UBR-1 deficiency leads to ivermectin resistance in Caenorhabditis elegans
Figures
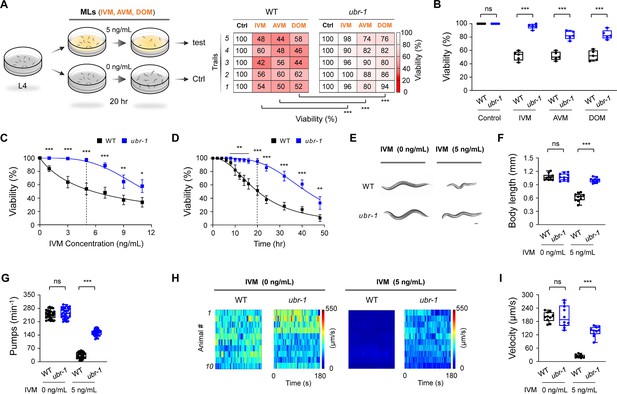
ubr-1 exhibits IVM-resistant phenotypes.
(A) Left: Schematic representation of the IVM resistance test in C. elegans. Well-fed L4 stage animals were transferred to plates containing OP50 bacteria with or without macrocyclic lactones (MLs) for 20 hr. Right: Representative grid plots illustrating the viability of wild-type and ubr-1 animals in response to ivermectin (IVM, 5 ng/mL), avermectin (AVM, 5 ng/mL), and doramectin (DOM, 5 ng/mL). We used shades of red to represent worm viability on each experimental plate (n=50 animals per plate), with darker shades indicating lower survival rates. The viability test was repeated at least five times (five trials). (B) Quantification analysis of the viability of wild-type and ubr-1(hp684) mutants exposed to different MLs. ubr-1(hp684) mutants exhibit resistance to various MLs. (C) Dose‒response curve depicting the viability of wild-type and ubr-1(hp684) mutants in the presence of varying concentrations of IVM. The IC50 was 5.7 ng/mL for the wild type and 11.6 ng/mL for the ubr-1 mutants. (D) Time-dependent effect of IVM exposure on animal viability. The IC50 values were 20.2 hr for the wild type and 42.1 hr for the ubr-1 mutants. (E) Representative images of worm size in the wild type and ubr-1(hp684) mutants with or without IVM treatment. Scale bar, 50 µm. (F) Quantitative analysis of body length in different genotypes with or without IVM. (G) Quantification of the average pharynx pump number in animals with or without IVM. (H) Raster plots illustrating the locomotion velocity of individual animals in the absence (0 ng/mL) and presence (5 ng/mL) of IVM. n=10 animals in each group. (I) Quantification of the average velocity in different genotypes with or without IVM treatment. ns, not significant, *p<0.05, **p<0.01, ***p<0.001 by Student’s t test. The error bars represent the SEM.
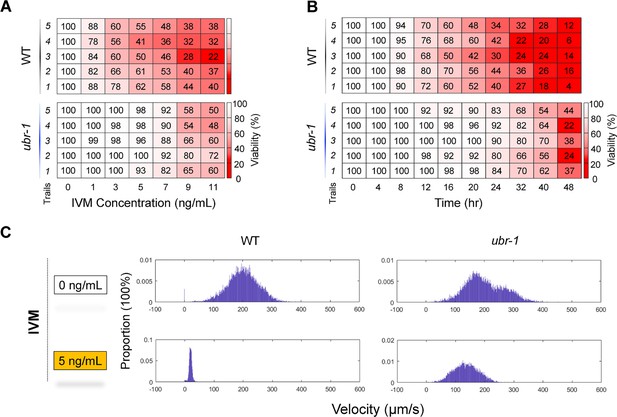
Dose- and time-dependent patterns of IVM resistance observed in ubr-1 mutants.
(A, B) Grid plots depicting the viability of wild-type and ubr-1 animals treated with different concentrations and durations of IVM. The IVM resistance of ubr-1 is clearly dose- and time- dependent. The shades of red represent worm viability, with darker shades indicating lower survival rates, based on 100 animals per plate and at least five trials. (C) Distribution of locomotion velocity of wild-type and ubr-1 mutants in the absence and presence of IVM (5 ng/mL).
