An opioid-like system regulating feeding behavior in C. elegans
Figures
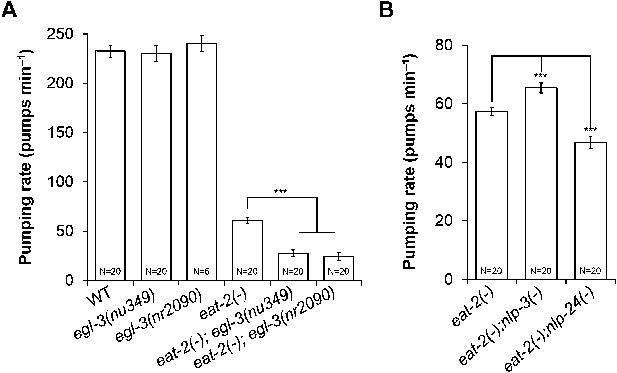
MC neurons and neuropeptides redundantly control pumping rate.
(A) Pumping rate in eat-2, egl-3 and eat-2; egl-3 mutants. egl-3 reduced pumping in eat-2 mutants. ***p < 0.001. (ANOVA + Tukey tests). (B) nlp-3 suppressed pumping and nlp-24 stimulated pumping in eat-2 mutants. ***Different from eat-2, p < 0.001 (ANOVA + Dunnett tests). In this and all figures, errors bars represent standard error.
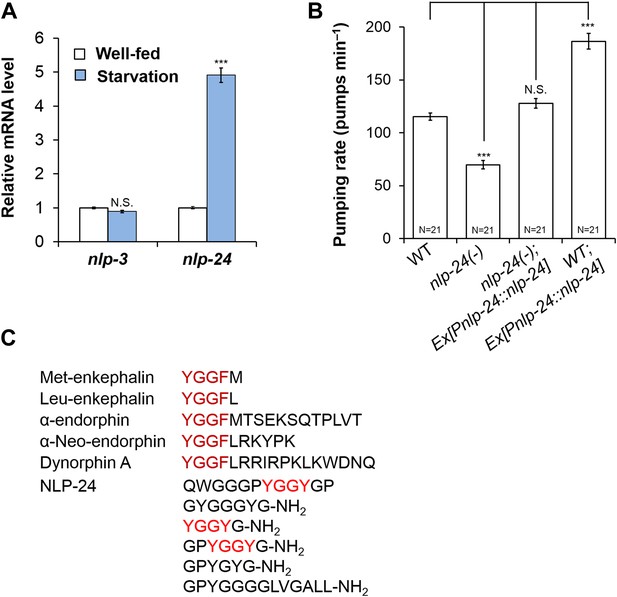
nlp-24 stimulates pumping in starved worms.
(A) nlp-3 and nlp-24 mRNA levels in well-fed and starved worms. qRT-PCR was performed to assess mRNA expression. Normalized with ama-1. ***Different from well-fed, p < 0.001 (two-way ANOVA on Ct with Bonferroni correction). (B) Pumping rate after 1 hr starvation. nlp-24 stimulated pumping during starvation. ***p < 0.001, N.S. not significant (ANOVA + Tukey tests). (C) NLP-24 and human endogenous opioid peptides.
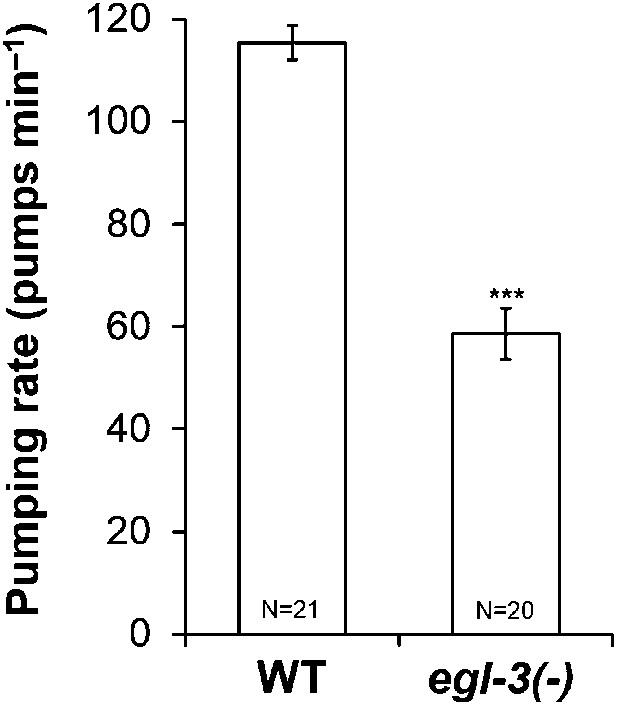
Neuropeptides stimulate pumping in starvation.
egl-3 mutants, lacking most active neuropeptides, pumped less than wild type worms during starvation. (***p < 0.001, Student's t-test.)
