Electrophysiology and morphology of human cortical supragranular pyramidal cells in a wide age range
Figures
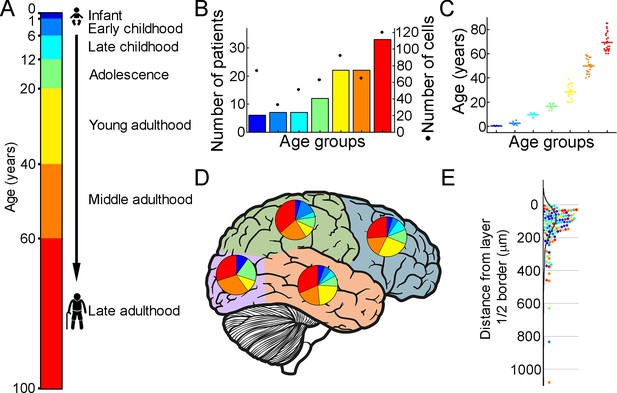
Illustration of the patient data on the samples utilized.
(A) Illustration of the defined age groups. (B) Number of patients involved in age groups, (n=6, 7, 7, 12, 22, 22, 33 from infant to late adulthood, respectively). Dots show the number of human layer 2/3 pyramidal cells in our dataset regarding the defined age groups (n=74, 33, 51, 63, 92, 66, 120 from infant to late adulthood, respectively). (C) Distributions of patient ages within age groups. (D) Brain model indicates the number of surgically removed tissues from the cortical lobes. Colors indicate age groups. (E) The distribution of recovered cell bodies distance from the L1/2 border.
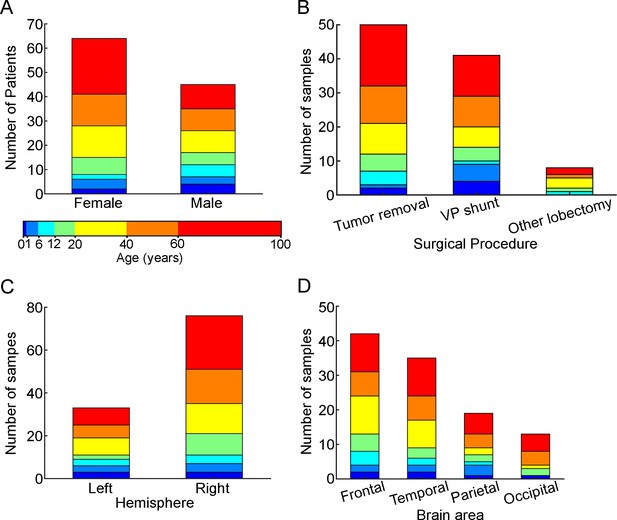
Patient metadata.
(A) Distribution of patients by gender (female: infant n=2, early childhood n=4, late childhood n=2, adolescence n=7, young adulthood n=13, middle adulthood n=13, late adulthood n=23, male: infant n=4, early childhood n=3, late childhood n=5, adolescence n=5, young adulthood n=9, middle adulthood n=9, late adulthood n=10), (B) surgical procedure (tumor removal: infant n=2, early childhood n=2, late childhood n=5, adolescence n=7, young adulthood n=13, middle adulthood n=12, late adulthood n=19, VP (ventriculoperitoneal) shunt: infant n=4, early childhood n=5, late childhood n=1, adolescence n=4, young adulthood n=6, middle adulthood n=9, late adulthood n=12, other: infant n=0, early childhood n=0, late childhood n=1, adolescence n=1, young adulthood n=3, middle adulthood n=1, late adulthood n=2), (C) hemisphere (left: infant n=3, early childhood n=3, late childhood n=3, adolescence n=2, young adulthood n=8, middle adulthood n=6, late adulthood n=8, right: infant n=3, early childhood n=4, late childhood n=4, adolescence n=10, young adulthood n=14, middle adulthood n=16, late adulthood n=25) and (D) brain area (frontal: infant n=2, early childhood n=2, late childhood n=4, adolescence n=5, young adulthood n=11, middle adulthood n=7, late adulthood n=11, temporal: infant n=2, early childhood n=2, late childhood n=2, adolescence n=3, young adulthood n=8, middle adulthood n=7, late adulthood n=11, parietal: infant n=1, early childhood n=3, late childhood n=1, adolescence n=2, young adulthood n=2, middle adulthood n=4, late adulthood n=6, occipital: infant n=1, early childhood n=0, late childhood n=0, adolescence n=2, young adulthood n=1, middle adulthood n=4, late adulthood n=5). Stacked columns are colored regarding the age groups shown on the colorbar.
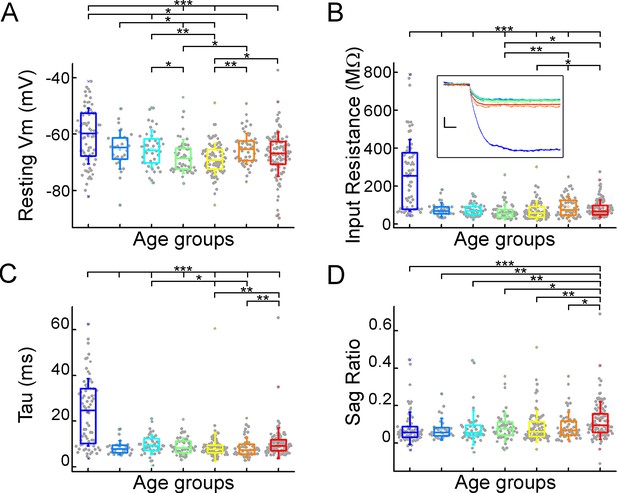
Subthreshold membrane properties vary across life stage.
