Cell type-specific transcriptomics of hypothalamic energy-sensing neuron responses to weight-loss
Figures
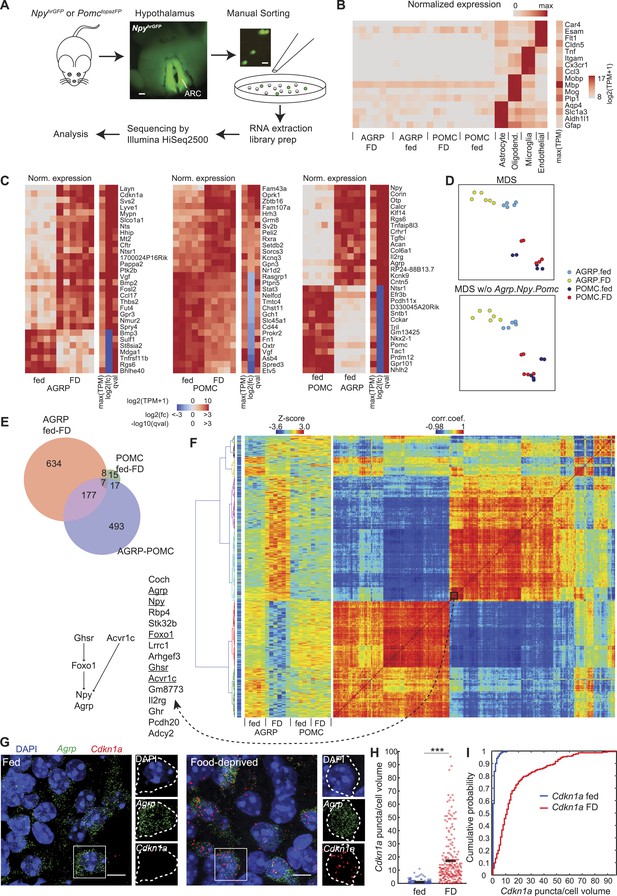
Cell type-specific transcriptomic profiling of starvation-sensitive neurons.
(A) Schema for dissection and sorting of fluorescent neurons from the hypothalamic arcuate nucleus (ARC) followed by cell type-specific RNA-Seq. Scale: ∼200 µm (left), ∼20 µm (right). (B) Expression levels of marker genes for astrocytes, myelin oligodendrocytes, microglia, and endothelial cells indicate high purity of AGRP and POMC samples. FD: 24-hr food-deprived. Left, for each sample, log expression levels for a single gene in each row normalized by maximum expression level of the transcript in any of the samples. Right, sidebar shows maximum expression level for each row (each transcript) across all samples. TPM: transcripts per million. (C) Top 30 differentially expressed genes (DEG) for: AGRP neurons, FD/fed; POMC neurons, FD/fed; AGRP/POMC neurons (fed). FD: 24-hr FD. As in (B), each row corresponds to a transcript where the expression level in each sample is normalized by maximum expressions on level in the row. The sidebars show maximum expression level [max(TPM)] across samples in each row (each transcript). In addition, log2(fold-change) [log2(fc)], and q-value (qval) for the differential expression across fasted/fed states (left and middle) or AGRP/POMC expression levels are shown. (D) Top, multidimensional scaling (MDS) projection of distance (1-corr.coef.) between samples. Bottom, MDS without Agrp, Npy and Pomc genes in the calculation. (E) Venn diagram for DEG between FD and fed conditions (AGRP FD-fed: red, POMC FD-fed: green) and between AGRP and POMC neurons (both fed, purple). Reported DEG required q-value <0.05, abs[log2(fc)] > 1 and mean CPM > 20 in at least 1 cell type/condition (see ‘Materials and methods’). (F) Hierarchical clustering of DEG. Matrix in the middle indicates standardized expression level for the samples (columns) and DEG (rows). The matrix on the right shows the correlation coefficients between genes, calculated across all samples. The colormap on the left indicates maximum TPM expression level in log2 scale, which ranges from 2.96 (blue) to 15.6 (red). Left, genes in a cluster that comprise known pathways that regulate Agrp and Npy expression. (G) Representative images of double single molecule fluorescence in situ hybridization (smFISH) for Agrp and Cdkn1a. Scale, 10 μm. (H, I) Population counts (bars: mean value) (H) and cumulative probability distributions (I) of Cdkn1a puncta per cell volume in AGRP neurons (p = 1.5e−53, Kolmogorov–Smirnov [ks]-test). ***p < 0.001. Fed, n = 189 cells; FD, n = 215 cells; 3 mice per condition.
