A peptide-neurotensin conjugate that crosses the blood-brain barrier induces pharmacological hypothermia associated with anticonvulsant, neuroprotective, and anti-inflammatory properties following status epilepticus in mice
Figures
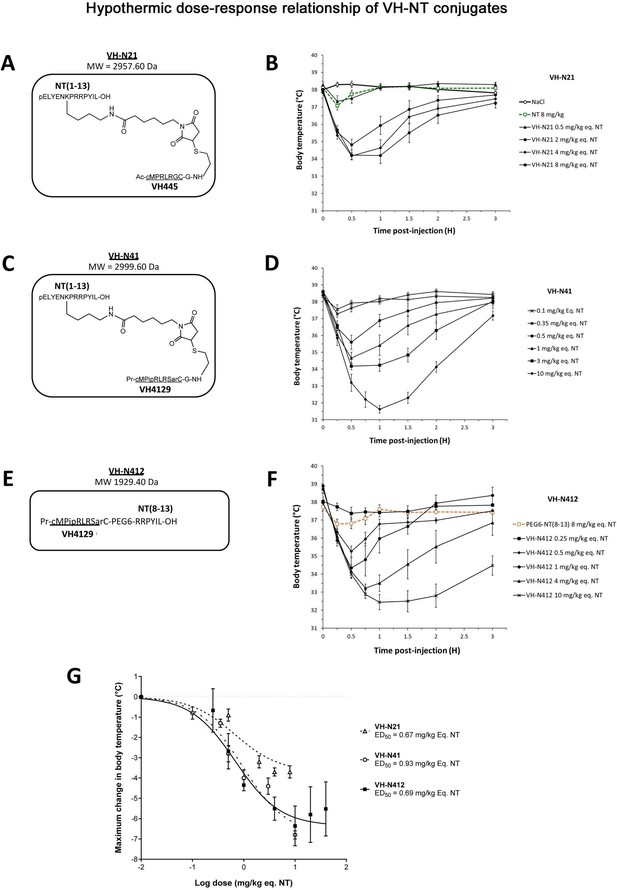
Hypothermic dose-response relationship of different VH-NT conjugates following single intravenous (i.v.) (bolus) injection in naïve Swiss (CD-1) mice.
(A, C, and E) Chemical structure and molecular weight of the VH-N21, VH-N41, and VH-N412 conjugates, containing the eight amino acid cyclic brain penetrating peptide that recognizes the LDLR (VH445 for VH-N21 and VH4129 for VH-N41 and VH-N412), and either the neurotensin (NT) tridecapeptide (VH-N21 and VH-N41) or its C-terminal NT(8–13) fragment (VH-N412). (B, D, and F) Hypothermic response to VH-N21, VH-N41, and VH-N412 conjugates in mice after single i.v. (bolus) injection at increasing dose levels. Core body (rectal) temperature was measured before (baseline) and at indicated times after injection. Data are presented as means ± SEM, n=4–8 per group. (G) Dose-response curves of VH-N21, VH-N41, and VH-N412 hypothermic response. ED50 values for each conjugate were estimated by plotting the response vs log[dose(mg/kg eq. NT)] followed by nonlinear regression (three parameters) using GraphPad Prism software.
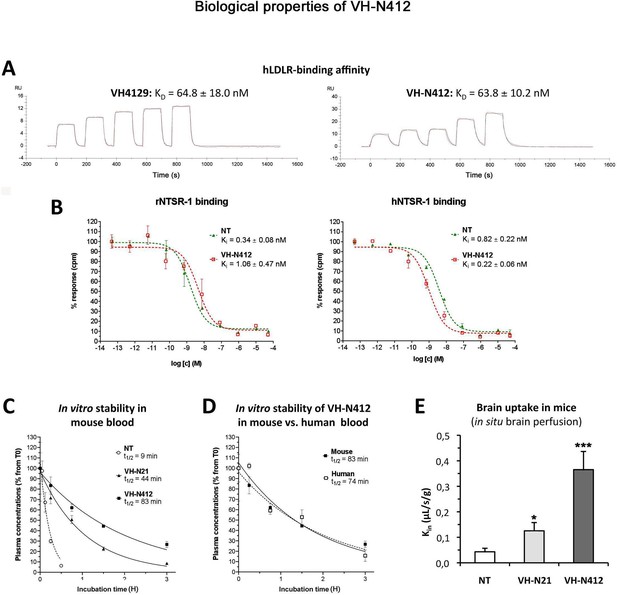
In vitro and in vivo biological properties of VH-N412.
(A) Surface plasmon resonance (SPR) sensorgrams of the free VH4129 and the VH-N412 compound on immobilized human LDLR (hLDLR). Red lines show the specific binding of molecules obtained after double subtraction of the signal measured on the control flow cell (without immobilized LDLR) and a blank run. Black lines show fit curves of the experimental data with a 1:1 binding model. The illustrated data are representative of two to five independent experiments. (B) Dose-response inhibition curves of tritiated NT, bound on hNTSR-1 or rNTSR-1 membrane extracts, in the presence of indicated concentrations of NT or VH-N412. Indicated Ki values were estimated from mean IC50 values obtained by logarithmic regression of experimental data. Data were plotted as means ± SD of biological duplicates. (C and D) Comparison of degradation rates for NT or peptide-NT conjugates in mouse blood. NT or peptide-NT conjugates were incubated in freshly collected mouse (C) or human (D) blood and analyzed using liquid chromatography-tandem mass spectrometry (LC-MS/MS) at indicated times in the plasma fraction. Data were plotted as means ± SD of n=3 biological replicates. T1/2 values were estimated from nonlinear regression (one-phase decay) of experimental data. (E) Blood-brain barrier (BBB) transport of tritium-labeled NT or peptide-NT conjugates using in situ brain perfusion in mice. Data were presented as mean ± SEM for three to six animals. Student’s t-test vs NT: *p<0.05, **p<0.01.
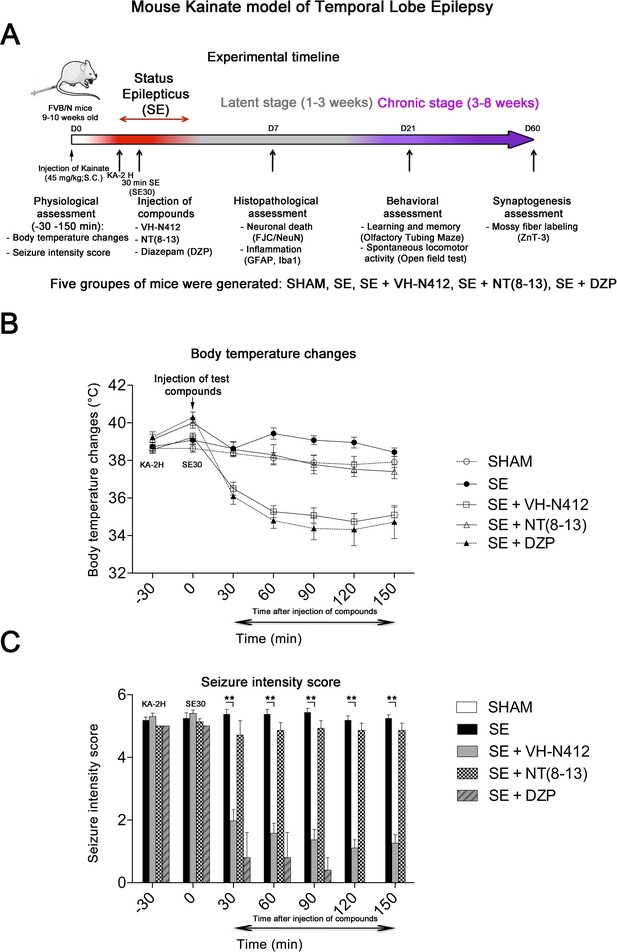
Effects of VH-N412 on body temperature and seizure intensity following status epilepticus (SE).
