 568, red), staining DNA with Dapi (blue). White scale bar, 10 µm. All pictures are representative of results obtained in three independent experiments. (D) KAP1 levels in MRC-5 Treated or not (NT) with Rapamycin (Rapa) Torin-1 (Torin) or KU55933 (KU), analyzed by western-blot. Results are representative of three independent experiments.
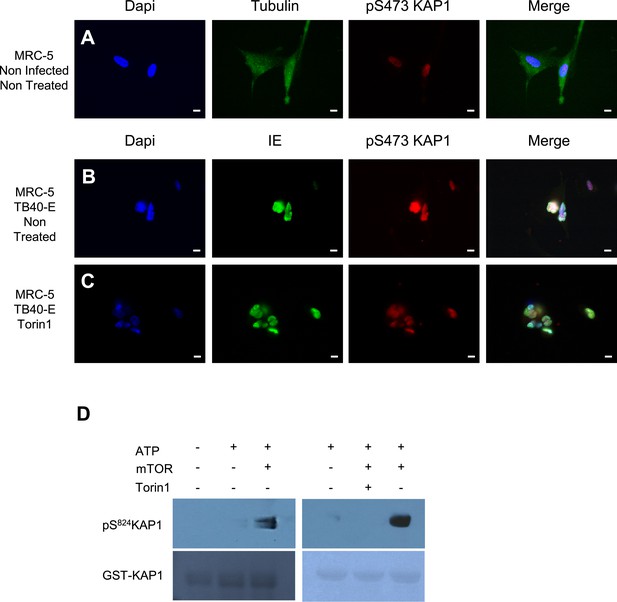
mTOR and HCMV-associated KAP1 phosphorylation.
(A–C) Confocal microscopy coupled to immunofluorescence was performed on MRC-5 (A–C), HCMV infected (TB40-E) or not (Non Infected) and treated or not (Non Treated) with mTOR inhibitors (Torin1). Staining was performed with antibodies against IE or anti-β-Tubulin (Alexa 488, green), and pS473KAP1 (Alexa 568, red), staining DNA with Dapi (blue). White scale bar, 10 µm. All pictures are representative of results obtained in three independent experiments. (D) In vitro phosphorylation of GST-KAP1 by recombinant mTOR, using Western blot and Ponseyu staining to detect pS824KAP1 and total KAP1, respectively.
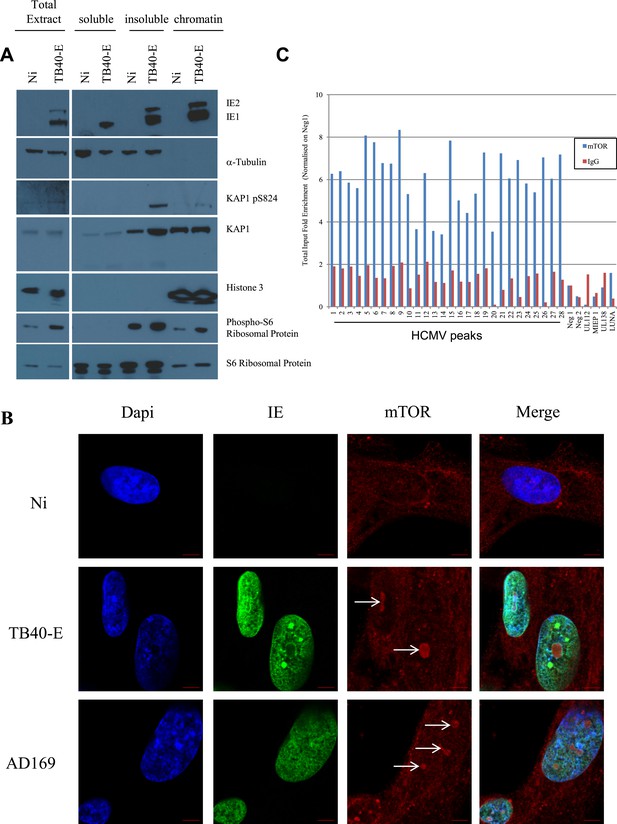
mTOR associates with the HCMV genome during lytic replication.
