Nemo-like kinase is a novel regulator of spinal and bulbar muscular atrophy
Figures
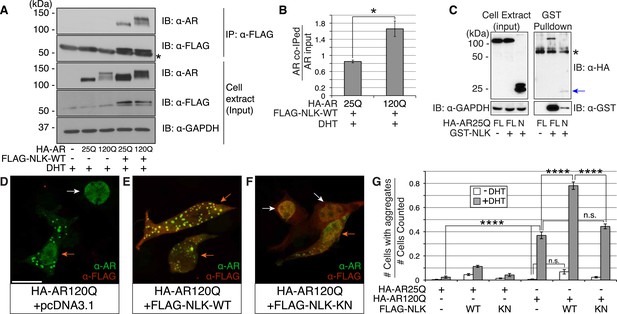
Nemo-like kinase (NLK) interacts with the mutant AR and enhances its aggregation.
(A) NLK interacts with the AR protein in NSC-34 cells treated with 10 nM DHT. IP: immunoprecipitation. IB: immunoblot. GAPDH was used as a loading control in this and all following analyses unless otherwise specified. Asterisk marks a band corresponding to the immunoglobin heavy chain. (B) Quantification of co-IPed AR over total AR in input. *p < 0.05 (t-test). n = 3 trials. Error bars are standard error of the mean in this and all following graphs unless otherwise specified. (C) NLK interacts with the N-terminal region of AR. Both full-length (FL) and an N-terminal fragment (N, arrow) of AR were pulled down with NLK. Asterisk marks a non-specific band. (D–G) NLK enhances the formation of mutant AR aggregates in a kinase activity-dependent manner. NSC-34 cells were treated with DHT as indicated and subjected to immunofluorescence using anti-AR N-20 (green) and anti-FLAG (red) antibodies to detect AR aggregation and NLK co-expression, respectively. NLK-WT: wild-type NLK. NLK-KN: kinase-dead NLK. Representative images of DHT-treated cells are shown in (D–F). Images of the non-DHT-treated and AR25Q-expressing cells can be found in the Figure 1—figure supplements 1–3. Scale bar in (D) is 20 μm and refers to all three images. Cells were scored as containing aggregates (orange arrows) or not (white arrows) and the ratio of aggregate-positive cells out of total scored is quantified in (G). n.s. = not significant, ****p < 0.0001 (ANOVA with Tukey's post-hoc analysis). n ≥ 3 trials.
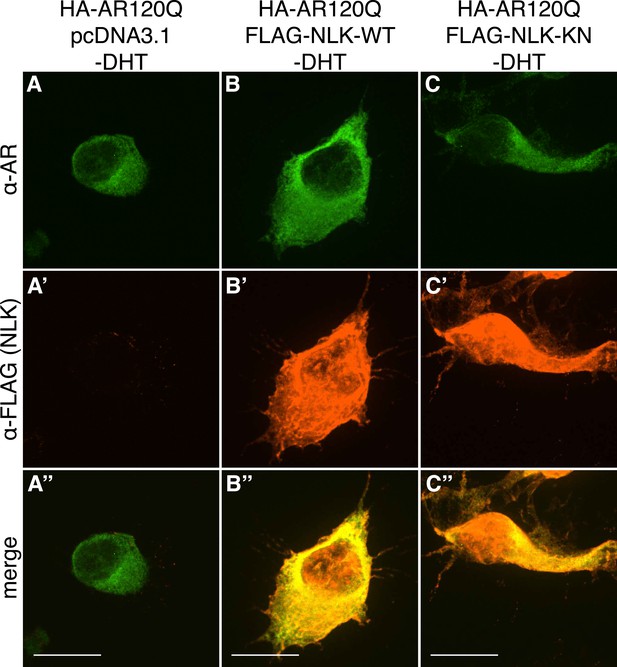
PolyQ-expanded AR120Q does not aggregate in the absence of DHT.
Representative z-stack images of NSC-34 cells that were transfected as indicated, treated with ethanol (as a negative control for DHT), and subjected to immunofluorescence using anti-AR N20 (green; A–C) and anti-FLAG (red; A′–C′) antibodies to detect AR and NLK expression, respectively. Merged images are shown in (A′′–C′′). In the absence of hormone, AR120Q shows diffuse cytoplasmic localization, while NLK localizes to both the cytoplasm and the nucleus. Scale bars in merged images are 25 μm. There is a variation in overall cell size with NSC-34 cells that is not obviously influenced by the transfection of AR or NLK.
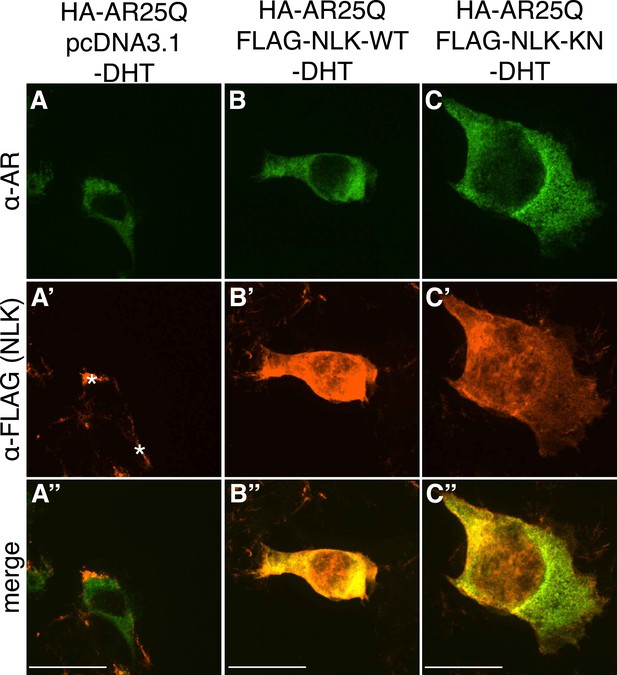
Non-pathogenic AR25Q shows diffuse cytoplasmic localization in the absence of DHT.
