Ventromedial hypothalamic neurons control a defensive emotion state
Figures
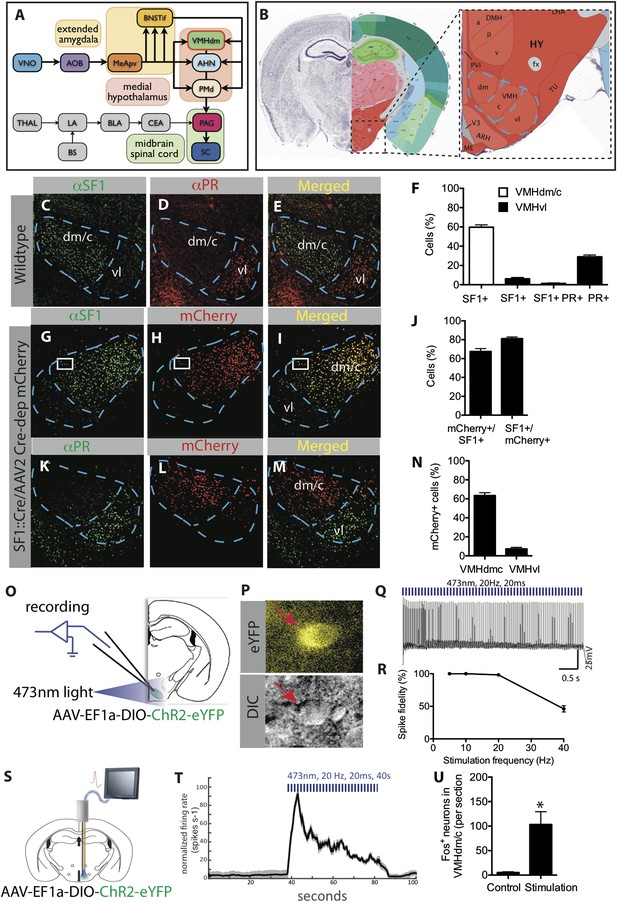
Characterization of SF1+ neurons and their optogenetic activation.
(A) Schematic illustrating brain circuits involved in defensive behaviors. (B) Coronal section of the mouse brain showing the location of VMHdm (Allen Brain Atlas). VMH is indicated by the blue outline. (C–E) Representative images of the VMH in a wild type mouse showing double-label immunostaining for SF1 (green) and progesterone receptor (PR), a marker of VMHvl neurons involved in social behaviors (red). (F) Percentage of cells in VMHdm/c (white bars) and VMHvl (black bars) that are SF1+ or PR+. n = 3 animals for each condition. (G–I) Representative images of VMH from an SF1-Cre transgenic mouse injected in VMH with a Cre-dependent AAV encoding mCherry (red) and immunolabeled with anti-SF1 antibody (green). (J) Percentage of overlap between VMHdm/c SF1+ cells and mCherry. n = 3 animals for each condition. (K–M) Representative images of VMH as in (G–I), double labeled for mCherry+ (red) and PR+ cells (green). (N) Percentage of total neurons that are mCherry+ in VMHdm/c and VMHvl (defined by domain of PR expression). n = 3 animals for each condition. (O) Schematic illustrating preparation for whole-cell patch clamp recordings of SF1+ neurons. (P) Representative photomicrograph of ChR2-eYFP-expressing (SF1+) cells (red arrow) patched for recording; DIC, differential interference contrast. (Q) Photostimulation-evoked spiking in neurons recorded as in (P). (R) Percent spike fidelity in (Q). n = 7 cells. (S) Schematic for in vivo electrophysiological response recordings from VMHdm/c in mice expressing ChR2 in SF1+ neurons. (T) Time-course of mean firing rate change in vivo in response to photostimulation. n = 6 units. (U) Average number of Fos+ neurons per section of VMHdm/c from photostimulated mice expressing ChR2 in SF1+ neurons. Control non-stimulated contralateral side within each animal. n = 4 animals for each condition. Values are represented as mean ± SEM. *p < 0.05.