(A) MRC-5 cells were infected 3 days (TB40-E) or not (NT) with HCMV and harvested for cell fractionation. IE1/2, α-Tubulin, KAP1, KAP1 pS824, Histone 3 and phosphorylated or not S6 ribosomal protein expression/localization in Total extract, cytosolic (soluble) nuclear (insoluble) and chromatin (chromatin) parts were analysed by Western-Blot. (B) Immunofluorescence by confocal microscopy was performed on MRC-5, HCMV infected (TB40-E, AD169) or not (Non Infected). Staining was performed with anti-IE (Alex-488, green), and anti-mTOR antibodies (Alexa 568, red). DNA was stained with Dapi (blue). Scale Bar in white represents 5 µm. (C) ChIP-PCR for indicated HCMV genomic regions were performed with anti-mTOR or control-IgG on material extracted from TB40-E-infected MRC-5. Results are presented as total input fold enrichment after HCMV Neg1 region normalization.
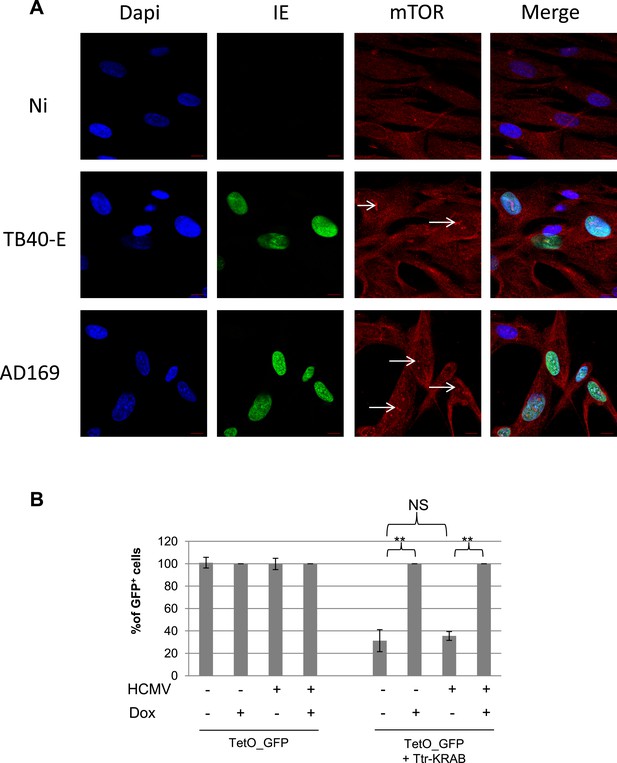
mTOR nuclear translocation and KAP1 activity in HCMV replicating cells.
(A) Confocal microscopy coupled to immunofluorescence was performed on MRC-5, infected (TB40-E, AD169) or not (Non Infected) with HCMV, using antibodies against IE- (Alex-488, green) or mTOR (Alexa 568, red), staining DNA with Dapi (blue). White scale bar, 10 µm. (B) MRC-5 cells were transduced with a lentiviral vector expressing GFP from the TetO-PGK promoter (TetO-GFP) with or without tTR-KRAB, infected or not with HCMV and treated or not with DOX as indicated. Results are presented as % of GFP positive cells, using non-infected and Dox treated cells as a 100% reference. (n = 3, **p < 0.01, error bars as s.d.).
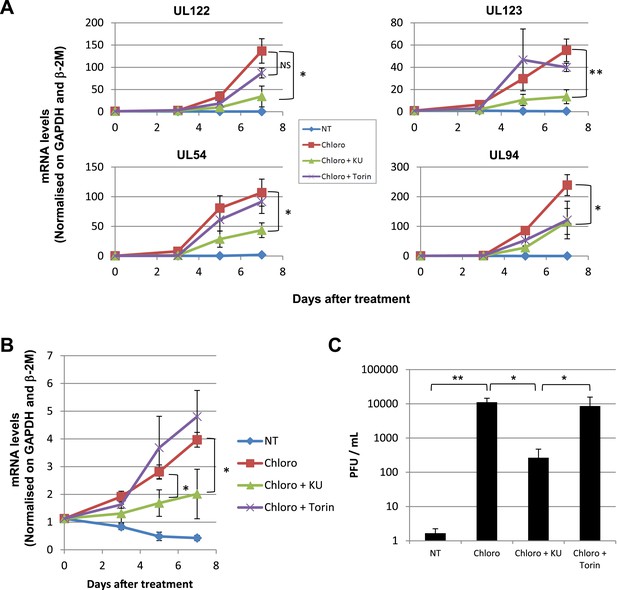
Forcing HCMV out of latency in CD34+ cells by KAP1 pharmacological manipulation.
