A novel GSK3-regulated APC:Axin interaction regulates Wnt signaling by driving a catalytic cycle of efficient βcatenin destruction
Figures
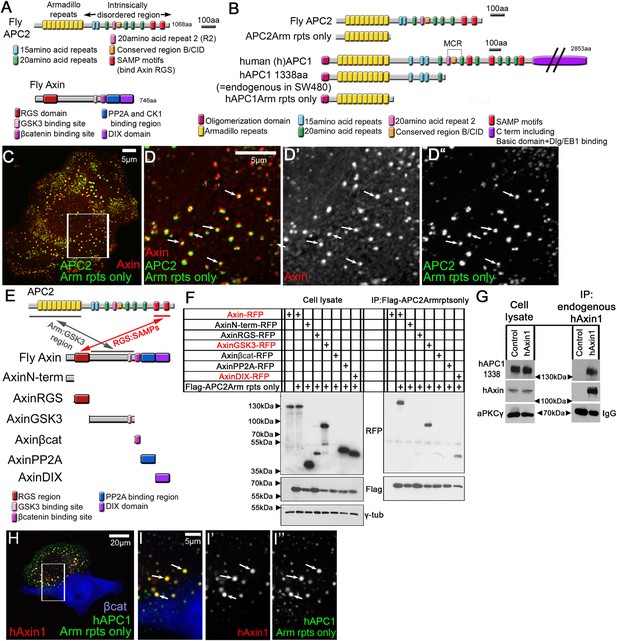
APC2's Arm rpts provide a second means of interacting with the Axin complex.
(A) Fly APC2 and Axin. (B) Constructs used. hAPC1-1338 = the endogenous truncated hAPC1 in SW480 cells. (C and D) SW480 cells coexpressing GFP-APC2Arm rpts only and Axin-RFP, which localize adjacent to one another (arrows). (D) Insets = box in (C). (E) Known and novel APC:Axin interaction sites (top) and Axin constructs (bottom). (F and G) IPs from SW480 cells. (F) APC's Arm rpts coIP with Axin's middle region that contains the GSK3 binding site. Axin's DIX domain was weakly detected. The βcat binding site fragment was not detected in either immunoblots or immunofluorescence (not shown), suggesting rapid degradation. (G) IP of endogenous hAxin1 or control Ig. Truncated endogenous hAPC1-1338 coIPs with hAxin1 at endogenous levels. (H) GFP-hAPC1Arm rpts only and hAxin1-RFP colocalize in puncta (arrows). (I) Insets = box in (H).
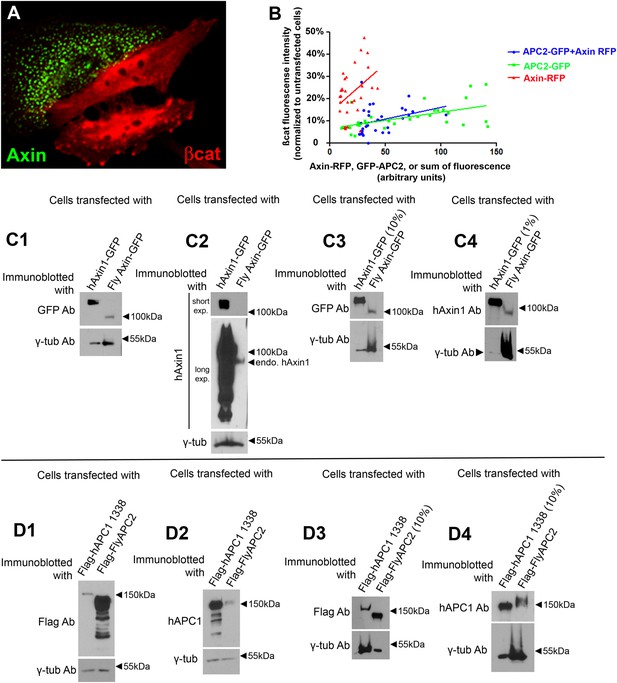
Assessing levels of over-expression of Axin and APC.
(A) When overexpressed in SW480 cells, fly Axin forms puncta. (B) Plot of immunofluorescence intensities in SW480 cells transfected with GFP-APC2, or Axin-RFP, or GFP-APC2 + Axin–RFP, and stained for βcat via antibody. βcat intensities of transfected cells were normalized to adjacent untransfected cells and plotted against the GFP, or RFP, or sum of GFP and RFP intensities. 10 cells were measured each time in 3 independent experiments. (C) Measuring the levels of Axin overexpression in SW480 cells. Immunoblot analysis of cells transfected with human Axin1-GFP (hAxin1-GFP) or fly Axin-GFP with the indicated antibodies. γ-tubulin was used as a loading control. (C1,C2) Equal volumes of cell lysate were loaded. (C3) 10% the amount of hAxin1-GFP lysate was loaded. (C4) 1% of the amount of hAxin1-GFP lysate was loaded. One representative immunoblot of 3 independent experiments. Details of calculations used are in the Results and Materials and methods—full data is in Table 1. (D) Measuring the levels of APC2 overexpression in SW480 cells. Immunoblot analysis of cells transfected with Flag-tagged truncated human APC1-1338 (see Figure 1B) or fly APC2 with the indicated antibodies. γ-Tubulin was used as a loading control. (D1,D2) Equal volumes of cell lysate were loaded. (D3) 10% the amount of Flag-flyAPC2 lysate loaded. (D4) 10% the amount of Flag hAPC1-1338 lysate was loaded. One representative immunoblot of 3 independent experiments.
