Nanoparticulate carbon black in cigarette smoke induces DNA cleavage and Th17-mediated emphysema
Figures
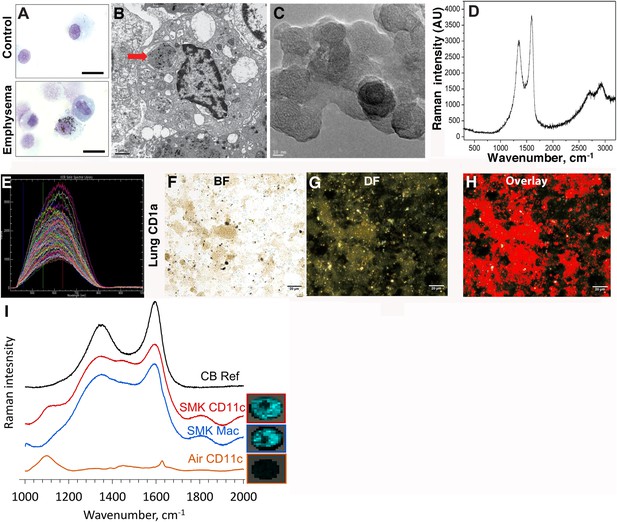
Carbon black (CB) deposition in the lungs of patients with emphysema.
(A) Representative images of lung CD1a+ cells from a smoker with emphysema and a control subject. Scale bar: 10 μm. (B) Lung CD1a+ cells from a patient with emphysema, detected by transmission electron microscopy (TEM). Arrow indicates black substance in the vesicles. Scale bar: 1 μm. (C) Structure of the residual black material from digested human emphysema lung tissue, detected by high-resolution transmission electronic microscopy (HRTEM). Scale bar: 10 nm. (D) Raman spectrum yielded by the black material in the cells. The bifid spectral peaks between 1000 and 2000 cm−1 are the typical Raman signature for CB. Representative hyperspectral image of lung CD1a+ cells from a patient with emphysema (E–H): a reference sample of nanoparticulate carbon black (nCB) was used to generate a signature spectral library (E) using CytoViva Hyperspectral Imaging System. Each colored spectra represents the spectral profile of a distinct area of the nCB sample, which were used in combination to map nCB present in cells. (F) Bright field (BF), (G) dark field (DF), and (H) overlay CB signature spectrum of lung CD1a+ cells. Positive signals were pseudo-colored red to aid visualization. Scale bar: 20 μm. (I) Raman spectrum yielded in lung CD11c+ and macrophages isolated from lungs of mice exposed to smoke for 4 months; CB reference (CB Ref) signal indicates solid CB sample. SMK: 4 months of cigarette smoke. Inset images for cell type correspond to Raman spectra indicating the subcellular localization of CB. The brightness of each 2 µm × 2 µm pixel, representing one spectrum, indicates the height of the graphitic band of CB at 1600 cm−1 compared to the background, such that brighter pixels indicate more CB.
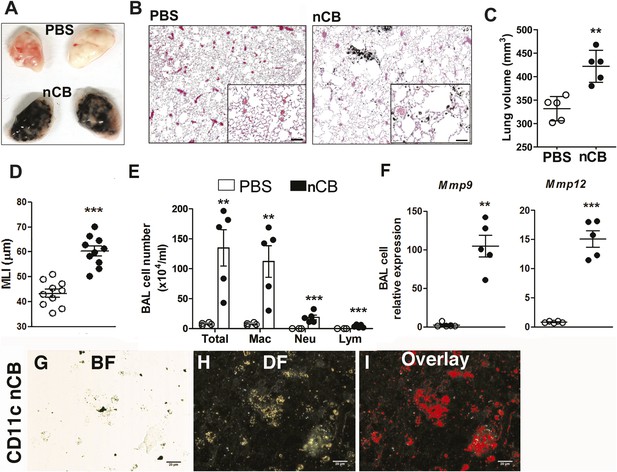
Carbon black-induced emphysema mouse model.
(A) Representative image of fresh lungs harvested from mice exposed to vehicle (PBS) or nanoparticulate carbon black (nCB) as described in Figure 2—figure supplement 1. (B) Representative Hematoxylin and eosin (H&E) staining of formalin-fixed lung sections. Scale bar: 100 μm. (C) Micro-CT quantification of lung volume. (D) MLI measurement was done on the same groups of mice. (E) Total and differential cell count in bronchoalveolar (BAL) fluid: macrophages (Mac), neutrophils (Neu), and lymphocytes (Lym). Quantitative PCR of Mmp9 and Mmp12 (F) gene expression in BAL cells isolated from PBS- or CB-challenged mice. Representative lung CD11c+ cells isolated from mice challenged with nCB under bright field (BF) (G), dark field (H), and overlap images (pseudo-red area) (I) signifying nCB signature spectrum. Scale bar: 20 μm. Data are mean ± SEM and representative of three independent experiments; ***p < 0.001, **p < 0.01 as determined by the Student's t-test; n = 5 per group.
