Generation of contractile actomyosin bundles depends on mechanosensitive actin filament assembly and disassembly
Figures
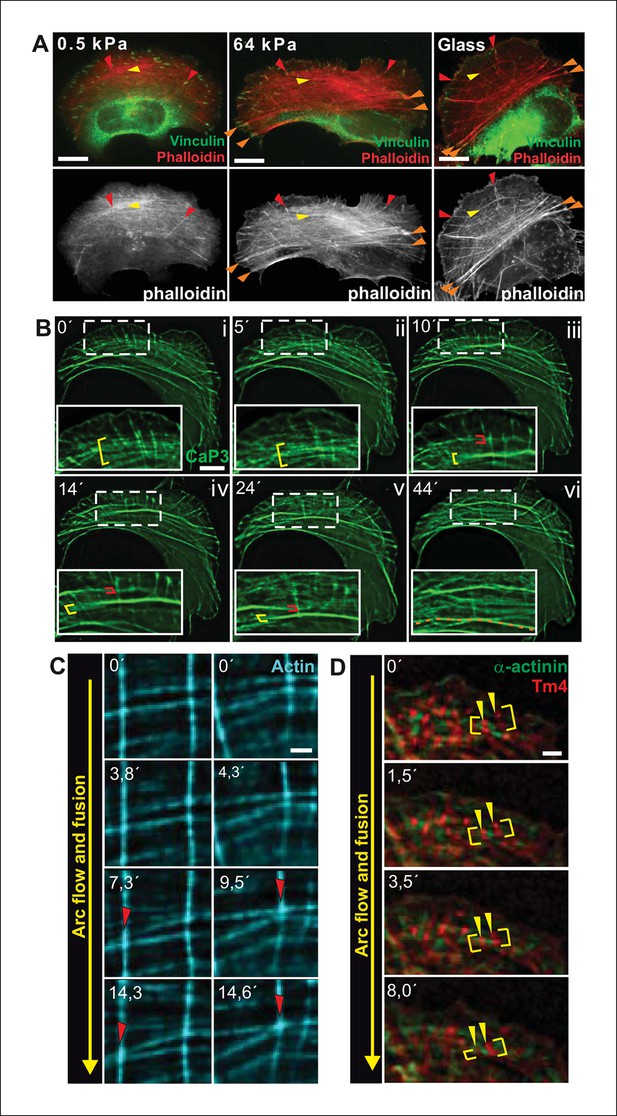
Transverse arcs fuse during centripetal flow to generate a contractile ventral stress fiber.
(A) U2OS cells have three subtypes of stress fibers: Dorsal stress fibers (red arrowheads), which are attached to focal adhesion at their distal end; Transverse arcs (yellow arrowheads), which are curved actomyosin bundles oriented parallel to the leading edge; Ventral stress fibers (orange arrowheads), which are thick contractile bundles connected to focal adhesions at both ends. All three stress fiber categories are present in cells grown on glass or on stiff (E = 64 kPa) silicone matrix, whereas assembly of contractile ventral stress fibers is compromised in cells grown on soft (0.5 kPa) matrix. (B) Live-imaging of U2OS cells expressing GFP-calponin-3 (CaP3) revealed that transverse arcs fuse with each other during centripetal flow to form thicker actomyosin bundles. Red and yellow brackets highlight fusing arcs, and the orange dashed line indicates the thick ventral stress fiber derived from the fusing arcs. (C) Arc fusion often initiates at the connection points of dorsal stress fibers and transverse arcs (indicated by red arrowheads). Two separate video frame series are shown in the panels. In the images, dorsal stress fibers and arcs are oriented vertically and horizontally, respectively. Stress fibers were visualized by expression of GFP-actin. Bar, 1 μm. (D) Live imaging of YFP-Tm4 and CFP-α-actinin-1 expressing U2OS cell reveals that homotypic coalescence of adjacent Tm4 and α-actinin foci occurs during arc fusion, thus allowing to retain the periodic pattern of transverse arcs. Yellow brackets indicate fusing arcs and yellow arrowheads highlight pairs of fusing α-actinin-1 foci. Bar, 1 μm.
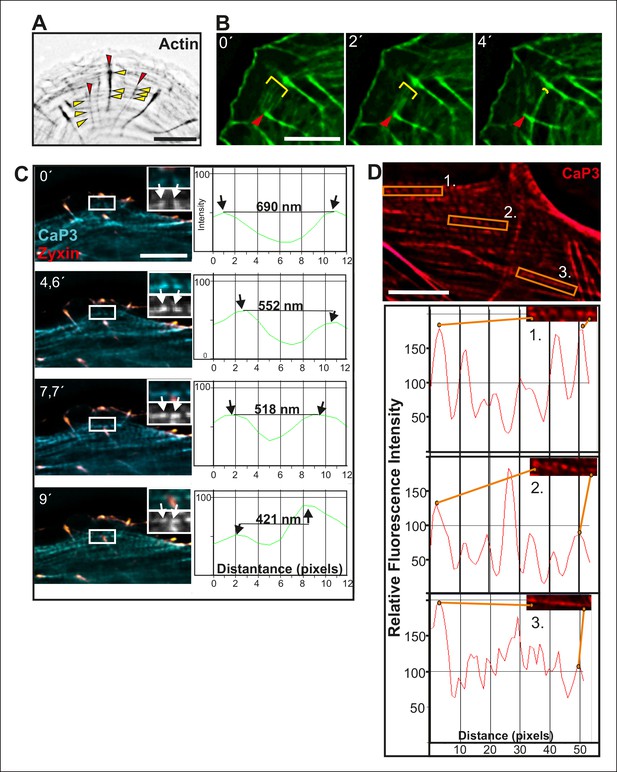
Fusion of transverse arcs.
(A) Dorsal stress fibers and transverse arcs have multiple connections points, thus forming a spider-net -like structure. Stress fibers were visualized by expression of GFP-actin. Red arrowheads indicate dorsal stress fibers and yellow arrowheads transverse arcs. Bar, 5 μm. (B) Connections between dorsal stress fibers and transverse arcs are formed early during the arc fusion process. The stress fiber network was visualized by expression of GFP-CaP3. Red arrowhead indicates the distal end of a dorsal stress fiber, and yellow brackets highlight arc fusion. Bar, 5 μm. (C) Frames from a movie of representative GFP-CaP3 and CFP-zyxin expressing cell demonstrates that the spacing between individual CaP3 foci decreases as the arc flows towards the cell center and fuses with adjacent arcs. Bar, 5 μm. (D) Also in fixed U2OS cells expressing GFP-CaP3, the distance between adjacent CaP3 foci correlates inversely with the distance of the arc from the cell edge. Bar, 5 μm.
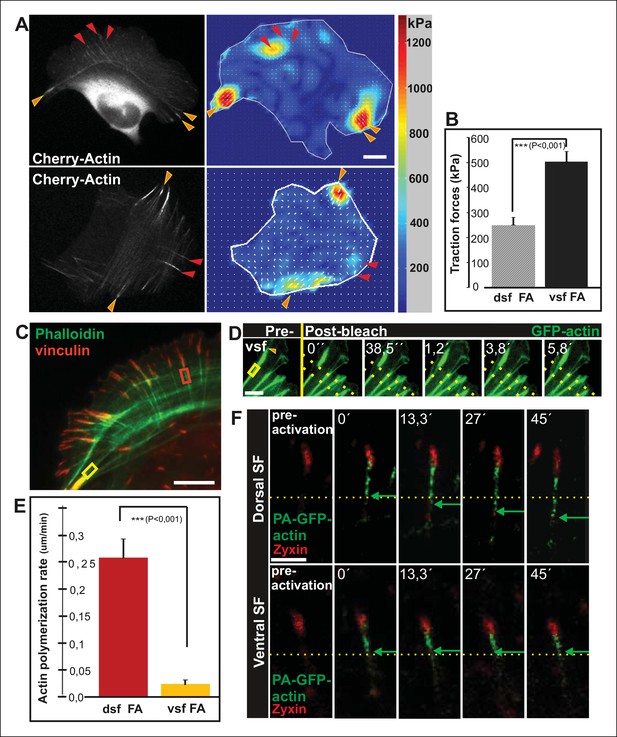
Vectorial actin polymerization at focal adhesions halts upon increased contractility and formation of ventral stress fibers.