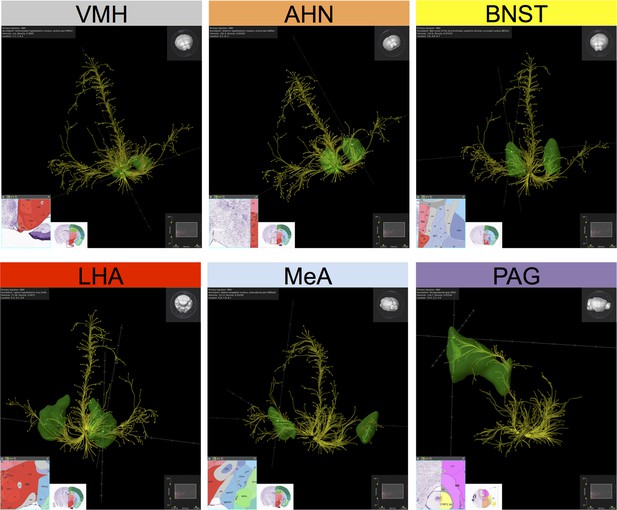
Projection profile of SF1+ (Nr5a1+) VMHdm/c neurons.
Yellow fibers are a computational representation of the projections of SF1+ neurons from VMHdm, revealed by the expression of a Cre-dependent GFP AAV reporter virus (Oh et al., 2014) sterotaxically injected into the VMHdm of NR5a1-Cre transgenic mice. Data were collected using serial two-photon tomography (Ragan et al., 2012) and deposited in the Allen Mouse Brain Connectivity Atlas (http://connectivity.brain-map.org). Source experiment: Nr5a1-Cre:VMH-LHA, Experiment 114290225 (http://connectivity.brain-map.org/?searchMode=source&sourceDomain=693&primaryStructureOnly=true&transgenicLines=177839331&initImage=TWO_PHOTON&experimentCoordinates=7100,6800,6200&experiment=114290225). Data were imported into Brain Explorer 2 (http://mouse.brain-map.org/static/brainexplorer), for computational reconstruction in 3D. Data were filtered to show the highest-density/highest intensity projections. Projection targets (green) and associated coronal sections from the Allen Brain Atlas (http://connectivity.brain-map.org/?searchMode=source&sourceDomain=693&primaryStructureOnly=true&transgenicLines=177839331&initImage=TWO_PHOTON&experimentCoordinates=7100,6800,6200&experiment=114290225) were identified by sampling terminals in high-density target sites, following which a volumetric rendering of relevant target structure was computationally superimposed on the projection map. A subset of the highest-density projection targets is illustrated. Individual panels are shown in slightly different 3D orientations to more clearly illustrate the pattern of projections to the indicated structure. The relative orientation of the entire brain in each panel can be viewed in the 3D volumetric model illustrated in the top right corner of each panel. Upper left panel depicts the VMH (injection site). Remaining panels depict the following projection sites: AHN, BNST, LHA, MeA, PAG. These illustrations are presented in accordance with AIBS policy and citation guidelines (see http://www.alleninstitute.org/terms-of-use/ and http://www.alleninstitute.org/citation-policy/). These projection profiles can be viewed in 3D here in these two supplemental Videos (Video 9, 10). Projections from Nr5a1+ neurons in VMH to the following structures are labeled: AHN (orange), CeA (blue-grey), CoA (turquoise), LHA (red), MeA (pale blue), PAG (violet), LA (green).
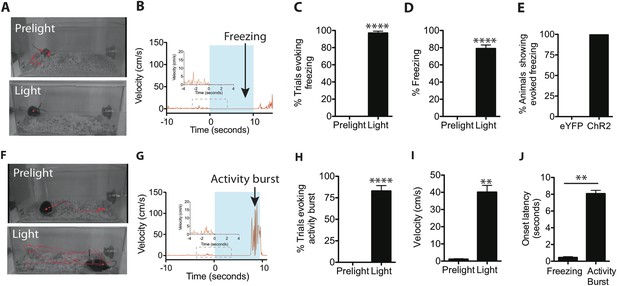
Optogenetic stimulation of SF1+ neurons induces freezing and/or activity bursts.