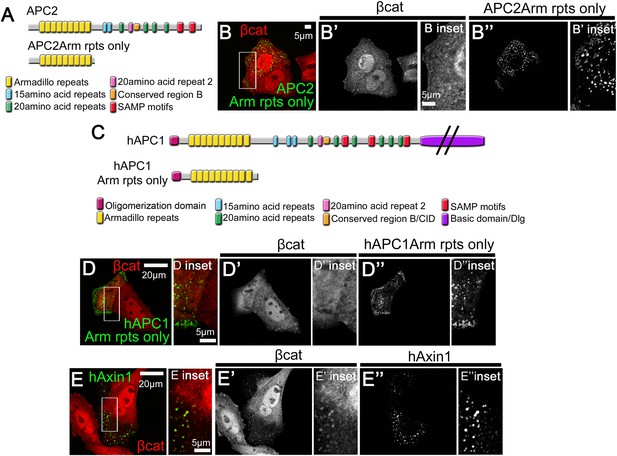
Human APC's Arm repeat domain colocalizes with Axin.
(A) Diagrams of fly APC2 and APC2Arm rpts only. (B) SW480 cells expressing GFP-APC2Arm rpts only. APC2Arm rpts only forms cytoplasmic puncta and is unable to reduce βcat levels (insets = box in B). (C) Diagrams of hAPC1 and hAPC1Arm rpts only mutant. (D) GFP-hAPC1Arm rpts only expressed in SW480 cells. hAPC1Arm rpts also forms cytoplasmic puncta and is unable to reduce βcat levels (insets = box in D). (E) GFP-hAxin1 expressed in SW480 cells forms cytoplasmic puncta, and does reduce βcat levels, but βcat accumulates in the puncta (insets = box in D).
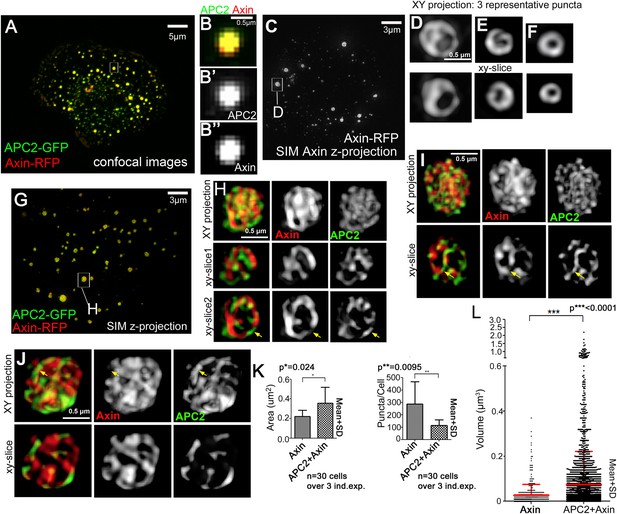
Axin and APC2 form structured macromolecular complexes in vivo.
SW480 cells. (A) Confocal image, GFP-APC2 and Axin-RFP. APC2 is recruited into Axin puncta. (B) Closeups, showing failure to resolve internal structure. (C–J) SIM super-resolution. (C) Axin-RFP alone. (D–F) Closeups, Axin complexes from different cells. D = punctum boxed in C. Axin cables assemble into spheres/sheets. (G) Cell coexpressing GFP-APC2 and Axin-RFP. (H–J) Closeups of APC2:Axin complexes from different cells. H = punctum boxed in G. Axin cables increase in complexity and APC2 forms cables intertwined with Axin (arrows). (K) Analysis of confocal images. Complexes formed by APC2 and Axin average nearly twice the cross-sectional area of complexes formed by Axin alone (left). Axin-expressing cells have twice as many complexes as cells coexpressing APC2 + Axin (right). Student's t-test. (L) Puncta volume in Axin expressing cells (n = 3) vs APC2 + Axin expressing cells (n = 11) showing volumes across puncta population. Volume differences expressing cells are consistent with area quantification in (K). ANOVA-Bonferroni was used.
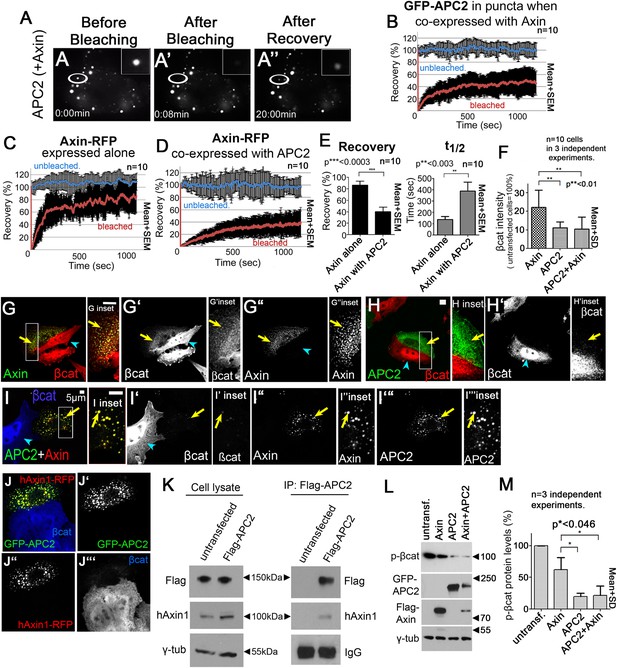
APC2 stabilizes Axin complexes and promotes efficient βcat destruction.