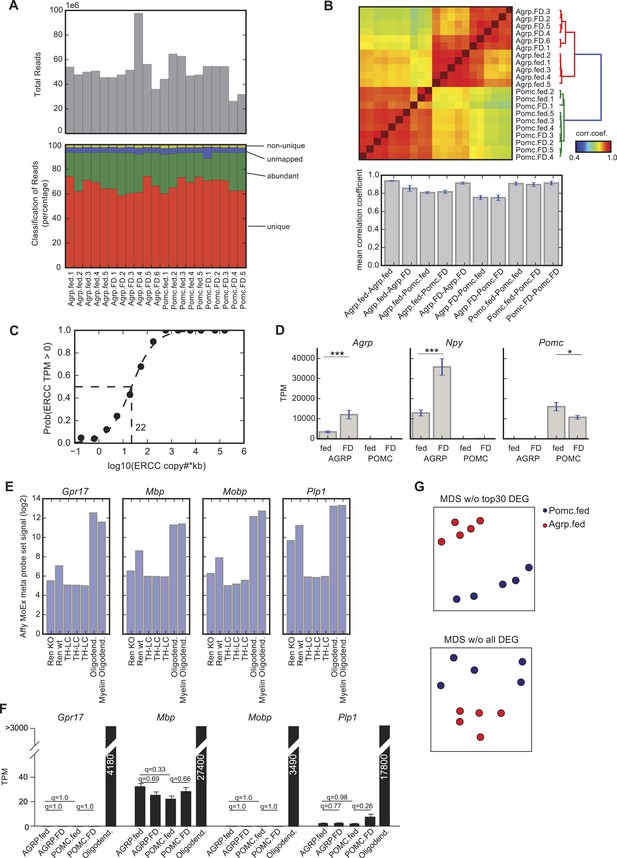
Comparison of AGRP and POMC neuron transcriptomic samples.
(A) Sequencing statistics. Upper panel shows the total reads for each sample. Lower panel shows classification of reads into uniquely mapped to the genome (unique), mapped to ribosomal RNA, mitochondrial genome, phiX or low complexity sequences (abundant), unmapped to neither of the above (unmapped), or mapped to the genome but to multiple locations (non-unique). (B) Upper panel shows the correlation coefficients calculated using robustly expressed genes (some group has average TPM > 20) and lower panel shows the average and standard deviation of correlation coefficients within group and between groups. (C) Sensitivity of the assay was calculated from ERCC spike-in data. 50–50% detection probability is obtained at 22 copies × kb transcripts. (D) Gene expression of Agrp, Npy and Pomc in AGRP and POMC neurons under fed and FD conditions. Data is mean ± s.e.m. ***q < 0.005, *q < 0.05. (E) Evidence for oligodendrocyte contamination in one of the fluorescence activated cell sorted AGRP neurons samples (Ren-wt) from Ren et al. (2012), which is compared to AGRP neuron-specific Foxo1 knockout (Ren-KO), as well as three samples of manually sorted Th-expressing neurons from the locus coeruleus (as an example of low contamination samples), and purified samples of oligodendrocytes or myelin oligodendrocytes (Cahoy et al., 2008). Gpr17, Mbp, Mobp, and Plp1 are all highly expressed in oligodendrocytes and are also found differentially expressed in the wildtype AGRP neuron sample from microarray data from Ren et al. (2012). Data from all seven samples were generated on the same Affymetrix mouse exon array platforms and processed together. Y-axis is standardized probe set signal from the arrays on a log2 scale. (F) The AGRP and POMC neuron samples generated in our study and processed by RNA-Seq are compared for oligodendrocyte markers and to RNA-Seq data from purified oligodendrocyte samples (Zhang et al., 2014a). There were no statistically significant differences in the oligodendrocyte in the groups that are compared in this study. (G) MDS projection of distance (1-corr.coef.) between AGRP and POMC neurons where correlation coefficients were calculated without the top 30 DEG (top) or with all DEG (based on criteria in ‘Materials and methods’) removed (bottom).