(A–D) Boxplots show resting membrane potential (A), input resistance (B), tau (C), and sag ratio (D) distributions in various age groups. Inset shows representative voltage traces from each group. Scale bar: 5 mV; 20 ms. Asterisks indicate significance (Kruskal–Wallis test with post-hoc Dunn test, *p<0.05, **p<0.01, ***p<0.001).
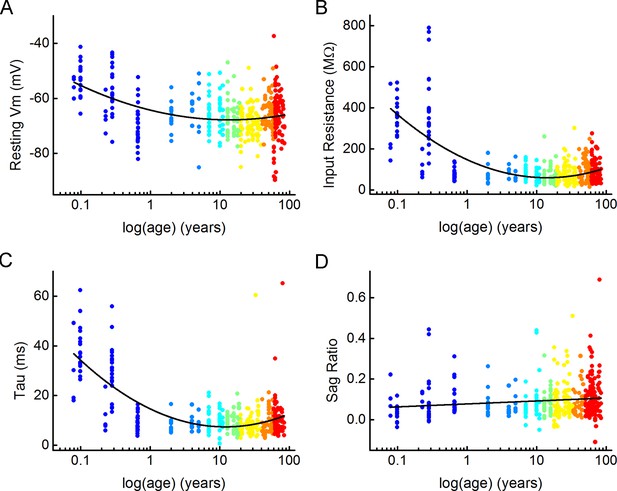
Distribution of subthreshold electrophysiological features with age.
(A–D) Scatter plots showing the passive electrophysiological characteristics: resting membrane potential (A), input resistance (B), tau (C), and sag ratio (D) throughout the lifespan. Dots are colored according to the age groups, age is represented in years on a logarithmic scale.
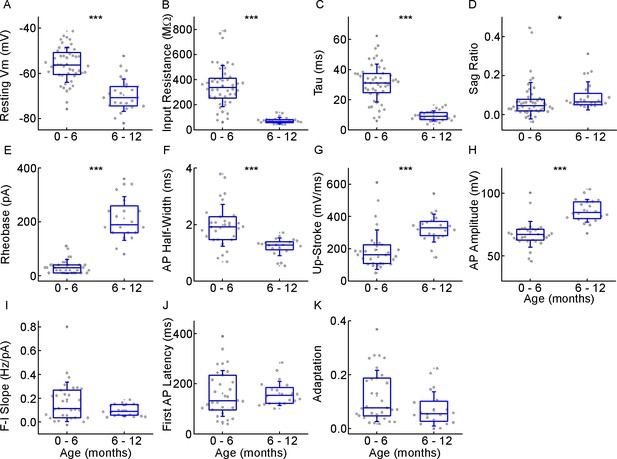
Electrophysiological differences during the first year of life.
(A–D) Boxplots showing differences in passive properties, resting membrane potential (A), input resistance (B), tau (C), and sag ratio (D) within the infant age group (*p<0.05, **p<0.01, ***p<0.001, two-sample t-test (A, B, C) or Mann-Whitney test (D)). (E–H) Differences in the action potential kinetics, rheobase (E), action potential (AP) half-width (F), up-stroke (G), and AP amplitude (H) between the cells from the first and the second half of the first year of life. Asterisks indicate significance (*p<0.05, **p<0.01, ***p<0.001, Mann-Whitney test). (I–K) Deviations in AP firing pattern parameters, F-I slope (H), first AP latency (I), and adaptation (J) during infancy.
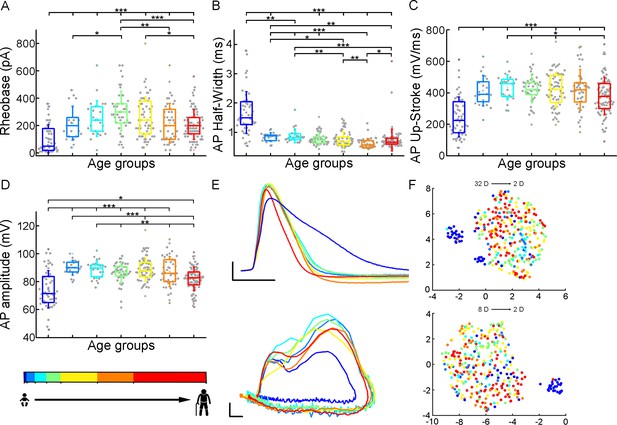
Age-related differences in the action potential kinetics.
(A–D) Boxplots show differences in rheobase (A), action potential half-width (B), action potential up-stroke (C), and action potential amplitude (D) between the age groups. Asterisks indicate statistical significance (*p<0.05, **p<0.01, ***p<0.001). (E) Representative action potentials aligned to threshold potential onset (scale: x-axis: 1 ms, y-axis: 20 mV) (top) and phase plots of the representative action potentials (APs) (scale: x-axis: 10 mV, y-axis: 100 mV/ms) (bottom). (F) Uniform Manifold Approximation and Projection (UMAP) of 32 (Table 4) (top) and eight selected electrophysiological properties (resting Vm, input resistance, tau, sag ratio, rheobase, AP half-width, AP up-stroke, and AP amplitude) (bottom) with data points for 331 cortical L2/3 pyramidal cells, colored with the corresponding age groups.