Representative z-stack images of NSC-34 cells that were transfected as indicated, treated with ethanol (as a negative control for DHT), and subjected to immunofluorescence using anti-AR N20 (green; A–C) and anti-FLAG (red; A′–C′) antibodies to detect AR and NLK expression, respectively. Merged images are shown in (A′′–C′′). In the absence of hormone, AR25Q shows diffuse cytoplasmic localization, while NLK localizes to both the cytoplasm and the nucleus. Asterisks in (A′) mark non-specific staining in the red channel. Scale bars in merged images are 25 μm. There is a variation in overall cell size with NSC-34 cells that is not obviously influenced by the transfection of AR or NLK.
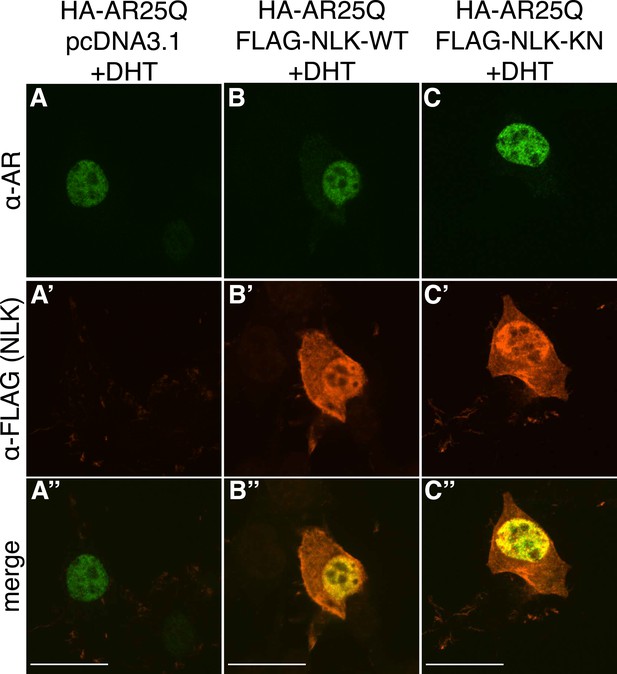
Non-pathogenic AR25Q undergoes nuclear translocation in response to DHT, but largely does not aggregate.
Representative z-stack images of NSC-34 cells that were transfected as indicated, treated with 10 nM DHT, and subjected to immunofluorescence using anti-AR N20 (green; A–C) and anti-FLAG (red; A′–C′) antibodies to detect AR and NLK expression, respectively. Merged images are shown in (A′′–C′′). In the presence of hormone, AR25Q shows nuclear localization, while NLK localizes to both the cytoplasm and the nucleus. Scale bars in merged images are 25 μm. There is a variation in overall cell size with NSC-34 cells that is not obviously influenced by the transfection of AR or NLK.
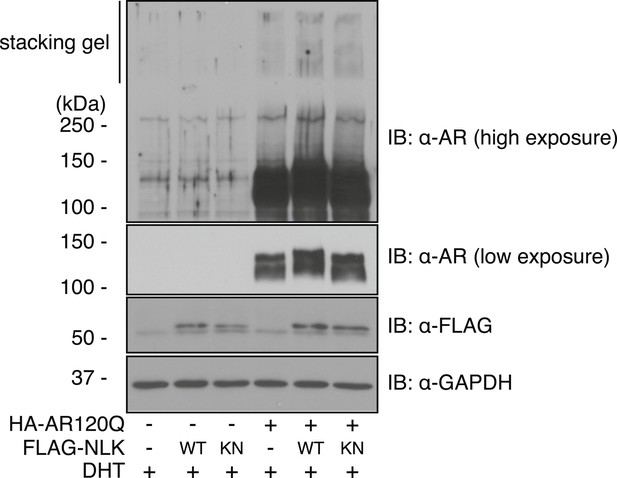
Mutant AR forms high molecular weight aggregates in the stacking gel of SDS–PAGE gels.
NSC-34 cells were transfected as indicated and treated with 10 nM DHT. High molecular weight AR aggregates can be detected as a smear in the stacking gel. This aggregation is only seen upon mutant AR expression and is increased with co-expression of NLK.
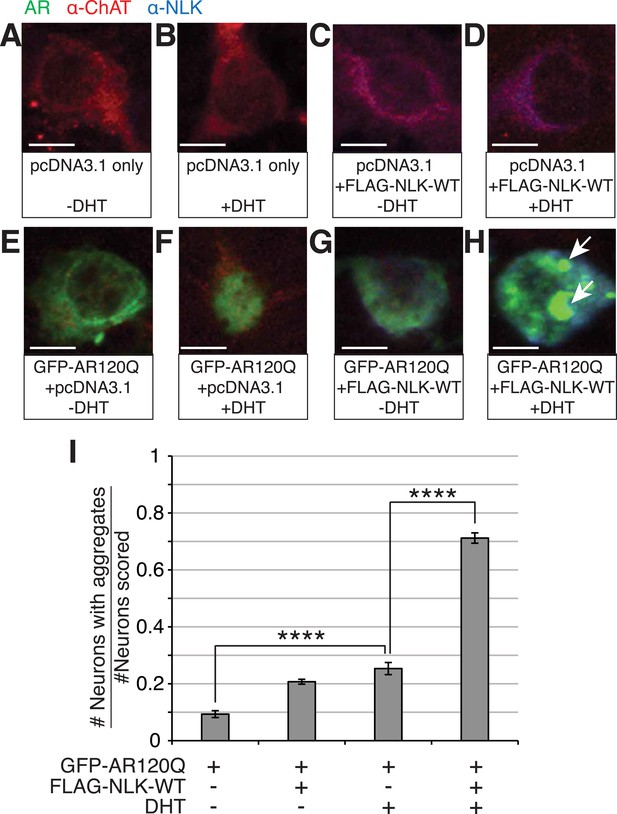
NLK increases mutant AR aggregation in primary motor neurons.