(A) Representative images of U2OS cells grown on 26 kPa polyacrylamide dishes with fluorescent nanobeads together with the corresponding force maps. Adhesions located at the ends of ventral stress fibers (orange arrowheads) apply stronger forces to their substrate compared to adhesions located at the ends of dorsal stress fibers (red arrowheads). Bar, 10 um. (B) Quantification of traction forces at adhesions located in the ends of dorsal and ventral stress fiber adhesions. Mean +/- SEM, n = 20 cells, 4–8 adhesions per cell. (C) Recovery of GFP-actin signal was measured next to dorsal stress fiber adhesions (dsf FA, red box) and ventral stress fiber adhesions (vsf FA, yellow box). Bar, 5 um. (D) Representative example of a fluorescence-recovery-after-photobleaching (FRAP) experiment performed on a GFP-actin expressing cell at a ventral stress fiber (vsf) region close to a focal adhesion. Yellow box indicates the photobleached region and the orange arrowhead the distal end of the ventral stress fiber. Scale, 3 um. (E) Quantification of the recovery speed (μm/min) for GFP-actin from focal adhesions located at the tips of dorsal stress fibers (dsf) or ventral stress fibers (vsf). Means +/- SEM, n (dorsal stress fibers) = 11; n (ventral stress fibers) = 17. (F) Activation of PA-GFP-actin in focal adhesions is followed by centripetal flow of photoactivated actin along the dorsal stress fiber. In contrast, PA-GFP-actin activated at a focal adhesion located at the tip of ventral stress fiber does not distribute from the adhesion to the stress fiber. Activated PA-GFP-actin is in green, focal adhesion marker mCherry-zyxin in red and the yellow dashed lines show the borders of the photoactivated region. Bar, 2,5 μm.
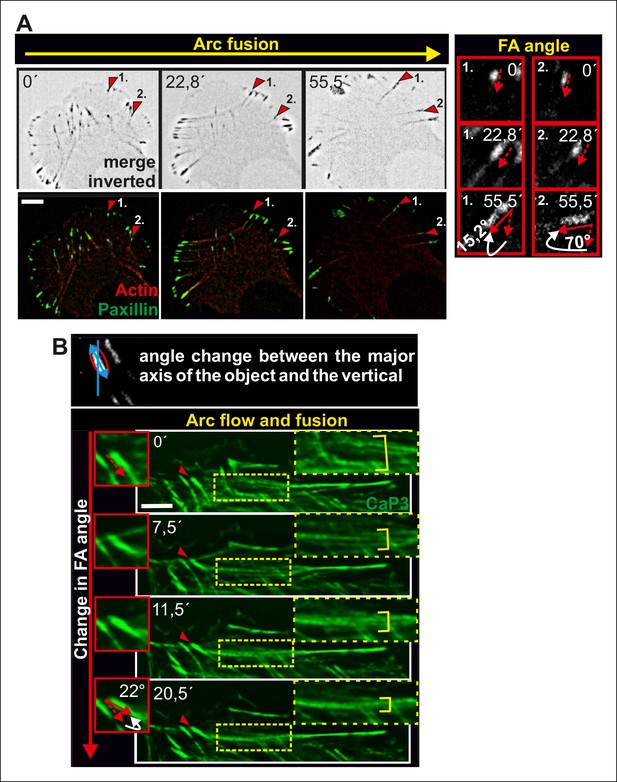
Alignment of focal adhesions linked to the ends of transverse arcs.
(A) Thick actomyosin bundles derived through arc fusion apply tension to focal adhesions leading to enlargement and alignment of those focal adhesions that are linked to the ends of the actomyosin bundle through dorsal stress fibers. Eventually, this results in a formation of a straight ventral stress fiber, where the focal adhesions are aligned along the direction of the actomyosin bundle. Focal adhesions and stress fibers were visualized by expression of GFP-paxillin and mCherry-actin, respectively. The two ‘terminal’ focal adhesions, whose alignments during the process are shown in the insets, are indicated by red arrowheads in the larger cell frames. Bars, 10 μm. (B) Fusion of transverse arcs into thicker actomyosin structures correlates with the alignment of the ‘terminal’ focal adhesions along the direction of the arc bundle. Stress fibers were visualized by expression of GFP-CaP3. Bar, 5 μm.
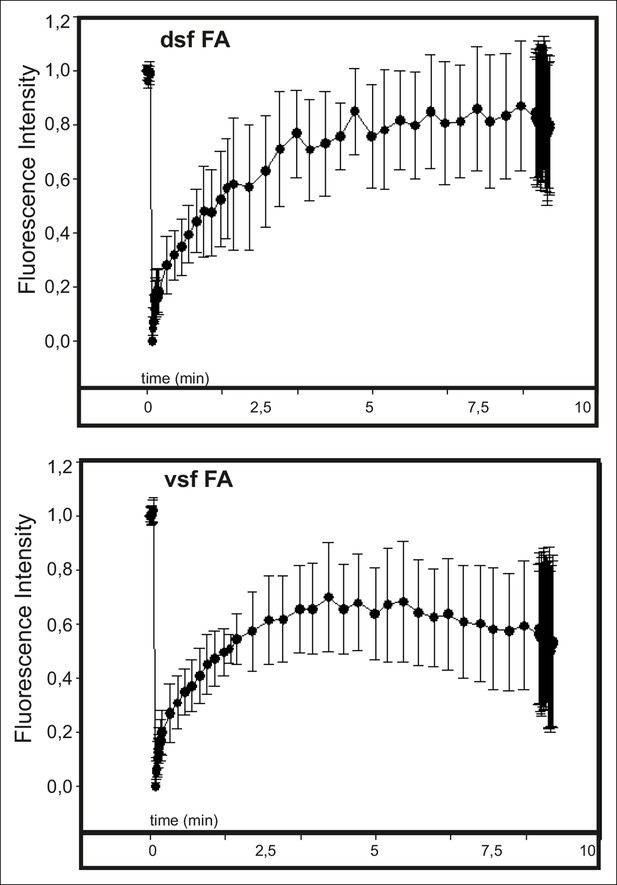
Actin dynamics in focal adhesions located at the tips of dorsal and ventral stress fibers.
The dynamics of GFP-actin in focal adhesions of U2OS cells were examined by fluorescence recovery after photobleaching (FRAP). Recovery curves with mean +/- SD are shown. n (dorsal stress fiber adhesions) = 11; n (ventral stress fiber adhesion) = 12.