(A) Stills, FRAP movie, SW480 cells transfected with GFP-APC2 (shown) and Axin-RFP. Inset = magnified APC2 signal in punctum. (B) APC2 recovers to ∼40% when in Axin puncta. Recovery curve (red); unbleached control (blue). (C) Axin expressed alone plateaus at ∼80%. (D and E) Axin is stabilized when coexpressed with APC2. (F) Total cell βcat fluorescent intensity normalized to untransfected cells (= 100%). APC2 or APC2 + Axin expression lead to stronger βcat reduction than Axin alone. (G–I) Indicated constructs expressed in SW480 cells. Insets = regions boxed. (G) GFP-Axin forms puncta and reduces βcat levels in this hAPC1 mutant cell line. βcat is detectable in puncta (arrows). (H) GFP-APC2 expressed alone is dispersed throughout the cell and βcat levels are low overall and in puncta. (I) Axin-RFP + GFP-APC2 coexpressed. βcat is reduced in APC2:Axin puncta (arrow) relative to puncta with Axin alone (G). (J) GFP-APC2 is recruited into puncta formed by human hAxin1-RFP. (K) Endogenous human hAxin1 co-IPs from SW480 cells with transfected Flag-APC2. Untransfected cells serve as a negative control. (L and M) Phospho-S33/37-βcat levels are more reduced when either APC2 or APC2 + Axin are expressed relative to Axin alone. (L) Immunoblot , transiently transfected SW480 cell extracts, centrifuged at 1000 rpm. (M) Quantification, phospho-S33/37-βcat protein levels from (L) and 2 replicates. Student's t-test.
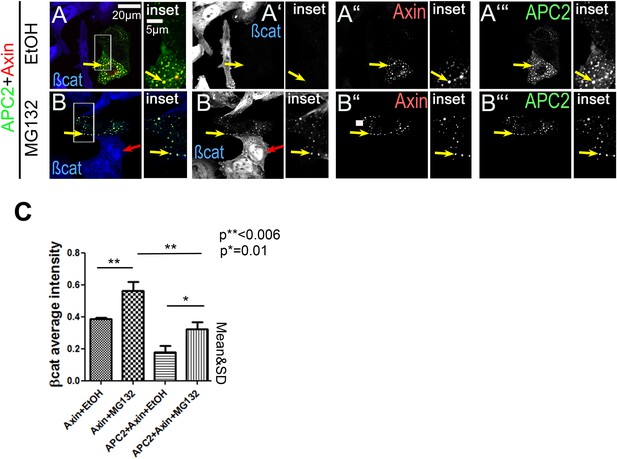
While proteasome inhibition reduces βcat destruction and causes βcat to detectably accumulate in APC2 + Axin puncta, it does not abolish the ability of APC2 to enhance Axin function in this regard.
(A and B) SW480 cells transfected with APC2 and Axin and treated with either ethanol as a control or with the proteasome inhibitor MG132. MG132 treatment elevates overall βcat levels and leads detectable accumulation in APC2 + Axin puncta (yellow arrows). Red arrows indicate untransfected cells. (C) Quantitation of total cell βcat fluorescent intensity normalized to untransfected cells (= 100%). MG132 treatment increases βcat levels in both Axin and APC2 + Axin treated cells. However, APC2 + Axin expression still leads to stronger βcat reduction than Axin alone.
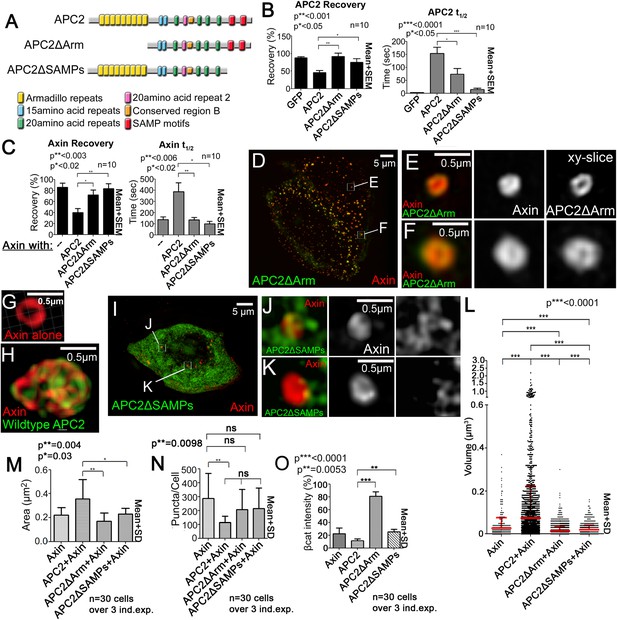
APC2’s Arm rpts and SAMPs each are required to stabilize APC2:Axin complexes.
(A) APC2 mutants. (B and C) FRAP analyses, SW480 cells. (B) APC2 needs both the Arm rpts and SAMPs to robustly associate with Axin puncta. Student's t-test. (C) Axin stabilization by APC2 is abolished when either APC2's Arm rpts or SAMPs are deleted. (D,K) SIM super-resolution images, SW480 cells expressing indicated constructs. (D–F) GFP-APC2ΔArm and Axin-RFP. (E–F) Close-ups, X–Y slice. Axin structure resembles complexes formed by Axin alone. (G and H) Axin-RFP puncta and APC2:Axin puncta for comparison. (I–K) GFP-APC2ΔSAMPs and Axin-RFP. (J,K) Close-ups. Axin does not form a complex internal structure when APC2 ΔSAMPs is expressed. (L) Puncta volume in SIM images of Axin (n = 3 cells), APC2 + Axin (n = 11), APC2ΔArm + Axin(n = 9), and APC2ΔSAMPs + Axin (n = 5) expressing cells. Deleting either the Arm rpts or the SAMPs inhibits APC2's ability to enhance puncta volume. ANOVA-Bonferroni. (M) Puncta area, confocal images. Area differences are consistent with volumes in (L). Student's t-test. (N) Puncta number, confocal images. Deleting Arm rpts or SAMPs in APC2 fails to decrease number of APC2:Axin puncta as does wildtype APC2. (O) APC mutants lacking the Arm rpts or SAMP motif show decreased ability to reduce βcat levels in SW480 cells. Quantification, total cell βcat fluorescent intensity normalized to untransfected cells.