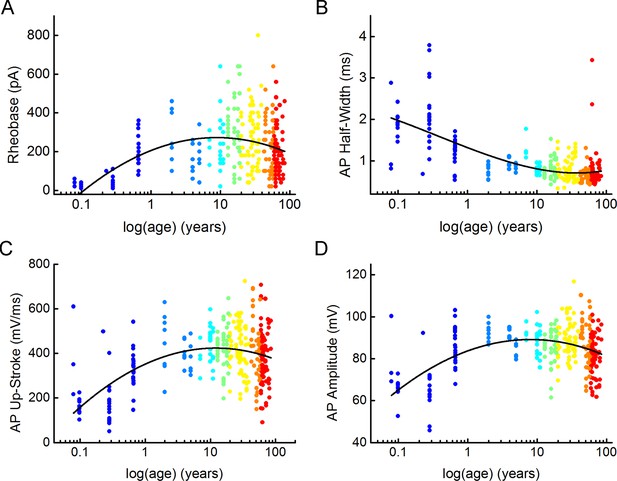
Distribution of suprathreshold electrophysiological features with age.
(A–D) Diagrams show changes through age in rheobase (A), action potential half-width (B), action potential up-stroke (C), and action potential amplitude (D). Dots are colored according to the age groups, age is represented in years on a logarithmic scale.
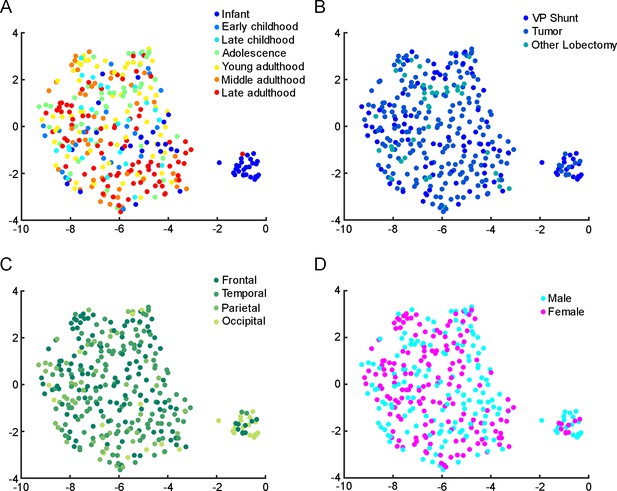
Relationship between patient metadata and electrophysiology.
Uniform Manifold Approximation and Projection (UMAP) of eight electrophysiological properties (resting Vm, input resistance, tau, sag ratio, rheobase, AP half-width, AP up-stroke, and AP amplitude) with data points for 331 cortical L2/3 pyramidal cells, colored with the corresponding age groups (A), surgical procedures (B), brain area (C), and gender (D).
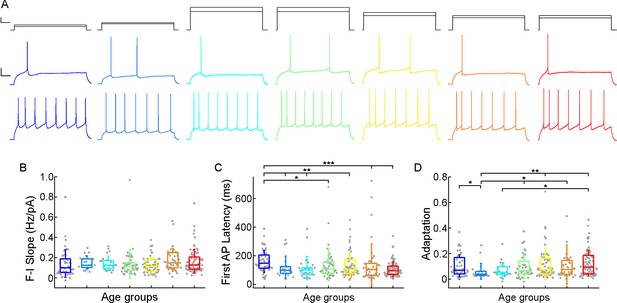
Age-dependency of the action potential (AP) firing pattern parameters.
(A) Representative membrane potential responses to an 800 ms long rheobase (middle) (left to right: infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, late adulthood), and increased current steps (bottom) colored respectively to the age groups. Scale bar top: 1 ms, 100 pA, bottom: 1 ms, 20 mV. (B–D) Boxplots show changes across the age groups in f-I slope (B), first AP latency (C), and adaptation of APs (D). Asterisks indicate statistical significance (*p<0.05, **p<0.01, ***<0.001).
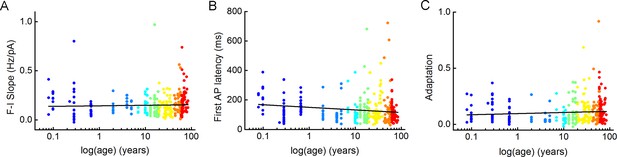
Distribution of firing pattern characteristics with age.
(A–C) Plots show F-I slope (A), first action potential (AP) latency (B), and adaptation of APs (C) depending on age. Dots are colored according to the age groups, age is represented in years on a logarithmic scale.
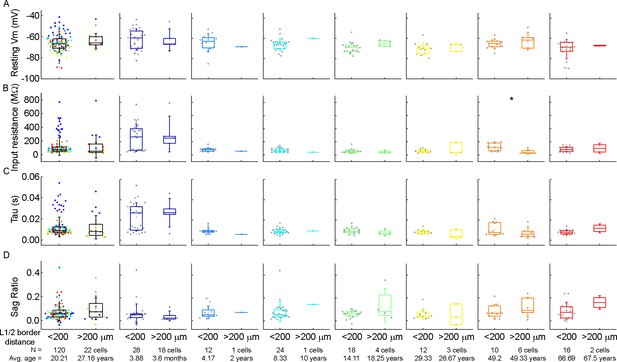
Comparison of the subthreshold properties of the cells as a function of their distance from the layer border.
(A–D) Boxplots show resting membrane potential (A), input resistance (B), tau (C), and sag ratio (D) of the pyramidal cells whose soma is located at a distance greater than and less than 200 μm from the L1 border. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood groups. Asterisks indicate significance (*p<0.05, Mann-Whitney test).