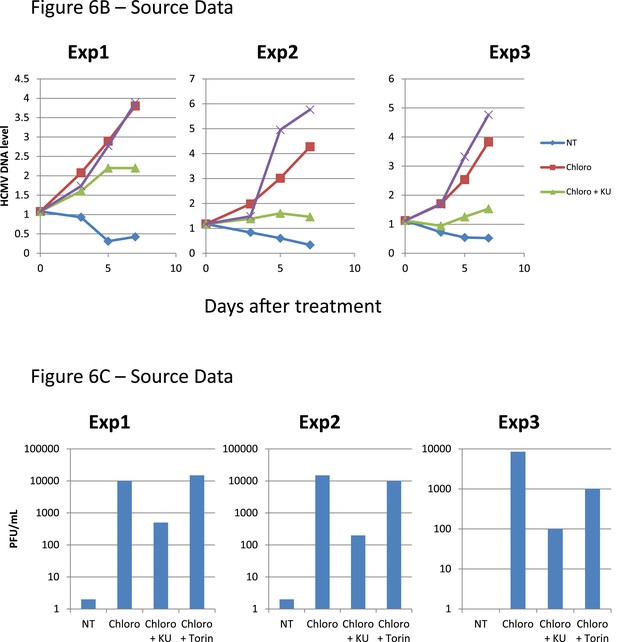
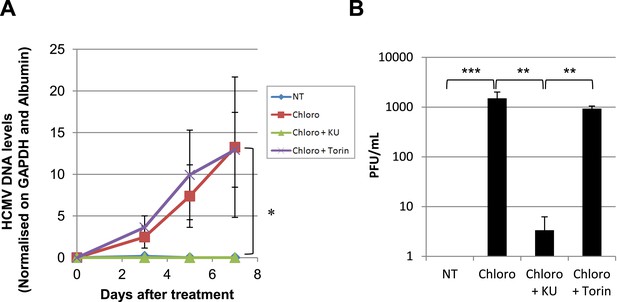
After 5 days of infection with TB40-E, CD34+ cells were treated three times (Day 0, 3 and 5) or not (NT) with Chloroquine (Chloro) alone or in combination with the mTor inhibitor Torin-1 (Torin) or the ATM inhibitor KU59933 (KU). (A) Indicated HCMV transcripts were quantified by RT-qPCR, using GAPDH and β-2 microglobulin for normalization. (B) HCMV DNA associated with the TB40-E-infected HSC was quantified by qPCR, normalizing with the GAPDH and albumin genes. Data are presented as average of three different experiments performed with cells from independent donors. (C) Supernatant from the TB40-E-infected CD34+ cells, harvested after 7 days of treatment and the viral production was quantified by classical plaque assay on MRC-5 fibroblasts. Results are presented as PFU/ml. Histogram represents an average of three different experiments performed with HSC from three independent donors (n = 3, *p < 0.05, **p < 0.01, error bars as s.d.). See Figure 6—figure supplements 5, 6 for all individual experiments.