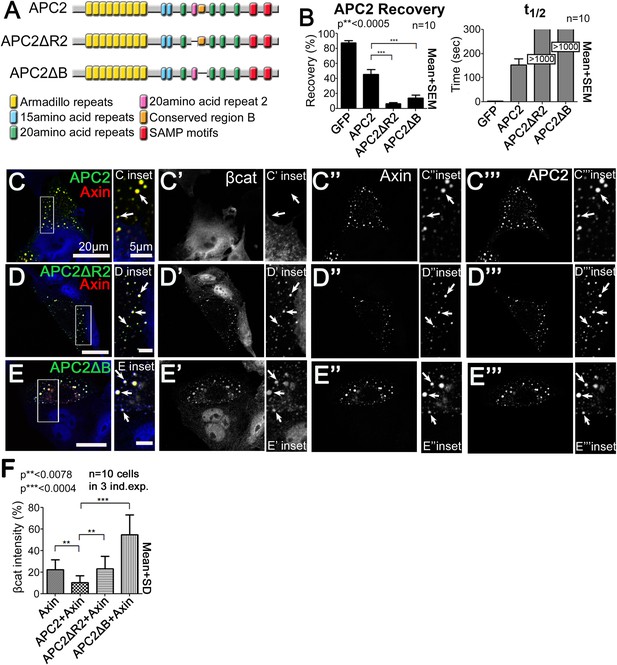
R2 and B regulate APC2 dynamics in the destruction complex, and regulate βcat removal from the destruction complex.
(A) APC2 mutants. (B–F) SW480 cells transfected with Axin-RFP and indicated GFP-APC2 constructs (B) FRAP assay. Deleting either R2 or B slows APC2's turnover in Axin puncta. (C–E) Axin-RFP, GFP-APC2 constructs, βcat (inset = boxes). (C) βcat is essentially undetectable in APC2:Axin puncta (arrows). (D) βcat accumulates in APC2ΔR2:Axin puncta (arrows). (E) βcat strongly accumulates in APC2ΔB:Axin puncta (arrows). (F) Deletion of R2 or B impair the ability of APC2 to aid Axin in reducing βcat fluorescent intensity.
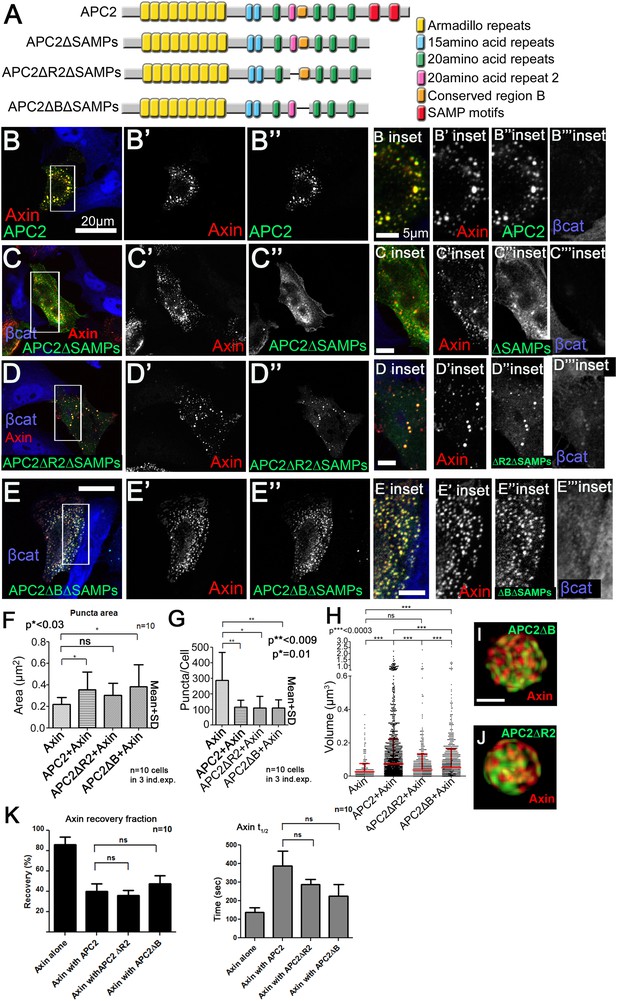
Colocalization of APC2's Arm repeat domain with Axin is controlled by R2 and B and APC2 without R2 or region B still stabilizes Axin complexes.