(A) Experimental timeline with the assessment of physiological, histopathological, behavioral, and synaptogenesis features associated with the mouse KA model of temporal lobe epilepsy. Five groups of mice were generated: SHAM, SE, SE+VH-N412, SE+NT(8–13), SE+DZP. (B) Mice were injected with KA, which induced stage 5 or stage 6 seizures after 2 hr, characteristic of SE, associated with hyperthermia as compared to all animal groups. VH-N412 was administered 30 min after SE onset at the dose of 4 mg/kg eq. NT caused significant hypothermia, which persisted at least 2 hr, similar to the effects of high-dose DZP (45 mg/kg) administered i.p. and used as positive control. SE+NT(8–13) had no effect on body temperature when administered 30 min after SE onset. (C) Hypothermia induced by VH-N412 was associated with a significant decrease of seizures in the SE+VH-N412 group, similar to DZP, while SE+NT(8–13) had no effect on seizure intensity.
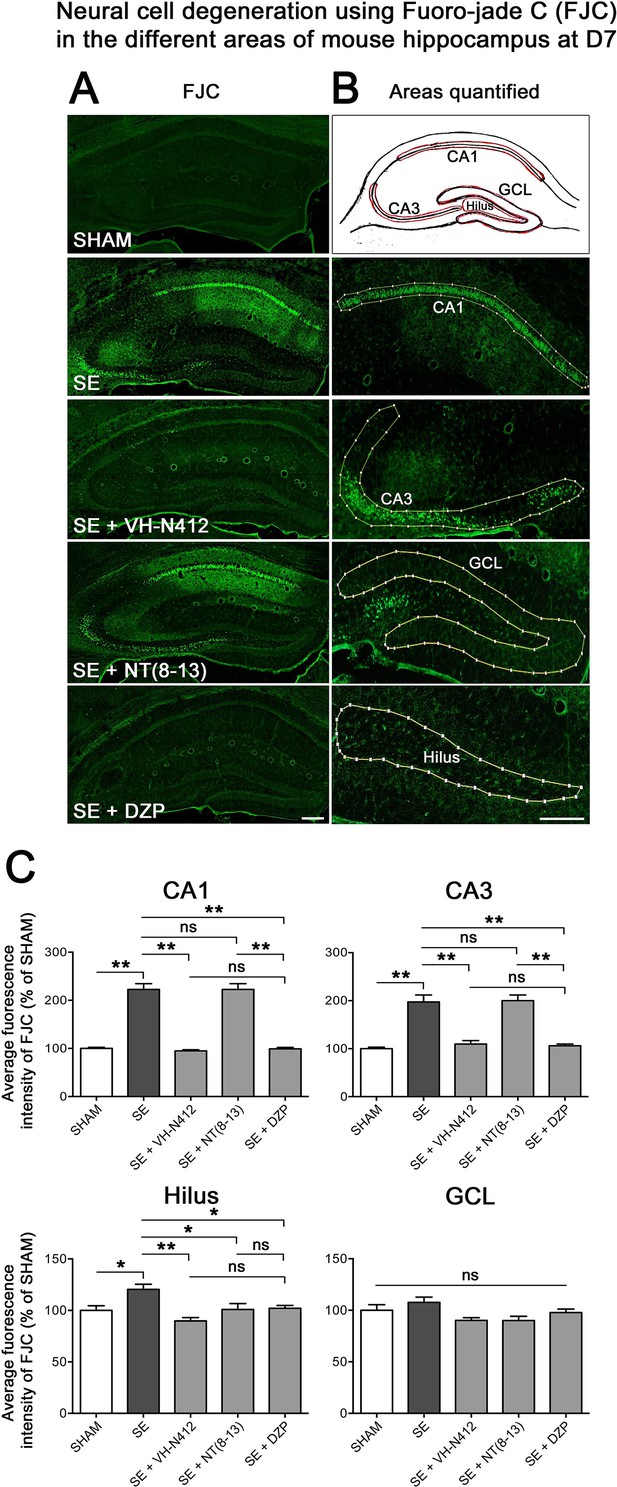
Effects of VH-N412 on neural cell degeneration following KA-induced status epilepticus (SE).
(A) Fluoro-Jade C (FJC) staining was used to assess the extent of neural cell damage in coronal sections of the dorsal hippocampal formation at D7 post-SE from SHAM, SE, SE+VH-N412, SE+ NT(8–13) and SE+ DZP animals. (B) The regions of interest were highlighted on the scheme, upper panel, and were traced to quantify FJC in the CA1, CA3, GCL, and the H. Scale bars: 200 μm in all panels. (C) Histograms comparing the mean intensities of staining for FJC in dorsal CA1, CA3, H, and GCL from SHAM, SE, SE+VH-N412, SE+NT(8–13), and SE+DZP animals. VH-N412 as well as DZP displayed significant protective effect in dorsal CA1, CA3, and H but not in GCL. Data were expressed as the average percentage ± SEM, normalized to the SHAM CTL. Asterisks indicate statistically significant differences: *p<0.05, **p<0.01 (Tukey’s test).
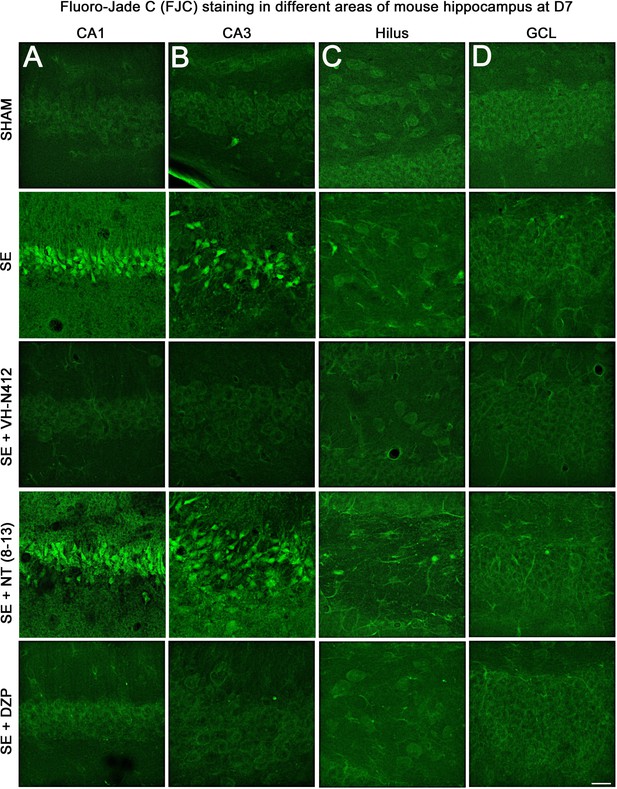
Representative examples of FJC staining in the different areas of mouse dorsal hippocampus.