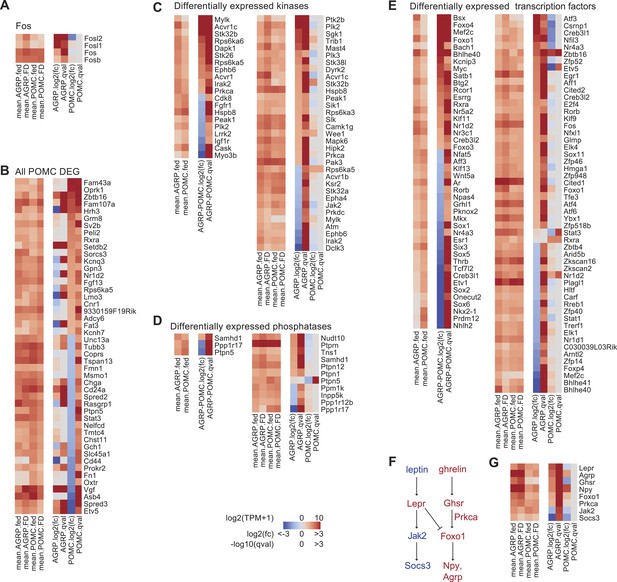
DEG in AGRP and POMC neurons with food deprivation.
(A) Gene expression of Fos and related genes. Left, mean expression level [log2(TPM)] of each transcript for each experimental group. Right, log2(fold-change) and q-values for differential expression between FD and fed states separately for AGRP and POMC neurons. (B) All DEG during food deprivation from POMC neurons. (C) Differentially expressed kinase genes between AGRP and POMC neurons (left) or AGRP or POMC neurons FD/fed (right). Differentially expressed kinase genes after food deprivation were over-represented in samples from AGRP but not in POMC neurons during food deprivation (AGRP: p = 0.0026, POMC: p = 0.66; hypergeometric test). (D) Differentially expressed phosphatase genes between AGRP and POMC neurons from fed mice (left) or separately from AGRP or POMC neurons comparing FD/fed groups (right). (E) Differentially expressed transcription factor genes between AGRP and POMC neurons from fed mice (left) or separately from AGRP or POMC neurons comparing FD/fed groups (right). Differentially expressed transcription factor genes were over-represented in samples from AGRP and POMC neurons during food deprivation (AGRP: p = 3.1e−5, POMC: p = 0.02; hypergeometric test). (F) Canonical signaling pathways in AGRP and POMC neurons for leptin and ghrelin signaling (red: upregulated, blue: downregulated in RNA-Seq dataset). (G) Expression changes for transcripts associated with leptin and ghrelin signaling pathways.
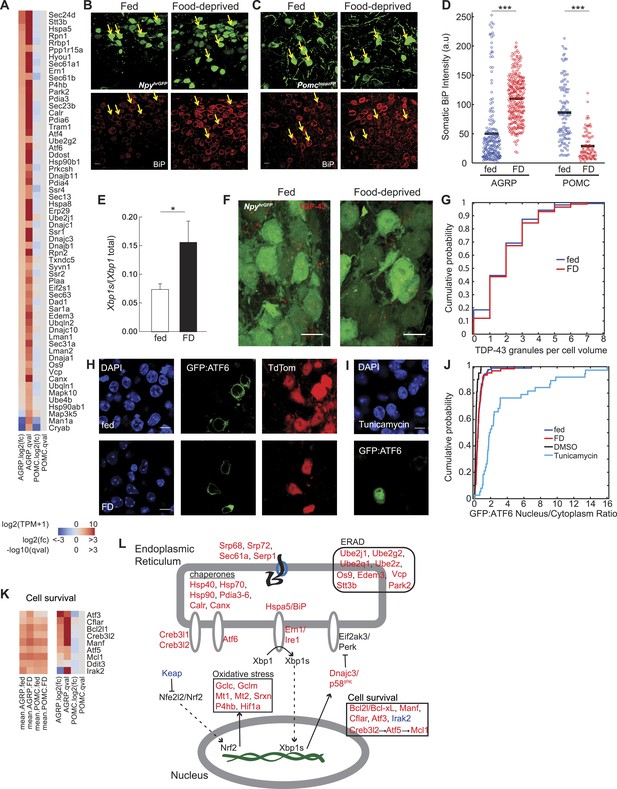
Food deprivation induces unfolded protein response in AGRP neurons.