(A) Diagram of APC2 constructs. (B) GFP-APC2 and Axin-RFP colocalize with one another in SW480 cells and reduce βcat levels (inset = box in B). (C) Deleting APC2's SAMPs reduces colocalization with Axin (inset = box in C). (D) Deleting R2 from APC2ΔSAMPs enhances colocalization with Axin (inset = box in D). (E) APC2ΔSAMPs lacking B colocalizes strongly with the Axin (inset = box in E). (F) Puncta area of APC2ΔR2 + Axin and APC2ΔB + Axin is similar to wildtype APC2 + Axin and differs from puncta formed by Axin alone. Analysis of confocal images, total of n = 30 cells. Student's t-test was used. (G) Wildtype APC2, APC2∆R2 and APC2∆B all reduce Axin puncta number. Quantitation of puncta number of confocal images used in (F). (H) Comparison of puncta volumes using SIM images reveals that APC2ΔR2 + Axin and APC2ΔB + Axin form puncta that are in between those formed by Axin alone and those formed by APC2 + Axin puncta. Volumes across puncta population. Axin (n = 3), APC2 + Axin (n = 11), APC2ΔR2 + Axin (n = 6), and APC2ΔB + Axin (n = 9). ANOVA-Bonferroni was used. (I,J) APC2ΔR2 + Axin and APC2ΔB + Axin puncta maintain complex internal structure. Closeups of puncta of SIM high resolution images of APC2ΔB + Axin (I) and APC2ΔR2 + Axin expressed in SW480 cells. (K) Deleting R2 or B does not significantly diminish the ability of APC2 to stabilize Axin in the destruction complex.
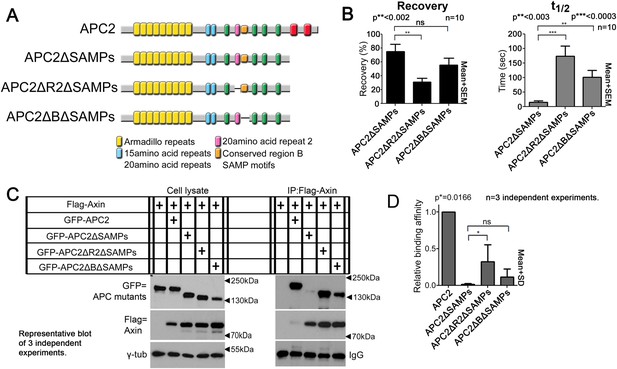
Association of Axin with APC2's Arm rpts is controlled by R2 and B.
(A) APC2 constructs. (B) Deleting either R2 or B in APC2ΔSAMPs slows APC2 recovery time and reduces recovery fraction. FRAP assay, SW480 cells transfected with GFP-tagged APC2 constructs and Axin-RFP. (C) IPs of indicated constructs. Deleting the SAMPs substantially decreases APC2:Axin coIP, but this is partially restored by deleting R2. (D) Quantification, >2 replicates, normalized to Axin pull down. Student's t-test.
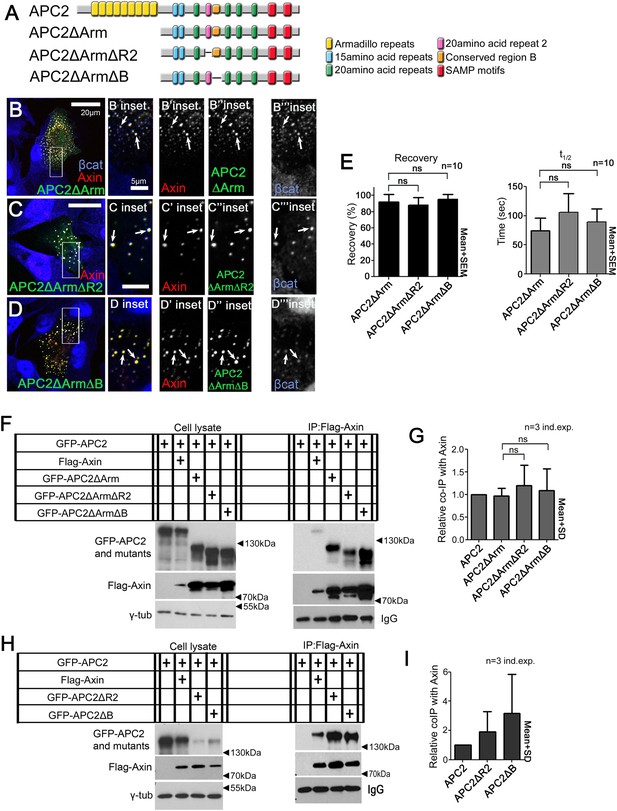
Binding of APC2's SAMP motif to Axin is not regulated by R2 and B.
(A) Diagram of APC2 constructs. (B) GFP-APC2ΔArm colocalizes with Axin-RFP in SW480 cells (inset = box in B). βcat is detectable in APC2ΔArm:Axin complexes. (C) Deleting R2 does not alter colocalization of APC2ΔArm with Axin (inset = box in C). βcat remains detectable in the APC2ΔArmΔR2:Axin puncta. (D) APC2ΔArm lacking B colocalizes with Axin (inset = box in D). βcat is detectable in the APC2ΔArmΔB:Axin complexes. (E) FRAP assay of GFP-APC2ΔArm mutants with Axin-RFP in SW480 cells. Deletion of either R2 or B does not alter turnover of APC2ΔArm. Student's t-test was used. (F) Co-Immunoprecipitations of GFP-APC2ΔArm mutants with Flag-Axin. IP of Axin via anti-Flag antibody. APC2 binding to Axin via the SAMP motifs is not altered when R2 or B are deleted. Representative blot of 3 independent experiments. (G) Quantification Co-IP of indicated APC2 mutants with Flag-Axin ((F) and 2 replicates), normalized to coIP with wild-type APC2. Binding via the SAMPs is not altered when R2 or B are deleted. Student's t-test was used. (H) Co-IP of indicated APC2 mutants with Flag-Axin. Deletion of either R2 or B in full length APC2 increases its association with Axin. Representative blot of 3 independent experiments. (I) Quantification, Co-IP of indicated APC2 mutants with Flag-Axin ((H) and 2 replicates), normalized to coIP with wild-type APC2. Student's t-test was used.