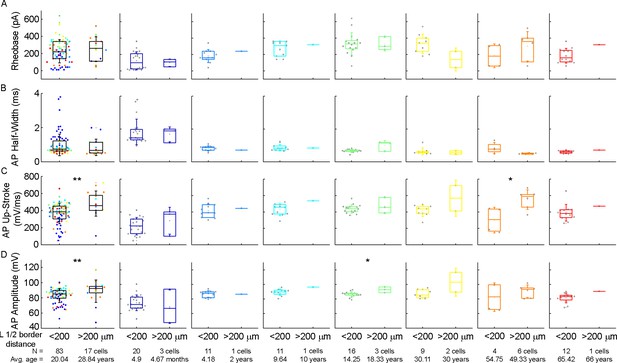
Comparison of the action potential properties of the cells as a function of their distance from the layer border.
(A–C) Boxplots show rheobase (A), action potential half-width (B), action potential upstroke (C), and action potential amplitude (D) of cells whose soma is located at a distance greater than and less than 200 μm from the L1 border. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood. Asterisks indicate significance (*p<0.05, **p<0.01, Mann-Whitney test).
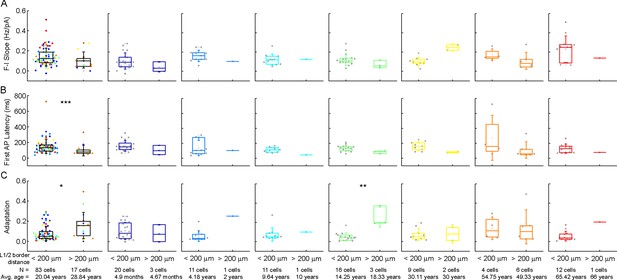
Comparison of the firing pattern characteristics of the cells as a function of their distance from the layer border.
(A–C) Boxplots show: F-I slope (A), latency of the first action potential (AP) (B), and adaptation of APs (C) of cells whose soma is located at a distance greater than and less than 200 μm from the L1 border. From left to right: data from all ages, infancy, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood. Asterisks indicate significance (*p<0.05, **p<0.01, Mann-Whitney test).
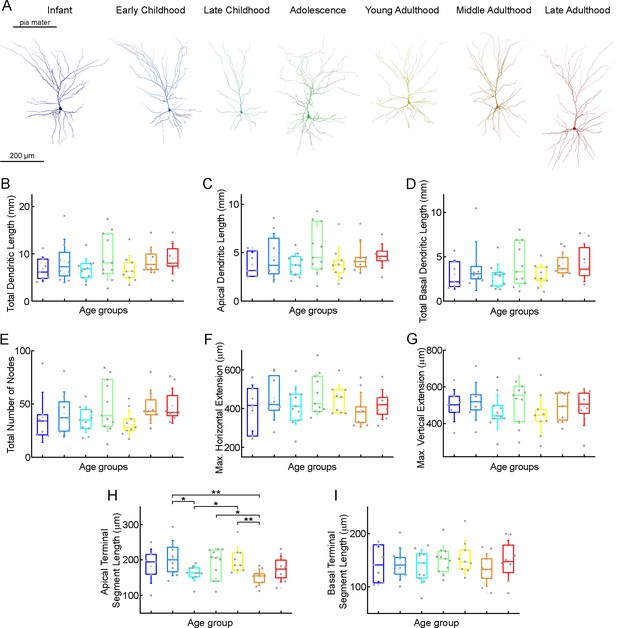
Morphological features of layer 2/3 pyramidal cells in different stages of life.
(A) Representative reconstructions of L2/3 pyramidal cells (from left to right) from infant (n=7), early childhood (n=8), late childhood (n=11), adolescence (n=11), young adulthood (n=9), middle adulthood (n=9), and late adulthood (n=8) patients. (B–I) Boxplots show summarized data from all the reconstructed cells (Figure 4—figure supplement 2) of total dendritic length (B), apical dendritic length (C), total basal dendritic length (D), the total number of nodes on the apical and basal dendrites (E), the maximal horizontal (F), and the maximal vertical (G) extension of dendrites, the average length of the apical (H), and basal (I) terminal dendritic segments. Asterisks indicate statistical significance (*p<0.05, **p<0.01, Kruskal-Wallis test with post-hoc Dunn test).

Human cortical L2/3 pyramidal cell dendritic reconstructions.
Reconstructions (n=63) of the examined human cortical pyramidal cells, from top to bottom: infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood age groups. Black lines represent the pia mater, gray dashed lines represent the L1-L2 border.
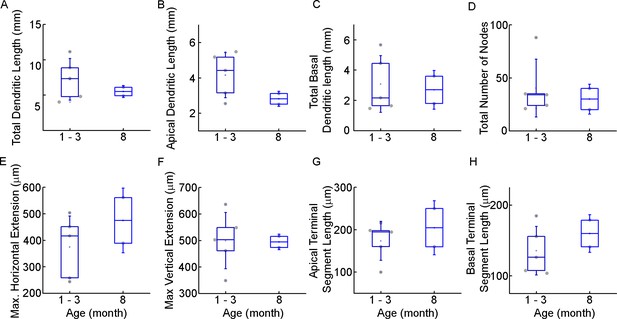
Morphological comparison of the examined infant cells.
(A–H) Boxplots show morphological features: total (A), apical (B), total basal (C) dendritic length, total number of nodes (D), maximal horizontal (E), and vertical (F) extension, average apical (G) and basal (H) terminal dendritic segment from infant patient during the first and second half of the first year of life.