(A) Representative tracking traces (red) of SF1-ChR2 expressing mice before (‘Prelight’) or during (‘Light’) optogenetic stimulation. Red dot in lower image reflects immobility of animal. (B) Representative velocity trace displaying light-elicited freezing (arrow) in a ChR2 mouse. Blue shading represents period of photostimulation. Inset, expanded view of region in dashed box. (C) Percentage of photostimulation trials evoking freezing. (D) Percentage of time spent freezing during photostimulation averaged across trials. (E) Percentage of ChR2-expressing or control eYFP-expressing animals showing photostimulation-evoked freezing behavior. n = 17–18 animals for each group. (F) Representative tracking traces of a SF1-ChR2 expressing mouse (‘Prelight’) or during (‘Light’) optogenetic stimulation. Wider spacing between points in ‘Light’ indicates higher velocity. (G) Representative velocity trace displaying light-induced activity burst behavior in an SF1-ChR2 mouse. Note period of freezing prior to activity burst. Inset, expanded view of region in dashed box. (H) Percentage of stimulation trials evoking activity bursts following freezing. (I) Average velocity during activity burst period. (J) Average onset latency for freezing vs activity burst. n = 9 animals for each condition. Values are displayed as mean ± SEM. ****p < 0.0001; ***p < 0.001; **p < 0.01; *p < 0.05.
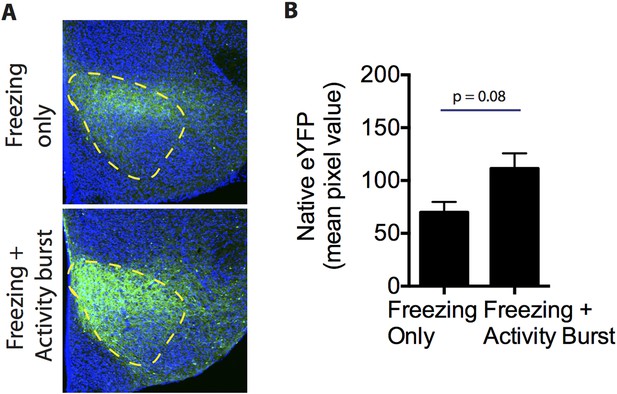
ChR2-eYFP quantification in the VMHdm/c of freezing only and freezing + activity burst groups.
(A) Representative images of ChR2-eYFP expression in the VMHdm/c for mice that showed freezing only (top) or freezing followed by an activity burst (bottom). (B) Average pixel value of native ChR2-eYFP expression in the VMHdm/c in the freezing only group and freezing + activity burst group. n = 5–6 animals for each condition. Values are displayed as mean ± SEM.
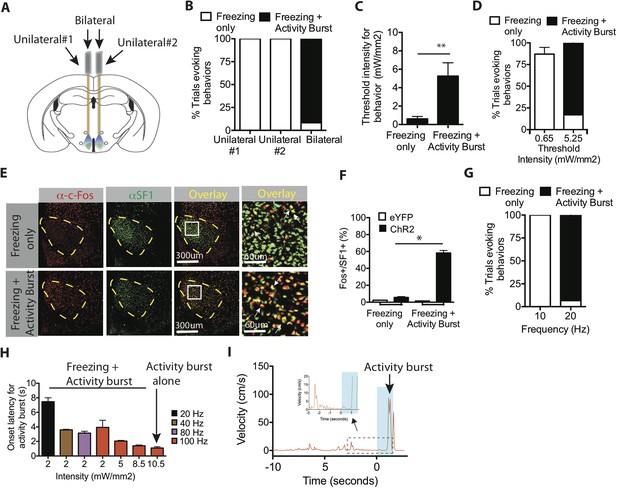
Optogenetic stimulation of SF1+ neurons induces freezing and/or activity bursts depending on strength and duration of photostimulation.