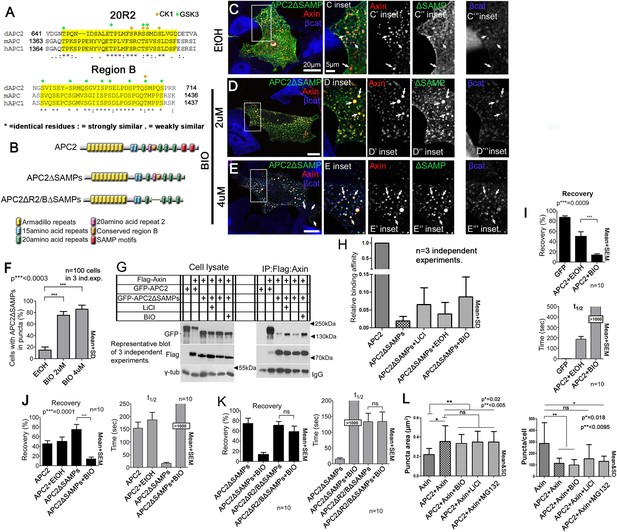
Axin:APC2 Arm rpts association is regulated by GSK3.
(A) R2 and B of Drosophila dAPC2, mouse mAPC1 and human hAPC1. Potential CK1(orange) and GSK3(green) phosphorylation sites. (B) APC2 mutants. (C–E) SW480 cells expressing GFP-APC2ΔSAMPs and Axin-RFP. Insets = boxes in C–E (C) Control (Ethanol treated (EtOH)). Deleting the SAMPs reduces APC2:Axin colocalization (arrows). (D) 2 μM BIO enhances APC2ΔSAMPs recruitment into Axin puncta. (E) Increasing BIO to 4 μm further boosts APC2ΔSAMPs recruitment into Axin puncta. (F) Quantification of (C–E). (G) CoIP of APC2∆SAMPs with Axin in SW480 cells ± LiCl or BIO. Full length APC2 is a control. Deleting the SAMPs drastically reduces coIP but GSK inhibition partially restores this. (H) Quantification of coIP in G, >2 replicates, normalized to Axin. (I) FRAP assay, SW480 cells transfected with Axin-RFP + GFP-APC2. GSK3 inhibition decreases APC2 dynamics. (J) GSK inhibition also slows APC2ΔSAMPs dynamics. (K) Deleting R2/B in APC2ΔSAMPs abolishes effect of GSK3 inhibition on dynamics. Student's t-test. (L) GSK3 inhibition with either BIO or LiCl does not further increase the size or decrease the number of APC plus Axin puncta, nor does treatment with the proteasome inhibitor MG132. Puncta area and puncta number, from confocal images.
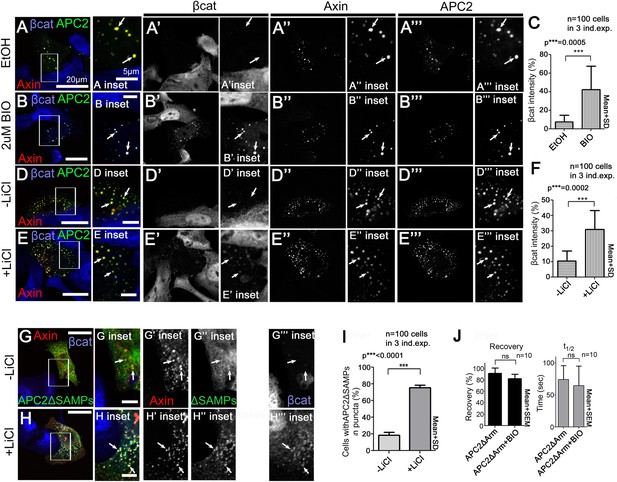
GSK3 regulates association of APC2's Arm repeats with the Axin complex.
(A, B, D and E) GFP-APC2 and Axin-RFP expressed in SW480 cells. βcat stained via antibody. (A) APC2 and Axin strongly colocalize in cytoplasmic puncta in cells treated with EtOH as control for BIO. (Inset = box in A) βcat levels in APC2:Axin complexes are very low. (B) GSK3 inhibition via BIO does not alter colocalization of APC2 with Axin, but leads to βcat accumulation in APC2:Axin puncta (inset = box in B). (C) Quantification of total βcat fluorescent intensity in (A and B). (D) In control cells for LiCl treatment, βcat levels in APC2:Axin complexes are very low (inset = box in D). (E) βcat accumulates in APC2:Axin complexes when GSK3 is inhibited via LiCl (inset = box in E). (F) Quantification of total βcat fluorescent intensity in (D and E). (G) GFP-APC2ΔSAMPs is diffuse and only associates weakly with Axin (inset = box in G) in control cells for LiCl treatment. (H) GSK3 inhibition with LiCl enhances association of APC2ΔSAMPs with Axin (inset = box in H). (I) Quantification of APC2ΔSAMPs colocalization with Axin (G vs H). Student's t-test was used. (J) FRAP assay in SW480 cells transfected with Axin-RFP and GFP-APC2ΔArm. GSK3 inhibition via BIO does not alter APC2's turnover rate when association is mediated by the SAMPs. Student's t-test was used.