(A) Log2(fold-change) [log2(fc)] and q-values for genes associated with endoplasmic reticulum (ER) localization (KEGG pathway: mmu04141) that are affected by food deprivation in AGRP (left columns) or POMC (right columns) neurons. (B, C) Representative images showing BiP-immunofluorescence from NpyhrGFP or PomctopazFP mice. Arrows: examples of fluorescently labeled (B) AGRP and (C) POMC neurons used for BiP quantification. Scale: 10 μm. (D) Population counts of BiP somatic intensity in AGRP or POMC neurons. AGRP.fed, n = 209; AGRP.FD, n = 283; POMC.fed, n = 121; POMC.FD, n = 92; 3 mice per condition. Bars: mean values. Rank-sum test. ***p < 0.001. (E) Fraction of spliced to total Xbp1 transcript isoforms in AGRP neurons. Unpaired one-tailed t-test. *p < 0.05. (F, G) Representative images (F) and cumulative probability distribution (G) of TDP43-immunoreactive-granules (red) in GFP-expressing AGRP neurons (p = 0.76, ks-test). Fed, n = 276; FD, n = 174; 2 mice per condition. Scale, 10 μm. (H–J) Representative images (H, I) of GFP:ATF6 expression AgrpCre;ai9(tdtomato) mice. Cumulative probability distribution (J) of the ratio of nuclear to cytoplasmic GFP fluorescence in AGRP neurons: fed vs FD (p = 0.44, ks-test) Fed, n = 92; FD, n = 63; 4 mice per condition; or AGRP neurons: DMSO vs tunicamycin (p = 2.9e−16, ks-test) DMSO, n = 42; Tunicamycin, n = 38; 1 mouse per condition. (K) Differentially expressed cell survival genes. Left, mean expression level [log2(TPM)] of each transcript of each experimental group. Right, log2(fold-change) and q-values for differential expression between FD and fed states separately for AGRP and POMC neurons. (L) Schematic for DEG in ER stress-associated pathways in AGRP neurons after food-deprivation. Red: upregulated expression, Blue: downregulated expression.
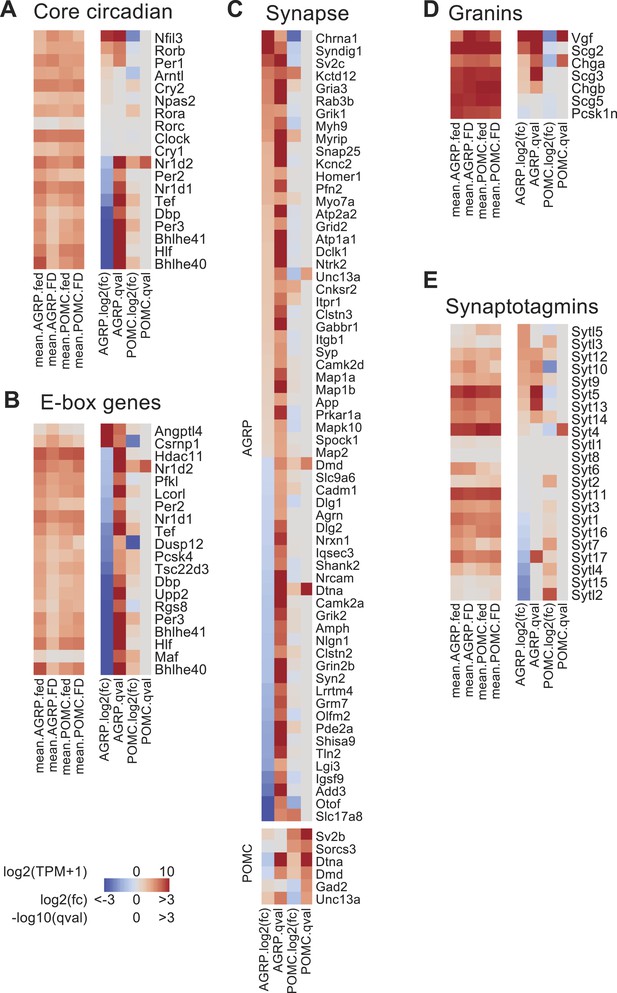
Changes in the expression of circadian and synapse-associated genes after food-deprivation.