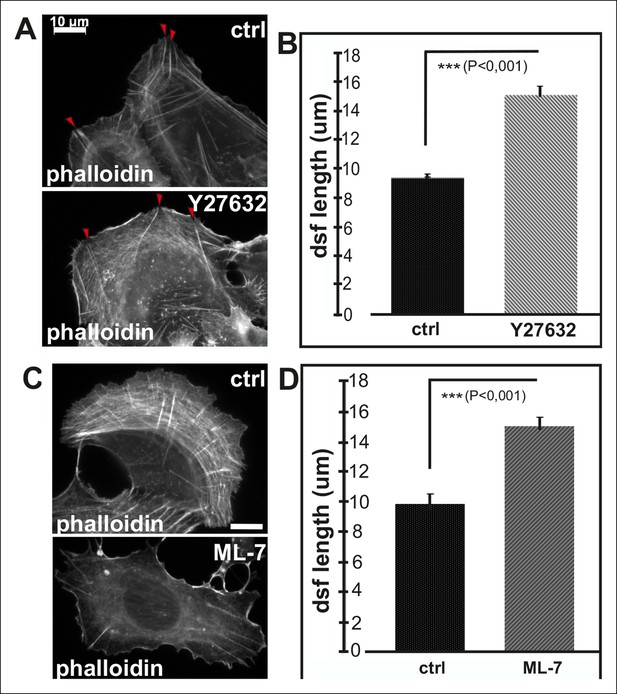
Inhibition of myosin light chain phosphorylation results in formation of abnormally long dorsal stress fibers.
(A) Examples of a control U2OS cell and a cell incubated for 2 h with ROCK kinase inhibitor, Y27632. This inhibitor results in an abnormal morphology of transverse arcs or their total disapparance. Y27632 –treated cells also contain fewer dorsal stress fibers, which appear abnormally long and thin. Red arrowheads indicate dorsal stress fibers. Bar, 10 μm. (B) Quantification of the lengths of dorsal stress fibers in control and Y27632 -treated cells. Mean lengths (+/- SD) of dorsal stress fibers from 20 cells is shown. (C) Inhibition of myosin light chain kinase (MLCK) by ML-7 leads to a loss of transverse arcs and concomitant defects in formation of ventral stress fibers. Bar, 10 μm. (D) Dorsal stress fibers are abnormally long in ML-7 treated cells. Mean length (+/- SD) of 55 dorsal stress fibers measured from control and ML-7 treated cells.
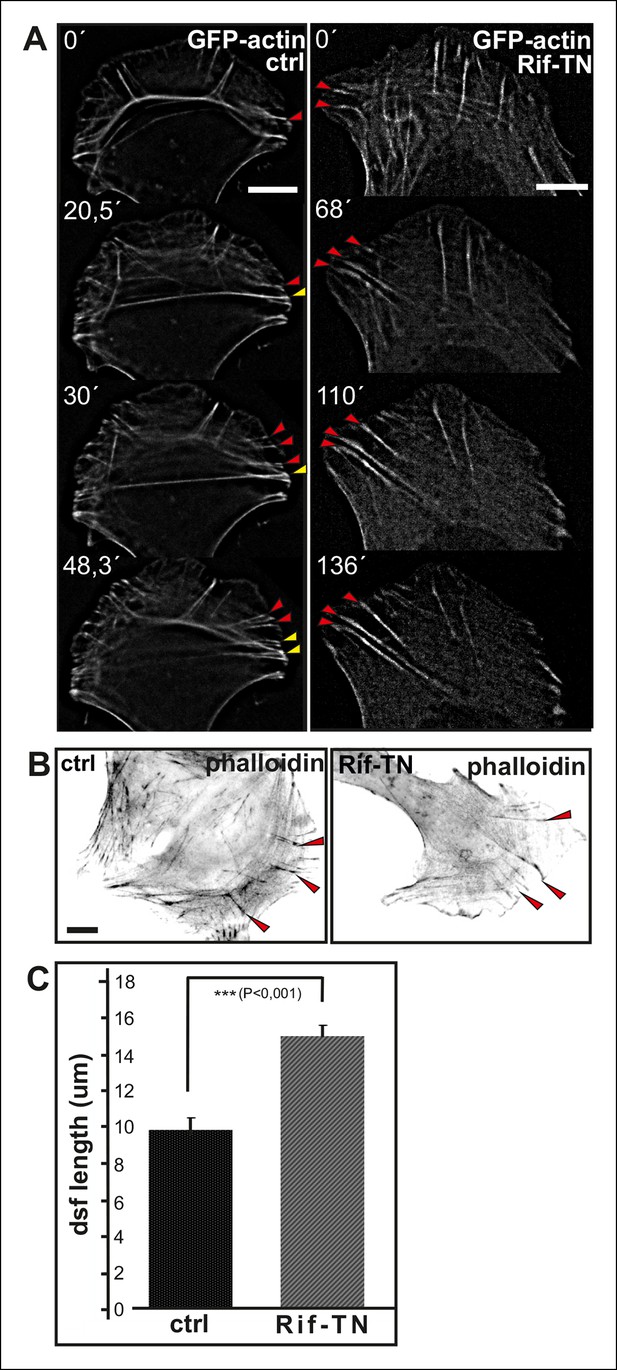
Expression of dominant inactive Rif leads to abnormal elongation of dorsal stress fibers.
(A) Video frames from a U2OS cells expressing GFP-actin alone (right panel) or GFP-actin and Rif-TN. In control cells, ventral stress fibers (yellow arrowhead) are generated through fusion of relatively short dorsal stress fibers (red arrows) and transverse arcs. Rif-TN expressing cells lack transverse arcs and concomitant formation of ventral stress fibers. Importantly, dorsal stress fibers in Rif-TN expressing cells are abnormally long and continue elongating through the observation period. Bar, 10 μm. (B) Images of phalloidin stained control and Rif-TN expressing cells. Dorsal stress fibers are indicated by red arrowheads. (C) Quantification of the lengths of dorsal stress fibers length from 20 control and Rif-TN expressing cells. Mean lengths of the dorsal stress fibers (+/- SEM) are shown.
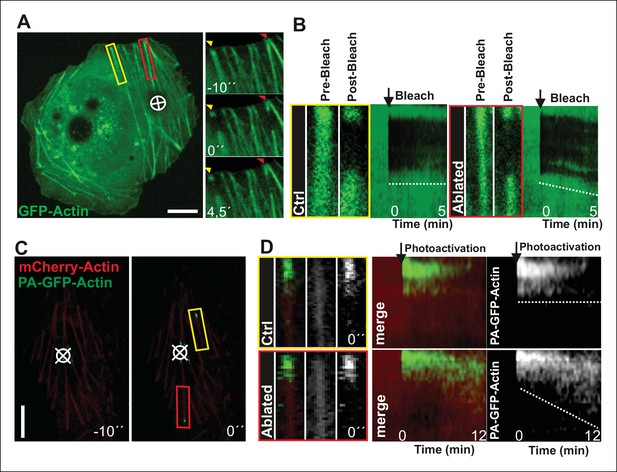
Local relaxation of ventral stress fibers induces vectorial actin polymerization at focal adhesion.
(A) The effect of tension on actin polymerization at focal adhesions was monitored by fluorescence recovery after photobleaching (FRAP) in laser-ablated (indicated by red box/arrowhead) and intact ventral stress fibers (indicated by yellow box/arrowhead). FRAP experiment was initiated 10 seconds after ablation. Symbol (¤) indicates the ablation site. (B) Kymographs recorded along the center of ablated and non-ablated ventral stress fiber regions (shown in the red and yellow boxes to the left from the kymograps) reveal that non-ablated and ablated fibers display differences in actin dynamics. In contractile control fibers, the rate of vectorial actin polymerization is slow (0,023 μm/min +/- 0,007 μm/min; SEM; n = 17), whereas relaxation induces vectorial actin polymerization from the adhesion located at the end of ablated ventral stress fiber (0,257 μm/min +/- 0,035 μm/min; SEM; n = 12). See also Figure 7B for a graphical representation of the data. (C) Photoactivation of GFP-PA-actin in contractile (yellow box) and relaxed (red box) ventral stress fibers. (D) Kymograph analysis performed along the center of indicated ventral stress fiber regions (shown in yellow and red boxes to the left from the kymographs) demonstrate induction of vectorial actin polymerization at the focal adhesion located in the end of an ablated ventral stress fiber. However, no detectable vectorial actin polymerization occurred in the non-ablated ventral stress fiber. Photoactivation was performed 10 s after ablation of the contractile stress fiber. Bars, 10 μm.