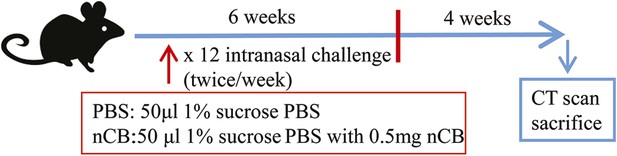
Schematic representation of nCB-induced lung inflammation and emphysema protocol.
Mice were lightly anesthetized with isoflurane and challenged with 50 µl of 107 ng/ml of CB or vehicle (PBS with 1% sucrose) twice weekly for 6 weeks; 4 weeks following the last challenge, mice underwent CT scan of chest and were euthanized.
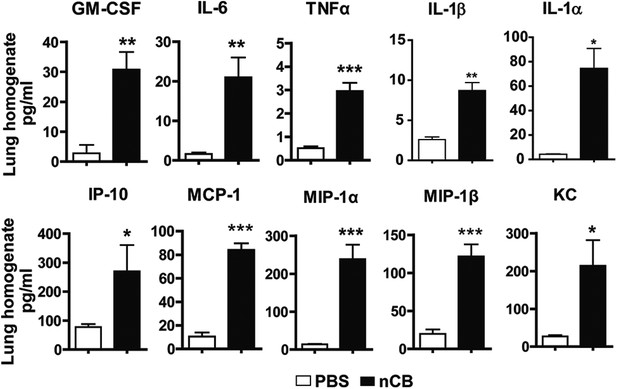
nCB induces pro-inflammatory cytokines and chemokines in the lung.
Concentration of pro-inflammatory cytokines and chemokines detected via MILLIPLEX Assay (Millipore, Billerica, MA) in the lung homogenate collected from mice in each group. n = 5 per group. ***p < 0.001, **p < 0.01, *p < 0.05 as determined by Student's t-test and data are mean ± SEM and representative of three independent experiments.
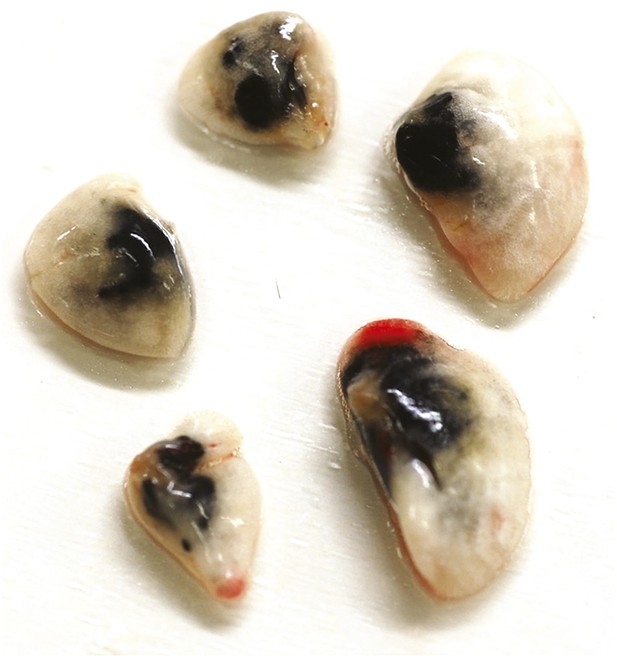
nCB persists in the lungs 18 months after the last challenge.
Representative image of left and right lobes of the lungs harvested from mice 18 months after the last nCB challenge.
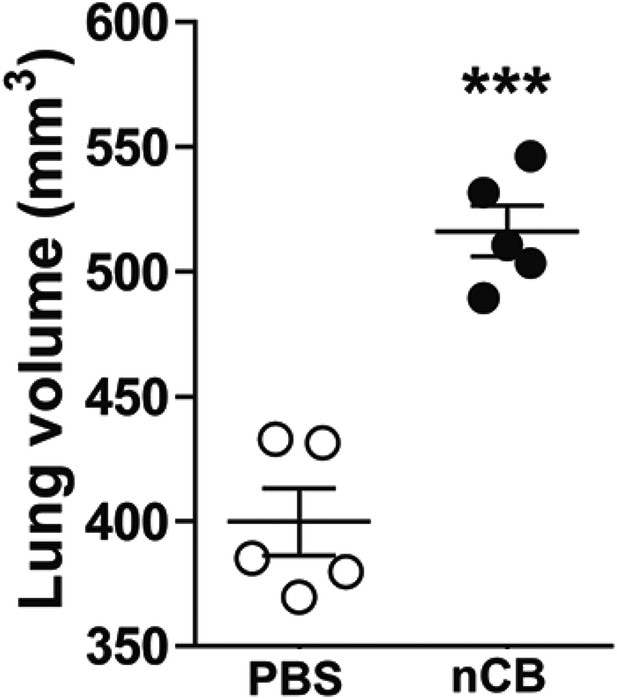
nCB-induced emphysema persists in the lungs.