(A) Gene expression changes for core circadian genes. Left, mean expression level [log2(TPM)] of each transcript of each experimental group. Right, log2(fold-change) and q-values for differential expression between FD and fed states separately for AGRP and POMC neurons. (B) E-box genes differentially expressed during food-deprivation. (C) DEG with synapse-localized functions (Gene Ontology: 0045202). Top, AGRP neurons: FD vs fed; bottom, POMC neurons FD vs fed. (D, E) Gene expression changes for granin genes (D) and synaptotagmins (E).
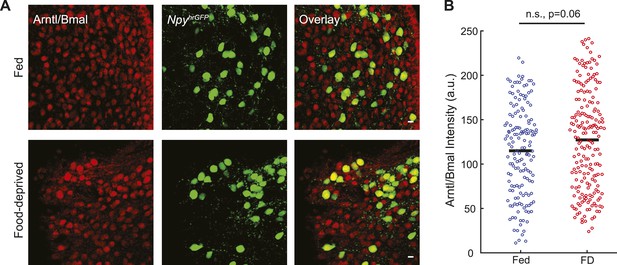
Arntl/Bmal expression in AGRP neurons.
(A) Representative images for Arntl/Bmal-immunofluorescence in AGRP neurons from fed and FD mice. Scale, 10 µm. (B) Arntl/Bmal immunofluoresence intensity (bars, mean values) in AGRP neurons from fed and FD mice. Fed, n = 159; FD, n = 196; 3 mice per condition.
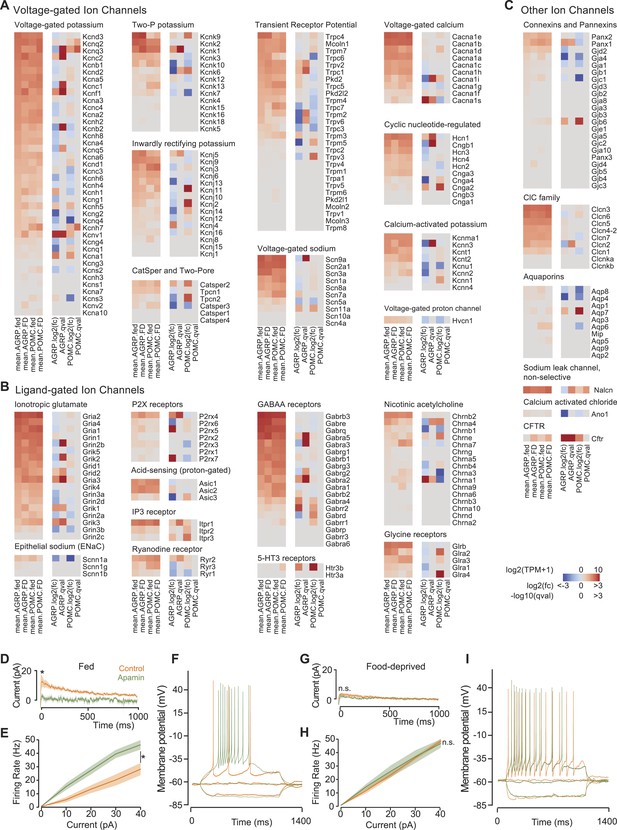
Ion channel gene expression in AGRP and POMC neurons.
(A–C) Gene expression for voltage-gated ion channels (A), ligand-gated ion channels (B), and other ion channels (C). For each colormap, left, mean expression level [log2(TPM)] of each transcript of each experimental group. Right, log2(fold-change) and q-values for differential expression between FD and fed states separately for AGRP and POMC neurons. (D) Tail currents elicited in AGRP neurons from fed mice by a voltage step from −30 mV to −60 mV in the absence (n = 5) and presence of apamin (n = 5). Unpaired t-test. *p < 0.05. Lines show mean, shading shows s.e.m. (E) Firing rate from current injection to AGRP neurons from fed mice in the absence (n = 8) and presence (n = 13) of apamin. Unpaired t-test. (F) Example of action potential firing in AGRP neurons from fed mice in response to −10, 0, +10 pA in the absence and presence of apamin. (G) Tail currents in AGRP neurons from FD mice (−apamin, n = 10; +apamin, n = 5). Unpaired t-test. n.s., p > 0.05. (H) Firing rate from AGRP neurons from FD mice (−apamin, n = 4; +apamin, n = 4). Unpaired t-test. (I) Example of action potential firing in AGRP neurons from FD mice (current injections: −10, 0, +10 pA).