(A–H) Primary motor neurons were transfected with GFP-tagged AR120Q, FLAG-tagged NLK-WT, or a pcDNA3.1 empty vector control and treated with 10 μM DHT. Aggregation was analyzed by immunofluorescence at 9 days in vitro (DIV). An antibody to choline acetyltransferase (ChAT) was used to confirm motor neuron identity and is shown in red. GFP-AR120Q is shown in green and NLK co-expression (as detected by an NLK antibody) is in blue. All images were collected using identical confocal settings. In the absence of DHT, AR localizes to the cytoplasm (E, G), while DHT induces its nuclear translocation (F) and its aggregation, which is enhanced by NLK (H). Arrows mark aggregates, which can be detected in both the nucleus and cytoplasm. Scale bars are 10 μm. (I) The number of neurons containing aggregates out of total scored was quantified and averaged over different regions of the plate. At least 140 neurons were scored per condition. ****p < 0.0001 (ANOVA with Tukey's post-hoc analysis).
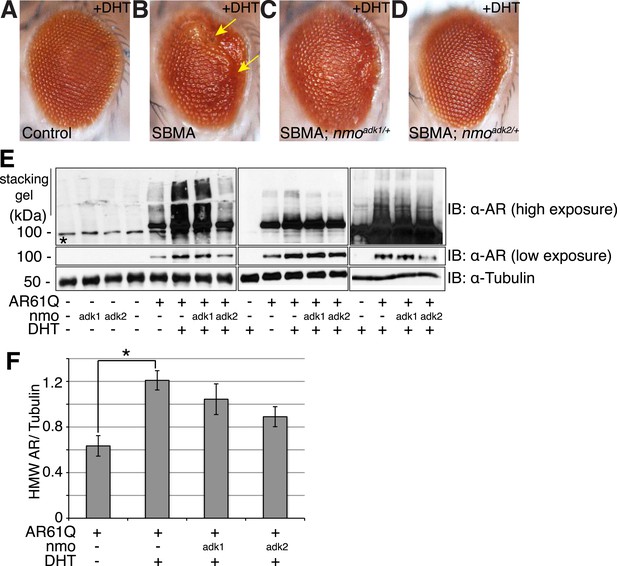
NLK genetically interacts with the mutant AR in Drosophila.
Loss of one nmo allele suppresses mutant AR-mediated SBMA phenotypes in Drosophila. (A–D) Light microscopy of adult Drosophila eyes is shown. In (B), arrows mark a DHT-dependent retinal degeneration phenotype along the posterior margin. Flies were raised at 30°C and genotypes are as follows: (A) GMR-Gal4/+; UAS-EGFP/+, (B) GMR-Gal4, UAS-AR61Q/+, (C) GMR-Gal4, UAS-AR61Q/+; nmoadk1/+, (D) GMR-Gal4, UAS-AR61Q/+; nmoadk2/+. For all panels, experiments were repeated multiple times and representative images are shown. (E) Western blots from three different trials show the aggregation of the mutant AR as a smear in the stacking gel at high exposure. Lower exposure reveals the AR61Q monomer at the expected size of around 110 kDa. Asterisk marks a non-specific band present in all lanes. (F) High molecular weight (HMW) or aggregated AR was quantified as compared to the tubulin loading control and averaged over trials. *p < 0.05 (ANOVA with Tukey's post-hoc analysis). n ≥ 3 trials.
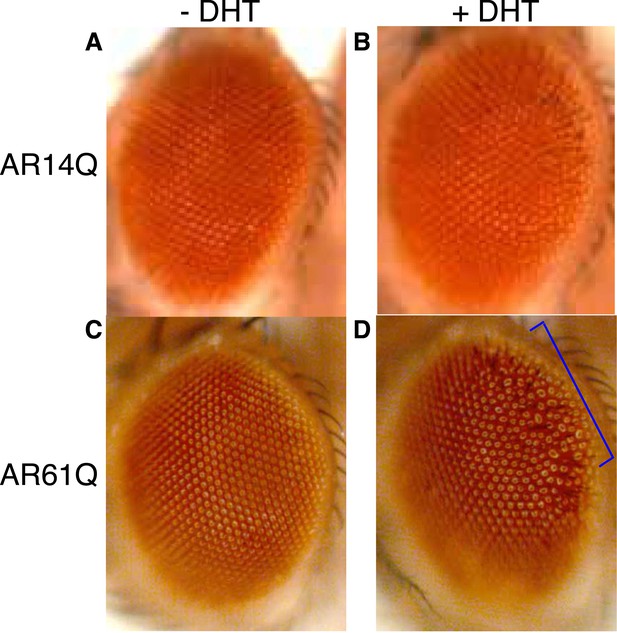
Expression of a full-length AR protein in the Drosophila eye results in polyQ- and DHT-dependent retinal degeneration phenotypes.