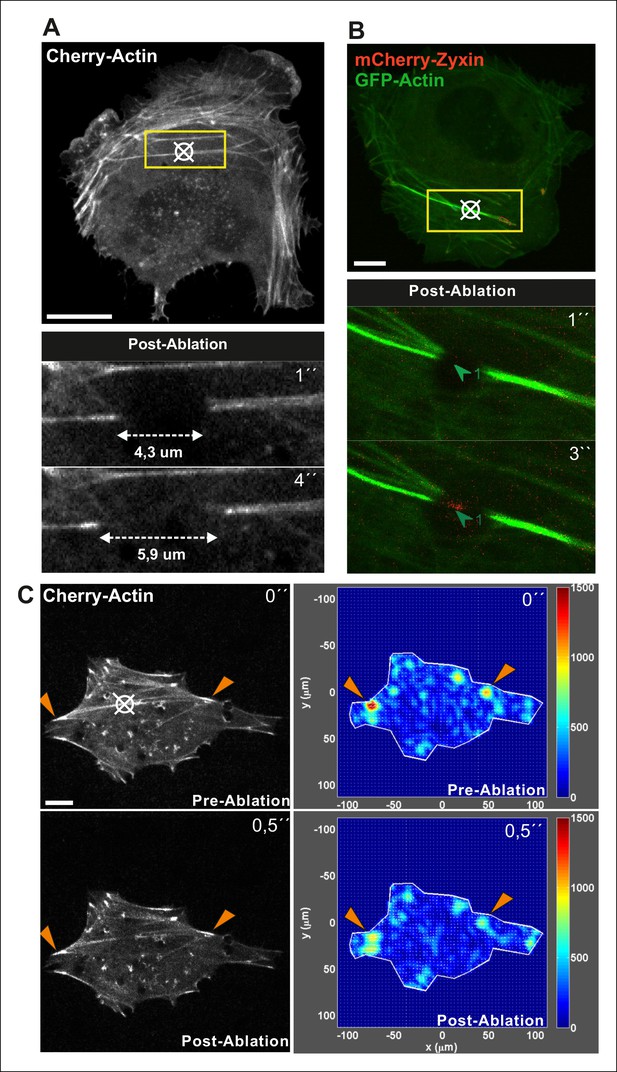
Method for laser ablation of ventral stress fibers.
Ablation of contractile stress fibers was achieved by using full laser power of 405 laser line (3I Marianas microscope) for 3 x 200 ms pulses. Symbol (¤) indicates ablation sites. Breakage of the fibers was confirmed by following retraction of mCherry-actin bundle with 561 channel for 10 seconds (A) or by immediate recruitment of a repair protein zyxin to the ablation site (B). Bar, 10 um. (C) Ablation of a ventral stress fiber leads to an immediate decrease in cell-mediated forces at focal adhesions located at the tips of the stress fiber. A representative example is shown in the figure, and quantification of the data from six adhesions at the ends of ablated stress fibers from three different cells revealed 27,3 +/- 7,0 (SD)% decrease in tension at the area around focal adhesions. Traction forces were measured both before, and 0,5 seconds after ablation. Bar, 10 um.
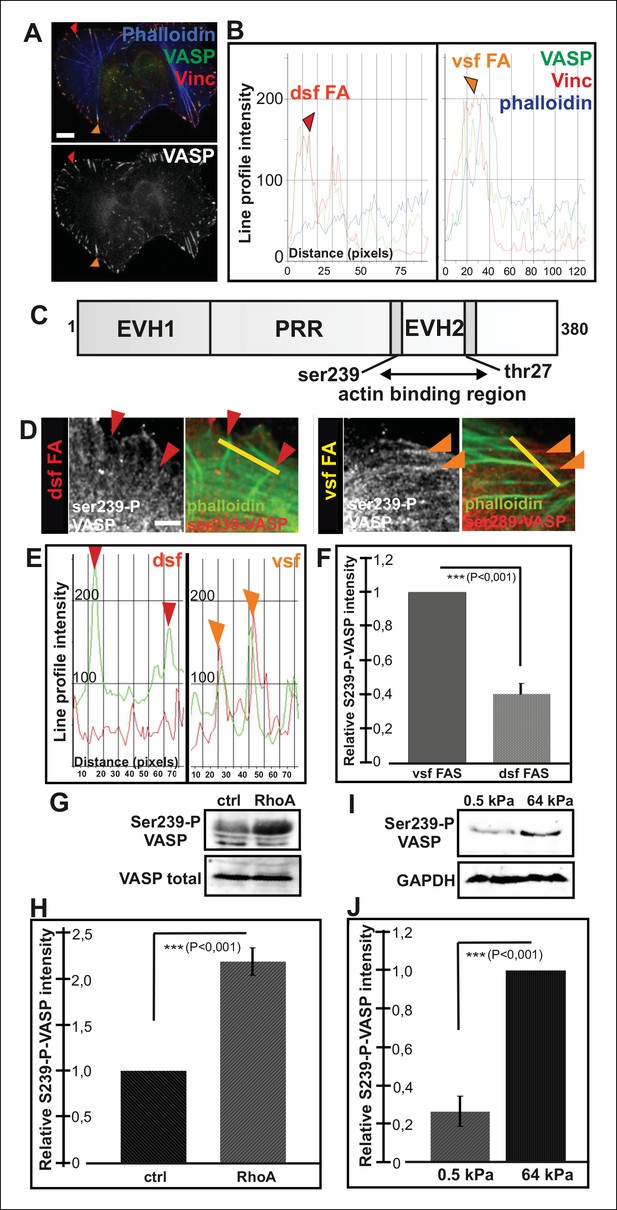
Increased VASP phosphorylation in focal adhesions located at the tips of ventral stress fibers.
(A) VASP localizes to focal adhesions at the tips of both dorsal and ventral stress fibers. Focal adhesions positioned at the tips of dorsal (dsf FA) and ventral stress fibers (vsf FA) are indicated by red and orange arrowheads, respectively. Bar, 10 μm. (B) Line profile intensity graphs of the adhesions (highlighted in panel A) and adjacent stress fiber regions show similar localizations of VASP and vinculin in focal adhesions positioned at the tips of dorsal and ventral stress fibers. (C) The domain structure of VASP. Phosphorylation of VASP at Ser239 and Thr278 inhibits its actin polymerization activity. (D) Localization of phospho-Ser239 VASP in focal adhesions at the tips of dorsal (red arrowheads) and ventral stress fibers (orange arrowheads). Bar, 5 μm. (E) Line profile intensities along the yellow lines (indicated in panel D) demonstrating that phospho-Ser239 VASP is enriched at the tips of ventral stress fibers, but not at the tips of dorsal stress fibers. phospho-Ser239-VASP–red; actin-green (F) Quantification of the relative fluorescence intensity ratio of phospho-Ser239-VASP: total VASP in focal adhesions located at the tips of dorsal (dsf FAs) and ventral (vsf FAs) stress fibers. The obtained intensity value from ventral stress fibers was set to 1. Mean intensity values (+/- SEM) of 21 adhesions are shown. (G) Western blot analysis demonstrating that expression of dominant active Rho-14V expression leads to an increase in the total Ser239 phosphorylation levels of VASP. (H) Quantification of the relative phospho-Ser239-VASP: total VASP ratios in control and Rho-14V transfected cells. The ratio in control cells was set to 1 and the mean values (+/- SEM) from three separate experiments are shown. (I) Western blot analysis demonstrating increased phospho-Ser239-VASP levels in cells grown on stiff (64 kPa) martix compared to cells grown on compliant (0.5 kPa) matrix. (J) Quantification of the relative phospho-Ser239-VASP: total VASP ratios in cells grown on soft (0.5 kPa) or rigid (64 kPa) matrices. The phospho-Ser239-VASP: total VASP ratio in cells from stiff matrix was set to 1 and the mean values (+/- SEM) from three separate experiments are shown.