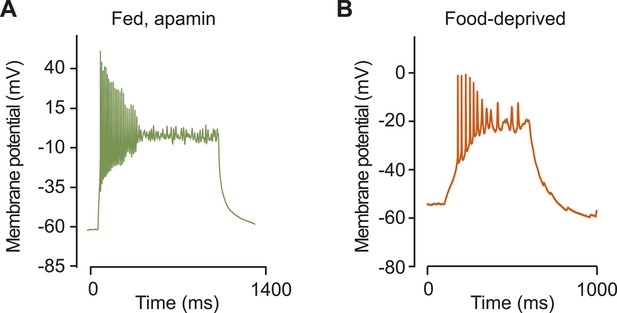
Burst firing in AGRP neurons with apamin or food deprivation.
(A) Burst firing and plateau potential from current injection (+45 pA) in AGRP neurons from a well-fed mouse after apamin block of SK channels. (B) Burst firing and plateau potential from current injection (+10 pA) in an AGRP neuron from a FD mouse.
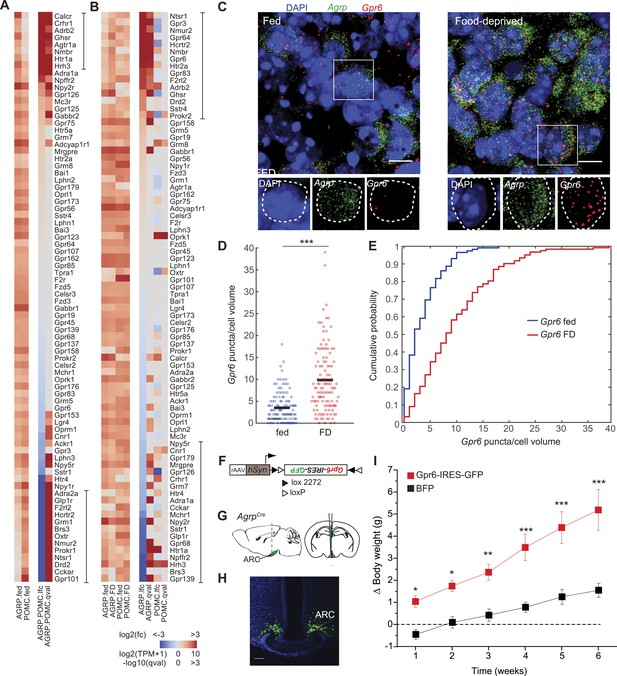
G-protein coupled receptors regulated by food-deprivation.
(A, B) All GPCR genes expressed (TPM > 20) in at least one group, sorted by log2(fold-change) between AGRP and POMC neurons (A) or AGRP or POMC neurons FD/fed (B). Bars indicate genes with >10-fold change (A) or >twofold change (B). (C) Double smFISH for Agrp and Gpr6. Scale, 10 μm. (D, E) Population counts (bars: mean values) (D) and cumulative probability distributions (E) of Gpr6 puncta per cell volume in AGRP neurons (p = 4.8e−10, ks-test). Fed, n = 115; FD, n = 122; 3 mice per condition. (F) Cre-dependent viral vector for cell type-specific Gpr6 overexpression in AGRP neurons. hSyn: synapsin promoter. Black and white triangles denote heterotypic loxP sites for stable inversion of Gpr6-IRES-GFP. (G) Schematic for viral transduction and cell-type specific overexpression of Gpr6 in AgrpCre mice. (H) Representative image showing Gpr6-IRES-GFP-transduced AGRP neurons. Scale, 100 µm. (I) Body weight change from pre-injection weight (starting age: 8 weeks) in AgrpCre mice expressing Gpr6-IRES-GFP or BFP (2-way ANOVA, one factor repeated measures, transgene: F1,65 = 19.6, p < 0.001; time: F5,65 = 30.1, p < 0.001; interaction: F5,65 = 4.2, p = 0.002). Holm-Sidak correction for multiple comparisons. AGRPGpr6 n = 9 mice, AGRPBFP n = 7 mice. Data is mean ± s.e.m. *p < 0.05, **p < 0.01, ***p < 0.001.