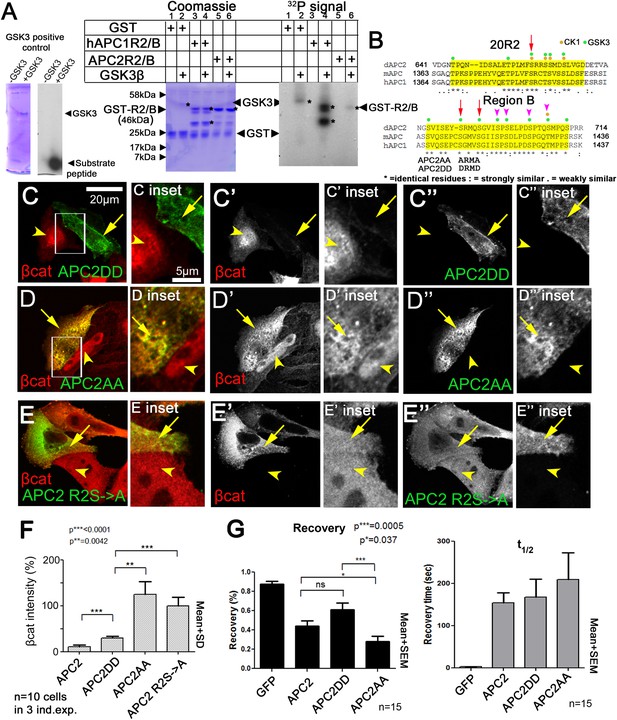
Mutating putative phosphorylation sites in B disrupts APC2 function.
(A) R2/B of human or fly APCs can be phosphorylated by human GSK3. In vitro kinase assay, GSK3 substrate peptide (positive control, left panels), GST-tagged humanAPC1R2/B or fly APC2R2/B fragments. GST was a negative control. Left of each pair: Coomassie stained gel, right: Phosphorylation detected using P32. Asterisks (*) indicate Coomassie-stained bands that align with P32-labeled proteins. In the presence of GST alone, GSK3 autophosphorylates (lane 2). HumanAPC1R2/B is strongly phosphorylated (lane 4) while fly APC2R2/B was more weakly phosphorylated (lane 6). Representative of two experiments. (B) R2 and B of Drosophila dAPC2, mouse mAPC1 and human hAPC1. Potential CK1 (orange) and GSK3 (green) phosphorylation sites. Red Arrow in R2 = serine mutated to alanine in mutant assessed in panel E. Red arrows in B = serines mutated to aspartic acid or alanine in APC2AA or APC2DD. Magenta arrowheads = additional serines mutated in 4 serine and six serine mutations (data not shown). (C) APC2DD (2 serines in B changed to aspartic acid; arrows in B) effectively reduces βcat levels in SW480 cells (arrow vs arrowhead). Inset = box in A. (D) APC2AA (2 serines in B changed to alanine, arrows in B) is unable to target βcat for destruction (arrow vs arrowhead). (E) APC2 R2S->A (single serine in R2 changed to alanine, arrow in B) is unable to target βcat for destruction (arrow vs arrowhead). (F) Quantification, total βcat fluorescent intensity. Student's t-test. (G) FRAP, Axin-RFP + GFP-APC2 constructs. APC2AA reaches a lower recovery plateau than either wild-type APC or APC2DD.
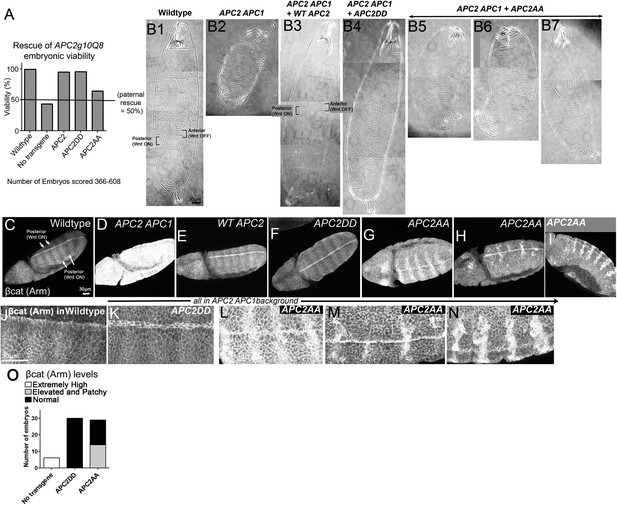
Blocking potential phosphorylation at 2 conserved serines in B disrupts APC2 function in the fly.
APC2DD and APC2AA (Figure 8B) were expressed with the endogenous APC2 promoter in APC2 APC1 maternal/zygotic double mutants. (A) APC2 APC1 maternal/zygotic double mutants die as embryos (50% of embryos are zygotically rescued). APC2DD rescues embryonic viability as well as wildtype APC2. In contrast, APC2AA has only weak rescue ability. (B) Cuticles. (B1) Wildtype. Note pattern of anterior denticles (Wnt inactive) and posterior naked cuticle (Wnt active). (B2) Loss of APC2 and APC1 leads to denticle loss and expanded naked cuticle. (B3) Wildtype APC2 fully restores Wnt regulated cell fates of alternating denticles and naked cuticle. (B4) APC2DD similarly restores cell fates. (B5–B7) APC2AA largely fails to restore Wnt-regulated cell fates, and thus most cells secrete naked cuticle. Images = range of rescue ability. (C–I) βcat (fly Armadillo (Arm)) levels. Stage 9–10 embryos. (J–N) Close-ups of C–I. (C,J) Wildtype. Striped pattern of βcat indicative of Wg (fly Wnt) active and Wg inactive regions. (D) Loss of APC1 and APC2 leads to uniform very high levels of βcat. (E) WT APC2 restores normal βcat regulation, with higher levels in cells receiving Wg signal, and lower levels in other cells. However, Wg signal does not elevate βcat levels to those seen in embryos lacking functional APC. (F and K) APC2DD also rescues normal βcat regulation. (G–I and L–N) APC2AA restores some Wnt responsiveness, but βcat levels are elevated in all cells and especially elevated in a subset of cells receiving Wnt signal. (O) Quantification, embryos blind-scored.