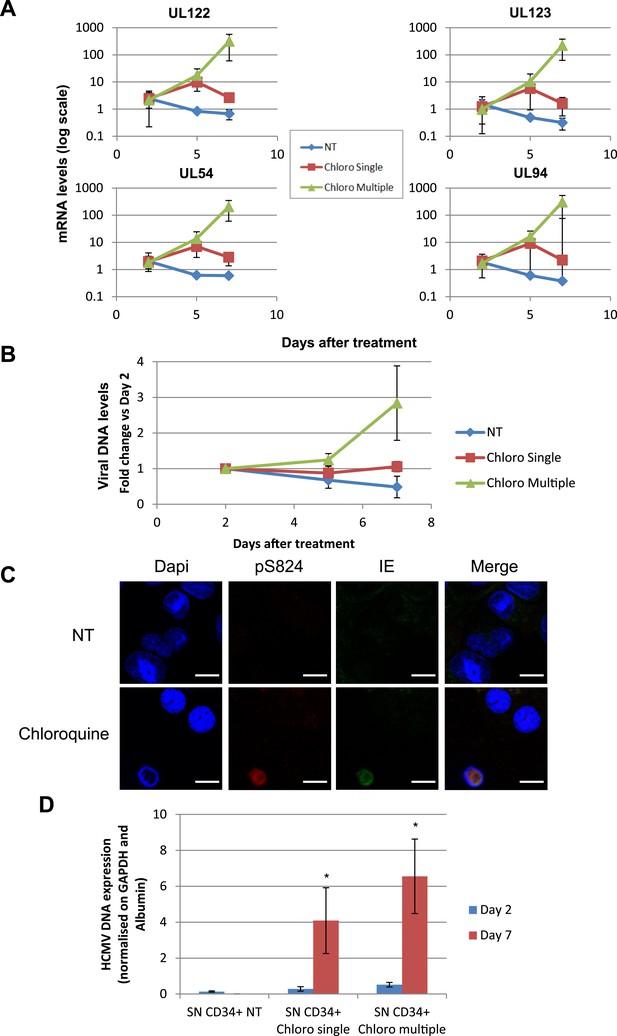
HCMV can be forced out of latency in CD34+ cells by pharmacological manipulation.
After 5 days of infection with TB40-E, CD34+ cells were treated or not (mock) with chloroquine in single or multiple doses as indicated. Indicated HCMV transcripts were quantified by RT-qPCR, using GAPDH and β-2 Microglobulin for normalization (A). HCMV DNA associated with the TB40-E-infected HSC was quantified by qPCR, normalizing with the GAPDH and albumin genes (B). Data are presented as average of three different experiments performed with cells from independent donors. (C) Immuno-fluorescent staining was performed on CD34+ cells for pS824KAP1 (pS824), HCMV IE1-2 (IE), or total KAP1 (KAP1), using Dapi for DNA. White scale bar, 10 µm. All pictures are representative of three independent experiments. (D) Supernatant from the TB40-E-infected CD34+ cells, harvested after 7 days of treatment, was used to infect MRC-5 fibroblasts, which were assessed for their viral DNA content 2 and 7 days later by qPCR. Results are presented as HCMV DNA level normalized for GAPDH and Albumin. Histogram represents an average of three different experiments performed with HSC from three independent donors (n = 3, *p < 0.05, error bars as s.d.).
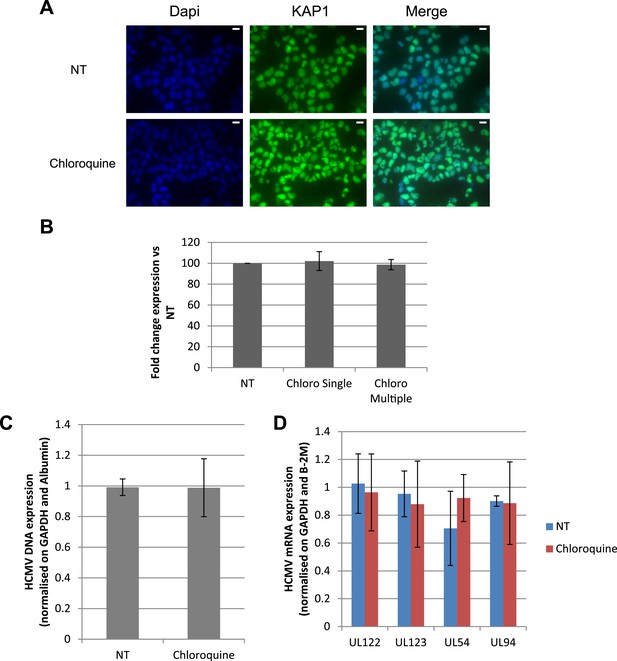
HCMV can be forced out of latency in CD34+ cells by pharmacological manipulation.