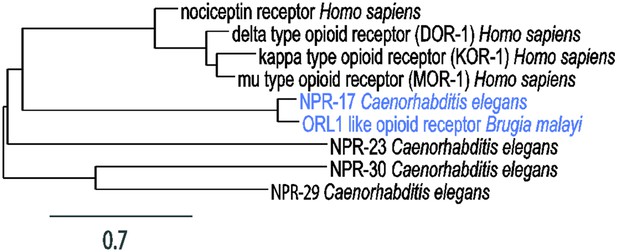
Phylogenetic analysis of opioid receptors.
Branch length is proportional to evolutionary distance. The diagram was constructed using Phylogeny.fr (phylogeny.lirmm.fr/) (Dereeper et al., 2008). Accession numbers of sequences used in the alignment: NP_001186948.1 (nociceptin receptor Homo sapiens), NP_000902.3 (delta-type opioid receptor (DOR-1) Homo sapiens), NP_000903.2 (kappa-type opioid receptor (KOR-1) Homo sapiens), NP_000905.3 (mu-type opioid receptor (MOR-1) Homo sapiens), NP_498743.2 (NPR-17 Caenorhabditis elegans), NP_490815.2 (NPR-23 Caenorhabditis elegans), NP_497125.2 (NPR-30 Caenorhabditis elegans), NP_499038.1 (NPR-29 Caenorhabditis elegans), XP_001900553.1 (ORL1-like opioid receptor Brugia malayi).
nlp-3 pumping rate after 1 hr starvation.
nlp-3 did not change pumping rate in starved worms (N.S. not significant, Student's t-test.).
Well-fed nlp-24 mutant pumping rate.
nlp-24 mutants pumped at a rate similar to wild type on food. (N.S. not significant, Student's t-test.)
Opioids control pumping through npr-17.
(A) Effect of morphine on pumping. (B) Effect of naloxone on pumping rate. Adult hermaphrodites were starved for 1 hr and tested. Morphine stimulates pumping and naloxone inhibits it in wild-type worms; neither drug affects npr-17(tm3210) mutants. ***Different from 0 mM, p < 0.001; N.S. not significantly different from 0 mM (two-way ANOVA, concentration effect). The effects of morphine and naloxone on wild-type are significantly different from their effects on npr-17, p < 0.001 (two-way ANOVA, genotype × concentration interaction).
Effect of naloxone on pumping rate in npr-17(tm3225) mutant worms.
Adult hermaphrodites were starved for 1 hr and tested. Naloxone did not affect npr-17(tm3210) mutants. ***Different from 0 mM, p < 0.001; N.S. not significantly different from 0 mM (two-way ANOVA, concentration effect). The effect of naloxone on wild-type is significantly different from its effect on npr-17(tm3225), p < 0.001 (two-way ANOVA, genotype × concentration interaction). Wild-type data are the same as in Figure 3B.
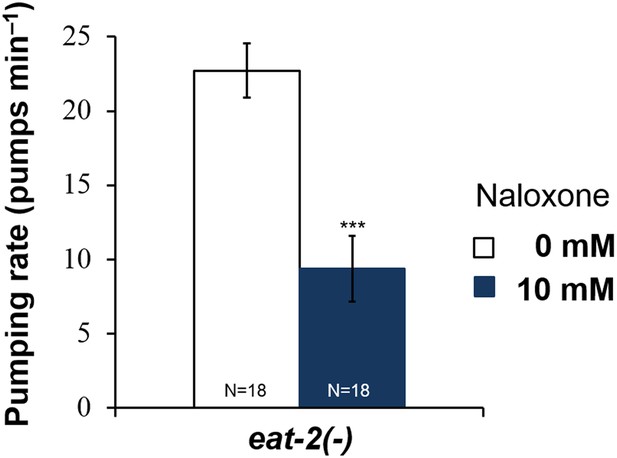
Naloxone reduces the pumping rate of eat-2 mutants.
Naloxone reduced the pumping rate in eat-2 mutants, which lack MC → muscle neurotransmission. (***p < 0.001, Student's t-test.)