(A, B) Expression of a full-length AR transgene with 14Q (wild-type, AR14Q) in the fly eye does not produce a recognizable phenotype on the exterior fly eye, regardless of DHT treatment. (C, D) Expression of a full-length AR transgene with 61Q (mutant, AR61Q) results in a ‘rough’ eye phenotype along the posterior margin of the eye (bracket) in the presence of DHT. Flies were raised at 25°C. Phenotypes were consistent over multiple trials. Genotypes are as follows: (A) GMR-Gal4/UAS-AR14Q [without DHT] (B) GMR-Gal4/UAS-AR14Q [with DHT], (C) GMR-Gal4/UAS-AR61Q [without DHT], (D) GMR-Gal4/UAS-AR61Q [with DHT].
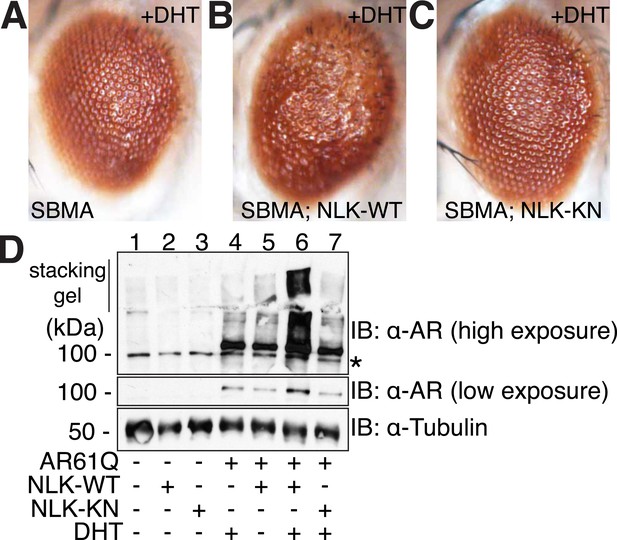
NLK modulates mutant AR phenotypes in Drosophila in a kinase activity-dependent manner.
(A–C) Light microscopy of adult Drosophila eyes is shown. Flies were raised at 30°C and genotypes are as follows: (A) GMR-Gal4, UAS-AR61Q/UAS-EGFP, (B) GMR-Gal4, UAS-AR61Q/UAS-NLK-WT, (C) GMR-Gal4, UAS-AR61Q/UAS-NLK-KN. (D) Mutant protein aggregation is shown by immunoblot with indicated genotypes. Aggregated mutant AR protein can be detected as a smear in the stacking gel at higher exposures, while the AR61Q monomer expresses at around 110 kDa and can be seen at lower exposures. Asterisk marks a non-specific band present in all lanes. For all panels, experiments were repeated multiple times, and representative images are shown.
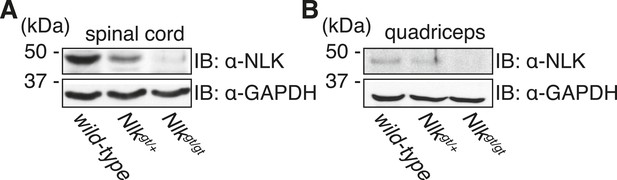
Nlkgt mice show reduced NLK expression in the spinal cord and skeletal muscle.
Whole spinal cord (A) and quadriceps (B) extracts from indicated genotypes were immunoblotted with a NLK antibody. Mice heterozygous for Nlkgt show a 50% reduction in protein expression, while mice homozygous for the allele show an approximately 90% reduction. GAPDH was used as a loading control.
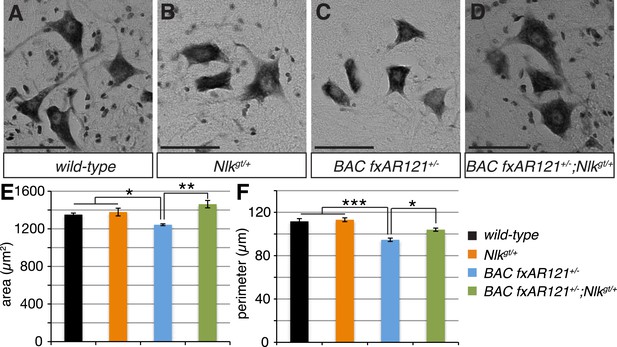
Loss of one copy of Nlk improves the pathogenic change in motor neuronal soma size in SBMA mice.
(A–D) Spinal cord cross-sections from the L4–L5 region were stained with cresyl violet (nissl stain) to visualize the spinal motor neuron cell bodies. Representative images from the anterior horn region of 40-week-old mice are shown. Scale bars are 50 μm. (E, F) The average motor neuron area (E) and perimeter (F) were measured and averaged over genotype. n = 2, 4, 4, 3 per genotype, respectively. Over 100 neurons were scored per animal. *p < 0.05, **p < 0.01, ***p < 0.001 (ANOVA with Tukey's post-hoc analysis).
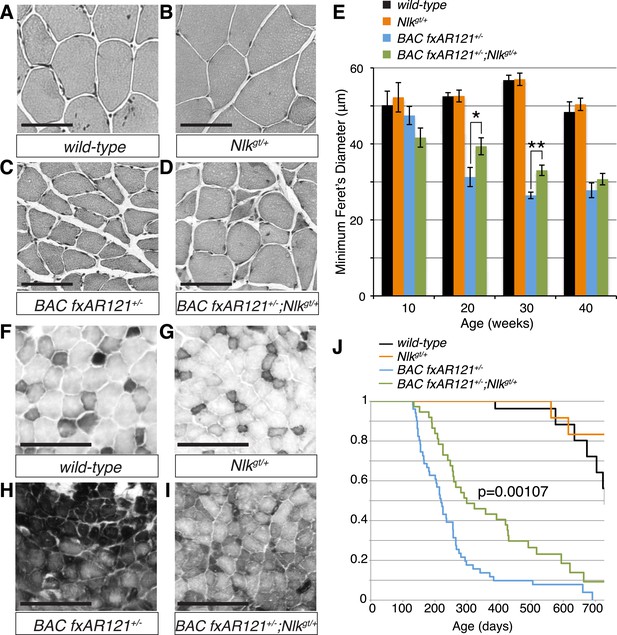
Loss of one copy of Nlk significantly rescues SBMA phenotypes in mice.