(A) CA1, (B) CA3, (C) H and (D) GCL, at D7 post-SE from SHAM, SE, SE+VH-N412, SE+ NT(8–13), and SE+DZP animals. Scale bar: 20 μm in all panels.
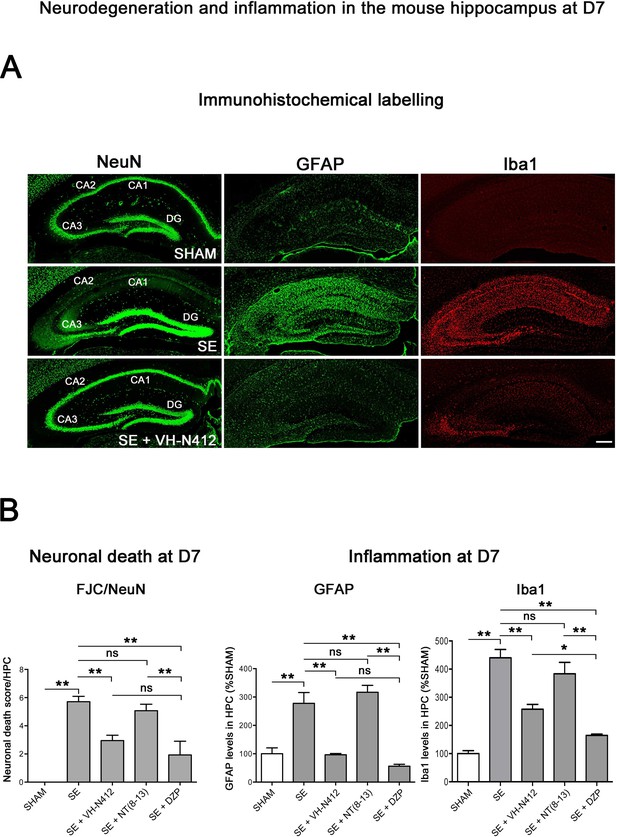
Neuroprotective and anti-inflammatory effects of VH-N412 following SE.
(A) Immunohistochemical labeling was used to assess the extent of brain damage in coronal sections of the dorsal hippocampus from SHAM, SE, SE+VH-N412, SE+NT(8–13), and SE+DZP animals at D7 post-SE. Left panels show neurons labeled with the anti-NeuN antibody directed against a neuronal-specific nuclear protein in all animals. Middle and right panels show inflammation assessed with anti-GFAP and Iba1 antibodies to monitor astrocytic and microglial reactivity respectively. Scale bar: 200 μm in all panels. In SHAM animals, a basal labeling for GFAP and Iba1 was detected in the hippocampus. In SE animals, a strong activation of glial cells occurred in all hippocampal layers. This inflammatory response was nearly abolished when VH-N412 was administered 30 min after SE onset. (B) Histograms comparing the mean neuronal death score, the mean GFAP, and Iba 1 levels in the dorsal hippocampus of SHAM, SE, SE+VH-N412, SE+NT(8–13), and SE+DZP animals. NeuN and FJC labeling were used to quantify neuronal death and the effects of VH-N412 (left histogram). The neuronal death score was expressed as the mean scores ± SEM. GFAP and Iba1 labeling levels allowed quantification of glial inflammation, which was expressed as the average percentage ± SEM normalized to CTL SHAM. In SHAM animals, no neuronal death was observed in the hippocampus (score 0). In SE animals, significant neuronal death was observed in CA1–3 pyramidal cell layers and H. Neurodegeneration observed in SE animals was significantly decreased when VH-N412 or DZP were administered 30 min after SE onset, but no changes were observed when NT(8–13) was administered. Asterisks indicate statistically significant differences: *p<0.05, **p<0.01 (Tukey’s test).
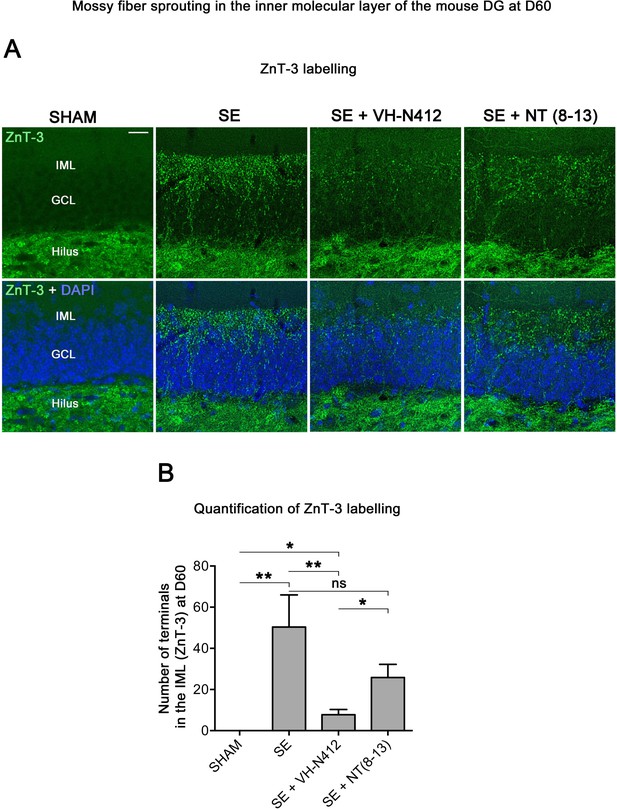
VH-N412 reduces mossy fiber sprouting in the hippocampus 8 weeks (D60) after SE.
(A) The effects of VH-N412 on mossy fiber sprouting was assessed 8 weeks after induction of SE with immunohistochemical labeling for the zinc vesicular transporter 3 (ZnT-3). In SHAM animals, mossy fiber terminals were only present in the H. In SE animals, in addition to ZnT-3 staining present in the H, mossy fiber terminals were also observed within the IML. Scale bar: 20 μm in all panels. (B) Semiquantitative analysis revealed that ZnT-3 staining in the IML at D60 was significantly reduced in animals administered with VH-N412 30 min after SE onset, but unchanged when NT(8–13) was administered. Asterisks indicate statistically significant differences: *p<0.05, **p<0.01 (Tukey’s test).