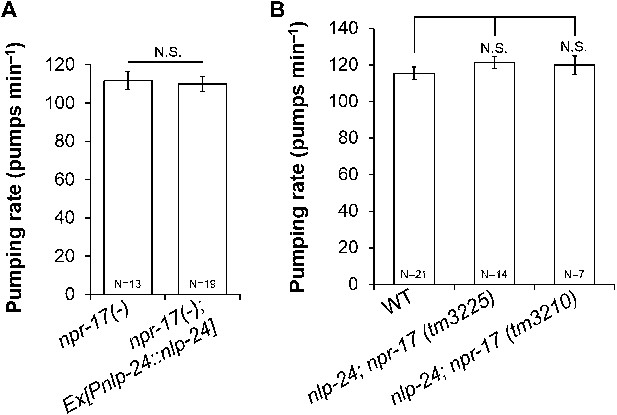
nlp-24 regulates pumping through npr-17.
(A) Overexpressed nlp-24 in npr-17 mutants did not stimulate pumping. (B) In the npr-17 background, nlp-24 mutations have no effect on pumping rate in starved worms. (N.S. not significant, Student's t-test).
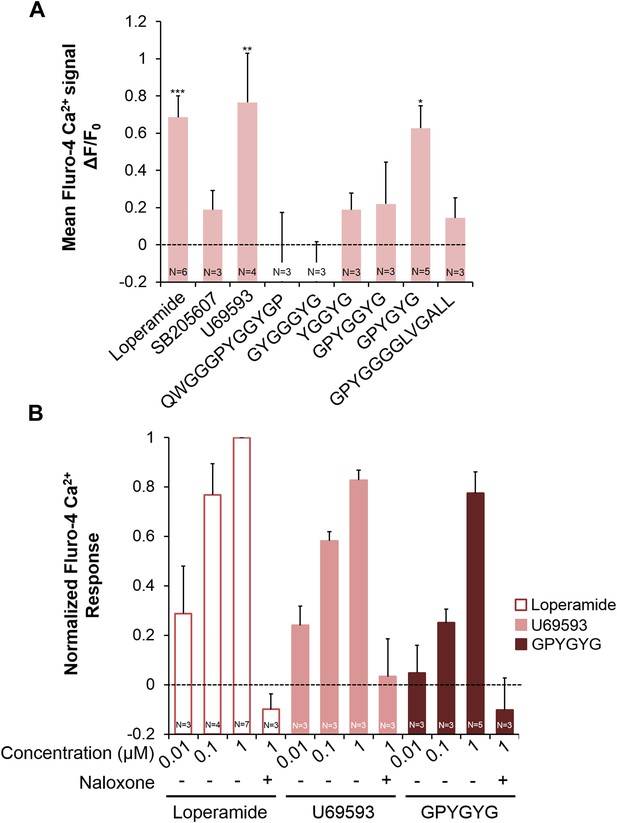
Activities of NLP-24 peptides and opioid agonists on NPR-17.
Opioid agonists and NLP-24 peptide-mediated changes in intracellular calcium were measured with the calcium detector Fluo-4. (A) Calcium responses in human embryonic kidney 293 (HEK-293) cells transfected with NPR-17 and Gα15. All compounds were tested at 1 μM. Loperamide (μ agonist), SB205607 (δ agonist), U69593 (κ agonist) and GPYGYGamide activated NPR-17. Different from 0, *p < 0.05, **p < 0.01, ***p < 0.001, two-way ANOVA + sequential Bonferroni correction. (B) Dose-response curves of HEK-293 cells transfected with NPR-17 and Gα15. Loperamide, U69593 and NLP-24 peptide GPYGYGamide induced NPR-17 activation in a dose dependent manner, and these responses were suppressed by opioid blocker naloxone (10 μM). Responses were normalized to 1 μM loperamide. The loperamide effect is significant at p < 0.001, and the effects of U69593 and GPYGYGamide at p < 0.01, two-way ANOVA + sequential Bonferroni correction. Pooling the results from A and B, the effect of GPYGYGamide is significant at p < 0.001 (Stouffer's weighted Z-score test).