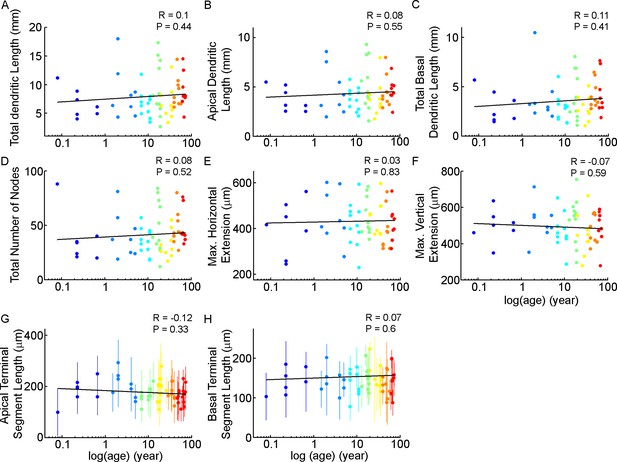
Distribution of morphological features with age.
(A–F) Scatterplots show the distribution of total (A), apical (B), total basal dendritic length (C), total number of bifurcations (D), maximum horizontal extension (E), maximum vertical extension of the dendritic arborisation (F). The age is represented in years on a logarithmic scale. G-H: Scatterplots show the average apical (G) and basal (H) terminal segment length of the cells, dots indicate the mean segment length of cells, vertical lines show the standard deviation.
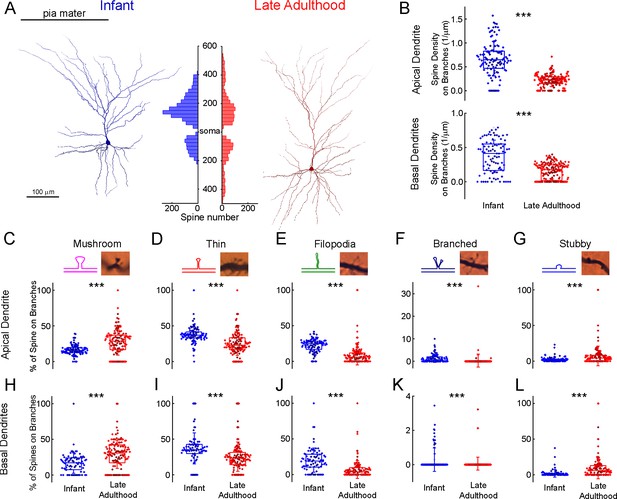
Comparison of dendritic spine densities in pyramidal cells from infant and late adulthood samples.
(A) Anatomical 3D reconstruction of human L2/3 pyramidal cells from the infant (left), and the late adulthood (right) age groups. The histogram (in the middle) demonstrates the distribution of dendritic spines on the two representative cells according to their distance from the soma (μm) on the apical and basal dendrites. (B) Boxplots of the average spine densities on the apical (top), and basal (bottom) dendritic branches from the n=3 infant (blue) and n=3 late adulthood (red) L2/3 pyramidal cells. The symbols are color-coded by the 6 individual cells.(C–G) The plots show the distribution of mushroom (C), thin (D), filopodium (E), branched (F), and stubby (G) dendritic spine types on the apical dendrites of the reconstructed infant (n=3, blue) and late adult (n=3, red) pyramidal cells. Top, schematic illustration and representative images of the examined dendritic spine types. Center, age-dependent distribution of spine types. Asterisks indicate significance (*p<0.05, **p<0.01, ***p<0.001). (H–L) Same as C-G but on basal dendrites.
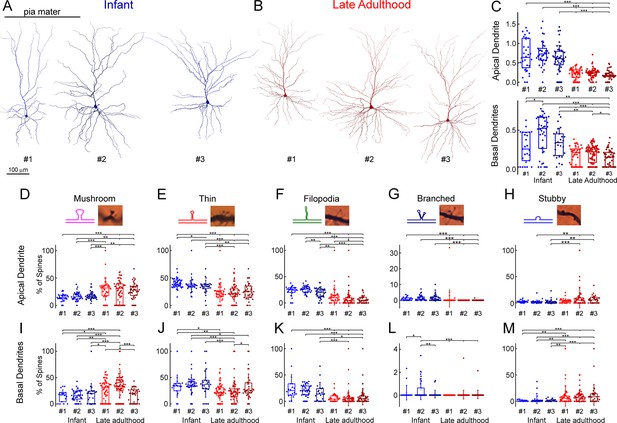
Comparison of dendritic spine densities in pyramidal cells from all samples.
(A) Anatomical 3D reconstructions of the examined n=3 human L2/3 pyramidal cells from the infant age group. (B) Anatomical reconstructions (n=3) from the late adulthood group. (C) Boxplots showing the spine density on the six individual cells, the infant pyramidal cell shown with blue, the late adulthood pyramidal cell with red (cells are numbered according to panels A and B), on the apical (top) and the basal (bottom) dendrites. Asterisks indicate significance (*p<0.05, **p<0.01, ***p<0.001, Kruskal-Wallis test with post-hoc Dunn test). (D–H) The plots show the distribution of mushroom (D), thin (E), filopodium (F), branched (G), and stubby (H) dendritic spine types on the apical dendrites of the reconstructed infant (n=3, blue) and late adult (n=3, red) pyramidal cells. Top, schematic illustration and representative images representation of the examined dendritic spine types. The width of the inserted images is 5 μm. Bottom, spine distributions on the individual cells that were examined. Asterisks indicate significance (*p<0.05, **p<0.01, ***p<0.001, Kruskal-Wallis test with post-hoc Dunn test). (I–M) Same as D-H but on basal dendrites.