(A) Immuno-fluorescent staining was performed on CD34+ cells for total KAP1 (KAP1), using Dapi for DNA. White scale bar, 10 µm. All pictures are representative of three independent experiments. (B) Percentage of CD34+ cells after 7 days of treatment was quantified by FACS with a PE-anti-CD34 antibody. Results are presented of fold change expression vs NT of three independent experiments. HCMV DNA (C) and mRNAs (D) were quantified by qPCR in MRC-5 cells infected for 5 days with TB40-E in the presence or absence of chloroquine. Results are presented as HCMV DNA or mRNA levels normalized for GAPDH and Albumin for DNA, GAPDH and β-2M for mRNA.
HCMV can be forced out of latency in CD34+ cells by pharmacological manipulation.
(A) RT-qPCR analysis of indicated HCMV transcripts in cord blood CD34+ cells infected with the TB40-E strain 7 days prior to being transduced or not (NT) with lentivectors expressing (shKAP1) or not (empty) a small hairpin RNA against Kap1. RT-qPCRs were performed 7 days after lentiviral transduction treated or not (NT) with chloroquine as described in Figure 6. Results are presented as average of fold change expression vs NT after GAPDH and β-2M normalization (n = 4, *p < 0.05, **p < 0.01, error bars as s.d.). (B) HCMV production in CD34+ cells was quantified by plaque assay on MRC-5 cells. Results are presented as average of PFU/ml (n = 3, *p < 0.05, **p < 0.01, error bars as s.d.).
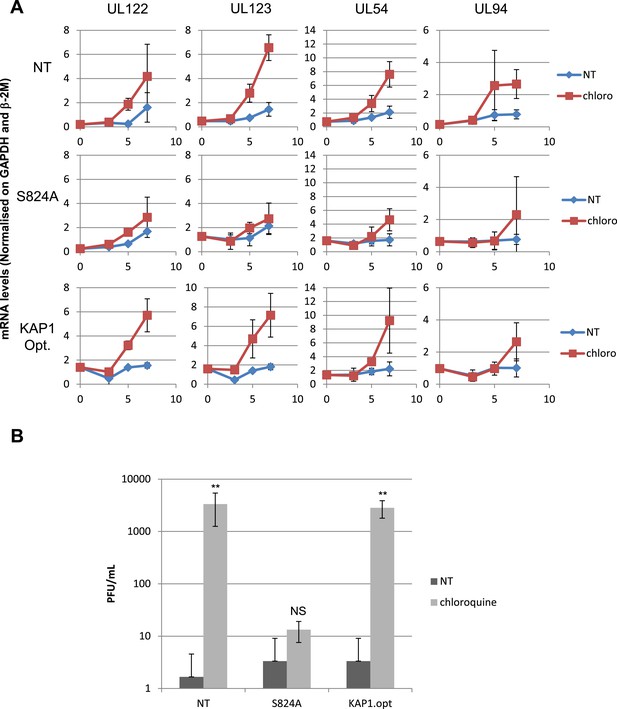
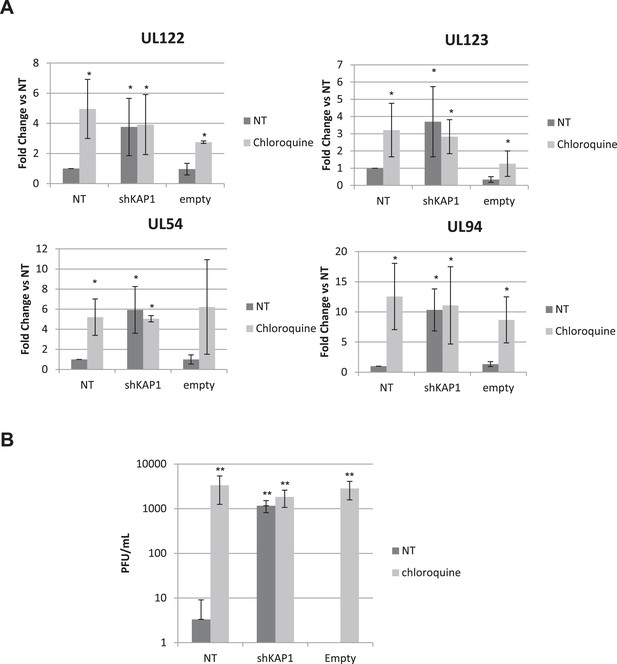
HCMV can be forced out of latency in CD34+ cells by pharmacological manipulation.