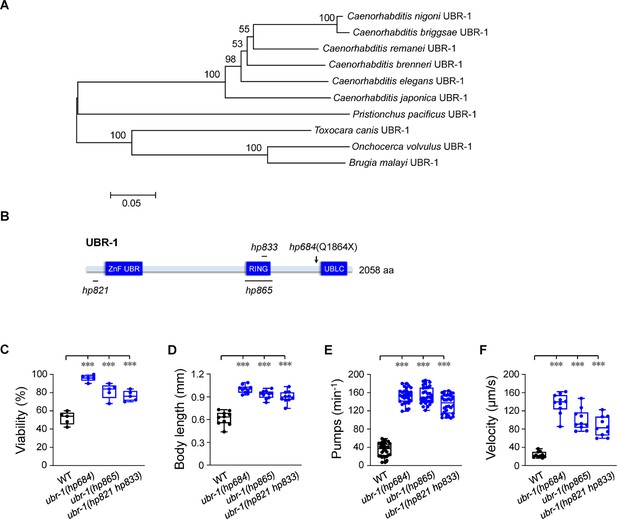
IVM resistance across various ubr-1 mutant alleles.
(A) UBR-1 phylogenetic tree predicting potential functional conservation of ubr-1 in pathogenic helminthes. (B) Structure of the UBR-1 protein with significant mutants used in this study. Three predicted functional domains (ZnF UBR, RING, UBLC) and amino acid substitutions in C. elegans alleles are denoted. (C–F) Quantification of viability, body length, pharyngeal pumps, and locomotion velocity in different ubr-1 mutant alleles in the presence of IVM (5 ng/mL). All the mutants exhibited varying degrees of IVM resistance. For the viability test in (C), n=50 animals per plate, which was repeated at least 5 times (5 trials), n≥10 animals in (D–F). ***p<0.001 by one-way ANOVA. Error bars, SEM.
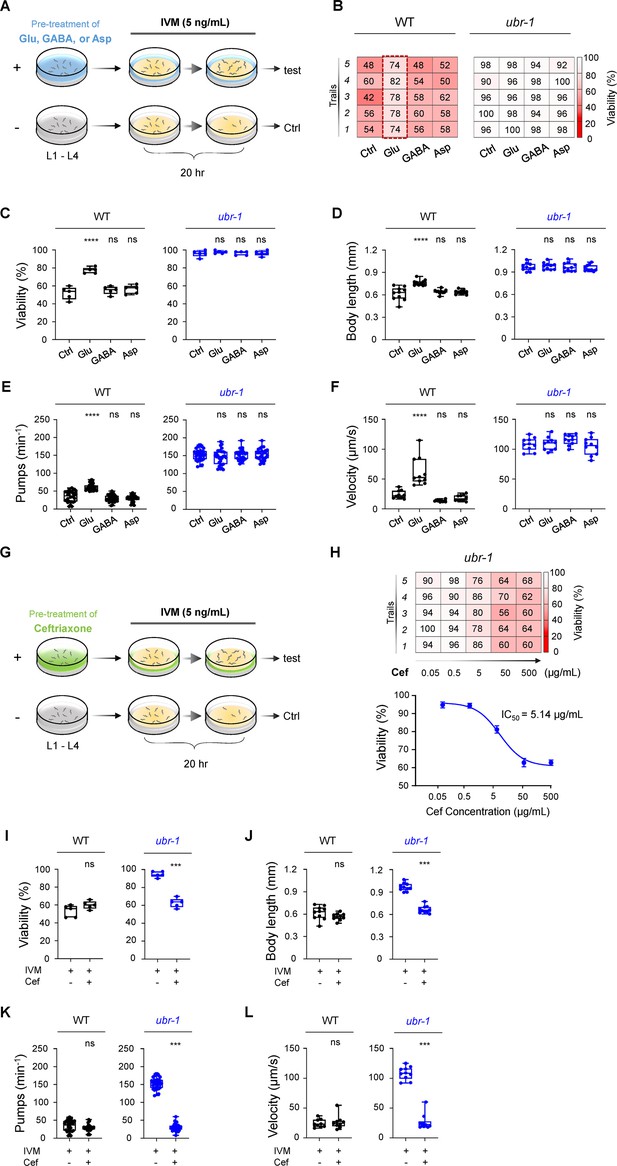
Glutamate mimics IVM resistance in wild-type N2 animals, and ceftriaxone fully reverses IVM resistance in ubr-1 mutants.