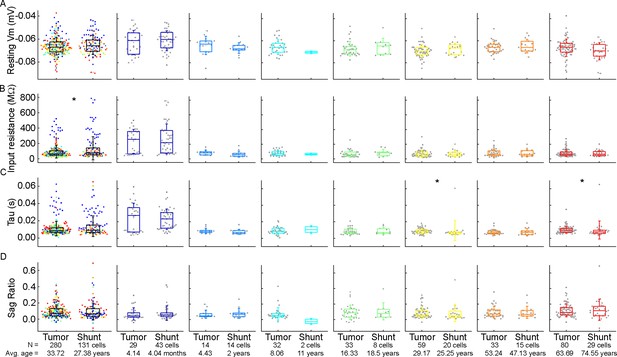
Comparison of subthreshold electrophysiological properties of the examined cells from patients with tumor removal or ventriculoperitoneal (VP) shunt surgical procedures.
(A–D) Boxplots show resting membrane potential (A), input resistance (B), tau (C), and sag ratio (D) from patients with tumor removal (tumor) or VP shunt (shunt) surgical procedures. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood groups. Asterisks indicate significance (*p<0.05, Mann-Whitney test).
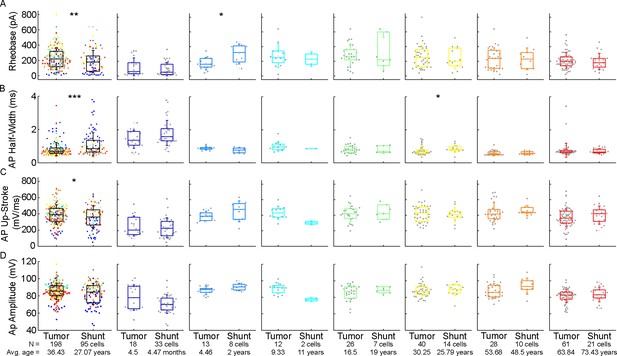
Comparison of action potential properties of the examined cells from patients with tumor removal or ventriculoperitoneal (VP) shunt surgical procedures.
(A–D) Boxplots show rheobase (A), action potential half-width mean (B), action potential up-stroke (C) and action potential amplitude (D) from patients with tumor removal (tumor) or VP shunt (shunt) surgical procedures. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood groups. Asterisks indicate significance (*p<0.05, **p<0.01, ***p<0.001, Mann-Whitney test).
Comparison of firing pattern characteristics of the examined cells from patients with tumor removal or ventriculoperitoneal (VP) shunt surgical procedures.
(A–C) Boxplots showing: F-I slope (A), first action potential (AP) latency (B), and adaptation of APs (C) from patients with tumor removal (tumor) or VP shunt (shunt) surgical procedures. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood groups. Asterisks indicate significance (*p<0.05, **p<0.01, Mann-Whitney test).
Morphological comparison of the examined cells from patients with tumor removal or ventriculoperitoneal (VP) shunt surgical procedures.
(A–H) Boxplots showing morphological features: total (A), apical (B), total basal (C), dendritic length, total number of nodes (D), maximal horizontal (E), and vertical (F) extension, average apical (G) and basal (H) terminal dendritic segment from patients with tumor removal (tumor) or VP shunt (shunt) surgical procedures. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, middle adulthood, and late adulthood groups. Asterisks indicate significance (*p<0.05, Mann-Whitney test).
Comparison of subthreshold electrophysiological properties of the examined cells from female and male patients.
(A–D) Boxplots show resting membrane potential (A), input resistance (B), tau (C), and sag ratio (D) from female and male patients. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood groups. Asterisks indicate significance (*p<0.05, **p<0.01 Mann-Whitney test or two-sample t-test).
Comparison of action potential properties of the examined cells from female and male patients.
(A–D) Boxplots show rheobase (A), action potential half-width mean (B), action potential up-stroke (C) and action potential amplitude (D) from female and male patients. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood groups. Asterisks indicate significance (*p<0.05, **p<0.01, ***p<0.001, Mann-Whitney test or two-sample t-test).
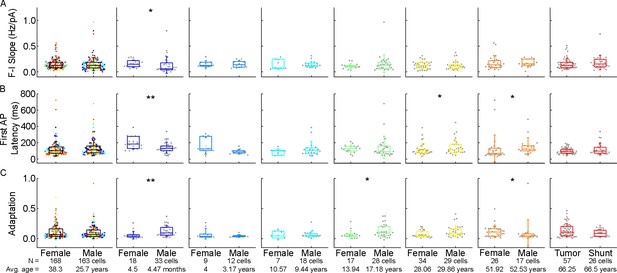
Comparison of firing pattern characteristics of the examined cells from female and male patients.
(A–C) Boxplots showing: F-I slope (A), first action potential (AP) latency (B), and adaptation of APs (C) from female and male patients. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, young adulthood, middle adulthood, and late adulthood groups. Asterisks indicate significance (*p<0.05, **p<0.01, Mann-Whitney test or two-sample t-test).

Morphological comparison of the examined cells from female and male patients.
(A–H) Boxplots showing morphological features: total (A), apical (B), total basal (C), dendritic length, total number of nodes (D), maximal horizontal (E), and vertical (F) extension, average apical (G), and basal (H) terminal dendritic segment from female and male patients. From left to right: data from all age groups, infant, early childhood, late childhood, adolescence, and late adulthood groups.