(A) RT-qPCR analysis of indicated HCMV transcripts in CD34+ cells infected with the TB40-E strain, 3 days after transduction or not (NT) with lentiviral vectors expressing or not (empty) an shRNA (shKAP1) against Kap1, together with a vector expressing an shRNA-resistant allele of the S824 KAP1 phospho-resistant mutant (S824A) or a wild type KAP1 (KAP1.opt) lentiviral vector. After 5 days of infection with TB40-E, CD34+ cells were treated three times (Day 0, 3 and 5) or not (NT) with Chloroquine (Chloro). Indicated HCMV transcripts were quantified by RT-qPCR, using GAPDH and β-2 microglobulin for normalization. (B) Supernatant from the TB40-E-infected CD34+ cells, harvested after 7 days of treatment and the viral production was quantified by classical plaque assay on MRC-5 fibroblasts. Results are presented as PFU/ml. Histogram represents an average of three different experiments performed with HSC from three independent donors (n = 3, **p < 0.01, error bars as s.d.).
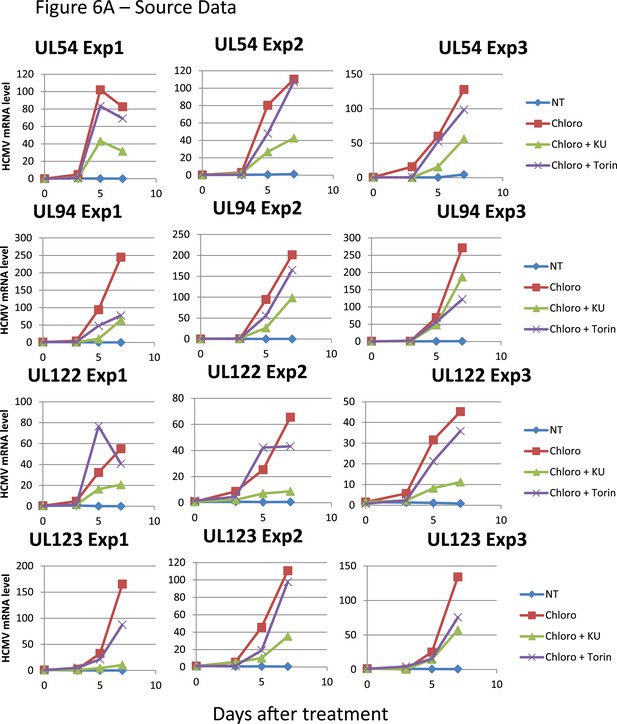
HCMV can be forced out of latency in CD34+ cells by pharmacological manipulation.
Figure 6A source datas.
HCMV can be forced out of latency in CD34+ cells by pharmacological manipulation.
Figure 6B,C source datas.
Inducing KAP1 phosphorylation releases HCMV from latency in monocytes.
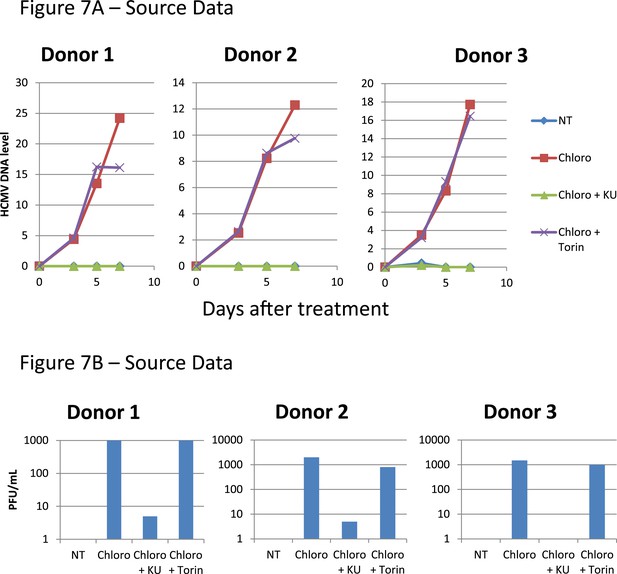
Monocytes from HCMV seropositive donors were purified and treated three times (Day 0, 3 and 5) or not (NT) with Chloroquine (Chloro) alone in combination with Torin-1 (Torin) or KU59933. (A) HCMV DNA associated with the monocytes was quantified by qPCR, normalizing with the GAPDH and albumin genes. Data are presented as average of three different experiments performed with cells from independent donors. (B) Supernatant from the monocytes, harvested after 7 days of treatment and the viral production was quantified by classical plaque assay on MRC-5 fibroblasts. Results are presented as PFU/ml. Histogram represents an average of three different experiments performed with HSC from three independent donors (n = 3, *p < 0.05, **p < 0.01, error bars as s.d.). See Figure 7—figure supplement 2 for all individual experiments.