Micro-CT quantification of lung volume in mice rested for 7 month after the last nCB or PBS challenge. ***p < 0.001 as determined by the Student's t-test, and data are mean ± SEM.
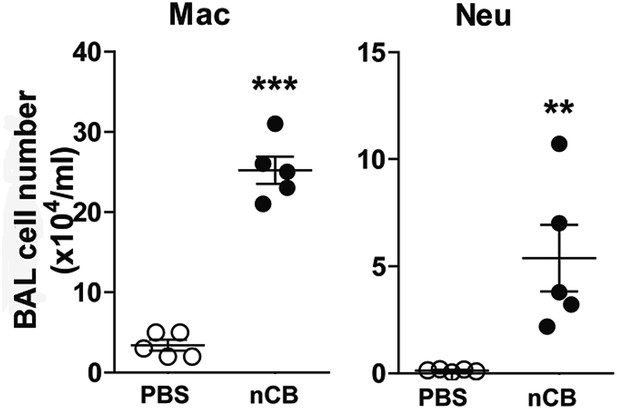
nCB-induced immune cell infiltration persists in the lungs.
BAL fluid analysis of the mice rested for 7 month after the last nCB or PBS challenge showing the cell number of macrophages (Mac) and neutrophils (Neu). ***p < 0.001, **p < 0.01 as determined by the Student's t-test, and data are mean ± SEM.
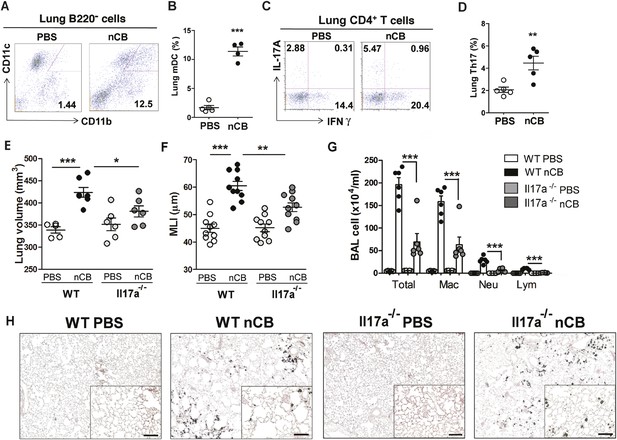
nCB promotes Th17 responses.
Representative staining (A) and cumulative analysis (B) of the percentage of CD11c+CD11bhigh cells in lung B220− cell subset. Representative intracellular staining (C) and cumulative analysis (D) of IL-17A+ cells expressing lung CD4+ T cell (Th17) subset. (E) Micro-CT quantification of lung volume in WT and Il-17a−/− mice. (F) Lung MLI was determined in the same group of mice. (G) BAL fluid analysis of the indicated groups of mice showing the total cells including macrophages (Mac), neutrophils (Neu), and lymphocytes (Lym). ***p < 0.001, **p < 0.01, *p < 0.05 as determined by the one-way ANOVA and Bonferroni's multiple comparison test. N = 4 to 6 per group. Data are mean ± SEM. (H) Representative H&E staining of formalin-fixed, 5-μm lung sections in indicated groups of mice. Scale bar: 100 μm.
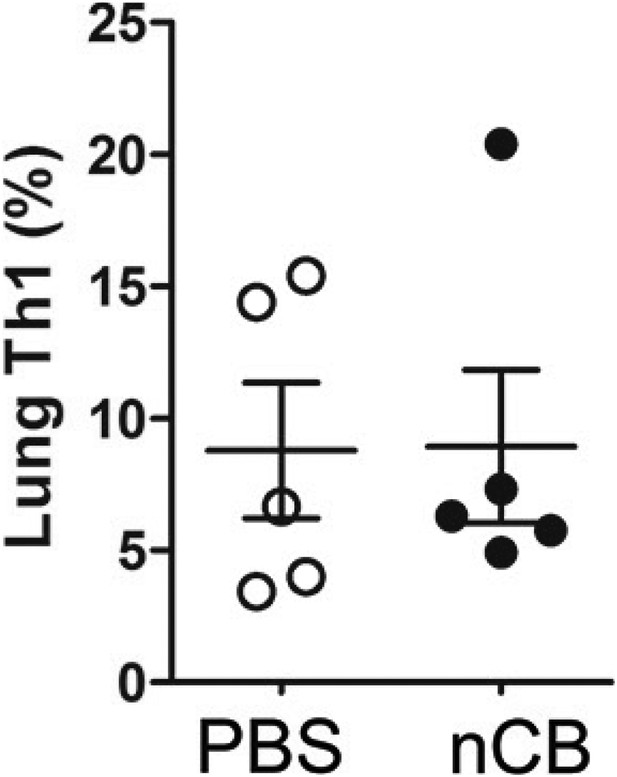
nCB did not induce Th1 responses.
Cumulative analysis of intracellular cytokine staining of IFNγ in lung CD4+ T cell subsets in PBS or nCB-challenged mice. Data are mean ± SEM and representative of three independent experiments.