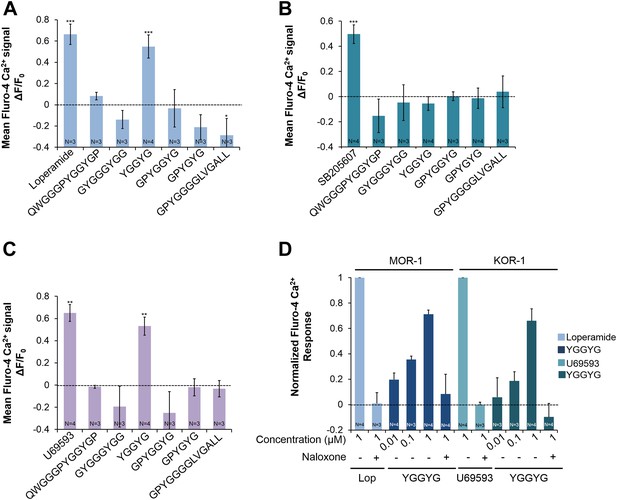
Activities of NLP-24 peptides and agonists on MOR-1, DOR-1 and KOR-1.
Opioid agonist and NLP-24 peptide-mediated changes in intracellular calcium were measured by the calcium detector Fluo-4. Agonists and peptides were tested at 1 μM. (A) Calcium responses in HEK-293 cell transfected with MOR-1 and Gα15. (B) Calcium responses in HEK-293 cell transfected with DOR-1 and Gα15. (C) Calcium responses in HEK-293 cell transfected with KOR-1 and Gα15. ***p < 0.001, **p < 0.01, *p < 0.05. (Two-way ANOVA with Bonferroni correction.) (D) Dose-response curves of HEK-293 cells transfected with MOR-1 or KOR-1 and Gα15. NLP-24 peptide YGGYGamide activated MOR-1and KOR-1, and these responses were suppressed by the opioid blocker naloxone (10 μM). MOR-1 responses were normalized to 1 μM loperamide and KOR-1 to 1 μM U69593. The effects of YGGYGamide on MOR-1 and KOR-1 are significant at p < 0.001 (two-way ANOVA).
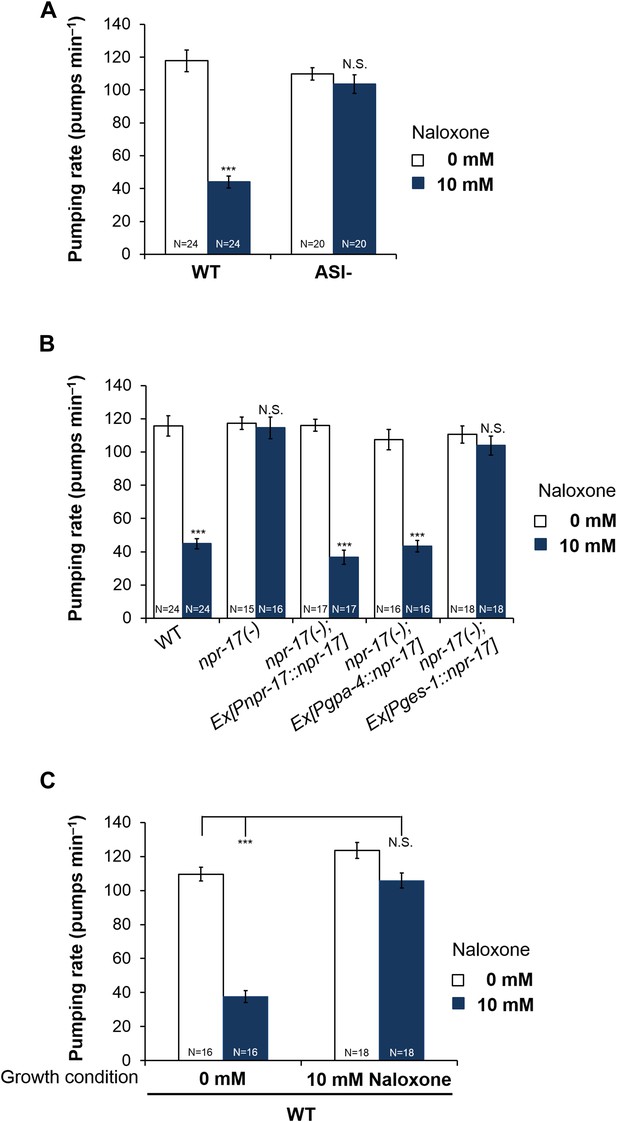
ASI neurons are required for opioid-mediated feeding control.