(A) Modified schematic representation of the pretreatment approach in the IVM resistance test in C. elegans. Synchronized L1 animals were cultured on plates containing OP50 bacteria and pretreated with glutamate (Glu, 5 mM), γ-aminobutyric acid (GABA, 5 mM), or aspartate (Asp, 5 mM) until they reached the L4 stage. Pretreated animals were then transferred to plates containing additional IVM (5 ng/mL) for 20 hr. (B) Representative grid plots illustrating the viability of wild-type and ubr-1 animals pretreated with different glutamate metabolites. (C–F) Quantitative analysis of viability, body length, pharyngeal pump rate, and locomotion velocity in wild-type and ubr-1 mutants following exposure to various glutamate metabolites. Wild-type worms treated with glutamate displayed resistance to IVM. However, glutamate did not affect the IVM resistance of the ubr-1 mutant. ns, not significant, ****p<0.0001 by one-way ANOVA in C-F. (G) Diagram illustrating the ceftriaxone pretreatment procedure in the IVM resistance test. Synchronized L1 animals were cultured on OP50-fed plates with or without ceftriaxone (50 μg/mL) until the L4 stage. The animals were subsequently transferred to plates seeded with additional IVM (5 ng/mL) for 20 hr. (H) Upper: Representative grid plots depicting the viability of ubr-1 animals at different concentrations of ceftriaxone. Bottom: Dose‒response curve illustrating the amount of ceftriaxone required for viability in ubr-1(hp684) mutants (IC50=5.1 μg/mL). (I–L) Quantification of viability, body length, pharyngeal pumping, and locomotion velocity in the wild type and ubr-1 mutants in the absence and presence of ceftriaxone. Ceftriaxone fully restored the sensitivity of ubr-1 mutants to IVM, comparable to the levels observed in wild-type N2 animals. ns, not significant, ***p<0.001 by Student’s t test in I-L. Error bars represent SEM.
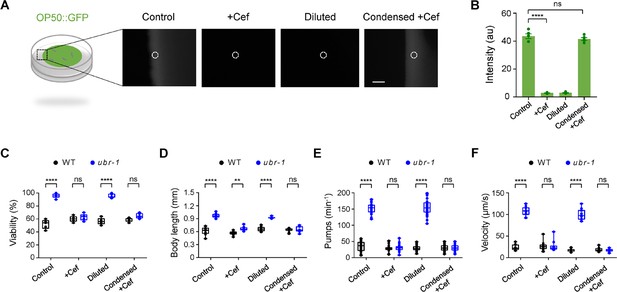
Ceftriaxone restores IVM sensitivity in ubr-1 mutants through mechanisms beyond its antibacterial properties.
(A) OP50 bacteria expressing GFP (OP50::GFP) were used to quantitatively analyze the antibiotic activity of ceftriaxone under the same experimental conditions. Left: Schematic diagram of the NGM plate with OP50::GFP. Right: OP50::GFP fluorescence intensity under different culture conditions. Control, normal NGM plates with standard culture conditions;+Cef, normal OP50::GFP seeded plate with ceftriaxone (50 μg/mL); Diluted, 15 x diluted OP50::GFP seeded plates; Condensed +Cef, 20 x condensed OP50::GFP seeded plate with ceftriaxone (50 μg/mL). (B) Quantification of OP50::GFP fluorescence intensity in plates grown under different culture conditions. (C–F) Quantification of all IVM resistance phenotypes under different OP50 levels. ubr-1’s IVM resistance is independent of the diluted OP50. Increasing OP50 levels also did not alter the ability of Cef to restore the IVM sensitivity of ubr-1. For the viability test in (C), n=50 animals per plate, which was repeated at least five times (five trials), n≥10 animals in (C–E). Black plots: wild-type, blue plots: ubr-1 mutants. ns, not significant, **p<0.01, ****p<0.0001 by one-way ANOVA. Error bars, SEM.