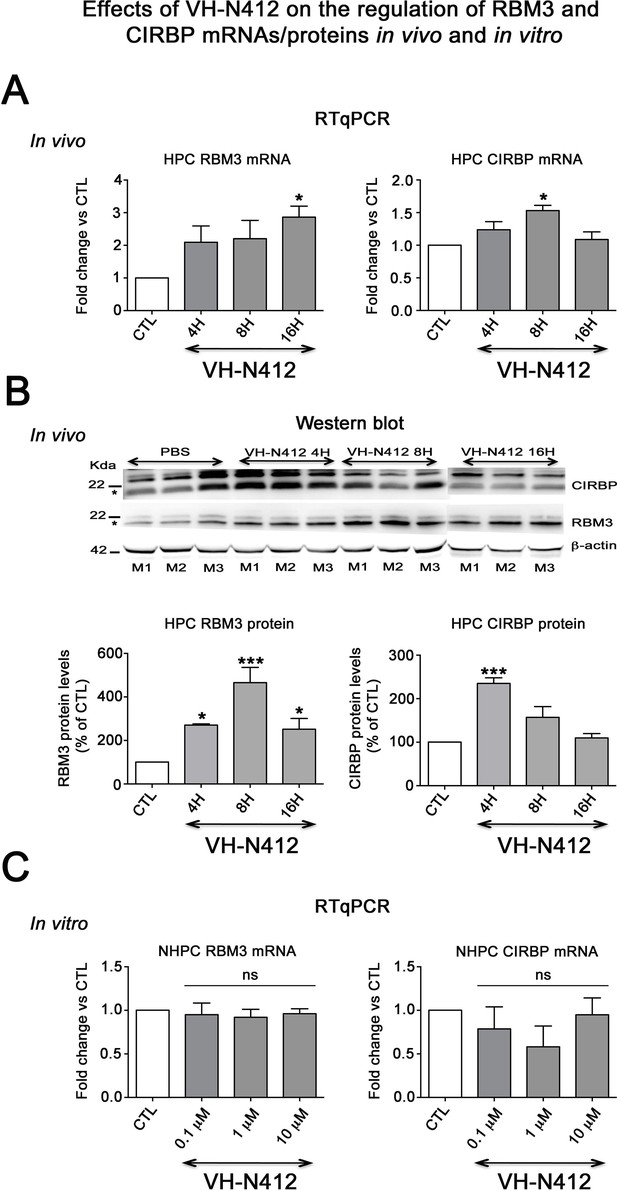
Effects of VH-N412 on the regulation of RBM3 and CIRBP mRNAs/proteins in the hippocampus and in vitro.
(A) Histograms related to analyses of RBM3 and CIRBP mRNA levels in the hippocampal tissue of CTL and mice treated with VH-N412 at 4, 8, and 16 hr by RT-qPCR. Note that VH-N412 treatment increased the expression of RBM3 at 16 hr while CIRBP peaked at 8 hr compared to CTL mice. (B) Western blots of CIRBP and RBM3 protein expression in VH-N412-treated mice at the same time points as above. Note that CIRBP and RBM3 were expressed predominantly at their expected molecular weight (MW) of ~18 kDa (stars). However, bands of higher MW between ~22 kDa (RBM3) and~25 kDa (CIRBP) were also detected in agreement with Rosenthal et al., 2017; Zhu et al., 2024. Similar patterns of expression of the higher MW bands compared to the ~18 kDa band strongly suggest that they correspond to either different isoforms for each protein or result from post-translational modifications. Protein levels were normalized with β-actin (~42 kDa, star) in all samples, and samples from mice treated with VH-N412 at 4 (n=3; lanes 4–6), 8 (n=3; lanes 7–9), and 16 hr (n=3; lanes 10–12) were normalized with CTL mice treated with PBS (CTL PBS; n=3; lanes 1–3). The graphs correspond to quantification of the western blots. RBM3 is upregulated at 4, 8, and 16 hr in VH-N412-treated mice, whereas CIRBP is upregulated at 4 hr after treatment. These results indicated that in vivo, VH-N412-induced hypothermia was associated with a regulation of cold stress proteins. (C) In vitro quantification of mRNA levels of RBM3 and CIRBP analyzed by RT-qPCR after treatment with different concentrations of VH-N412 (0.1, 1, and 10 μM) of cultured hippocampal neurons. VH-N412 did not regulate RBM3 and CIRBP mRNA at all concentrations used in our cultured hippocampal neurons.
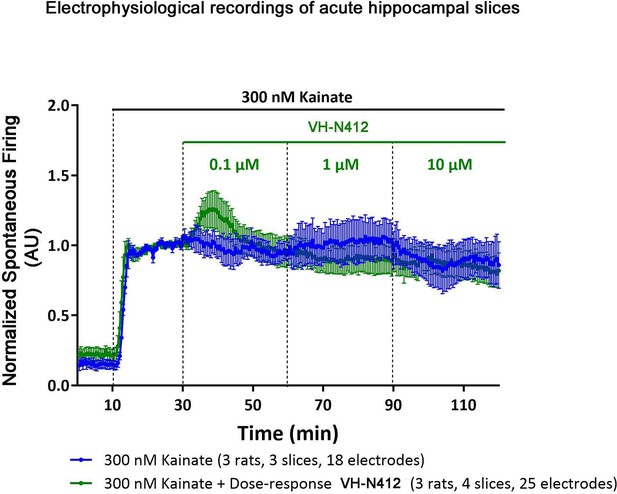
MEA electrophysiological recordings of acute hippocampal slices stimulated with KA in the presence or absence of VH-N412.
KA (300 nM) and VH-N412 (0.1, 1, and 10 μm) were added to the perfusion solution to assess the effects of VH-N412 compound on KA-induced increase of neuronal firing. Individual data from independent experiments were pooled and the mean values of the normalized spike rates (± SEM) were plotted as a function of time (before and after exposure to VH-N412). The control values (KA alone) were averaged from 3 rats, 3 slices, and 18 electrodes. The dose-response curves from the KA+VH-N412-treated slices were averaged from 3 rats, 4 slices, and 25 electrodes. In control conditions (in blue), the normalized firing rate was 0.86±0.17 at the end of the experiment. When VH-N412 was applied at increasing doses of 0.1, 1, and 10 μM over a 30 min period (in green), the KA-induced increase in firing rate in CA1 did not change significantly.
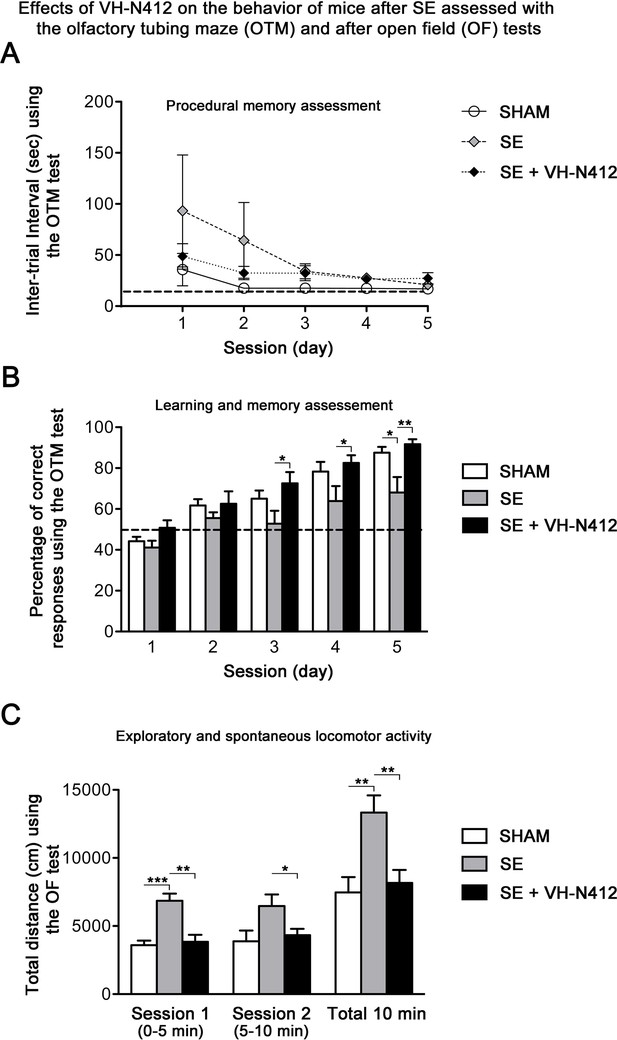
Following SE, VH-N412 preserved hippocampus-dependent learning and memory and normal locomotor activity.