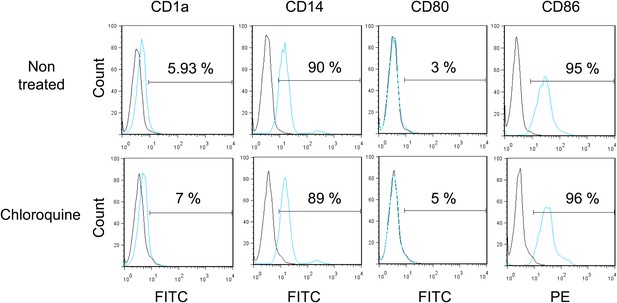
Monocytes do not differentiate during pharmacological reactivation of HCMV.
After 7 days of treatment with chloroquine, monocytes were examined by FACS for the surface expression of CD1a, CD14, CD80 and CD86, analyzing results with the FlowJo software. Graphs are representative of experiments performed with cells from three different HCMV-seropositive donors.
Monocytes do not differentiate during pharmacological reactivation of HCMV.
Figure 7 source datas.
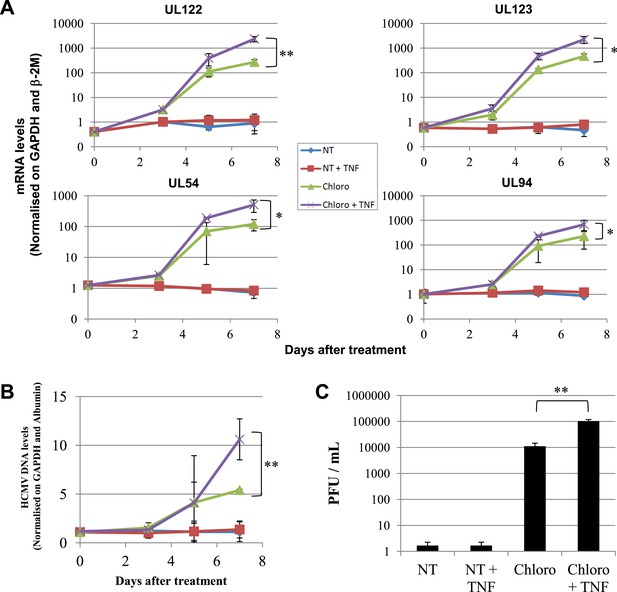
Combining KAP1 phosphorylation and NF-κB induction increases HCMV activation from latently infected HSC.
After 5 days of infection with TB40-E, CD34+ cells were treated three times (Day 0, 3 and 5) or not (NT) with chloroquine (Chloro) in combination or not with 5 ng/ml of recombinant TNF-α (TNF). (A) Indicated HCMV transcripts were quantified by RT-qPCR, using GAPDH and β-2 microglobulin for normalization. (B) HCMV DNA associated with the TB40-E-infected HSC was quantified by qPCR, normalizing with the GAPDH and albumin genes. Data are presented as average of three different experiments performed with cells from independent donors. (C) Supernatant from the TB40-E-infected CD34+ cells, harvested after 7 days of treatment and the viral production was quantified by classical plaque assay on MRC-5 fibroblasts. Results are presented as PFU/ml. Histogram represents an average of three different experiments performed with HSC from three independent donors (n = 3, *p < 0.05, **p < 0.01, error bars as s.d.). See Figure 8—figure supplements 2, 3 for all individual experiments.