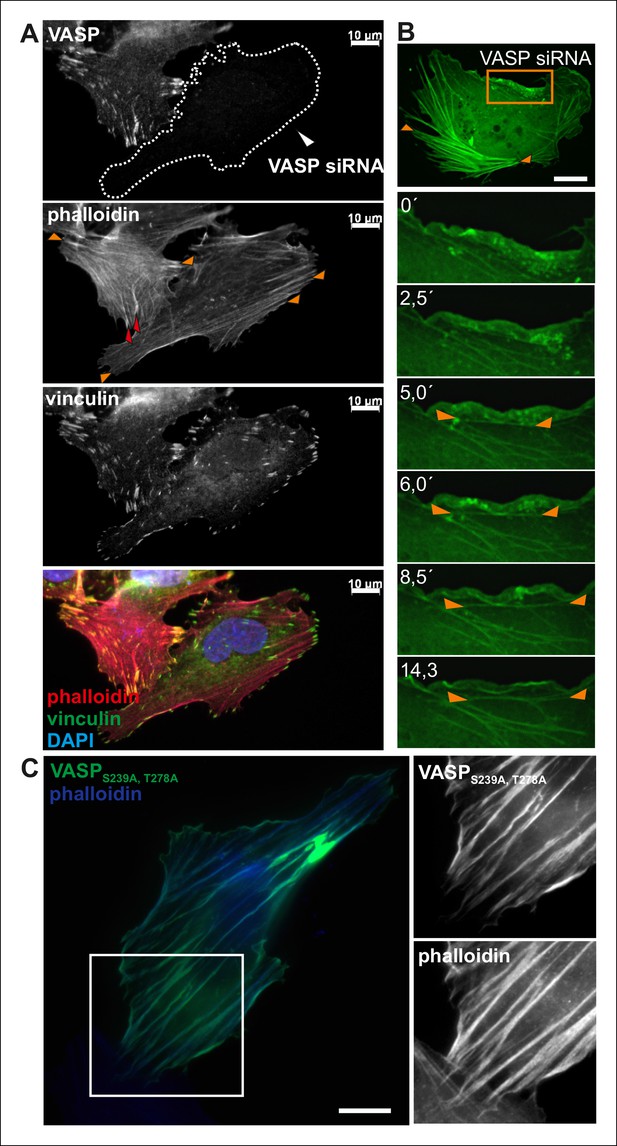
VASP regulates the elongation of dorsal stress fibers.
(A) Depletion of VASP from U2OS cells by siRNA leads to loss of dorsal stress fibers (examples indicated by red arrowheads in the control cell), but these cells still contain focal adhesions and ventral stress fibers (examples are indicated by orange arrowheads in the VASP-depleted and control cell). However, the ventral stress fibers are abnormally aligned in VASP knockdown cells. The cells were stained with VASP antibody, vinculin antibody (to visualize focal adhesions) and phalloidin (to visualize F-actin). The VASP depleted cell is surrounded by a dashed line. Bar, 10 μm. (B) VASP depleted cells still exhibit ventral stress fibers, but they are predominantly poorly aligned and form abnormal clusters (orange arrowheads). Time-lapse frames from a movie of VASP-depleted U2OS cell expressing GFP-actin also demonstrate that in the absence of VASP (and visible dorsal stress fibers), ventral stress fibers (orange arrowheads) form from transverse arcs very close to the lamellipodium. Bar, 10 μm. (C) Over-expression of constitutively active (Ser239Ala;Thr278Ala) VASP affects elongation of stress fibers and often leads to a formation of abnormally curly actin filament bundles (visualized by phalloidin staining). Bar, 10 μm.
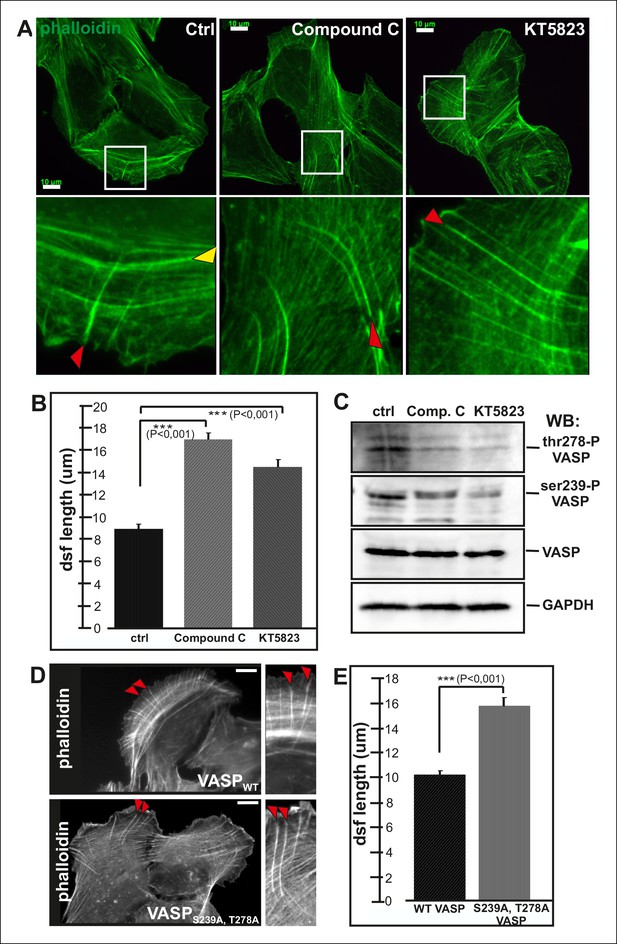
Elongation of dorsal stress fibers is regulated by VASP phosphorylation.
(A) U2OS cells treated with cAMPK and PKA inhibitors, compound C and KT5823, display abnormally long dorsal stress fibers (red arrows). Also fusion of arcs appears defective in compound C and KT5823 treated cells, because thick actin bundles (yellow arrowhead in the control cell) are largely absent from these cells. Actin filaments were visualized by phalloidin. Bar, 10 μm. (B) Quantification of dorsal stress fiber lengths (μm) from control cells and cells treated with compound C or KT5823. Mean lengths (+/- SEM) of 40 dorsal stress fibers from each sample are shown. (C) Western blot demonstrating that lysates of compound C or KT5823 -treated cells display decreased phosphorylation of VASP at Ser239 and Thr278. GADPH and total VASP were probed as loading controls. (D) Expression of constitutively active VASP mutant (Ser239Ala;Thr278Ala) induces formation of abnormally long dorsal stress fibers, whereas similar phenotype was not observed in wild-type VASP expressing cells. Bar, 10 μm. (E) Quantification of dorsal stress fiber lengths from cells expressing wild-type and Ser239Ala;Thr278Ala mutant VASP. Mean lengths (+/- SEM) of 55 dorsal stress fibers measured from both samples are shown.