(A and B) Learning and memory performance was assessed from 3 to 4 weeks after SE using the olfactory tubing maze (OTM). (A) Illustrates the mean ITI between the 12 trials in the OTM (in seconds, ± SEM). The dashed line indicates the minimum fixed ITI (15 s). There was no difference between groups on the ITI. (B) Mean percentage of correct responses obtained in the OTM during five training sessions of 12 trials per day. The dashed line denotes the chance level (%). From the second session, all animal groups had learned and memorized the test tasks. Only after the fifth session did the epileptic SE mice (n=6) show a significant impairment in memorization and learning while SE mice treated with VH-N412 showed similar performance to that of SHAM mice (n=10). (C) Locomotor activity using the open field (OF) test. (C) Illustrates the mean traveled distance in centimeters during two consecutive 5 min sessions (sessions 1 and 2) using OF test. SE mice (n=6) displayed a strong and significant hyperactivity in comparison with SHAM (n=10) while SE+VH-N412 mice (n=10) exhibited significant reduced exploratory and spontaneous locomotor activity, similar to that observed in SHAM mice.
-
Figure 9—source data 1
Uncropped immunoblot exposures displayed in Figure 9B.
Molecular weight ladders (black) depict separation range. Black boxes represent cropping as shown in Figure 9B. Membranes were physically cut (or not) following transfer and immunoblotted with different antibodies. White stars indicate the approximate size of different proteins of interest. Raw images are available on Dryad at https://doi.org/10.5061/dryad.nzs7h451x.
- https://cdn.elifesciences.org/articles/100527/elife-100527-fig9-data1-v1.tif
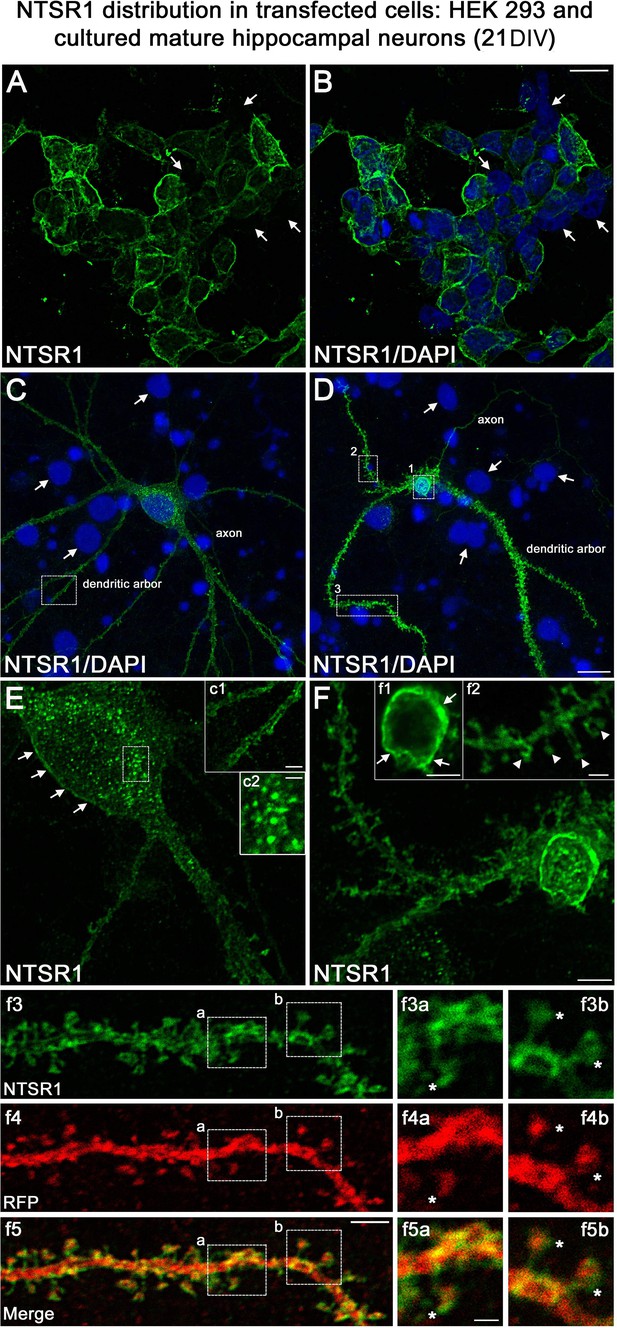
Validation of NTSR1 antibody.
The specificity of the goat polyclonal NTSR1 antibody was assessed by using transfection experiments for 43 hr followed by immunocytochemistry in different cell types including (A and B) human HEK 293 cells and (C and D) rat cultured mature hippocampal neurons (21 DIV). Cell nuclei were labeled with DAPI (blue). Both HEK 293 and hippocampal neurons displayed stronger NTSR1 immunolabeling (green) after transfection with a plasmid construct encoding rat NTSR1 (see cells double labeled for NTSR1 and DAPI), compared to non-transfected cells (see arrows, cells labeled for DAPI but not for NTSR1). Moreover, both types of cells exhibited high NTSR1 immunostaining within the cell body with a punctate pattern (see c2) and at the plasma membrane (see arrows in E, F, c1, and f1) as expected for receptor localization. The axons, the dendritic arbors, and their protuberances (see arrowheads in f2) of hippocampal neurons were also immunostained. f3 and f4 correspond to the dendritic portion of a neuron overexpressing NTSR1 (green) and RFP (red). RFP was used to outline the morphology of neurons including the dendrites and their dendritic spines. f5 corresponds to the merge of panels f3 and f4. Panels f3a and f3b correspond to the high magnification of NTSR1 labeling in two distinct areas of a dendrite (boxed in f3, f4, f5). Panels f4a and f4b correspond to RFP labeling in these same areas. Panel f5a corresponds to the merge of f3a and f4a. Panel f5b corresponds to the merge of f3b and f4b. Double immunostaining of NTSR1/RFP confirmed that NTSR1 was located in dendritic spines. However, some of the NTSR1 immunolabeling was slightly shifted relative to RFP (see stars in panels f3a to f5b) suggesting NTSR1 localization in the cell membrane. Scale bars: 20 μm in A and B; 5 μm in C, D, E, F, c1, f1–f5; 2 μm in f3a–f5b.