(A) Schema illustrating unilateral vs bilateral optogenetic stimulation. Each mouse was implanted with bilateral optic fibers, and stimulation was delivered either to one side (‘unilateral #1’), the contralateral side (‘unilateral #2’) or to both sides (‘bilateral’). (B) Percent trials evoking freezing only (white) or freezing followed by an activity burst (black) in unilateral vs bilaterally stimulated ChR2 mice. **p < 0.01; Two-Way ANOVA, Bonferroni correction. n = 2 animals for each condition, each animal was stimulated either through one or the other of the two optic fibers (‘Unilateral #1, Unilateral #2’), or through both (‘Bilateral’). (C) Threshold stimulation intensity required to generate freezing alone or freezing followed by an activity burst, during the photostimulation period. n = 9 per condition. (D) Percentage of trials evoking freezing alone, or freezing followed by an activity burst, at respective stimulation intensities. **p < 0.0001; Two-Way ANOVA, Bonferroni correction. (E) Representative images of Fos+ (red) and SF1+ (green) neurons from animals exhibiting optogenetically induced freezing alone (upper), or freezing followed by an activity burst. Last column is higher magnification view of boxed area in adjacent ‘Overlay’ column. Arrow indicates cells double labeled for Fos+ (red) and SF1+ (green) (F) Percentage of SF1+ neurons that are Fos+ in eYFP (white) or ChR2 (black) mice following photostimulation trials eliciting freezing alone (‘Freezing’) or freezing followed by an activity burst (‘Activity Burst’). n = 3–5 mice per condition. (G) Percentage of trials evoking freezing alone, or freezing followed by an activity burst, in response to different photostimulation frequencies. n = 5 animals per condition. (H) Onset latency for activity burst as a function of increasing light intensity (x-axis) and frequency (colored bars). Arrow indicates condition that elicited activity burst without prior freezing. (I) Representative velocity trace displaying light-induced activity burst without preceding freezing. Arrow indicates expanded trace from boxed region. n = 2 animals for each condition. Values are displayed as mean ± SEM. ****p < 0.0001; ***p < 0.001; **p < 0.01; *p < 0.05.
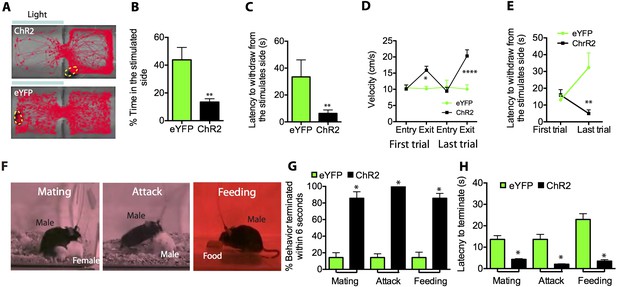
Optogenetic stimulation of SF1+ neurons induces aversion and interrupts ongoing consummatory behaviors.
(A) Representative tracking traces of ChR2 mouse (top) and eYFP control mouse (bottom) in a real-time place avoidance assay (RTPA). Photostimulation (blue bar) was delivered in a manual closed-loop manner depending on the animal's behavior (see text). (B) Percentage of total time (20 min) spent in stimulated side during 20-min trial. (C) Average latency to withdraw from the stimulated side. (D) Average velocity to enter or exit the stimulated side for the first (left) vs last trial (right). (E) Latency to withdraw from the stimulated side for the first vs last trials. n = 6–7 animals for each condition in D and E. (F) Sample video still frames taken from consummatory behavioral assays. (G) Percentage of indicated behavior episodes terminated by light stimulation during the behavior within 6 s of photostimulation onset. (H) Latency to terminate respective consummatory behavior during photostimulation. n = 4–6 mice per condition. Values are displayed as mean ± SEM. ****p < 0.001; ***p < 0.001; **p < 0.01; *p < 0.05.