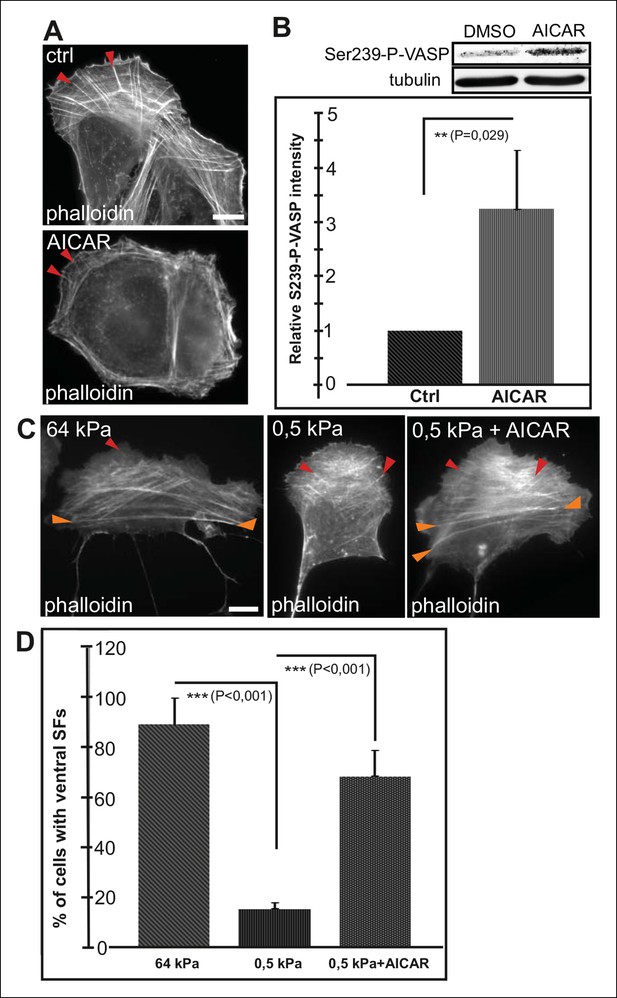
AMPK activation leads to maturation of contractile actomyosin bundles.
(A) U2OS cells grown on glass and treated with AMPK activator AICAR exhibited short dorsal stress fibers. Ventral stress fibers were typically localized close to cell edges, indicating early maturation of the contractile actomyosin bundles compared to control cells. (B) Western blot analysis of the corresponding samples showed elevation of Ser239-P-VASP in AICAR treated cells. A representative Western blot and quantification of three separate experiments (mean +/- SEM) are shown. (C) Activation of AMPK bypasses the need for stiff substrata in ventral stress fiber formation. U2OS cells grown on soft (0.5 kPa) matrix do not typically contain contractile ventral stress fibers, but their formation on soft matrix can be induced by AICAR. (D) Quantification of the amount of cells (%) containing ventral stress fibers. For each condition, 50–70 cells were analysed and the data are presented as mean +/- SEM.
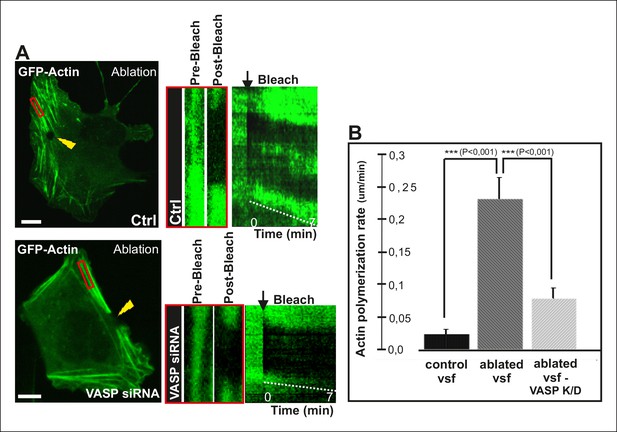
VASP-depletion leads to a decrease in tension-sensitive actin polymerization at focal adhesions.
(A) Actin polymerization at focal adhesions was monitored by fluorescence recovery after photobleaching (FRAP) in laser-ablated ventral stress fibers of control cells and VASP knockdown cells. FRAP experiments were initiated 10 seconds after ablation. Yellow arrowheads indicate the ablation sites and red boxes the regions of stress fibers that were followed for vectorial actin polymerization. Kymographs on the right were recorded along the center of the ablated ventral stress fibers. Release of tension induces vectorial actin polymerization from the adhesion located at the end of an ablated ventral stress fiber in a control cell, whereas the rate of vectorial actin polymerization was slower in an ablated ventral stress fiber in a VASP-depleted cell. Bar, 10 um. (B) Quantification of vectorial actin polymerization rates in intact ventral stress fibers, in ablated ventral stress fibers of control cells, and in ablated ventral stress fibers of VASP-depleted cells. Mean +/- SEM is shown; n (intact ventral stress fibers) = 17; n (ablated ventral stress fibers) = 12; n (ablated ventral stress fibers from VASP knockdown cells) = 17.
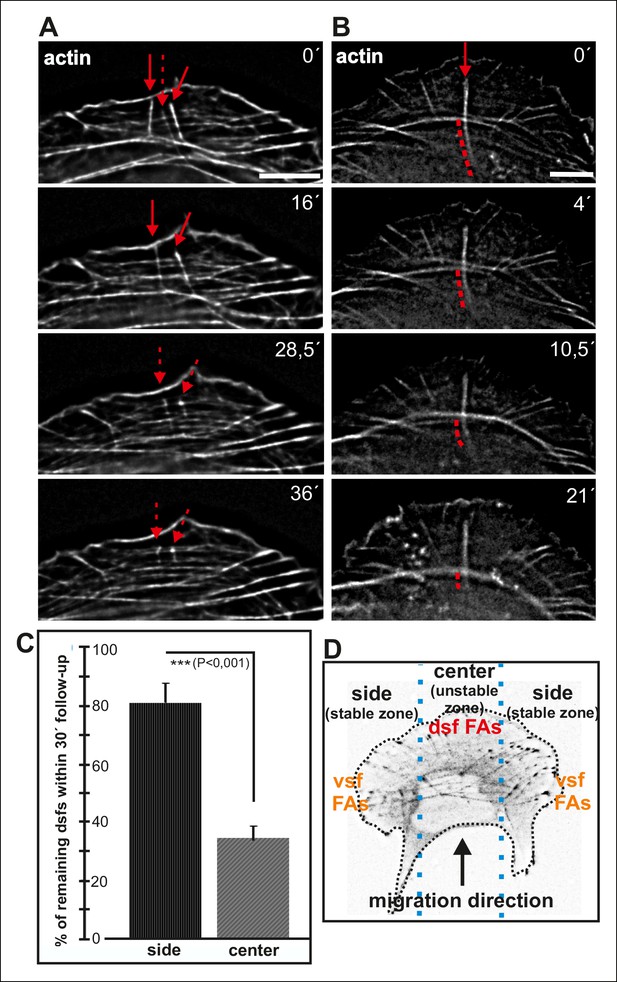
Dorsal stress fibers exhibit different lifespans depending on their interactions with the actomyosin network.
(A) Individual frames from a representative movie of GFP-actin expressing cell demonstrating the disassembly of non-contractile dorsal stress fibers located at the ‘unstable’ zone. Bar, 5 μm. (B) Frames from a movie of GFP-actin expressing cell displaying the disassembly of the non-contractile dorsal stress fiber region extending beyond the contractile transverse arc. Bar, 5 μm. (C) Quantification of the stability of dorsal stress fibers at different cell regions revealed that these actin bundles are more stable at the sides of the leading edge as compared to the central region of the leading edge. Amount of dorsal stress fibers (%), remaining after 30 min follow-up, is shown (mean +/- SD), n = 5 cells, 8–20 fibers per cell were analysed. (D) Representation of the ‘unstable’ and ‘stable’ dorsal stress fiber zones in U2OS cells.