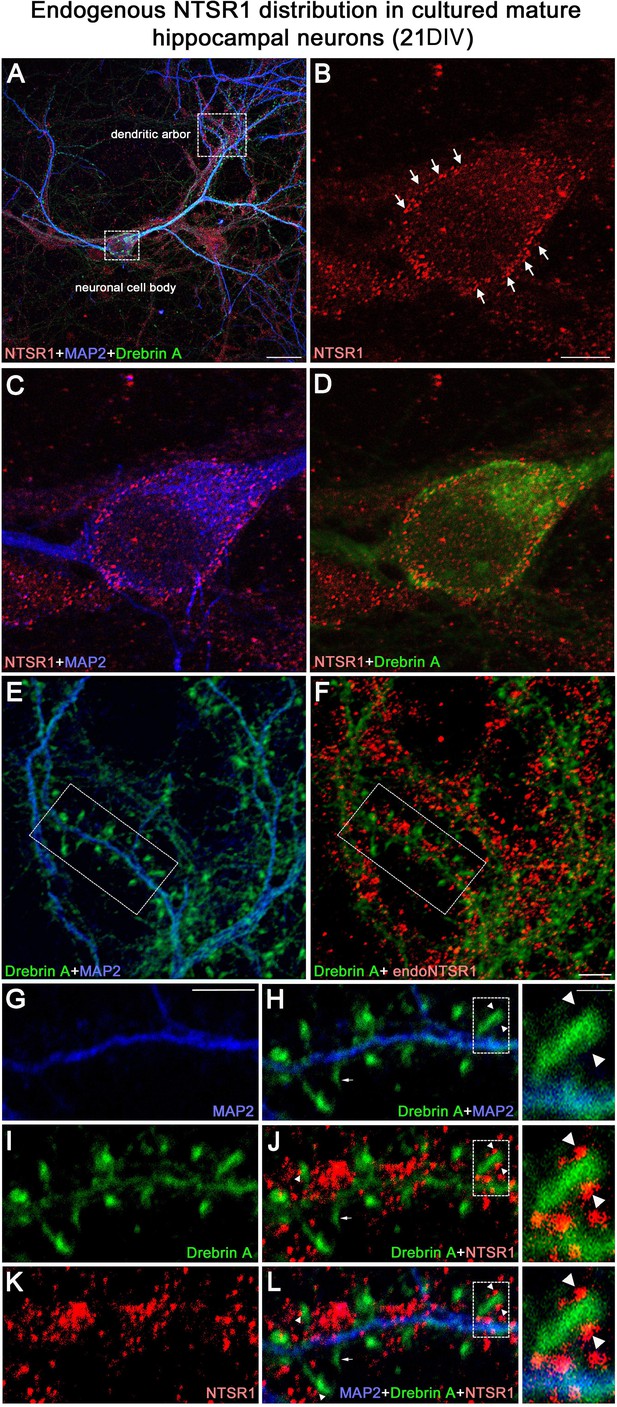
Expression of endogenous NTSR1 and its localization in mature cultures of hippocampal neurons.
Twenty-one-day-old (21 DIV) cultured hippocampal neurons were fixed and immunostained sequentially with antibodies against NTSR1 (red), MAP2 (blue), and drebrin E/A (green). Panels A, L and high magnification of the boxed area in L correspond to the merge of NTSR1/MAP2/drebrin. (E, H and F, J) Merge of MAP2/drebrin and NTSR1/drebrin respectively. At high magnification of the boxed-in area in A (pyramidal neuron cell body) and F (dendrites), mature cultured hippocampal neurons displayed endogenous NTSR1 with a punctate pattern (B, C, D, F, J, K, L, red) similar to observations in transfected cells (Figure 10). Enlargement of the boxed-in area in F and J illustrated that NTSR1 (red) was closely apposed to dendritic shafts and dendritic spines, presumably at the level of the cell membrane, as revealed by neuronal and dendritic shaft marker MAP2 (E and H, blue) and dendritic spine marker drebrin (I, J, and L, green, see arrows in high-magnification insets). Note that no NTSR1 immunostaining was observed in filopodia (see small white arrows in H, J, and L). Scale bars: 20 μm in A; 5 μm in B–L; 1 μm in boxed-in area in H, J, and L panels.
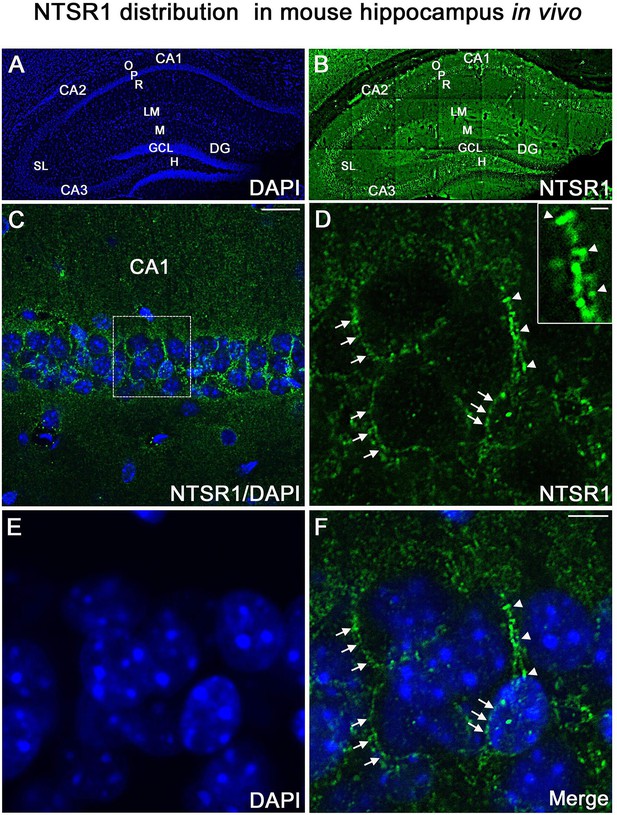
NTSR1 immunolabeling in mice hippocampal formation.
(A and B) Correspond to low-magnification pictures showing coronal section of the mouse dorsal hippocampus processed with DAPI (blue), used to highlight the different cell layers of the hippocampus and NTSR1 (green) antibody respectively. Moderate to strong NTSR1 immunolabeling levels were found in the stratum (O), (R), (LM), (SL), in pyramidal neurons of CA1, CA2, and CA3, M, H, and GCL of the DG. (C) Merge of DAPI and NTSR1 (green) of the CA1 area at high magnification. (D–F) High magnification of the boxed-in CA1 area illustrating pyramidal neurons immunolabeled with NTSR1 antibody (D, green) and counterstained with DAPI (E and F, blue). NTSR1 immunoreactivity was observed in the cell bodies, at the cell membranes (see arrowheads in E and F), as well as at the proximal dendrites of CA1 pyramidal neurons (see arrows in D and F). NTSR1 immunolabeling displayed a punctate pattern. Note that several dendritic protuberances displayed high levels of NTSR1 immunolabeling (see arrows in inset in D). (F) Merge of NTSR1/DAPI. Scale bars: 225 μm in A and B; 20 μm in C; 5 μm in D-F; 1 μm in inset in D.