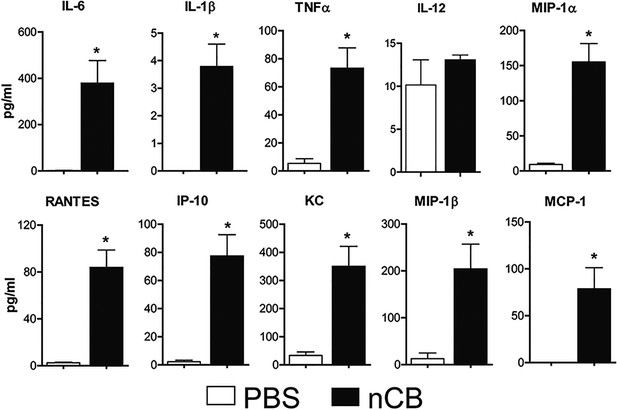
Lung APCs of nCB-challenged mice secrete Th17 cell-specific pro-inflammatory cytokines and chemokines.
Concentration of pro-inflammatory cytokines and chemokines detected by Multiplex system in the supernatant of overnight cultured of lung CD11c+ cells isolated from indicated groups.*p < 0.05 as determined by the Student's t-test. n = 3 per group. Data are mean ± SEM and representative of three independent experiments.
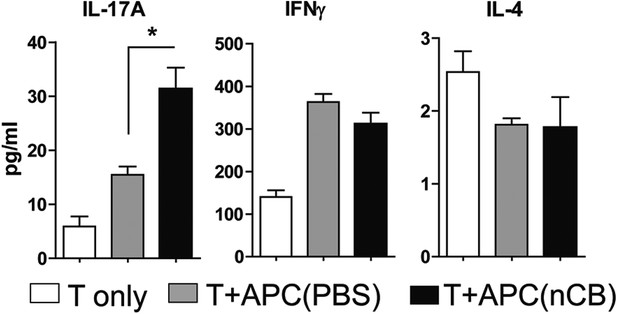
Lung APCs of nCB-challenged mice-induced Th17 responses.
Concentration of IL-17A, IFN-γ, and IL-4 expressed in the supernatant of lung CD11c+ cells isolated from CB- or PBS-challenged mice and co-cultured with splenic CD4+ T cells in the presence of anti-CD3 (1 μg/ml). *p < 0.05 as determined by the Student's t-test. n = 5 per group. Data are mean ± SEM and representative of three independent experiments.
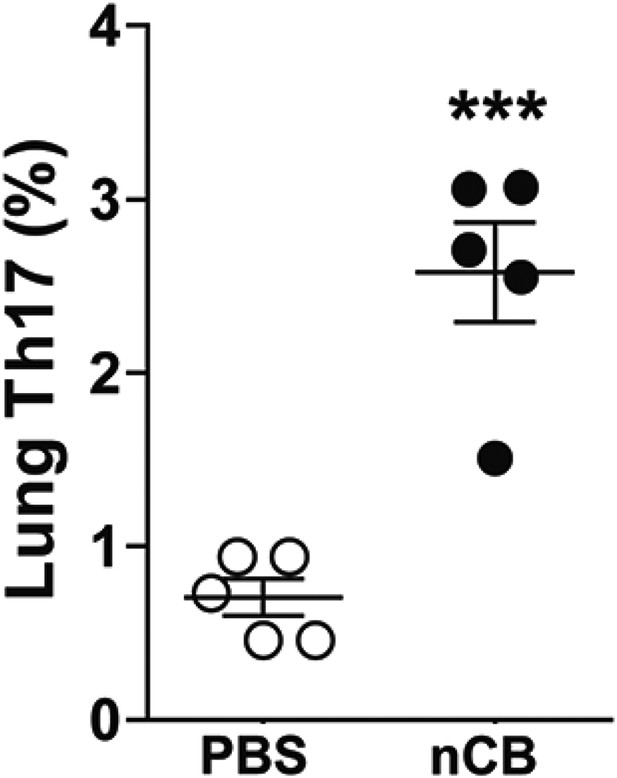
nCB-induced Th17 responses persist in the lungs.
Cumulative analysis of intracellular cytokine staining of IL-17A in lung CD4+ T cell subsets in the mice rested for 7 month after the last nCB or PBS. ***p < 0.001 as determined by the Student's t-test, and data are mean ± SEM.
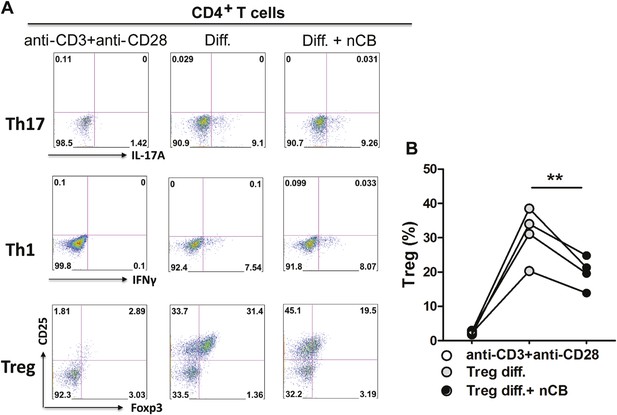
Direct effect of nCB on T helper cell differentiation in vitro.