(A–D) Mouse quadriceps sections of indicated genotypes were stained for hematoxylin and eosin and representative 30-week-old images are shown. Scale bars are 50 μm. (E) Quantification of the average minimum Feret's diameter of muscle fibers at ages indicated. *p < 0.05, **p < 0.005 (t-test). For 10 weeks, n = 3, 3, 4, and 3 per genotype, respectively. For 20 weeks, n = 4, 3, 5, and 5. For 30 weeks, n = 7, 5, 5, and 8. For 40 weeks, n = 2, 5, 4, and 3. More than 500 fibers were scored per animal. See also Figure 7—figure supplement 1. (F–I) Reduced NLK expression improves the defective NADH transferase activity pattern seen in BAC fxAR121+/− mouse muscle. Six littermate sets were compared and representative images at 30 weeks of age are shown. Scale bars are 200 μm. See also Figure 7—figure supplement 2. (J) Kaplan–Meier survival analysis shows a significant extension in the lifespan of BAC fxAR121+/− mice with a 50% reduction of NLK. p = 0.00107 (log rank test). n = 27, 27, 51, and 37 per genotype, respectively.
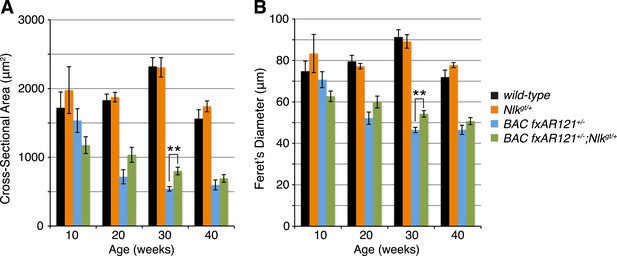
Loss of one copy of Nlk increases muscle fiber size in BAC fxAR121+/− mouse quadriceps.
Mouse quadriceps were stained for hematoxylin and eosin and the average cross-sectional area (A) and Feret's diameter (B) of muscle fibers were quantified at ages indicated. The BAC fxAR121+/−; Nlkgt/+ mice (green) have slightly larger fibers than their BAC fxAR121+/− littermate controls (blue). **p < 0.005 (t-test). For 10 weeks, n = 2, 3, 3, and 2 per genotype, respectively. For 20 weeks, n = 4, 3, 5, and 5. For 30 weeks, n = 7, 5, 6, and 8. For 40 weeks, n = 2, 5, 4, and 3. More than 500 fibers were scored per animal.
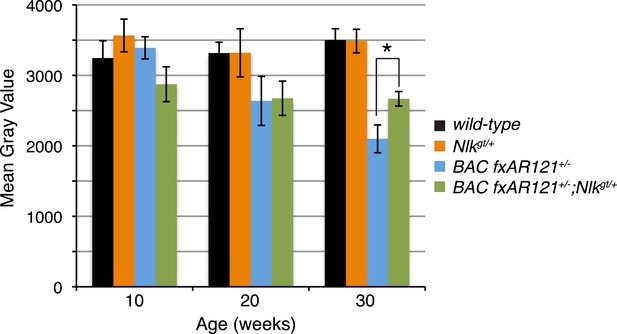
A 50% reduction in NLK expression reduces aberrant NADH transferase staining in 30-week-old SBMA mice.
Reduced NLK expression partially rescues the defective NADH transferase activity pattern seen in BAC fxAR121+/− mouse muscle. Representative images at 30 weeks are shown in Figure 7F–I. The average mean gray value of all images is quantified here with higher values corresponding to lower intensity staining. For 10 weeks, n = 3, 4, 4, and 3 per genotype, respectively. For 20 weeks, n = 4, 3, 5, and 5. For 30 weeks, n = 7, 5, 6, and 8. *p < 0.05 (t-test).
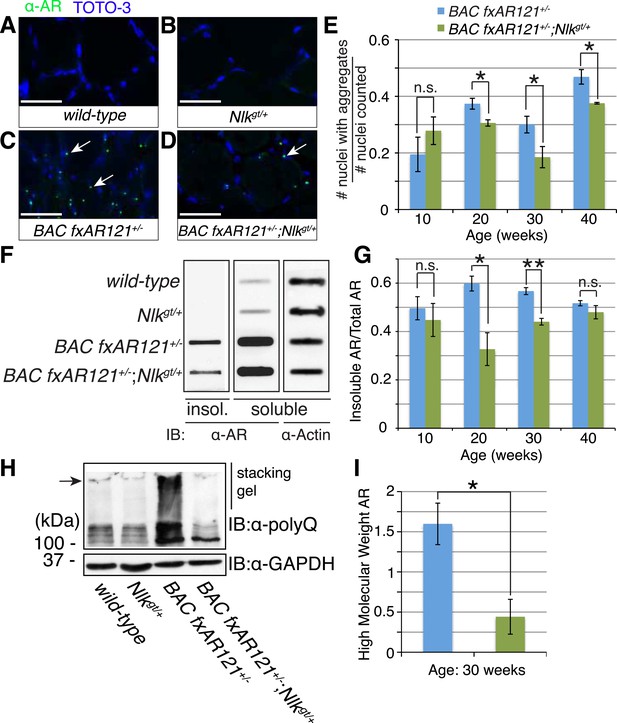
Loss of one copy of Nlk decreases mutant AR aggregation in mice.