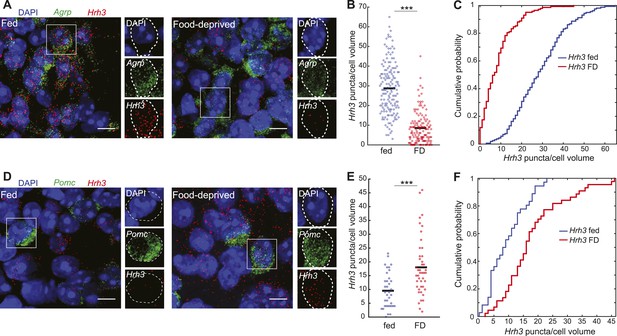
Opposite differential expression of Hrh3 in AGRP and POMC neurons after food deprivation.
(A–F) Representative images of double smFISH for Hrh3 and Agrp (A) or Pomc (B). Scale, 10 μm. Population counts (bars, mean values) (B, D) and cumulative probability distributions (C, E) of Hrh3 puncta per cell volume in AGRP and POMC neurons (p = 3.4e−34, p = 0.0037, respectively, ks-test). ***p < 0.05. AGRP fed, n = 159 cells; AGRP FD, n = 143 cells; POMC fed, n = 36 cells; POMC FD, n = 44 cells; 3 mice per condition.
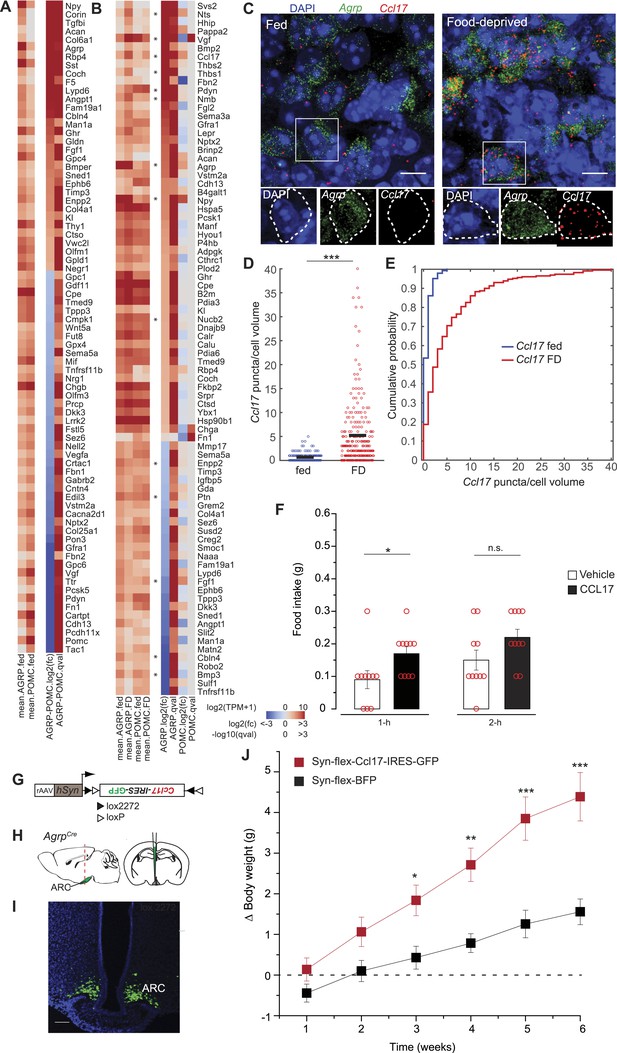
Secreted proteins regulated by food-deprivation.
(A, B) Secreted protein genes differentially expressed between AGRP and POMC neurons (A) or AGRP or POMC neurons FD/fed (B). Genes mentioned in the text are labeled with an asterisk. (C) Double smFISH for Agrp and Ccl17. Scale, 10 μm. (D, E) Population counts (bars: mean values) (D) and cumulative probability distributions (E) of Ccl17 puncta per cell volume in AGRP neurons (p = 1.5e−20, ks-test). Fed, n = 144; FD, n = 230; 3 mice per condition. (F) Food intake at start of light period 1-hr and 2-hr after intracerebroventricular injection of either saline or recombinant CCL17 (500 ng). Rank-sum test. (G) Cre-dependent viral vector for cell type-specific Ccl17 overexpression in AGRP neurons. hSyn: synapsin promoter. Black and white triangles denote heterotypic loxP sites for stable inversion of Ccl17-IRES-GFP. (H) Schematic for viral transduction and cell-type specific overexpression of Ccl17 in the brains of AgrpCre mice. (I) Representative image showing Ccl17-IRES-GFP-transduced AGRP neurons. Scale, 100 µm. (J) Body weight change from pre-injection weight (starting age: 8 weeks) in AgrpCre mice expressing Ccl17-IRES-GFP or BFP (2-way ANOVA, one factor repeated measures, transgene: F1,65 = 12.0, p = 0.004; time: F5,65 = 14.8, p < 0.001; interaction: F5,65 = 14.8, p < 0.001). BFP data is same as Figure 5I. Holm-Sidak correction for multiple comparisons. AGRPCcl17 n = 9 mice, AGRPBFP n = 7 mice. Data is mean ± s.e.m. n.s., p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001.