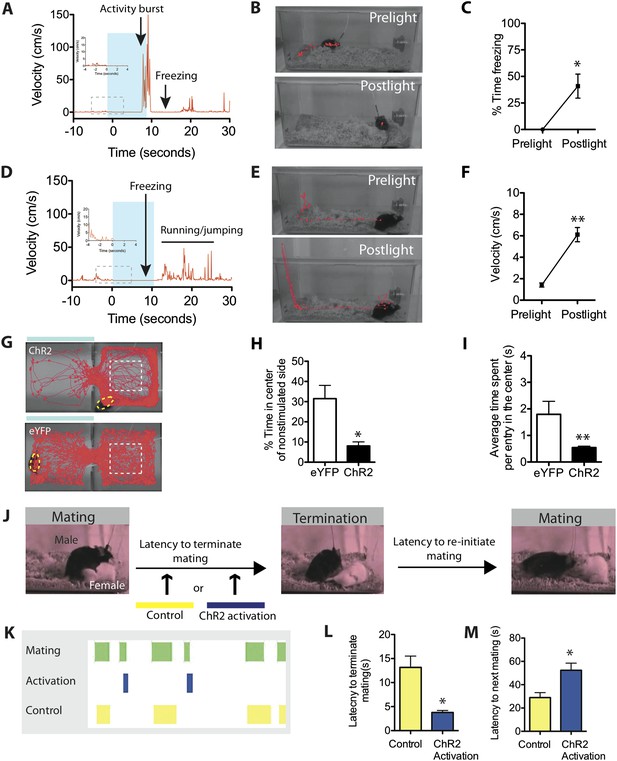
Stimulation of SF1+ neurons produces persistent defensive responses in multiple behavioral assays.
(A) Representative velocity trace for ChR2 mouse displaying light-induced activity burst and post-light freezing behavior (arrow). Note that freezing was observed during the photostimulation period prior to the activity burst (inset, boxed region). See also Figure 2G. (B) Representative tracking traces (red) for pre-light vs post-light behavior in ChR2 mice. (C) Average percentage of time spent freezing during the 10-s pre-light and post-light bins averaged across trials and all mice. (D) Representative velocity trace for ChR2 mouse displaying freezing during photostimulation and elevated locomotion/jumping during the post-stimulation period. (E) Representative tracking traces for pre-light and post-light behavior in a ChR2 mouse. Vertical trace on left side of cage indicates jump (‘Postlight’). (F) Average velocity during pre-light and post-light bins (10 s each) averaged across trials and mice. n = 9 animals for each condition. (G) Representative tracking traces of ChR2 mouse (top) and eYFP control mouse (bottom) in the RTPA assay. The white dashed box marks the center area of the non-stimulated side used to index thigmotaxic behavior. Modified from image in Figure 4A, to illustrate thigmotaxic behavior. (H) Percentage of total assay period (20 min) spent in the center of the non-stimulated side. (I) Average time spent in the center of the non-stimulated side, per individual entry. n = 6–7 animals for each condition. (J) Protocol to measure the resumption of mating behavior immediately following photostimulation of SF1+ neurons. (K) Representative raster plot illustrating mating episodes with ChR2 activation (blue) and control light (yellow) activation. The yellow wavelength does not activate ChR2 and is used as an internal control. (L) Latency to terminate mating following photostimulation with blue vs yellow light. (M) Latency to re-initiate mating after mating termination following blue vs yellow light. n = 7 animals for each condition. Values are displayed as mean ± SEM. **p < 0.01; *p < 0.05.
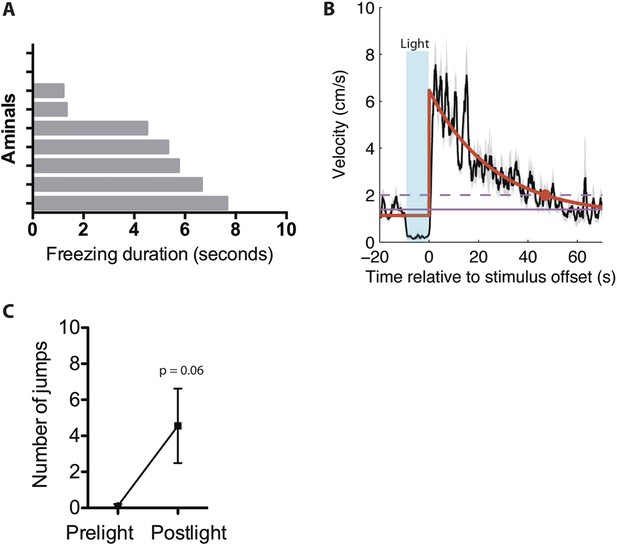
Persistent responses following light induced activity burst and freezing.