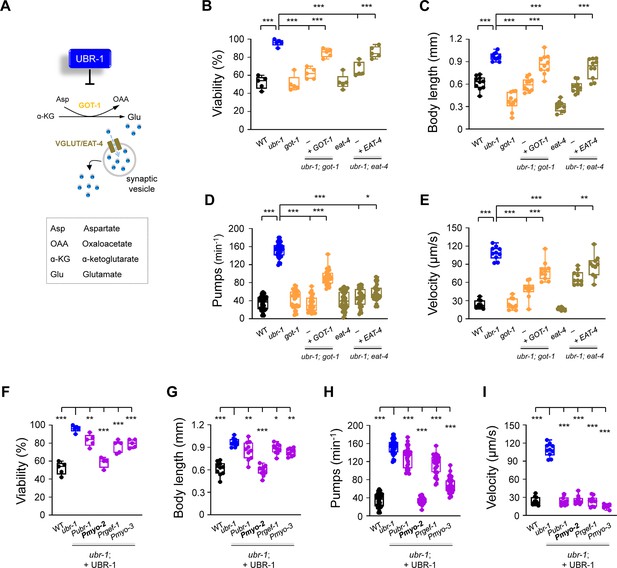
Deletion of got-1 and eat-4 suppresses ubr-1’s IVM resistance.
(A) Schematic of the pathway for glutamate synthesis through transaminase GOT-1 and loading via vesicular transport VGLUT/EAT-4. (B–E) Quantification of viability, body length, pharyngeal pump rate, and locomotion velocity in different genotypes. The removal of got-1 and eat-4 significantly restored sensitivity to IVM (5 ng/mL) in ubr-1 mutants, whereas the re-expression of GOT-1 (+GOT-1) and EAT-4 (+EAT-4) partially reinstated IVM resistance in the respective double mutants. (F–I) IVM resistance phenotypes were rescued by expression of UBR-1 (+UBR-1) driven by its own promoter (Pubr-1) or the pharynx muscle-specific promoter (Pmyo-2). For the viability test in F, n=50 animals per plate and repeated at least five times (five trials), n≥10 animals in G-I, *p<0.05, **p<0.01, ***p<0.001 by one-way ANOVA. All statistical analyses were performed against ubr-1 mutant. Error bars, SEM.
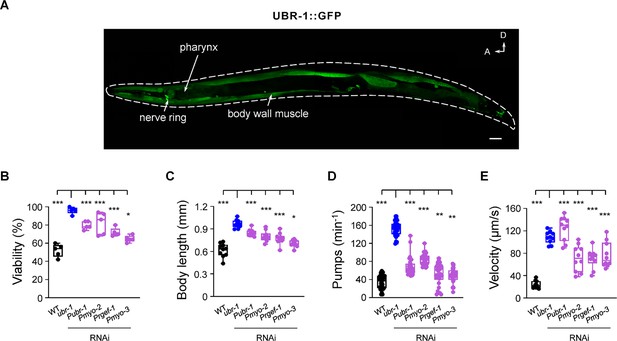
Knockdown of UBR-1 induces IVM resistance phenotypes.
(A) Expression of functional UBR-1::GFP, driven by its endogenous promoter, was observed predominantly in the pharynx, head neurons, and body wall muscles with weaker expression detected in vulval muscles and the intestine. Scale bar, 20 µm. (B–E) Double-stranded RNA (dsRNA) interference was used to suppress gene expression in specific tissues (Materials and methods). IVM resistance phenotypes were mimicked by RNAi knockdown of UBR-1 driven by its own promoter (Pubr-1), the pharynx muscle-specific promoter (Pmyo-2), the pan-neuronal promoter (Prgef-1) or body wall muscle (Pmyo-3). For the viability test in B, n=50 animals per plate and repeated at least five times (five trials), n≥10 animals in C-E, *p<0.05, **p<0.01, ***p<0.001 by one-way ANOVA. All statistical analyses were performed against ubr-1 mutant. Error bars, SEM.
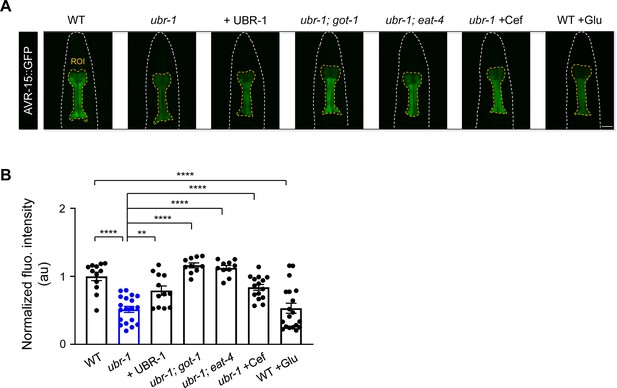
Downregulation of AVR-15 in ubr-1 mutants.