(A) Flow cytometric analysis of intracellular cytokine staining of IFN-γ (Th1), IL-17A (Th17), and Foxp3/CD25 surface expression (Tregs). Diff. is the differentiation conditions for Th1, Th17, and Tregs (as described in the methods). Y-axis of both Th1 and Th17 panel is empty channel. (B) Cumulative summary of four independent experiments for Treg differentiation. **p < 0.01 as determined by the one-way ANOVA and Bonferroni's multiple comparison test. Data are mean ± SEM.
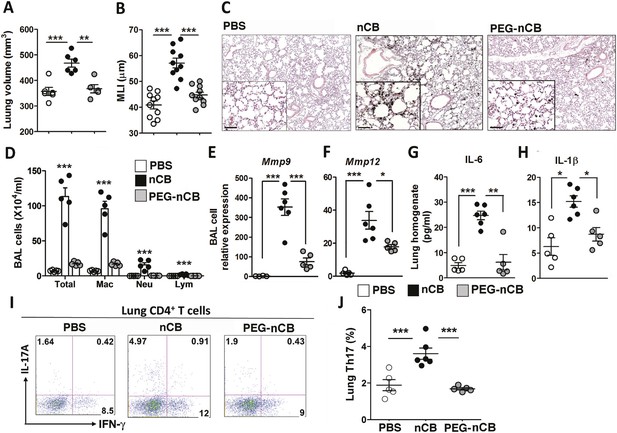
Hydrophobicity of nCB is important for its pathogenesis.
Micro-CT quantification of lung volume (A) and MLI measurement of lung morphometry (B) in vehicle (PBS), nCB, and PEG-nCB treated mice. (C) Representative H&E staining of lung sections Scale bar: 100 μm. (D) Total and differential cell count in bronchoalveolar (BAL) fluid; macrophages (Mac), neutrophils (Neu), and lymphocytes (Lym). Quantitative PCR of Mmp9 (E) and Mmp12 (F) gene expression in BAL cells isolated from the above group of mice. Lung homogenate collected from indicated groups of mice were measured for IL-6 (G) and IL-1β (H) by ELISA. Representative intracellular staining (I) or cumulative analysis (J) of Th17 cells in the lungs. ***p < 0.001, **p < 0.01, *p < 0.05 as determined by the one-way ANOVA and Bonferroni's multiple comparison test. n = 4 to 6 per group, and data are mean ± SEM and representative of two independent studies.
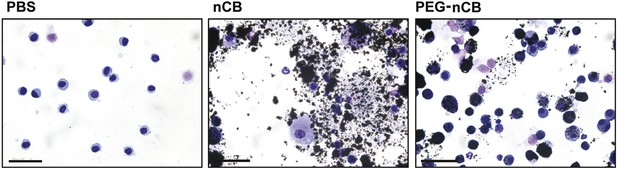
nCB-induced cell damage compared with PEG-nCB.
Representative image of H&E stained cytospin preparation of BAL cells isolated from indicated groups of mice. Scale bar: 50 μm. Data are representative of two independent studies.
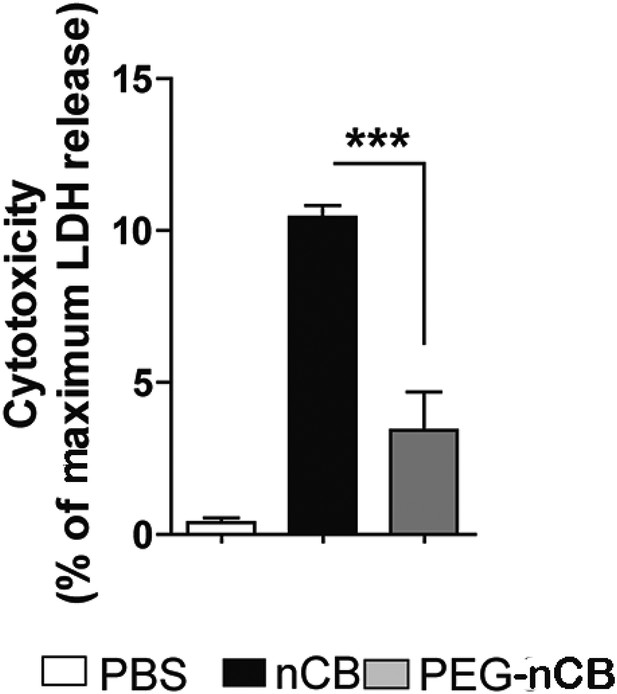
nCB-induced cell death compared with PEG-nCB.