(A–D) Nuclear AR aggregates (arrows) can be detected in quadriceps of mice expressing the BAC fxAR121 transgene (C, D), but not in controls (A, B). Representative 30-week-old samples are shown. Scale bars are 50 μm. Nuclei are marked with TOTO-3 in blue. (E) Quantification of the ratio of nuclei containing aggregates out of total nuclei counted; 300 to 500 fibers per mouse. n.s. = not significant, *p < 0.05 (t-test). For 10 weeks, n = 3 each. For 20 weeks, n = 5 each. For 30 weeks, n = 5 and 8, respectively. For 40 weeks, n = 4 and 3, respectively. (F) A representative filter trap assay blot from 20-week-old quadriceps samples. (G) The amount of insoluble (Insol.) AR out of total (Insol. + Soluble) was quantified. n.s. = not significant, *p < 0.05, **p < 0.005 (t-test). For 10 weeks, n = 3 each. For 20 weeks, n = 5 each. For 30 weeks, n = 4 each. For 40 weeks, n =4 and 3, respectively. (H) A representative blot shows mutant AR retained in the stacking gel of SDS-PAGE gels as high molecular weight aggregates (arrow). 30-week-old quadriceps samples are shown. An antibody to the polyQ region (1C2) was used. (I) Quantification of AR in the stacking gel normalized to loading control. *p < 0.05 (t-test). n = 3 for each genotype.
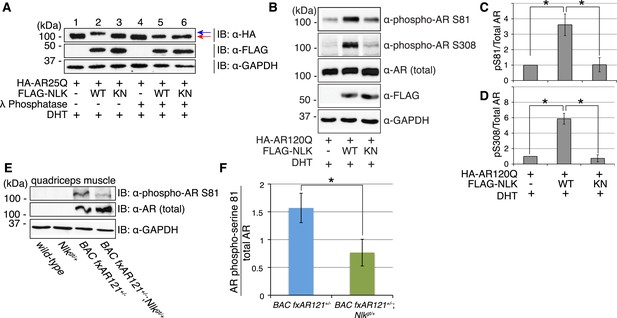
NLK influences the phosphorylation status of AR.
(A) NLK can induce the phosphorylation of AR in a cell culture system. AR25Q is shown here, but the same effect is seen with polyQ-expanded AR. (B–D) NLK can phosphorylate the mutant AR at S81 and S308. (C) Quantification of phospho-AR-S81 expression over total AR expression (as detected by AR-N20 antibody). (D) Quantification of phospho-AR-S308 expression over total AR expression. *p < 0.05 (t-test). n ≥ 4 trials. (E, F) NLK can affect mutant AR phosphorylation in SBMA mouse muscle in vivo. (E) Representative image of 30-week-old mouse quadriceps samples immunoblotted with phospho-AR-S81 antibody and an antibody to detect total AR. Only mutant AR protein is shown here, but a lower wild-type AR band can also be detected in all 4 genotypes. (F) Quantification of phospho-AR-S81 expression over total AR expression. *p < 0.05 (t-test). n = 7 and 9 for BAC fxAR121+/− and BAC fxAR121+/−; Nlkgt/+, respectively.
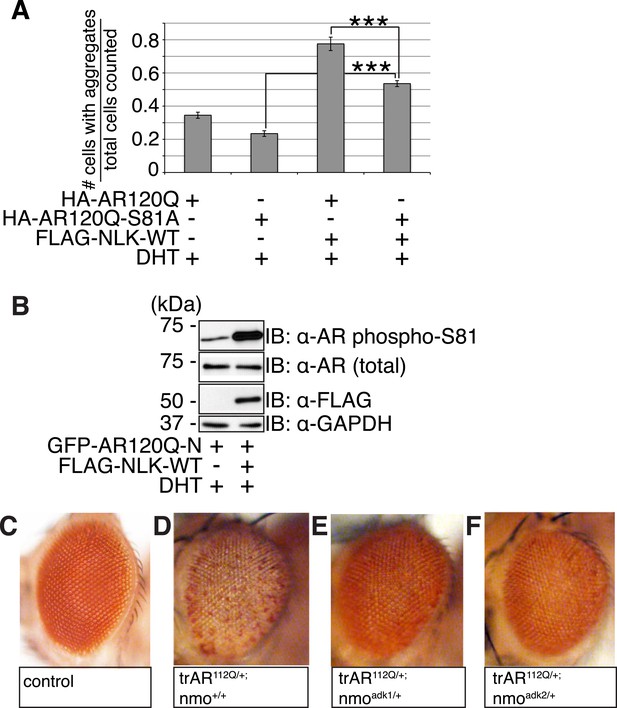
NLK regulates the aggregation and toxicity of mutant AR by influencing the phosphorylation of AR at residues including S81.