(A) Representative fluorescence intensity of AVR-15 in different genotypes. (B) Quantitative analysis of normalized fluorescence intensity revealed a reduction in AVR-15::GFP expression in ubr-1 mutants. The diminished AVR-15 levels were rescued by overexpression of UBR-1 and genetically suppressed in got-1 and eat-4 mutants. Pharmacological intervention with ceftriaxone (50 μg/mL) successfully reinstated the pharyngeal expression of AVR-15::GFP. Conversely, glutamate administration reduced AVR-15 expression in the wild-type N2 strain. ROI, region of interest; n≥10 animals in each group. **p<0.01, ****p<0.0001 by one-way ANOVA. Error bars, SEM.
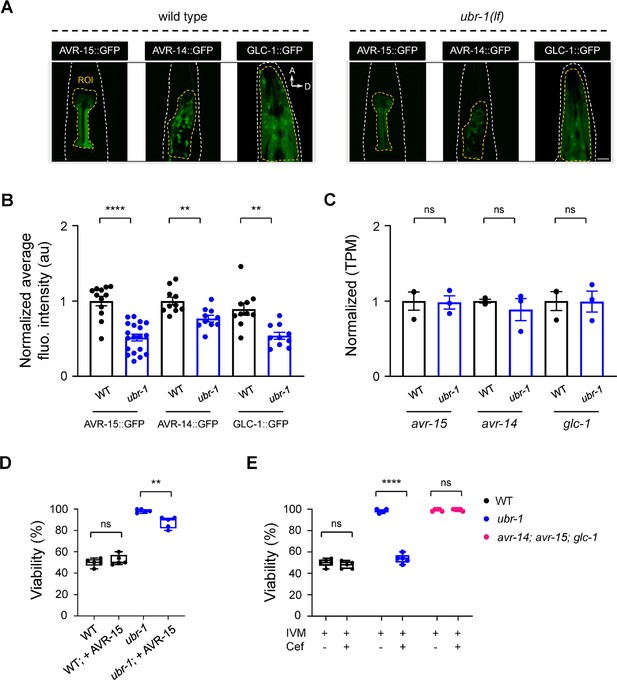
Downregulation of IVM-targeted GluCls in ubr-1 mutants.
(A) Expression pattern and representative fluorescence intensity of primary IVM-targeted GluCls in wild-type and ubr-1 animals. AVR-15::GFP was expressed primarily in the pharynx; AVR-14::GFP was expressed in head neurons; and GLC-1::GFP was expressed in the head muscles, pharynx and neurons. (B) Quantitative analysis of the normalized fluorescence intensity of these GluCls in different genetic backgrounds. The intensity of IVM-targeted GluCls was significantly reduced in ubr-1 mutants. (C) The normalized relative abundance of transcripts of the GluCl genes in wild-type and ubr-1 animals. (D) Overexpression of AVR-15 reduced the viability of ubr-1 mutant in IVM (5 ng/mL), thus partially restoring ubr-1’s IVM sensitivity. (E) Ceftriaxone (50 μg/mL) treatment rescued the IVM sensitivity in ubr-1, but not in avr-14; avr-15; glc-1 triple mutants. ROI, region of interest; n≥10 animals in each group. ns, not significant, **p<0.01, ****p<0.0001 by one-way ANOVA. Error bars, SEM.
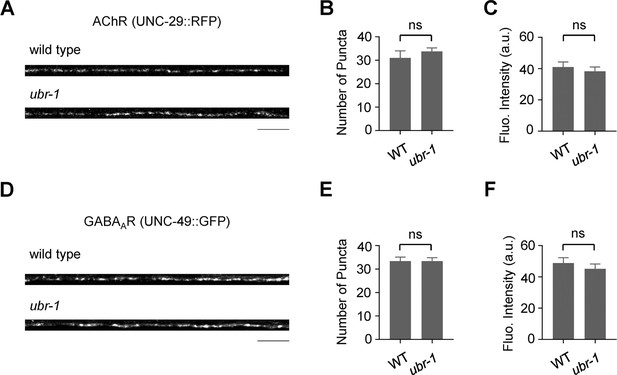
Localization and intensity of AChRs and GABAARs in ubr-1 mutants.