Lactate dehydrogenase (LDH) release from RAW 264.7 cells after 24 hr of the indicated treatment. Maximum LDH release was the amount of LDH released from lysed cells. ***p < 0.001 as determined by the one-way ANOVA and Bonferroni's multiple comparison test. n = 5 per group. Data are mean ± SEM and representative of two independent studies.
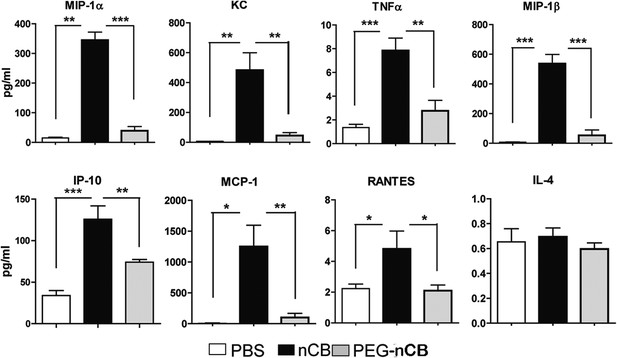
nCB-induced strong lung inflammation compared with PEG-nCB.
Multiplex analysis of pro-inflammatory cytokines and chemokines in lung homogenate of indicated groups. ***p < 0.001, **p < 0.01, *p < 0.1 as determined by the one-way ANOVA and Bonferroni's multiple comparison test. n = 5 per group. Data are mean ± SEM and representative of two independent studies.
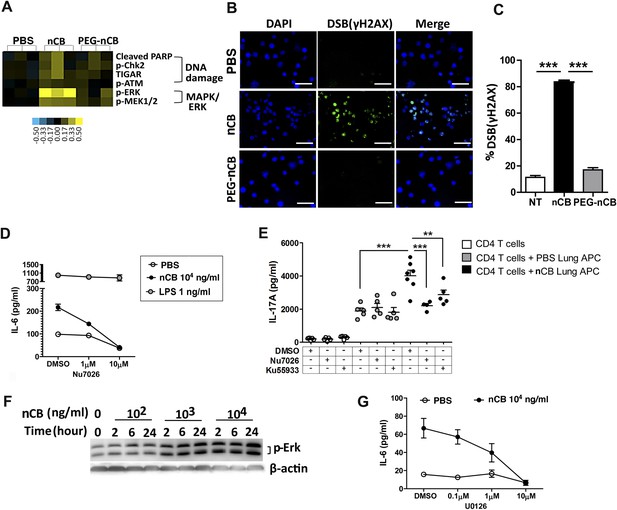
nCB activates APCs by the induction of DNA damage and Erk signaling.
(A) Heat map (reverse phase protein array) of protein expression and phosphorylation level in RAW 264.7 cells stimulated with vehicle (PBS), nCB (105 ng/ml), and PEG-CB (105 ng/ml). p: phosphorylated. Blue is relatively low (−0.5) and yellow high (0.5) based on log2 ratio of the value for expression level. (B) RAW 264.7 cells under indicated conditions immunostained for nuclear DNA (DAPI, blue) and anti-γH2AX (green) to detect double strand break (DSB). Scale bar: 50 μm. (C) Quantitative summary of panel B indicating the percentage γH2AX positive RAW cells in indicated groups. (D) IL-6 concentration detected by ELISA after 48 hr in the supernatant of MDDC treated with CB or LPS in the presence of increasing dose of Nu7026 or vehicle (DMSO). (E) IL-17A concentration detected by ELISA after 72 hr co-culture of splenic CD4 T cells and lung CD11c+ cell isolated from the mice after challenged with PBS or nCB and anti-CD3 (1 μg/ml) in the presence of Nu7026 (100 nM), Ku55933 (100 nM), or vehicle control (DMSO). (F) Western blot of protein extracted from BMDC treated with different concentration of nCB targeting phosphorylated-Erk. Data are representative of two independent experiments. (G) IL-6 concentration detected by ELISA in the supernatant of MDDC treated with nCB in the presence of increasing dose of U0126 (MEK1/2 inhibitor) for 48 hr. n = 4 to 7 per group and data are mean ± SEM and representative of two independent experiments (C, D, E, G). ***p < 0.001, **p < 0.01 as determined by the one-way ANOVA and Bonferroni's multiple comparison test.

Heat map depicting molecules whose expression and phosphorylation level differed when RAW 264.7 cells were treated with nCB compared with PBS or PEG-nCB treated groups detected by reverse phase protein array.
https://doi.org/10.7554/eLife.09623.021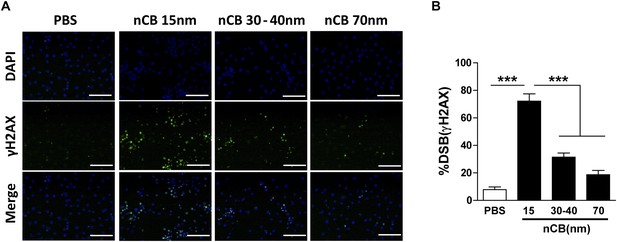
Larger nCB size correlates with weak induction of DNA double strand breaks (DSB).