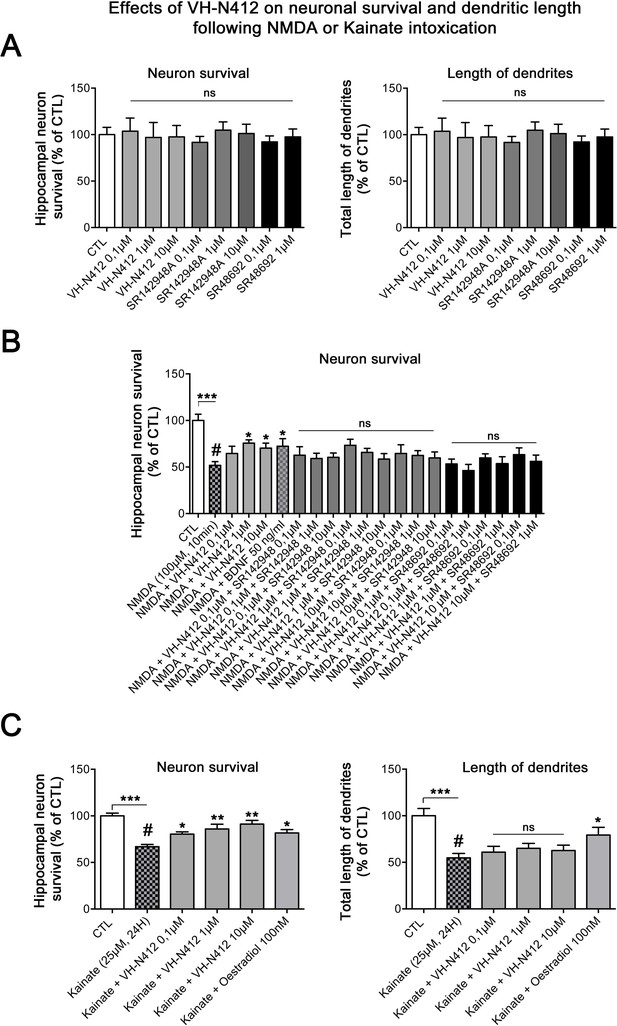
In vitro effects of VH-N412 on hippocampal neuronal survival and total dendrite length following NMDA or KA intoxication.
(A) Histograms illustrate the effects of VH-N412 and two NTSR antagonists, SR142948A and SR48692, on hippocampal neuronal survival (left histogram) and on total dendrite length (right histogram). These compounds alone showed no toxic effects on hippocampal neuronal survival and on total dendrite length at all concentrations used. (B) Illustrates the effects of VH-N412 on survival of primary hippocampal neurons injured by NMDA. VH-N412 promoted neuronal survival at 1 and 10 μM with similar potency as that of BDNF 50 ng/mL. Antagonizing NTSR by SR142948A and SR48692 blocks the neuroprotective effect of VH-N412 in the neuronal cultures. (C) Illustrates the effects of VH-N412 on hippocampal neuronal survival (left panel) and on total dendrite length (right panel) following KA intoxication (25 μM). VH-N412 promoted neuronal survival at all concentrations (0.1, 1, and 10 μM) with similar potency as that of oestradiol at 100 nM. Following KA injury, VH-N412 did not display any significant effects on the total length of dendrites while oestradiol did.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (M. musculus) | Swiss CD-1 mice | Janvier laboratories, France | Cat#: RjOrl:SWISS | Young adult males 5 week old |
| Strain, strain background (M. musculus) | FVB/N mice | Janvier laboratories, France | Cat#: FVB/NRj | Adult males 12-week-old |
| Strain, strain background (Rattus) | Sprague Dawley | Janvier laboratories, France | Cat#: RRID:MGI:5651135 | Young adult, 3–4 week old |
| Strain, strain background (Rattus) | Wistar | Janvier laboratories, France | Cat#: RjHan:WI | Young adult, 3–4 week old |
| Cell line (Homo-sapiens) | HEK293 | ECACC, Salisbury, UK | Cat#: RRID:CVCL_0045 | |
| Transfected construct (Homo-sapiens) | Ntsr1 (untagged ORF) - Human neurotensin receptor 1 (Ntsr1) plasmid construct | Origene, Rockville, Maryland, USA | Cat#:SKU SC313374 | |
| Transfected construct (Rattus) | Ntsr1 (untagged ORF) - Rat neurotensin receptor 1 (Ntsr1) plasmid construct | Origene, Rockville, Maryland, USA | Cat#:SKU RN202030 | |
| Biological sample | Human NTSR1 membrane homogenates | This paper | ||
| Biological sample | Rat NTSR1 membrane homogenates | This paper | ||
| Antibody | Neuronal nuclear antigen (NeuN) Clone A60 | Millipore, Darmstadt, Germany | Cat#: RRID:AB_2298772 | Dilution 1:1000 |
| Antibody | Mouse glial fibrillary acidic protein GFAP Clone GA5 | Millipore, Darmstadt, Germany | Cat#: MAB360 | Dilution 1:1000 |
| Antibody | Rabbit anti-ionized calcium binding adaptor molecule 1 Iba1 | Wako Pure Chemical Industries, Osaka, Japan | Cat#: 019–19741 | Dilution 1:1000 |
| Antibody | Rabbit anti-vesicular zinc transporter 3 (ZnT-3) | Synaptic System, Goettingen, Germany | Cat#: RRID:AB_2744656 | Dilution 1:500 |
| Antibody | Alexa 488-conjugated goat anti-rabbit IgG | ThermoFisher Scientific, Waltham, MA, USA | Cat#: RRID:AB_143165 | Dilution 1:200 |
| Antibody | Alexa 488-conjugated goat anti-mouse IgG | ThermoFisher Scientific, Waltham, MA, USA | Cat#: RRID:AB_346865 | Dilution 1:200 |
| Antibody | Rabbit anti-RBM3 | Proteintech, Manchester, UK | Cat#: 14363–1-AP | Dilution 1:500 |
| Antibody | Rabbit anti-CIRBP | Proteintech, Manchester, UK | Cat#: 10209–2-AP | Dilution 1:1000 |
| Antibody | Mouse anti-b-actin | Santa-Cruz, TX, USA | Cat#: SC1615 | Dilution 1:5000 |
| Antibody | Horseradish peroxidase-conjugated donkey anti-mouse antibody | Jackson ImmunoResearch, West Grove, PA, USA | Cat#: RRID:AB_2340770 | Dilution 1:10000 |
| Antibody | Horseradish peroxidase-conjugated donkey anti-rabbit antibody | Jackson ImmunoResearch, West Grove, PA, USA | Cat#: RRID:AB_10015282 | Dilution 1:10000 |
| Antibody | Goat polyclonal anti-NTSR1 (NTR1) | Santa-Cruz, TX, USA | Cat#: SC-7598 | Dilution 1:200 |
| Antibody | Mouse anti-MAP2, Clone HM-2 | Sigma-Aldrich (St. Louis, MO, USA) | Cat#: M-4403 | Dilution 1:1000 |
| Antibody | Rabbit anti-Drebrin E/A | Sigma-Aldrich (St. Louis, MO, USA) | Cat#: AB10140 | Dilution 1:1000 |
| Antibody | Donkey anti-goat IgG highly cross-adsorbed AlexaFluor A594 | Jackson ImmunoResearch, West Grove, PA, USA | Cat#: RRID:AB_2340433 | Dilution 1:800 |
| Sequence-based reagent | TaqMan probe, mouse CIRBP (Cold Inducible RNA Binding Protein) | ThermoFisher Scientific, Waltham, MA, USA | Cat#: Mn00483336 | |
| Sequence-based reagent | TaqMan probe, rat CIRBP (Cold Inducible RNA Binding Protein) | ThermoFisher Scientific, Waltham, MA, USA | Cat#: Rn00579806 | |
| Sequence-based reagent | TaqMan probe, mouse RBM3 (RNA Binding Motif Protein 3) | ThermoFisher Scientific, Waltham, MA, USA | Cat#: Mn00812518 | |
| Sequence-based reagent | TaqMan probe, rat RBM3 (RNA Binding Motif Protein 3) | ThermoFisher Scientific, Waltham, MA, USA | Cat#: Rn01525079 | |