(A, D) Visualization and fluorescence quantification of the acetylcholine receptor (UNC-29) and GABAA receptor (UNC-49) in both wild-type and ubr-1 animals. (B–C, E–F) Statistical assessment of the puncta count and mean fluorescence intensity for the respective receptors across various genetic backgrounds revealed no notable differences in the distributions of AChR and GABAAR in ubr-1 mutants. ns, not significant; unpaired Student’s t test. The error bars represent the SEM.
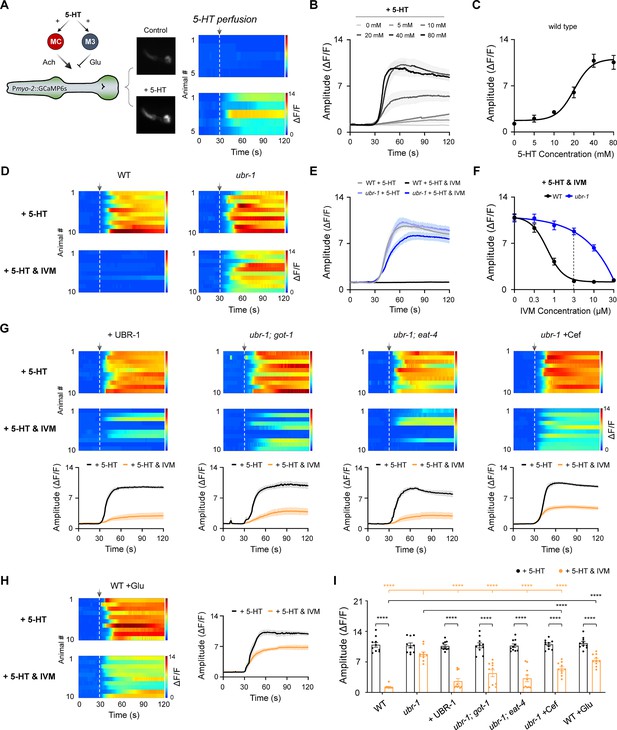
Reduced inhibition of serotonin-stimulated pharyngeal activity by IVM in ubr-1 mutants.
(A) Left: Schematic diagram of the pharynx motor circuit composed of an excitatory cholinergic motor neuron MC, an inhibitory glutamatergic motor neuron M3 and a group of compact muscles. Both types of motor neurons could be activated by serotonin (5-HT). Right: Representative pharynx muscle Ca2+ response and kymograph evoked by 5-HT (20 mM). The dashed lines aligned with the arrows denote the initiation of perfusion. (B) Average Ca2+ traces evoked by different concentrations of 5-HT. (C) Dose‒response curve of 5-HT for the pharynx muscle Ca2+ response in wild-type animals (EC50=20.1 mM). n≥5 animals in each group. (D) Baseline-subtracted kymograph of the 5-HT-evoked pharynx muscle Ca2+ response in wild-type and ubr-1 mutants. IVM (3 μM) completely inhibited the wild-type pharynx Ca2+ response, which was significantly reduced in ubr-1 mutants. (E) Average Ca2+ traces evoked by 5-HT in different genotypes with or without IVM. (F) The dose-dependent inhibitory effect of IVM on pharynx Ca2+ was attenuated in ubr-1 mutants (blue line) compared with wild-type animals (black line) (IC50=0.5 μM in WT, while IC50=10.1 μM in ubr-1). (G) The decreased IVM inhibition of serotonin-evoked pharynx activity in ubr-1 was rescued by the restoration of UBR-1 expression (+UBR-1), genetic removal of got-1 and eat-4, and pretreatment with ceftriaxone (+Cef, 50 μg/mL). (H) IVM-mediated inhibition of serotonin-evoked pharynx activity in wild-type animals was blocked by glutamate feeding (WT +Glu, 20 mM). n=10 animals in each group. (I) Quantification of the 5-HT-evoked peak amplitude of the pharynx muscle Ca2+ response in the different genotypes. Black stars: ****p<0.0001 was used to compare intragroup differences or pharmacological groups via Student’s t test; apricot stars: ****p<0.0001 was used to compare the differences with ubr-1 in the ‘+5 HT and IVM’ group via one-way ANOVA. Error bars, SEM.