(A) AR-S81 phosphorylation could contribute to the NLK effect on mutant AR aggregation. NSC-34 cells were transfected with indicated constructs and treated with 10 nM DHT. Quantification of the ratio of cells containing AR aggregates out of total counted is shown. ***p < 0.001 (ANOVA with Tukey's post-hoc analysis). n ≥ 3 trials. See also Figure 10—figure supplement 1. (B) NLK induces the phosphorylation of a 130 amino acid AR N-terminal fragment at S81 in NSC-34 cells. (C–F) Reduced expression of NLK suppresses the toxicity induced by a mutant AR fragment in a Drosophila model of SBMA. Two independent mutant alleles (adk1 and adk2) of nmo showed the same results. Flies were raised at 22°C and genotypes are as follows: (C) GMR-Gal4/+; UAS-EGFP/+, (D) GMR-Gal4/+; UAS-trAR112Q/+, (E) GMR-Gal4/+; UAS-trAR112Q/nmoadk1, (F) GMR-Gal4/+; UAS-trAR112Q/nmoadk2. More than 50 adult flies per genotype were observed at day 2 after eclosion, and five independent experiments were performed.
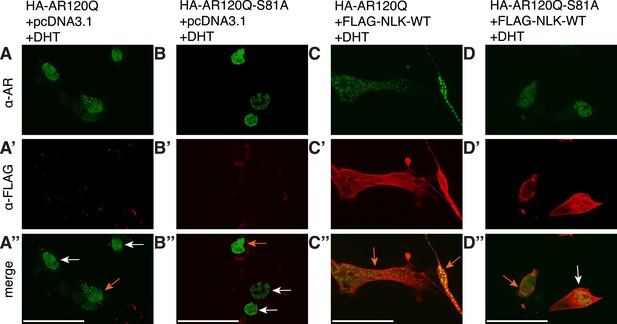
S81 phosphorylation contributes to NLK-mediated effects on AR aggregation.
Representative images of DHT-treated NSC-34 cells expressing HA-tagged AR120Q or AR120Q-S81A in the absence (A, B) and presence (C, D) of wild-type NLK (NLK-WT) co-expression. Cells were subjected to immunofluorescence using anti-AR N-20 (green; A-D) and anti-FLAG (red; A′-D′) antibodies to detect AR aggregation and NLK co-expression, respectively. Merged images are shown in (A′′-D′′). Scale bars are 50 μm. Cells were scored as containing aggregates (orange arrows) or not (white arrows). The ratio of aggregate-containing cells out of total scored is quantified in Figure 10A.
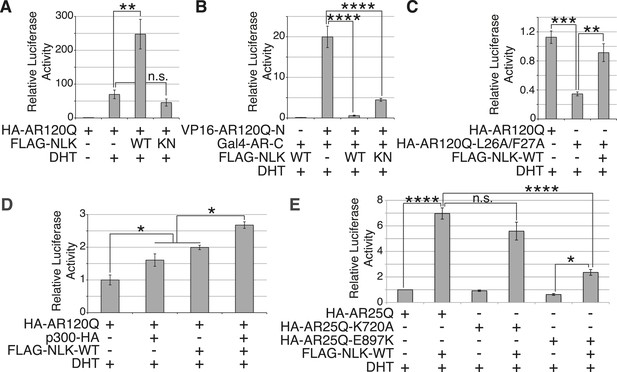
NLK promotes AR-mediated gene transcription by inhibiting the N/C interdomain interaction and promoting AF-2 cofactor binding.
(A) NLK increases AR-dependent gene transcription in a kinase activity-dependent manner in NSC-34 cells. n.s. = not significant, **p < 0.01 (ANOVA with Tukey's post-hoc analysis). n = 3 trials. (B) NLK inhibits the AR N/C interaction as measured by a mammalian two-hybrid assay in NSC-34 cells. ****p < 0.0001 (ANOVA with Tukey's post-hoc analysis). n ≥ 4 trials. See also Figure 11—figure supplement 2. (C) NLK can activate AR-dependent gene transcription in the absence of the N/C interaction in NSC-34 cells. **p < 0.01, ***p < 0.001 (ANOVA with Tukey's post-hoc analysis). n ≥ 3 trials. (D) NLK and p300 synergistically increase AR-mediated gene transcription in NSC-34 cells, suggesting NLK may promote AR-cofactor binding and function. *p < 0.05 (ANOVA with Tukey's post-hoc analysis). n = 5 trials. (E) NLK increases AR-mediated gene transcription via the AR AF-2 domain in NSC-34 cells. n.s. = not significant, *p < 0.05, ****p < 0.0001 (ANOVA with Tukey's post-hoc analysis). n = 4 trials.
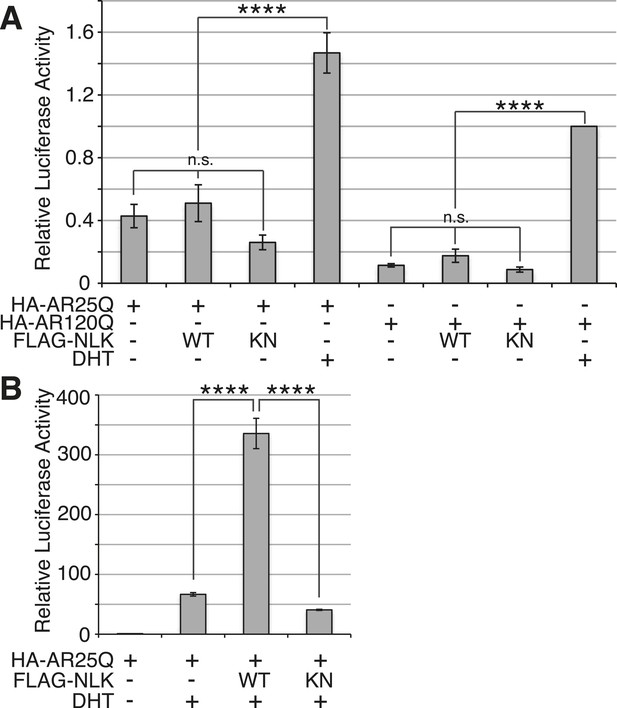
NLK does not induce AR transactivation in the absence of hormone.