(A) Average duration of freezing exhibited by individual ChR2 animals during post-stimulation period following stimulation-induced activity burst. n = 9 animals. (B) Trial-averaged velocity plot for ChR2 mice displaying pre-light, light-induced freezing, and post-light elevated locomotion behavior. In black, the trial-averaged velocity following stimulus offset. The solid pink line indicates the time-averaged velocity computed over the 10-s window prior to stimulus onset; the dashed pink line is + one standard deviation of the trial-averaged velocity in the same window. In red, we fit an exponential of the form a + b*exp(−t/tau) to the trial-averaged velocity starting at t = 0 (time of stimulus offset). The fit exponential had a time constant of 25.8 s; the trial-averaged velocity took 47.03 s to decay to within one standard deviation of the baseline velocity prior to stimulation (marked by red dot.) (n = 28 trials). Values are displayed as mean ± SEM. (C) Average number of jumps exhibited in the 20-s time bins pre-light and post-light for ChR2 mice. n = 9 animals for each condition. Values are displayed as mean ± SEM.
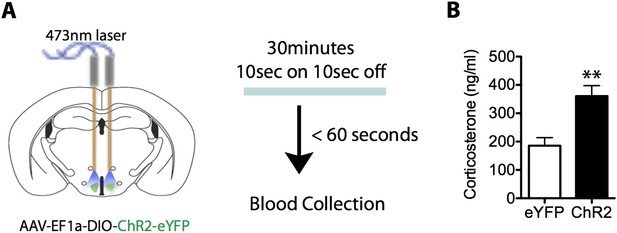
Activation of SF1+ neurons produces an increase in neuroendocrine responding.
(A) Illustration of testing for stimulation induced changes in corticosterone. (B) Optogenetic stimulation of SF1+ neurons increases serum corticosterone levels in ChR2 mice compared to eYFP controls. n = 11–12 animals for each condition.
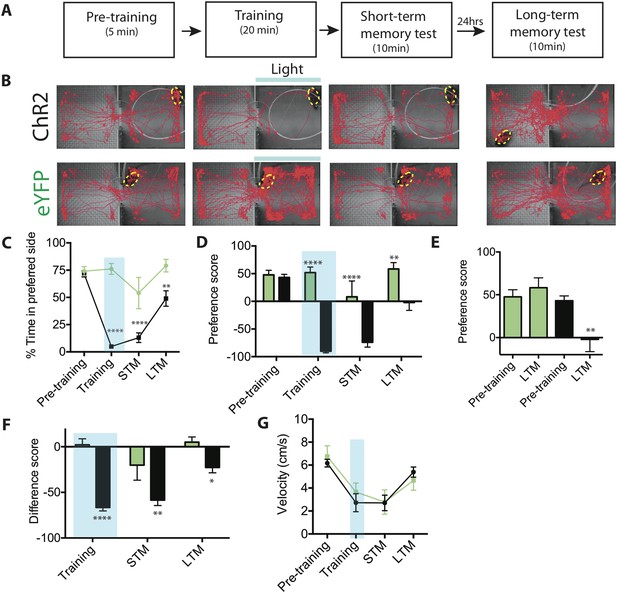
Activation of SF1+ neurons produces conditioned place avoidance learning and memory.