Thickness of cortical layer 1 at different life stages.
(A) Boxplot shows the thickness of layer 1. (B) Scatter plot shows the distribution of L1 thickness measured on the reconstructed cells. Age is shown in years on a logarithmic scale, dots are color-coded according to the corresponding age groups.

(A) Uniform Manifold Approximation and Projection (UMAP) of 8 selected electrophysiological properties (resting Vm, input resistance, tau, sag ratio, rheobase, AP half-width, AP up-stroke, and AP amplitude) with data points for 331 cortical L2/3 pyramidal cells, colored with the corresponding age groups. (B) UMAP colored by k-means clustering with 7 clusters, red crosses represent the centroids of the clusters.

Age-dependence of afterhyperpolarization and AP threshold.
(A-B) Boxplots show the differences in afterhyperpolarization (AHP) amplitude (A) and AP threshold (B) between age groups. Asterisks indicate statistical significance (* P < 0.05, ** P < 0.01, *** P < 0.001, Kruskal-Wallis test with post-hoc Dunn test). (C-D) Scatter plots show AHP amplitude (C) and AP threshold (D) across the lifespan. Age is shown on a logarithmic scale, dots are colored according to the corresponding age group.
Tables
Subthreshold membrane properties.
| Subthreshold properties | InfantN=72 | Early childhoodN=28 | Late childhoodN=45 | AdolescenceN=54 | Young adulthoodN=89 | Middle adulthoodN=56 | LateAdulthoodN=113 |
|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | |
| Resting Vm (mV) | –60.64±9.86 | –65.44±6.82 | –65.17±6.68 | –67.86±5.94 | –68.69±5.54 | –65.14±5.5 | –67.05±7.86 |
| Input resistance (MΩ) | 257.25±188.06 | 75.27±37.5 | 74.61±34.29 | 64.79±41.82 | 70.45±46.9 | 90.14±54.37 | 81.14±46.36 |
| Tau (ms) | 23.88±14.7 | 8.49±3.08 | 9.73±4.24 | 8.99±3.71 | 8.76±6.33 | 8.53±4.24 | 10.39±6.63 |
| Sag ratio | 0.079±0.87 | 0.075±0.058 | 0.082±0.092 | 0.086±0.076 | 0.09±0.084 | 0.092±0.073 | 0.12±0.1 |
Action potential and firing pattern parameters.
| Suprathreshold properties | InfantN=51 | Early childhoodN=21 | Late childhoodN=25 | AdolescenceN=45 | Young adulthoodN=63 | Middle adulthoodN=43 | LateAdulthoodN=83 |
|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | |
| Rheobase (pA) | 104.51±103.18 | 218.1±123.27 | 252.8±131.17 | 306.22±147.21 | 262.9±148.06 | 219.07±145.52 | 207.71±107.83 |
| AP half-width (ms) | 1.68±0.71 | 0.84±0.15 | 0.88±0.24 | 0.78±0.22 | 0.76±0.27 | 0.62±0.17 | 0.74±0.38 |
| AP up-stroke (mV/ms) | 247.43±127.27 | 413.68±97.11 | 434.75±78.32 | 424.69±87.58 | 418.49±110.33 | 419.15±129.29 | 379.22±118.45 |
| AP amplitude (mV) | 74.62±13.4 | 90±5.23 | 88.82±6.35 | 86.31±7.36 | 88.81±8.52 | 87.28±10.77 | 82.02±8.49 |
| F-I slope (Hz/pA) | 0.142±0.137 | 0.152±0.066 | 0.144±0.071 | 0.139±0.141 | 0.127±0.08 | 0.176±0.113 | 0.165±0.117 |
| First AP latency (ms) | 161.26±76.81 | 121.36±76.88 | 115.29±81.27 | 136.69±109.78 | 132.42±84.68 | 140.61±143.43 | 106.39±52.31 |
| Adaptation | 0.103±0.087 | 0.061±0.062 | 0.068±0.06 | 0.105±0.1 | 0.119±0.118 | 0.114±0.153 | 0.124±0.1 |
Morphological characteristics across the age groups.
| Morphological properties | InfantN=7 | Early childhoodN=8 | Late childhoodN=11 | AdolescenceN=11 | Young adulthoodN=9 | Middle adulthoodN=9 | Late adulthoodN=8 |
|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | |
| Total dendritic length (mm) | 6.74±2.56 | 8.5±4.56 | 6.62±2.29 | 9.27±4.82 | 6.76±2.9 | 8.7±2.7 | 8.96±3.22 |
| Apical dendritic length (mm) | 3.77±1.24 | 4.57±2.42 | 3.78±1.14 | 5.24±2.51 | 3.88±1.8 | 4.52±1.63 | 4.64±1.25 |
| Basal dendritic length (mm) | 2.97±1.63 | 3.93±2.74 | 2.84±1.33 | 4.04±2.44 | 2.88±1.27 | 4.18±1.35 | 4.31±2.13 |
| Total number of nodes | 37.42±23.58 | 40.88±20.54 | 35.36±12.07 | 48.36±24.23 | 32.11±12.38 | 47.33±15.57 | 48.38±16.47 |
| Max. horizontal extension (μm) | 403.33±118.55 | 455.24±114.42 | 417.2±98.1 | 472.2±104.51 | 448.3±75.94 | 393.7±90.29 | 420.93±77.49 |
| Max. vertical extension (μm) | 498.12±87.85 | 521.21±102.88 | 463.83±94.63 | 529.58±129.28 | 447.41±114.17 | 488.02±81.02 | 490.45±100.53 |
| Apical terminal length (μm) | 182.42±48.17 | 204.83±49.62 | 162.47±21.5 | 189.59±42.54 | 199.68±36.2 | 151.23±23.3 | 174.62±36.76 |
| Basal terminal length (μm) | 142.54±32.43 | 142.69±30.31 | 138.7±31.22 | 150.32±26.09 | 155.29±32.15 | 131.6±30.44 | 148.96±38.42 |
Examined electrophysiological properties.