(A) Effect of naloxone on pumping. Worms genetically engineered to lack ASI neurons were not affected. ASI neurons were genetically ablated by the recCaspase method (Chelur and Chalfie, 2007; Beverly et al., 2011) (ASI−) ***Different from 0 mM, p < 0.001; N.S. not significantly different from 0 mM (two-way ANOVA, concentration effect). The effect of naloxone on wild-type is significantly different from its effect on npr-17, p < 0.001 (two-way ANOVA, genotype × concentration interaction). (B) npr-17 expression in ASI neurons is required for opioid-mediated feeding control. npr-17 mutants were not affected by naloxone, but this phenotype was rescued with by expression of npr-17 under control of the ASI-specific promoter gpa-4. ***Different from 0 mM, p < 0.001, N.S., not significantly different from 0 mM (two-way ANOVA, concentration effect). The interaction between genotype and concentration is significant at p < 0.001 for Pges-1::npr-17 rescued vs WT, but not Pnpr-17::npr-17 or Pgpa-4::npr-17 rescued. The interaction between genotype and concentration is significant at p < 0.001 for Pnpr-17::npr-17 and Pgpa-4::npr-17 rescued vs npr-17, but not Pges-1::npr-17 rescued. (C) Chronic effect of naloxone on pumping. Worms grown to adulthood with or without 10 mM naloxone plate were then tested with or without 10 mM naloxone. ***p < 0.001; N.S. not significantly different. The stimulatory effect of taking naloxone away from worms grown in the presence of naloxone is significant at p < 0.05. (ANOVA + Tukey tests).
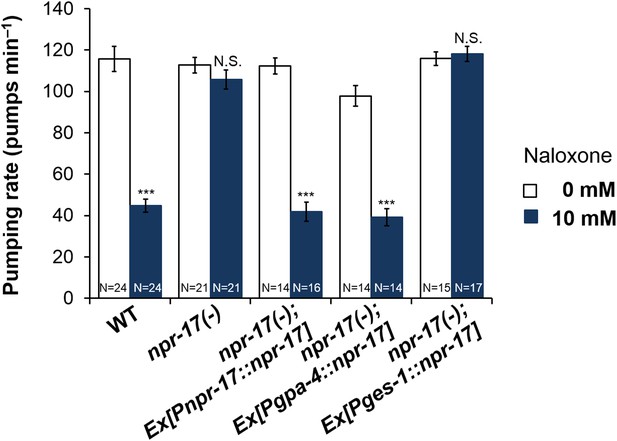
npr-17 in ASI neurons are required for opioid-mediated feeding control.
npr-17(tm3225) mutants were not affected by naloxone. This phenotype was rescued by npr-17 expression under the ASI specific promoter, gpa-4. ***Different from 0 mM, p < 0.001, N.S., not significantly different from 0 mM (two-way ANOVA, concentration effect). The interaction between genotype and concentration is significant at p < 0.001 for Pges-1::npr-17 rescued vs WT, but not Pnpr-17::npr-17 or Pgpa-4::npr-17 rescued. The interaction between genotype and concentration is significant at p < 0.001 for Pnpr-17::npr-17 and Pgpa-4::npr-17 rescued vs npr-17, but not Pges-1::npr-17 rescued. Wild-type and npr-17(tm3225) data reproduced from Figure 3B and Figure 3—figure supplement 1.
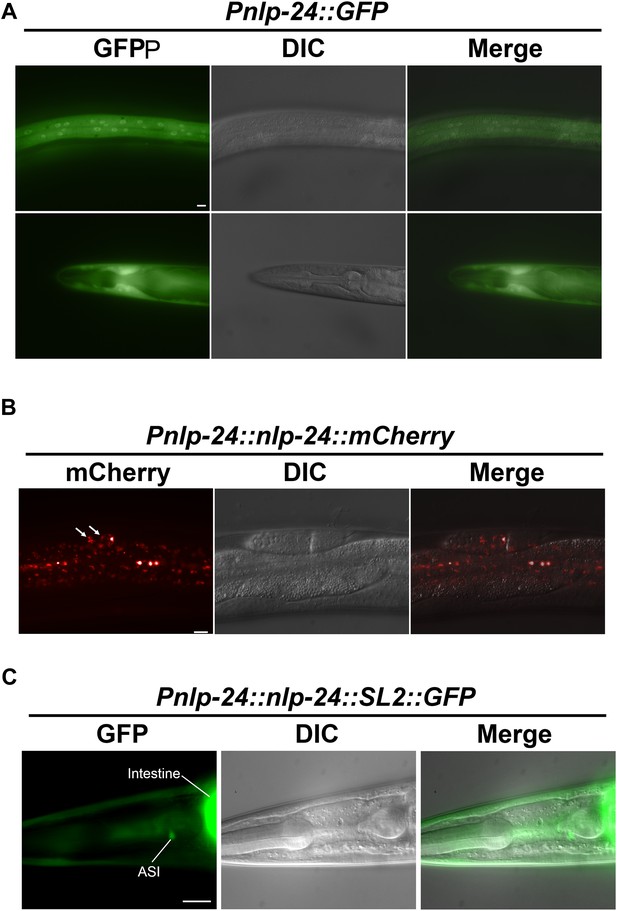
nlp-24 expression.