(A) Protocol for conditioned place avoidance assay. Pre-training phase used to determine each animal's initially preferred side. (B) Representative tracking traces during pre-training, training, short-term memory (STM) and long-term memory (LTM) test phases as indicated in overlying schematic. Blue bar represents light delivery on the initially preferred side exhibited by each individual animal. (C) Percentage of total time for each phase (see panel A) spent in initially preferred side during pre-training, training (blue shading), STM test, and LTM test. Animals spontaneously spent ∼75% of their time in one of the two chambers (defined as the initially preferred side), during pre-training period. (D) Preference score (percent total time spent in the initially preferred minus the initially non-preferred side), for each experimental phase. Negative value indicates that the animal spends more time in the initially non-preferred side, than in the initially preferred side. Preference score measures distribution of animals between the two chambers during a given testing phase. (E) Preference scores from (D) replotted for comparison of pre-training vs LTM scores for ChR2-expressing (black bars) vs eYFP control mice (green bars). (F) Difference scores (percent time spent in the initially preferred side during each respective phase minus the percent time spent in the same side during the pre-training phase) for training, STM and LTM tests. Difference score measures change in time spent in initially preferred side during pre-training phase vs a given testing phase. Difference score during LTM test is ∼38% of that measured during STM test, indicating some retention of avoidance conditioning. (G) Mean velocity during each experimental phase revealed no differences. n = 7–10 animals for each condition. All values are displayed as mean ± SEM. ****p < 0.001; ***p < 0.001; **p < 0.01; *p < 0.05.
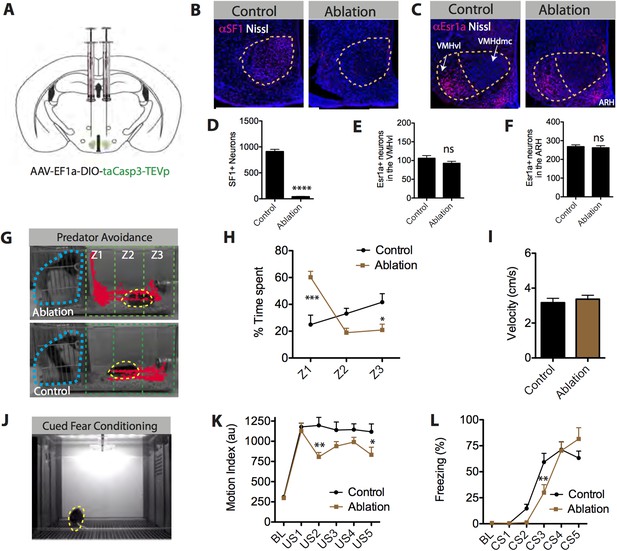
SF1+ neurons are necessary for predator aversion and conditional fear.
(A) Schematic for bilateral injection of Cre-dependent apoptotic effector virus into the VMHdm/c of SF1-Cre mice. (B) Representative images of SF1+ neurons (red) in control and SF1+ ablated mice. (C) Representative images of Esr1a+ (VMHvl) neurons (red) in control and SF1+ ablated mice. (D) Number of total SF1+ neurons in VMHdm/c of control vs SF1+ ablated mice. (E) Number of total Esr1a+ neurons in VMHvl of control vs SF1+ ablated mice. (F) Number of total of Esr1a+ neurons in the arcuate nucleus (ARH) of control and SF1+ ablated mice. n = 4–6 animals, 3–4 sections per injection site. (G) Representative tracking traces of an SF1+ ablated mouse (top) and a control mouse (bottom) in a predator avoidance task. The rat predator (constrained within a mesh cage) is outlined in blue and the mouse in yellow. (H) Percentage of total test time (3 min) spent by mice in each zone, with Z1 representing the closest zone and Z3 the furthest. (I) Average velocity across entire predator avoidance test. n = 5–7 animals for each condition. (J) Still video frame from the cued fear conditioning assay. Mice were given 5 tone-footshock pairings. (K) Average motion index (au) units during the two-minute, pre-conditioning baseline (‘BL’) period and during the activity burst elicited by each footshock-US. (L) Average percent time spent freezing during baseline (BL period) and during each 30 s tone-CS presentation preceding delivery of footshock. n = 9–10 animals for each condition. Values are displayed as mean ± SEM. ***p < 0.001; **p < 0.01; *p < 0.05.