(A) RAW 264.7 cells were treated with vehicle (PBS), nCB (105 ng/ml) with different size in diameter for overnight and immunostained for nuclear (DAPI) (blue) and anti-γH2AX (green) to detect double strand break (DSB). Scale bar: 100 μm. (B) Quantitative summary of panel A indicating the percentage γH2AX-positive RAW cells in indicated groups. ***p < 0.001 as determined by the one-way ANOVA and Bonferroni's multiple comparison test. n = 3 fields per group. Data are mean ± SEM.
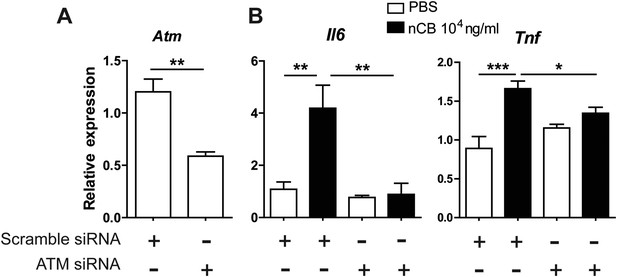
ATM is required for nCB-induced inflammatory factor upregulation in RAW cells.
(A) Atm mRNA expression in RAW 264.7 cells transfected with scramble or Atm siRNA. (B) Il6 and Tnf-α mRNA expression in RAW cells transfected with scramble or Atm siRNA followed by overnight treatment with nCB or vehicle (PBS). ***p < 0.001, **p < 0.01, *p < 0.1 as determined by One-way ANOVA and Bonferroni's multiple comparison test. n = 4 per group. Data are mean ± SEM and representative of two independent studies.
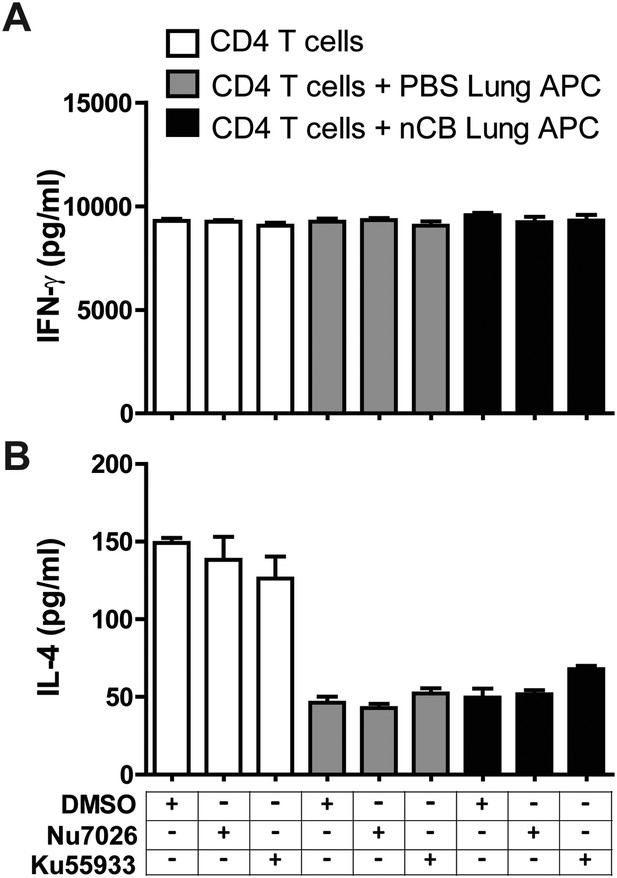
Inhibition of DNA damage does not affect Th1 or Th2 responses.
IFN-γ (A) and IL-4 (B) concentrations were measured using ELISA in the supernatant of anti-CD3 (1 μg/ml) treated CD4+ T cells co-cultured with lung CD11c+ cell isolated from the mice challenged with PBS or nCB in the presence of vehicle (DMSO) or inhibitors of DNA damage (Nu7026 or Ku55933 at 100 nM). n = 5 per group, and data are mean ± SEM.
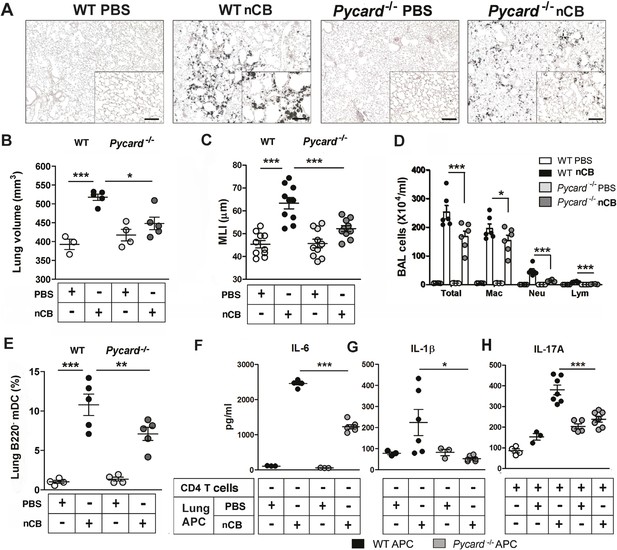
ASC-mediated inflammasome pathway is required for nCB-induced Th17 responses and emphysema.