(A) nlp-24::GFP transcriptional reporter expression. nlp-24 was expressed in hypodermis. (B) nlp-24::mCherry translational reporter expression. NLP-24::GFP was found in coelomocytes (white arrows). (C) NLP-24::SL2::GFP translational reporter expression. NLP-24 was expressed in ASI neurons and intestine. Scale bar = 10 μm.
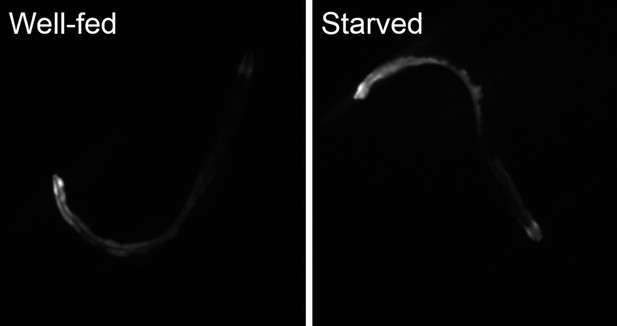
Starvation induces nlp-24 expression in intestine.
NLP-24::SL2::GFP expression is increased by starvation.
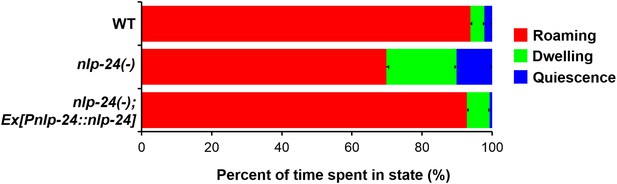
nlp-24 stimulates roaming behavior in starved worms.
Behavioral states of adult worms on plates without food. A single worm was starved for 30 min, then recorded for 1 hr. Locomotion was analyzed as described previously (Gallagher et al., 2013). Starvation increased roaming behavior in wild type. nlp-24 mutants had reduced roaming behavior compared to wild type, and an nlp-24 transgene rescued this phenotype. N = 22 worms for wild type, 27 for nlp-24 mutants, and 19 for nlp-24 rescued worms. All nlp-24 state probabilities are significantly different from the corresponding probabilities for wild type and the rescued strain, p < 0.001, with the exception of the Q state for nlp-24 vs wild type, for which p < 0.01. There are no significant differences between wild type and nlp-24 rescued. (Mann–Whitney U-test with sequential Bonferroni correction).
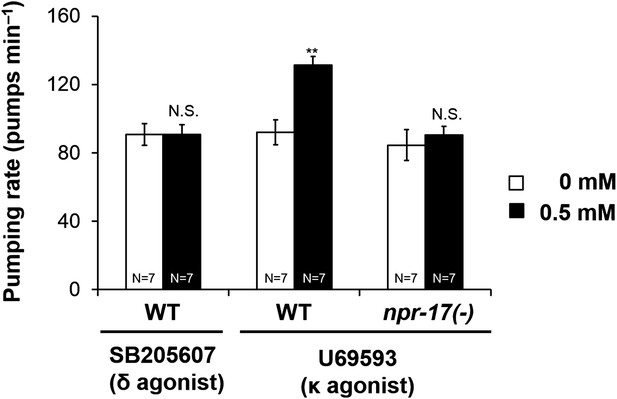
Opioid agonist effects in C. elegans.
The δ agonist SB205607 did not affect pumping. κ agonist U69594 stimulated pumping in starved wild-type worms but not npr-17 mutants. **p < 0.01. N.S. not significant. (Two-way ANOVA).
Additional files
-
Supplementary file 1
Genes that affect the growth when knocked down by RNAi.
- https://doi.org/10.7554/eLife.06683.021
-
Supplementary file 2
Primers for transgenic worms.
- https://doi.org/10.7554/eLife.06683.022
-
Supplementary file 3
Primers for RNAi.
- https://doi.org/10.7554/eLife.06683.023