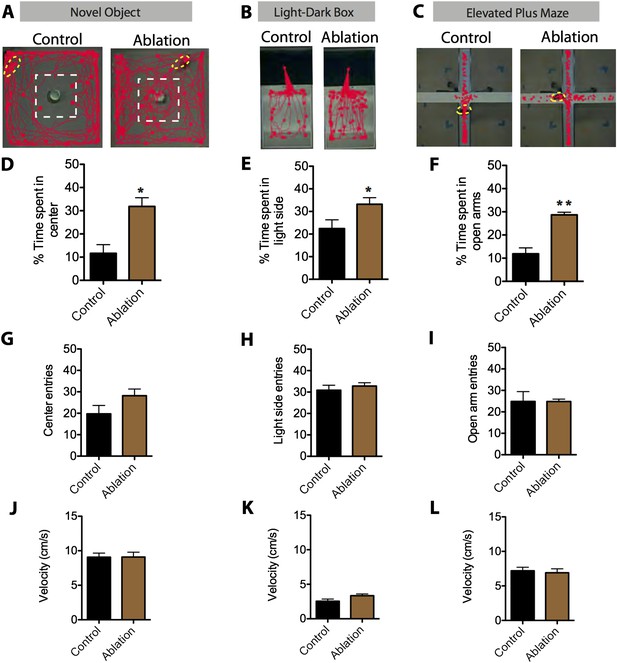
SF1+ neurons are necessary for anxiety.
(A) Representative tracking traces in the novel object test for control (left) and ablated (right) mice. Mice are outlined in yellow. The dashed white box marks the center of the chamber. (B) Representative tracking traces in the light–dark box test. Note higher density of traces in the light side for the Ablation condition, in comparison to the control. (C) Representative tracking traces in the elevated plus maze. (D) Percentage of time spent in the center of the novel object test. (E) Percentage of time spent in the light side of the light–dark box. (F) Percentage of time spent in the open arms of elevated plus maze. n = 5–7 animals for each condition. (G) Center entries in the novel object test for control (black bars) and SF1-ablated (brown bars) mice. (H) Stimulated-side entries in the light–dark box assay. (I) Total open arm entries in the elevated plus maze. (J) Average velocity in the novel object test. (K) Average velocity in the light–dark box. (L) Average velocity in the elevated plus maze. n = 5–7 animals for each condition. Values are displayed as mean ± SEM. ***p < 0.001; **p < 0.01; *p < 0.05.
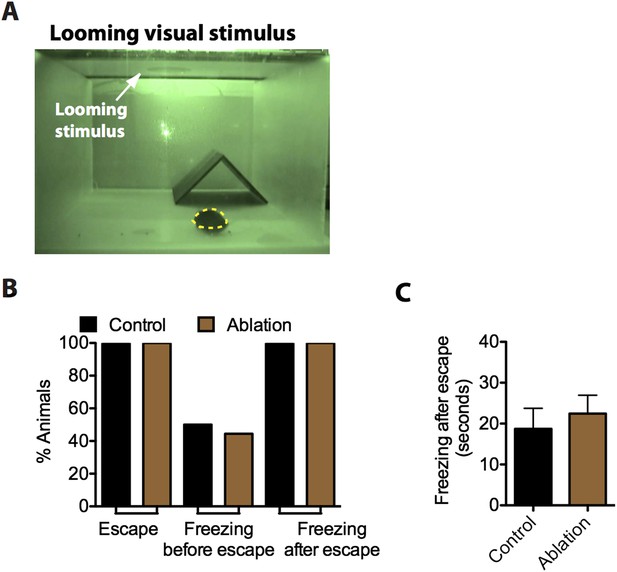
SF1+ neurons are not necessary for defensive responses elicited by looming visual stimuli.
(A) Still video frame image of looming assay box. The experimental mouse is labeled in yellow, a nest occupies the corner and a computer monitor for displaying the looming visual stimulus (i.e., a disk) is mounted directly above the apparatus. Mice are placed in the looming assay apparatus and given a 10-min acclimation period followed by a series of 10 looming stimulus presentations in 10 s. (B) Percentage of animals showing escape and freezing behaviors in response to the looming stimulus. (C) Duration of freezing following escape into the hide. n = 9–10 animals for each condition.