| Resting Vm | The membrane potential of the neuron, measured directly after attaining the whole-cell configuration with no current (if a holding current was used during the recording we compensated the resting membrane potential with the injected current). |
| Input resistance | To calculate input resistance the mean of all hyperpolarizing current produced voltage steps were used. |
| Tau | To calculate time constant the mean of all hyperpolarizing current produced voltage steps were used, measured between 0–63%. |
| Sag ratio | The ratio of the maximal deflection and the steady-state membrane potential during a –100 pA current step. |
| Rheobase | The minimal current step that elicited the first spike. |
| AP half-width | The width of the AP at half amplitude. |
| AP up-stroke | The mean of all the maximum values of dV/dt between the action potential onset and the action potential peak from each elicited APs of the cell. |
| AP amplitude | Average amplitude of all APs, from threshold to peak. |
| F-I slope | The slope of the line fitted to the data of the AP firing frequency versus stimulus intensity. |
| First AP latency | The duration from the start of the stimulus until the first AP under the rheobasic current step. |
| Adaptation | The average adaptation of the interspike interval between consecutive APs. |
| Rebound | The difference between the steady-state membrane potential and the maximum deflection after a hyperpolarizing current step. |
| Rebound-Sag ratio | The ratio of the rebound and sag amplitudes. |
| Avg. AP number | Average number of elicited APs per sweep. |
| AP threshold | Mean of all the voltage values at AP threshold. |
| Ap rise time | Mean time between the threshold and the peak of all APs. |
| AHP amplitude | Average amplitude of all afterhyperpolarization. |
| AHP length | Average duration of 0 (AHP minimum) to 90% of all the AHP. |
| Voltage at max.dV/dt | Average voltage value at the maximum of the AP dv/dt over all APs |
| Velocity at min. dV/dt | Average velocity value at the minimum of the AP dv/dt from all APs |
| Voltage at min. dV/dt | Average voltage value at the minimum of the AP dv/dt from all APs |
| AP peak | Average of AP maximum voltages |
| ISI mean | Average interspike interval from all sweeps containing at least three APs |
| AP amplitude accommodation | The difference of the first and last AP amplitude in a sweep. |
| AP half-width accommodation | The difference of the first and last AP half-width in a sweep. |
| AP threshold accommodation | The difference of the first and last AP threshold in a sweep. |
| Average ISI | The mean value of interspike intervals in a sweep. |
| AP amplitude adaptation | The average adaptation of the AP amplitude between consecutive APs. |
| AP half-width adaptation | The average adaptation of the AP half-width between consecutive APs. |
| AP threshold adaptation | The average adaptation of the AP threshold between consecutive APs. |
| AHP area | Integral of the AHP from the minimum value to 90%, using trapezoidal method. |
| ADP amplitude | The amplitude of the afterdepolarization, the average difference between the minimum value of the AHP and the threshold of the next AP. |
| Electrophysiological parameters | Age P-value | Age group P-value | Gender P-value | Gender (all age groups) | Brain area P-value | Hemisphere P-value | Surgical procedure P-value | Surgical procedure (all age groups) |
|---|---|---|---|---|---|---|---|---|
| Statistical analysis | GAMM | Kruskal-Wallis | GAMM | Pairwise* | GAMM | GAMM | GAMM | Pairwise** (tumor-shunt) |
| Resting Vm (mV) | 0.95 | 0.68 | 0.0498 | 0.025 | 0.11 | |||
| Input resistance (M) | 0.75 | 0.2 | 0.02 | |||||
| Tau (ms) | 0.44 | 0.03 | 0.09 | 0.28 | ||||
| Sag ratio | 0.44 | 0.003 | 0.09 | 0.34 | ||||
| Rheobase (pA) | 0.68 | 0.13 | 0.016 | 0.4 | 0.87 | |||
| AP half-width (ms) | 0.31 | 0.61 | ||||||
| AP up-stroke (mV/ms) | 0.83 | 0.03 | 0.02 | 0.012 | 0.03 | |||
| AP amplitude (mV) | <2*10–16 | 0.42 | 0.86 | 0.11 | 0.74 | 0.14 | ||
| F-I slope | 0.055 | 0.055 | 0.97 | 0.6 | 0.02 | 0.07 | 0.7 | 0.6 |
| First AP latency (ms) | 0.01 | 0.59 | 0.17 | 0.15 | 0.89 | 0.8 | 0.71 | |
| Adaptation | 0.03 | 0.032 | 0.46 | 0.95 | 0.002 | 0.28 | 0.34 | 0.88 |
Additional files
-
Supplementary file 1
Patients’ data for all subjects used in this study.
- https://cdn.elifesciences.org/articles/100390/elife-100390-supp1-v1.xlsx
-
Supplementary file 2
Number of dendritic spines of the examined pyramidal cells.
- https://cdn.elifesciences.org/articles/100390/elife-100390-supp2-v1.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/100390/elife-100390-mdarchecklist1-v1.docx