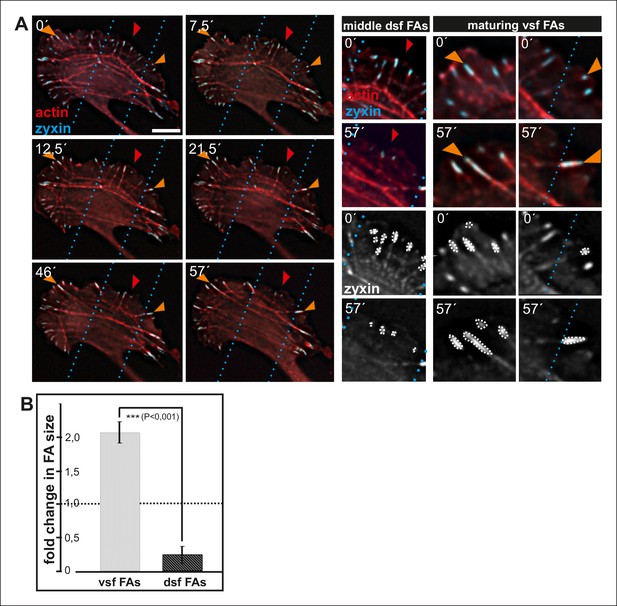
Enlargement and lifespan of focal adhesions depends on their location in migrating cells.
(A) Dorsal stress fiber-attached focal adhesions on the sides of a migrating cell (orange arrowheads) become more prominent in size and participate in formation of contractile ventral stress fibers. In contrast, dorsal stress fibers and focal adhesions in the central region of the leading edge (red arrowheads), which do not participate in the formation of ventral stress fibers, diminish in size and eventually disappear. Image frames are from movies of U2OS cells expressing GFP-actin and CFP-zyxin. Bar, 10 um. (B) Quantification of the areas of individual focal adhesions (fold change in comparison to the initial area of the adhesion) located at the tips of dorsal stress fibers and maturing ventral stress fibers. Cells were imaged for one hour and the mean values (+/- SD) of focal adhesions from 7 cells are shown.
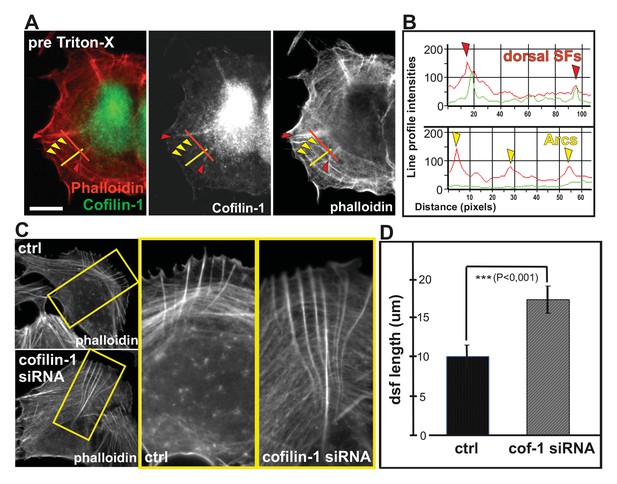
Cofilin-1 promotes disassembly of non-contractile dorsal stress fibers.
(A) Endogenous cofilin-1 localizes to dorsal stress fibers (red arrowheads) but it is absent from contractile arcs (yellow arrowheads) as shown by phalloidin and anti-cofilin-1 staining of a U2OS cell treated with Triton-X 100 prior to PFA fixation. Bar, 10 μm. (B) Line intensity profiles show incorporation of cofilin-1 into dorsal stress fibers (dsf) but not to the contractile ventral stress fibers (vsf). Cofilin (green); Actin (red). (C) Depletion of cofilin-1 leads to an appearance of abnormally long dorsal stress fibers and defects in the fusion of transverse arcs. Bar, 10 μm. (D) Quantification of the lengths of dorsal stress fibers (μm) in control and cofilin-1-depleted cells. Mean lengths (+/- SEM) of 50 dorsal stress fibers from control and cofilin-1 RNAi cells are displayed in the graph.
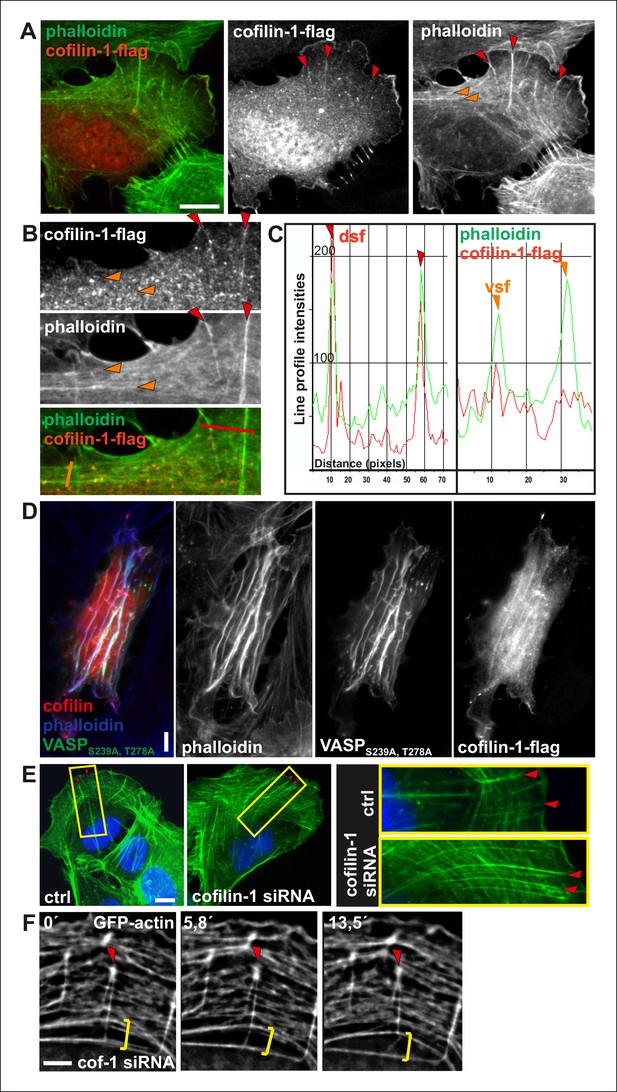
Cofilin-1 localizes to dorsal stress fibers and affects their turnover.
(A) Flag-tagged cofilin-1 localizes to non-contractile dorsal stress fibers, whereas it is absent from contractile ventral stress fibers and transverse arcs. Cofilin-1 (red) was visualized by anti-flag antibody and F-actin (green) by phalloidin staining. Bar, 10 μm. (B) Magnifications of a flag-tagged cofilin-1 expressing cell. The regions selected for the line intensity profiles from dorsal and ventral stress fibers are indicated by red and orange lines, respectively. (C) Line intensity profiles show incorporation of cofilin-1 into dorsal stress fibers (dsf) but not to the contractile ventral stress fibers (vsf). (D) Expression of constitutively active Ser239Ala;Thr278Ala mutant VASP leads to formation of curly actomyosin structures. Cofilin-1 localizes to these structures. (E) Cofilin-1-depleted cells show abnormally long and thick dorsal stress fibers as well as impaired fusion of transverse arcs into thick contractile ventral stress fibers. Stress fibers were visualized with phalloidin staining. Bar, 10 μm. (F) Time lapse frames from a cofilin-1-depleted U2OS cell, transfected with GFP-actin. Depletion of cofilin-1 leads to formation of abnormally stable dorsal stress fibers, which appear to prevent arc fusion, but continue to grow between adjacent arcs. Red arrowhead indicates a dorsal stress fiber and yellow brackets indicate two arcs that are not able to fuse. Bar, 5 μm.