(A) NSC-34 cells were transfected as indicated and treated with DHT or vehicle only, and AR transactivation activity was measured by a dual-luciferase assay using an AR-responsive reporter. In the absence of hormone, NLK does not induce the transactivation activity of AR. n.s. = not significant, ****p < 0.0001 (ANOVA with Tukey's post-hoc analysis). n = 4 trials. (B) NLK increases the activity of the wild-type AR25Q in NSC-34 cells in a kinase activity-dependent manner. ****p < 0.0001 (ANOVA with Tukey's post-hoc analysis). n = 3 trials.
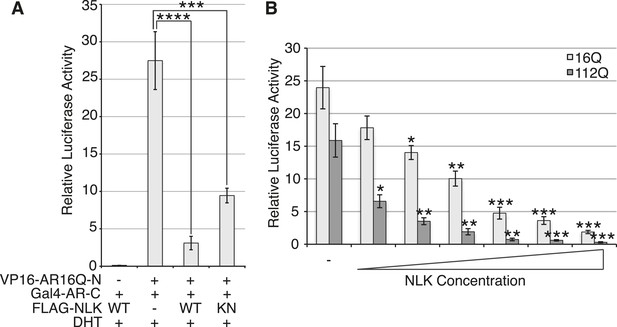
NLK dose-dependently inhibits the AR N/C interaction.
The AR N/C interaction was measured by a mammalian two-hybrid assay in DHT-treated NSC-34 cells. Interaction between the two AR fusion constructs (i.e., N/C interaction) was measured via the activity of a Gal4-dependent luciferase reporter normalized over a renilla luciferase control. (A) NLK inhibits the wild-type AR N/C interaction. This only partially depends upon the kinase activity of NLK. ***p < 0.001, ****p < 0.0001 (ANOVA with Tukey's post-hoc analysis). n ≥ 3 trials. (B) NLK dose dependently inhibits the AR N/C interaction. VP16-AR N-terminal fragments of indicated glutamine (Q) length were co-transfected with Gal4-AR-C and increasing amounts of FLAG-NLK-WT and cells were treated with 10 nM DHT. Even low levels of NLK are able to significantly repress the N/C interaction of polyQ-expanded AR. Asterisks in (B) refer to the comparison of indicated sample with the minus NLK control. *p < 0.05, **p < 0.005, ***p < 0.0005 (t-test). n ≥ 3 trials.
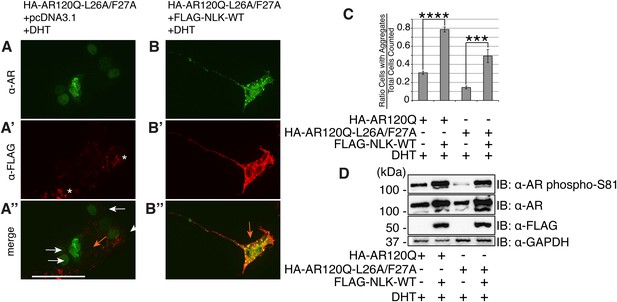
NLK can increase mutant AR aggregation and phosphorylation independent of an N/C interaction.
(A–C) NLK increases the aggregation rate of the N/C interaction-defective mutant AR (HA-AR120Q-L26A/F27A). NSC-34 cells were transfected as indicated and treated with 10 nM DHT. AR aggregation was detected via immunofluorescence using anti-AR N20 (green; A, B). NLK co-expression was detected via immunofluorescence using anti-FLAG (red; A′, B′). Merged images are shown in (A′′, B′′), where an orange arrow indicates a cell with aggregates and white arrow indicates one without. Asterisks mark non-specific staining in the red channel in (A′). Scale bar in (A′′) is 50 μm and applies to all panels. The ratio of cells containing aggregates out of total counted is quantified in (C). ***p < 0.001, ****p < 0.0001 (ANOVA with Tukey's post-hoc analysis). n = 3 trials. (D) NLK increases AR phosphorylation at S81 in the absence of an N/C interaction. Data were consistent over multiple trials, and a representative immunoblot is shown.
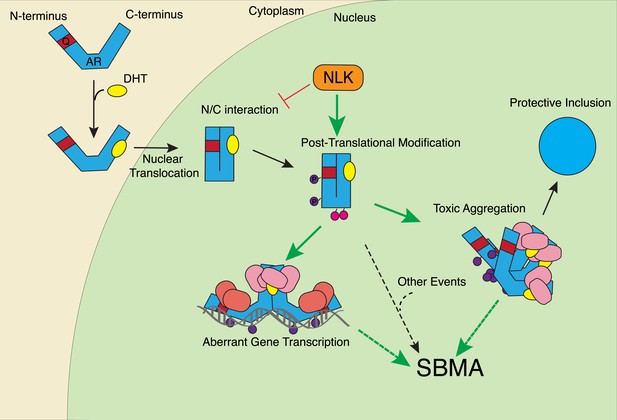
A potential model for the role of NLK in SBMA pathogenesis.
NLK can induce the phosphorylation of the polyQ-expanded AR, which influences its aggregation and contributes to its toxicity in SBMA models. NLK can also regulate the ability of the mutant AR to act as a transcription factor, which would enhance any aberrant AR-mediated gene transcription that contributes to SBMA pathology. A combination of these toxic mechanisms and others could ultimately result in the degeneration and pathology characteristic of SBMA. These events occur downstream of AR ligand binding and nuclear translocation. In addition, NLK may inhibit the AR N/C interaction to promote AR AF-2 cofactor binding.