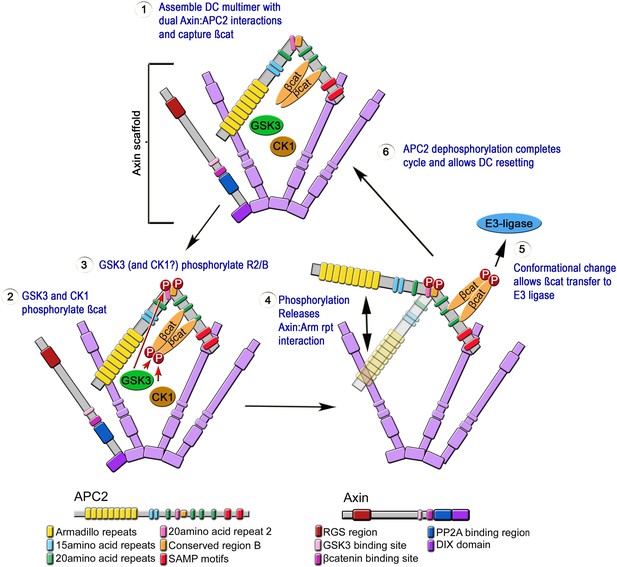
Speculative Model of APC2's catalytic cycle inside the destruction complex.
(1) APC2 assembles with Axin via its Arm rpts and SAMPs. (2) APC2-bound βcat is phosphorylated by CK1 and GSK3. (3) GSK3 and CK1 phosphorylate R2/B region in APC2. (4) This induces a conformational change that releases APC2's Arm rpts from Axin. (5) APC2-bound βcat is released to the E3-ligase—alternately an APC–βcat complex is released. (6) Dephosphorylation resets the cycle.
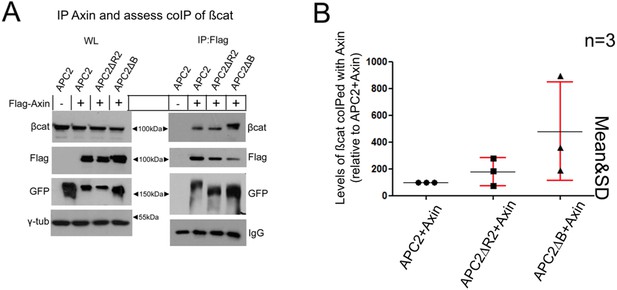
lnvestigating whether deleting R2 or B alters colP of þcat with Axin in SW480 cells. (A) Expression of Flag-Axin with indicated GFP-tagged APC2 constructs followed by lP of Flag-Axin and examination of the level of ßcat that colPs. Representative immunoblot of 3 independent experiments. (B) Quantification of 3 independent co-lPs. þcat levels were normalized to Flag-Axin. Levels of colPed ßcat were higher in 2 of 3 experiments with APC26R2 and in all three experiments with APC26B.
Videos
3D reconstruction of SIM superresolution image of Axin-RFP expressed in SW480 cells (see also Figure 2D).
Volume view from Imaris 5.5 was used for reconstruction.
3D reconstruction of SIM superresolution image of Axin-RFP (see also Figure 2E).
https://doi.org/10.7554/eLife.08022.0093D reconstruction of SIM superresolution image of Axin-RFP (see also Figure 2F).
https://doi.org/10.7554/eLife.08022.0103D reconstruction of SIM superresolution image of GFP-APC2 and Axin-RFP expressed in SW480 cells (see also Figure 2H).
Volume view from Imaris 5.5 was used for reconstruction.
3D reconstruction of SIM superresolution image of GFP-APC2 and Axin-RFP (see also Figure 2I).
https://doi.org/10.7554/eLife.08022.0123D reconstruction of SIM superresolution image of GFP-APC2 and Axin-RFP (see also Figure 2J).
https://doi.org/10.7554/eLife.08022.013Tables
Quantitation of relative expression levels of transfected versus endogenous APC and Axin
| Summary Axin overexpression | |||
|---|---|---|---|
| Experiment | I | II | III |
| Ratio GFP-FlyAxin to GFP-hAxin1 | 1.04 | 0.11 | 0.48 |
| Ratio GFP-hAxin1 to endo hAxin1 | 33.87 | 431.67 | 62.82 |
| Transfection efficiency | 30% | 42% | 36% |
| Fold overexpression level of GFP-FlyAxin to endo hAxin1 | 117.41 | 113.06 | 83.76 |
| Summary APC2 overexpresssion | |||
|---|---|---|---|
| Experiment | I | II | III |
| Ratio Flag-FlyAPC2 to Flag-hAPC1-1338 | 17.31 | 138.45 | 28.02 |
| Ratio Flag-hAPC1-1338 to endo hAPC1-1338 | 25.17 | 5.86 | 14.69 |
| Transfection efficiency | 30% | 33% | 25% |
| Fold overexpression level of Flag-FlyAPC2 to endo hAPC1-1338 | 1452.30 | 2458.54 | 1646.46 |