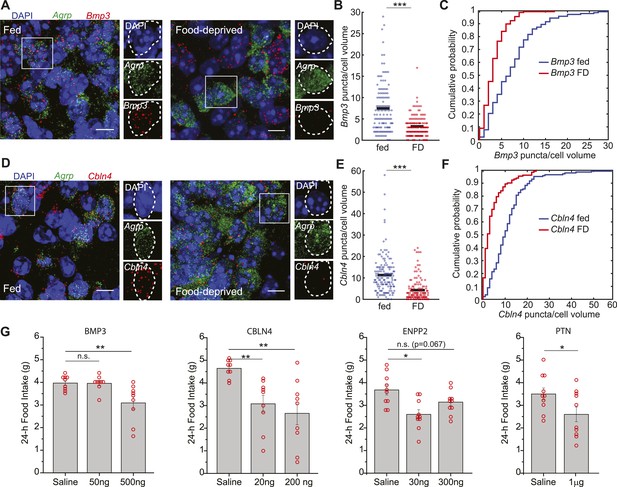
Secreted proteins that are downregulated in AGRP neurons with food-deprivation reduce food intake.
(A–F) Double smFISH for Agrp and (A) Bmp3 or (D) Cbln4. Scale, 10 μm. Population counts (B, E) and cumulative probability distributions (C, F) for Bmp3 and Cbln4 (p = 9e−12 and p = 6.8e−17, ks-test). Bmp3 fed, n = 142; Bmp3 FD, n = 157 cells; Cbln4 fed, n = 145; Cbln4 FD, n = 132; 3 mice per condition. (G) Mean food intake (24 hr) after intracerebroventricular injection of either saline or recombinant BMP3 (ANOVA, F2,24 = 7.5, p = 0.003), CBLN4 (ANOVA, F2,24 = 8.0, p = 0.002), ENPP2 (ANOVA, F2,27 = 7.3, p = 0.003), or Pleiotrophin (unpaired t-test). Holm-Sidak correction for multiple comparisons. Data is mean ± s.e.m. n.s., p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001.
Tables
Gene annotation enrichment analysis of differentially expressed genes
| Pathway | −log(p-value) |
|---|---|
| Agouti related protein | |
| Leptin signaling | 4.9 |
| Glutamate signaling/Axonal guidance/Ephrin/Rho GTPase | 3.5 |
| Endoplasmic reticulum stress/Oxidative stress | 3.2 |
| G-protein coupled receptor signaling | 3.0 |
| Circadian rhythm signaling | 2.7 |
| Sperm motility | 1.8 |
| Proopiomelanocortin | |
| Gαi signaling | 6.3 |
| Tetrahydrobiopterin biosynthesis (Gch1) | 2.1 |
| Zymostrerol biosynthesis (Msmo1) | 1.8 |
Additional files
-
Supplementary file 1
Gene expression data from RNA-Seq. Spreadsheet showing, for each gene, analyzed gene expression data for AGRP and POMC neurons. TPM values for each gene across all samples are provided. Comparisons for AGRP vs POMC gene expression in the fed state(.AgPo) as well as AGRP food-deprived (FD) vs fed (.agrp) and POMC food-deprived (FD) vs fed (.pomc) are also shown. CPM: counts per million, TPM: transcripts per million, fc: fold-change calculated as described in ‘Materials and methods’, lfc: log2(fc), qval: Benjamini-Hochberg corrected p-value as described in ‘Materials and methods’.
- https://doi.org/10.7554/eLife.09800.016
-
Source code 1
Matlab script for measuring fluorescence intensity from confocal stack image volumes.
- https://doi.org/10.7554/eLife.09800.017