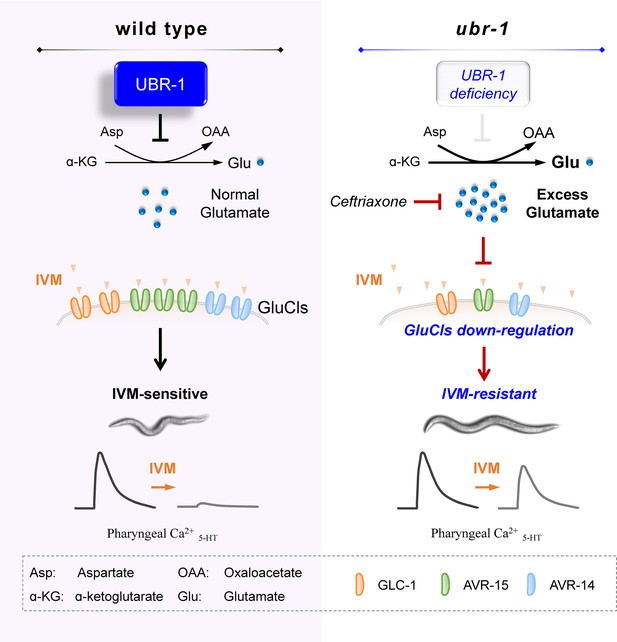
A working model.
In wild-type animals, functional UBR-1 helps maintain balanced glutamate levels by inhibiting transaminase GOT-1 activity. In contrast, in ubr-1 loss-of-function mutants, the absence of GOT-1 inhibition leads to excessive glutamate production. This glutamate excess, which can be reduced by ceftriaxone treatment, induces compensatory downregulation of IVM-targeted GluCls. This downregulation results in a diminished inhibitory response to IVM in the pharyngeal region, ultimately causing IVM resistance in ubr-1 mutants.
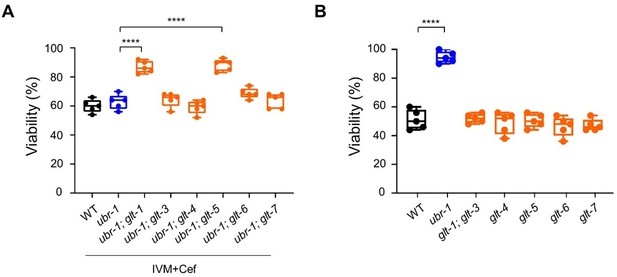
Glutamate transporter removal inhibits ceftriaxone-mediated restoration of ivermectin sensitivity in ubr-1.
(A) Compared to the ubr-1 mutants, the ubr-1; glt-1 and ubr-1; glt-5 double mutants show enhanced ivermectin resistance under ceftriaxone treatment. (B) The glt mutants do not show resistance to ivermectin. ****p < 0.0001; one-way ANOVA test.
Videos
Locomotion and body-length phenotypes of the wild type and ubr-1 mutants before and after treatment with IVM.
Representative free-moving worms of the wild type and ubr-1(hp684) mutants with or without IVM (50 ng/ml) treatment for 20 hr. The video shows the worm heading left. Scale bar, 50 µm.
Additional files
-
Supplementary file 1
Mutants and transgenic strains used in this study.
- https://cdn.elifesciences.org/articles/103718/elife-103718-supp1-v1.xlsx
-
Supplementary file 2
A list of plasmids used in this study.
- https://cdn.elifesciences.org/articles/103718/elife-103718-supp2-v1.xlsx
-
Supplementary file 3
Sequence of primers used in this study.
- https://cdn.elifesciences.org/articles/103718/elife-103718-supp3-v1.xlsx
-
Source data 1
Raw data used for statistical analysis in each panel.
- https://cdn.elifesciences.org/articles/103718/elife-103718-data1-v1.zip
-
MDAR checklist
- https://cdn.elifesciences.org/articles/103718/elife-103718-mdarchecklist1-v1.docx