(A) Representative H&E staining of lung sections from WT and Pycard−/− mice exposed to nCB or vehicle (PBS) as described in Figure 2—figure supplement 1. Scale bar: 100 μm. (B) Micro-CT quantification of lung volume in indicated groups of mice. (C) Lung MLI measurement in the same group of mice. (D) Total and differential cell count in bronchoalveolar (BAL) fluid: macrophages (Mac), neutrophils (Neu), and lymphocytes (Lym). (E) Relative abundance of lung mDCs (CD11c+CD11bhigh) isolated from whole lung tissue in the same group of mice. IL-6 (F) and IL-1β (G) concentrations detected by ELISA in the supernatant of lung CD11c+ cells isolated from indicated group of mice after overnight culture. (H) IL-17A concentration detected by ELISA in the supernatant of splenic CD4+ T cells co-cultured with lung CD11c+ cells isolated from indicated group of mice for 3 days in the presence of anti-CD3 (1 μg/ml). ***p < 0.001, **p < 0.01, *p < 0.05 as determined by the one-way ANOVA and Bonferroni's multiple comparison test; n = 3 to 7 per group, and data are mean ± SEM and representative of two independent studies.
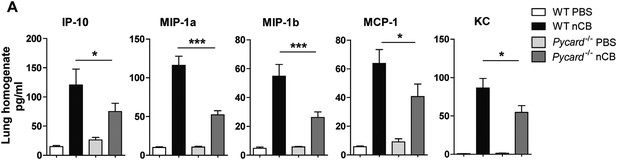
Pycard−/− mice produce less pro-inflammatory chemokines in the lungs in response to nCB challenge.
(A) Multiplex detection of indicated chemokines in freshly harvested lung homogenate from different groups of mice. ***p < 0.001, *p < 0.05 as determined by the one-way ANOVA and Bonferroni's multiple comparison test. n = 5 per group. Data are mean ± SEM and representative of two independent studies.
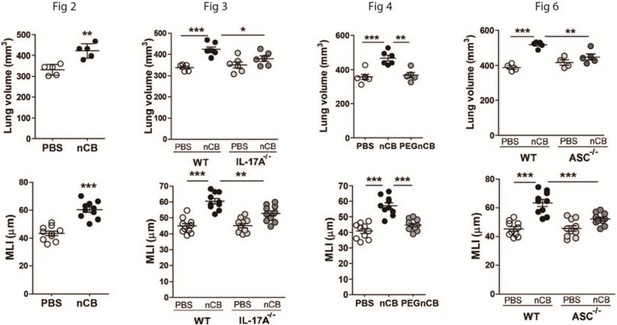
Lung volume and MLI figures without zero suppression.
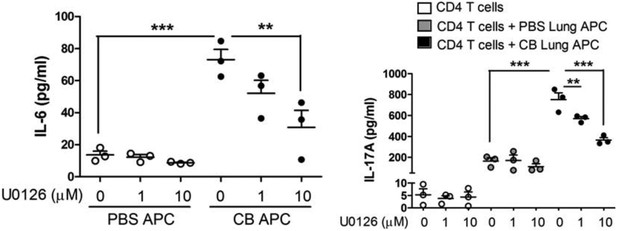
Inhibition of Erk phosphorylation reduces IL-17A and IL-6.
CD11c+ lung cells were isolated from lungs of mice exposed to PBS or nCB as described in the manuscript and treated with increasing concentration of U0126 for two days (left panel). Using the same conditions cells were co-cultured with congenic splenic T cells (1:10 ratio) and antiCD3 (1μg/ml) in the presence of increasing concentration of U0126 for three days (right panel). Concentration of IL-17A, and IL-6 were measured using ELISA. ** p<0.01, *** p<0.001 as determined by the one-way ANOVA and Bonferroni’s Multiple Comparison test.
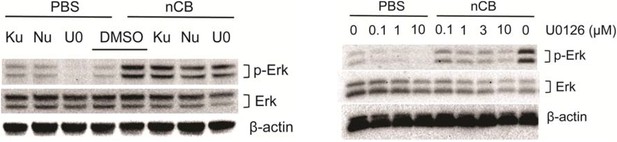
Nu7026 and U0126 inhibit Erk phosphorylation.
BMDCs were treated with PBS or nCB in the presence of different inhibitors as indicated (Ku: Ku55933, 3μM; Nu: Nu7026, 3μM; U0: U0126, 0.1μM) for 24 hours and phosphorylated Erk (p-Erk), total Erk and β-actin were measured using western blot of cell lysates (left panel). Inhibition of p-Erk was examined using the same system in the presence of increasing concentration of U0126 (right panel).