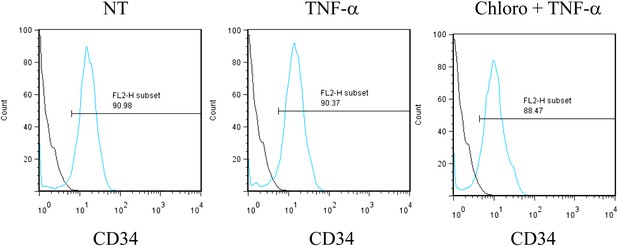
Pharmacological activation of HCMV does not trigger CD34+ cells differentiation.
After 7 days of treatment with chloroquine and TNF-α, cells were analyzed by FACS for CD34 surface expression. Representative of experiments performed on cells from three different donors.
Pharmacological activation of HCMV does not trigger CD34+ cells differentiation.
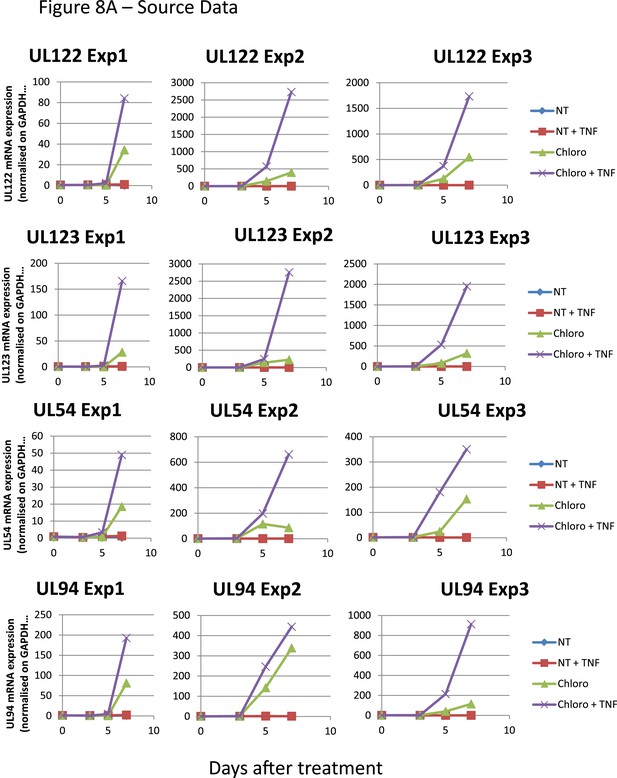
Figure 8A source datas.
Pharmacological activation of HCMV does not trigger CD34+ cells differentiation.
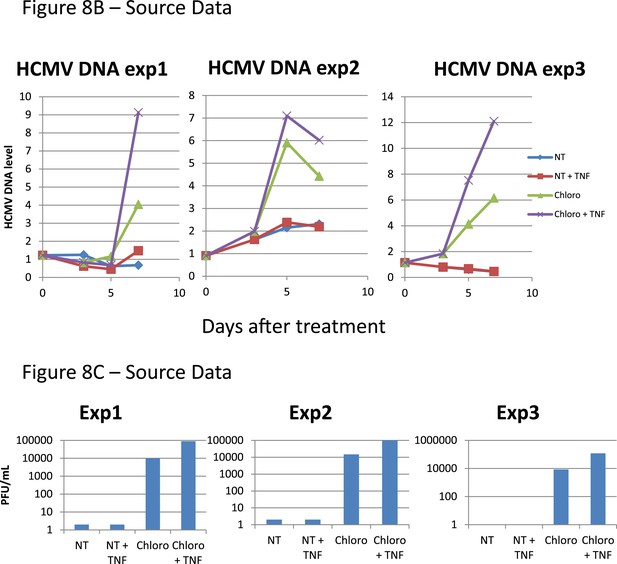
Figure 8B,C source datas.
Additional files
-
Supplementary file 1
Localization of KAP1 peaks on HCMV TB40-E genome. GenBank: EF999921.1.
- https://doi.org/10.7554/eLife.06068.031
-
Supplementary file 2
Primers used in this work and their sequences.
- https://doi.org/10.7554/eLife.06068.032
-
Source code 1
Custom-made PERL program.
- https://doi.org/10.7554/eLife.06068.033