| Sequence-based reagent | TaqMan probe, mouse GAPDH (Glyceraldehyde-3 phosphate dehydrogenase) | ThermoFisher Scientific, Waltham, MA, USA | Cat#: Mn01253033 | |
| Sequence-based reagent | TaqMan probe, rat GAPDH (Glyceraldehyde-3 phosphate dehydrogenase) | ThermoFisher Scientific, Waltham, MA, USA | Cat#: Rn01253033 | |
| Peptide | peptide 22: [cMPRLRGC]c | This paper | Malcor et al., 2012 | |
| Peptide | Ac-cMPRLRGC-G-OH | This paper | ||
| Peptide | Ac-[cMPRLRGC]c-G-OH | This paper | ||
| Peptide | pELYENKPRRPYIL-OH | This paper | ||
| Peptide | VH-N21 | This paper | ||
| Peptide | VH04129 [cM”Pip”RLR”Sar”C]c | This paper | ||
| Peptide | VH-N41 | This paper | ||
| Peptide | VH-N412 | This paper | ||
| Peptide | [3H]-neurotensin | Perkin Elmer | https://resources.perkinelmer.com/lab-solutions/resources/docs/sht_008241d_01.pdf | |
| Recombinant protein | Recombinant human LDLR (His-tagged) | Sino Biological (Beijing, China). | Cat. #: 10,231 H08H | |
| Commercial assay or kit | DC protein assay kit | Bio-Rad, Hercules, CA, USA | Cat. #: 5000111 | |
| Commercial assay or kit | Nucleospin RNA plus kit | Macherey Nagel, Allentown, PA, USA | Cat. #:740984.50 | |
| Commercial assay or kit | ECL chemiluminescence kit | GE Healthcare | Cat. #: 28980926 | |
| Chemical compound | Fmoc-Gly-Wang Resin (100–200 mesh) | Iris Biotech | Cat. #: WAA11313 | |
| Chemical compound | Fmoc-Leu-Wang Resin (100–200 mesh) | Iris Biotech | Cat. #: WAA11316 | |
| Chemical compound | Lipofectamine 2000 | ThermoFisher Scientific, Waltham, MA, USA | Cat. #: 11668027 | |
| Chemical compound | Kainic acid (kainate, KA) | Abcam, Cambridge, UK | Cat. #: ab120100 | |
| Chemical compound | Doliprane (Paracetamol) | Sanofi, Gentilly, France | Doliprane gel, 2.4% | |
| Chemical compound | Diazepam (ValiumR) | Roche, Basel, Switzerland | 5 mg/ml | |
| Chemical compound | Pentobarbital (Nembutal) | Sigma-Aldrich (St. Louis, MO, USA) | Cat. #: 76-74-4 | 700 mg/kg |
| Chemical compound | Fluoro-Jade C (FJC) | Sigma-Aldrich (St. Louis, MO, USA) | Cat#: AG325 | Dilution 1:10000 |
| Chemical compound | BDNF | PAN-Biotech, Aidenbach, Germany | Cat. #:CB-1115001 | |
| Chemical compound | Oestradiol | Sigma-Aldrich (St. Louis, MO, USA) | Cat. #: E9875 | |
| Chemical compound | N-Methyl-D-aspartic acid (NMDA) | Sigma-Aldrich (St. Louis, MO, USA) | Cat. #:6384-92-5 | |
| Software, algorithm | ImageJ softwares | Free access software | https://imagej.net/ij/ | |
| Software, algorithm | LabVIEW software | National Instruments (NI) Austin, TX 78759–3504 USA | https://www.ni.com/fr-fr/shop/product/labview.html?srsltid=AfmBOoo7GoVIL1yty98ffFmJFGqN8m64ST5WMFlMol6lBqQDYAtbLPcs | |
| Software, algorithm | Viewpoint VideoTrack version 3.0 | Lyon, France | https://www.viewpoint.fr/product/rodent/rodents-behavior-monitoring/videotrack | |
| Software, algorithm | 7500 v2.0 software | Applied Biosystems | https://www.thermofisher.com/fr/fr/home/technical-resources/software-downloads/applied-biosystems-7500-real-time-pcr-system.html | |
| Software, algorithm | biostaTGV software | Free access software, | http://biostatgv.sentiweb.fr | |
| Software, algorithm | IBM SPSS Statistics for Windows, Version 20.0. | IBM Corp. Released 2011. Armonk, NY, USA | https://www.ibm.com/support/pages/downloading-ibm-spss-statistics-20 | |
| Software, algorithm | Zen software | Zeiss, Jena Germany | https://www.zeiss.com/microscopy/en/products/software/zeiss-zen.html | |
| Software, algorithm | Developer software used with InCell 2000 image system | GE Healthcare, Pittsburgh, PA, USA | https://download.cytivalifesciences.com/cellanalysis/download_data/incell/incell_docs/IN%20Cell%20Analyzer%202200%20User%20Manual.pdf |
Additional files
-
Supplementary file 1
Analytical characterization of peptide-neurotensin (NT) conjugates.
- https://cdn.elifesciences.org/articles/100527/elife-100527-supp1-v1.docx
-
Supplementary file 2
Hypothermic response to intravenous (i.v.) administration of peptide-neurotensin (NT) conjugates in mice.
- https://cdn.elifesciences.org/articles/100527/elife-100527-supp2-v1.docx
-
Supplementary file 3
Comparison of mean body temperature changes between SHAM, status epilepticus (SE), SE+VH-N412, SE+neurotensin (NT) (8–13), and SE+diazepam (DZP).
- https://cdn.elifesciences.org/articles/100527/elife-100527-supp3-v1.docx
-
Supplementary file 4
Comparison of mean seizure intensity score between SHAM, status epilepticus (SE), SE+VH-N412, SE+neurotensin (NT) (8–13), and SE+diazepam (DZP).
- https://cdn.elifesciences.org/articles/100527/elife-100527-supp4-v1.docx
-
Supplementary file 5
Mouse and rat TaqMan probes used for qPCR analysis.
- https://cdn.elifesciences.org/articles/100527/elife-100527-supp5-v1.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/100527/elife-100527-mdarchecklist1-v1.docx