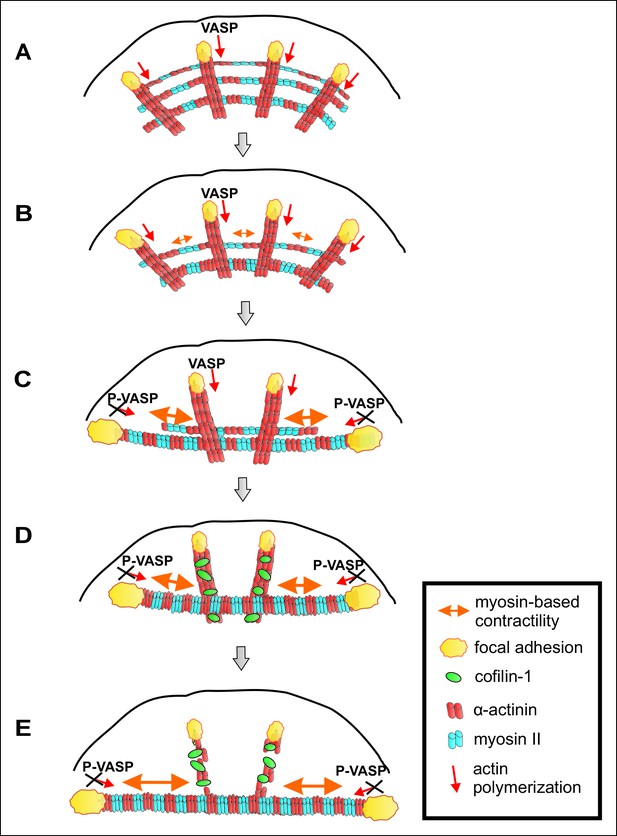
A working model for mechanosensitive generation of ventral stress fibers in U2OS cells.
(A) Dorsal stress fibers elongate through vectorial actin polymerization from focal adhesions located at the leading edge of the cell, and form a spider net -like structure with multiple transverse arcs. At least Dia1 formin and VASP are involved in vectorial actin polymerization and consequent dorsal stress fiber elongation from focal adhesions. (B) Arcs flow along the elongating dorsal stress fibers towards the cell center, and fuse with each other to form thicker and more contractile actomyosin bundles. (C) Tension provided by the contraction of arcs is mediated through dorsal stress fibers to those focal adhesions that are linked to the end of the arc. This leads to enlargement of ‘terminal’ adhesions and their alignment along the direction of the contractile arc bundle. Tension provided by myosin II –driven contractility of the arc inhibits vectorial actin polymerization in ‘terminal’ focal adhesions, and this is at least partially mediated by VASP phosphorylation. Consequently, elongation of the actomyosin bundle ceases, thus allowing its efficient contractility. Please note that focal adhesions are likely to be composed of many actin filament populations, and for simplicity only the one undergoing vectorial actin polymerization and thus promoting stress fiber elongation is shown in the model. (D) Cofilin-1 specifically binds to and promotes the disassembly of non-contractile dorsal stress fibers, which are connected to the central regions of the arc and thus do not participate in the formation of the ventral stress fiber. (E) Whereas non-contractile stress fibers are disassembled by cofilin-1, contractile stress fibers are protected from tension-sensitive cofilin-1–induced severing. Eventually, this leads to the formation of a contractile ventral stress fiber, which is connected to one large focal adhesion at its each end and aligned perpendicularly to the direction of lamellipodium extension.
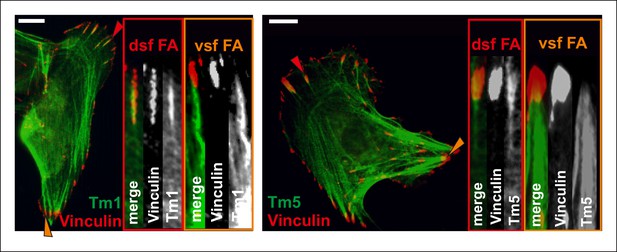
Differential localizations of Tm1 and Tm5NM1 in focal adhesions at the tips of dorsal and ventral stress fibers.
Tm1 and Tm5 localize to focal adhesion at the tips of dorsal stress fiber sites (dsf FAs), while they are not enriched in focal adhesions located at the tips of contractile ventral stress fiber adhesions (vsf FAs). Cells expressing GFP-tagged tropomyosins were stained with a focal adhesion marker vinculin. Red and orange arrowheads indicate focal adhesions at the tips of dorsal and ventral stress fibers, respectively. Bars 10 um.
Videos
Fluoresence recovery after photobleaching (FRAP) of GFP-actin at dorsal stress fiber-associated focal adhesions (FAs).
GFP-actin at FAs of dorsal stress fibers were bleached with 100% laser power of 488 laser line for 1 ms. Signal of GFP-actin displayed relatively uniform recovery at the site of focal adhesion. Duration of the movie is 8,5 min and the display rate is 10 frames/second.
Fluoresence recovery after photobleaching (FRAP) of GFP-actin at ventral stress fiber-associated focal adhesions (FAs).
GFP-actin at FAs of dorsal stress fibers were bleached with 100% laser power of 488 laser line for 1 ms. Signal of GFP-actin displayed relatively uniform recovery at the site of focal adhesion. Duration of the movie is 8,5 min and the display rate is 10 frames/second.
Control movie on stress fiber dynamics in GFP-actin expressing U2OS cell.
U2OS cells were transfected with GFP-actin 24 hr before imaging. Images were acquired every 15 s. Display rate is 15 frames/second and total duration is 73,3 min.
Expression of dominant-inactive Rif causes uncontrolled dorsal stress fiber elongation.
U2OS cells were transfected with Rif-TN and GFP-actin 24 hr prior to imaging. Loss of proper contractile structures due to Rif-TN expression causes abnormal elongation of the dorsal stress fibers as visualized by GFP-actin. Images were acquired every 15 s. Display rate is 15 frames/s, total video duration 53,5 min.
Effects of cofilin-1 depletion on stress fiber dynamics.
Cofilin-1 depleted U2OS cells were transfected with GFP-actin 24 h prior to imaging. Lack of cofilin-1 leads to elongated dorsal stress fibers and impairs transverse arc fusion. Images were acquired every 15 s. Display rate is 15 frames/s, total video duration 62,5 min.
VASP-depleted cells lack dorsal stress fibers and have poorly aligned contractile actomyosin bundles.
VASP-depleted U2OS cells were transfected with GFP-actin one day prior to imaging. Images were acquired every 15 s. Display rate is 15 frames/s, total video duration 21,3 min.
Ablation of contractile ventral stress fibers does not affect the position of focal adhesions.
An example of a laser-ablated ventral stress fiber from a U2OS cell co-expressing mCherry-Actin and GFP-Zyxin. Five captures, every 1 s, were taken before the ablation, and retraction of the ventral stress fiber was followed for 10 x 1 s before bleaching the region below the focal adhesion. Recovery of mCherry-Actin signal was detected at the bleached area of the relaxed fiber. Display rate is 10 frames/s, and the total duration of the video is 9 min. Please note, that position of the focal adhesion (indicated by GFP-Zyxin) is not significantly affected by ablation of the associated stress fiber.





