Early changes in the properties of CA3 engram cells explored with a novel viral tool in mice
Figures
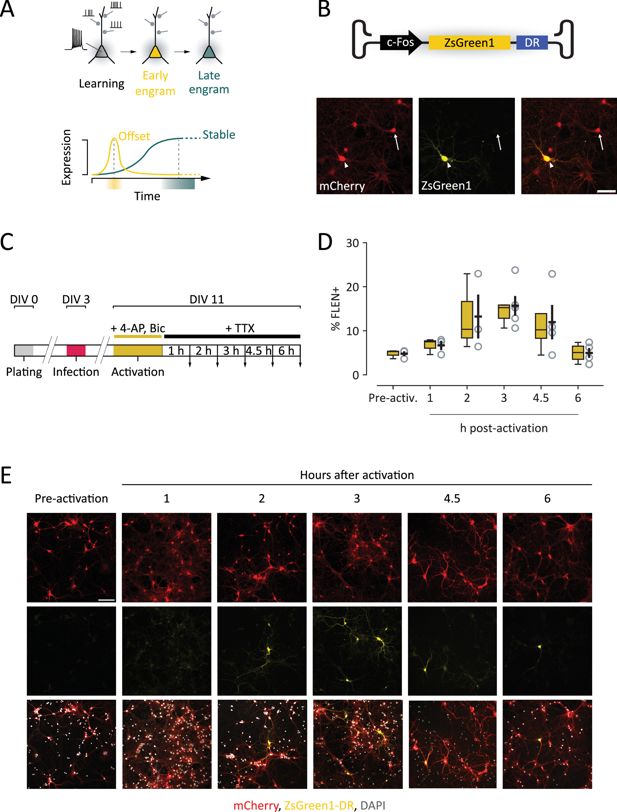
Development of a novel viral tool to study early changes in the properties of CA3 engram cells.
(A) Scheme representing the maturation of engram neurons during memory formation. Top panel: during learning, a neuron receives numerous synaptic inputs, resulting in sequences of action potentials. This activity triggers the rapid expression of immediate early genes (IEGs), marking the neuron as part of an engram (yellow). As the engram matures (green), IEG expression is not detectable anymore, yet the neuron retains established engram properties. Bottom panel: expression function of IEG-based construct to investigate and compare early and late engram properties. A construct with rapid offset (yellow) best captures engrams shortly after learning, whereas a progressively expressing construct best captures more mature engrams. (B) FLEN construct (c-Fos.ZsGreen1-DR) encapsulated in AAV2/9 vector. Bottom panel: representative images showing ZsGreen1 fluorescence in c-Fos+ neurons (arrowhead) and c-Fos− neurons (arrow). Scale bar = 50 µm. (C) Timeline of in vitro experiments to determine FLEN expression and offset time course. After infection (DIV3) and activation (DIV11), samples are washed with a blocking medium and images are acquired at different intervals (downward arrows). (D) Percentage of ZsGreen+ vs. all transduced neurons (mCherry+ cells) at the different intervals following activation. (E) Representative images of activated cultured neurons at 1, 2, 3, 4.5, and 6 hr after activation. Scale bar = 100 µm.

In vivo FLEN labeling and decay time course following a salient experience.
(A) FLEN and an infection marker viral vector are injected bilaterally into CA3. Mice undergo contextual fear conditioning (CFC), and sections are collected (downward arrows) 1.5, 3, 4.5, 6, and 12 hr following CFC. As a control, sections are obtained from mice left undisturbed in their home cage (HC). (B) Left: one-time CFC experiment scheme. Mice explore an arena for 5 min, and three foot-shocks are given at 180, 210, and 270 s after the start of the test (red line). Top-right: paired comparison of the percentage of time mice spent freezing during training and when replaced in the conditioning arena 1 hr after CFC. Bottom-right: comparison of the percentage of time mice spent freezing during training, when placed in a different neutral environment and 24 hr after CFC. These experiments show that the CA3-dependent shock-context association is rapidly learned (within 1 hr), is highly environment-selective, and is retained for 24 hr. (C) Representative images of CA3 neurons expressing the infection marker (mCherry+, indicated by arrow) and neurons expressing both the infection marker and FLEN (ZsGreen1+/mCherry+, indicated by arrowhead). The latter are considered FLEN+ neurons. Scale bar = 50 µm. (D) Percentage of ZsGreen+ CA3 neurons over the total number of mCherry+ neurons at different intervals following activation. (E) Representative sections of CFC-trained mice compared to HC untrained mice at different intervals following CFC. Dashed lines outline the dentate gyrus (DG) cellular layer, while the dashed segment indicates the separation between CA3 and CA2. Scale bar = 100 µm. (F) Left panel: schematic representation of neuronal position along the proximodistal axis analysis of CA3. The CA3 pyramidal layer was straightened and the x position of FLEN+ cells was normalized on a 0 (closer to DG) to 1 (closer to CA2) scale. Right panel: comparison of normalized proximodistal frequency distribution of CA3 FLEN+ cells placement in HC mice (gray) and 3 hr post-CFC mice (yellow). Bold lines represent the median of the respective distributions. HC median is displayed in the CFC plot to highlight distribution difference. Statistical signficance: *p<0.05; ***p<0.001.

FLEN expression in different behavioral conditions.
(A) Left panel: context-only and immediate-shock behavioral test layout. In the context-only (CO) task, mice freely explore an arena without being conditioned; mice subjected to the immediate-shock (IS) experience a long (8 s) foot-shock immediately after being placed in the conditioning cage, preventing them from exploring the environment and forming contextual memories. (B) Right panel: plot comparing the percentage of FLEN+ cells in the home cage group (from Figure 2D) and the context-only and immediate-shock groups. * p<0.05.
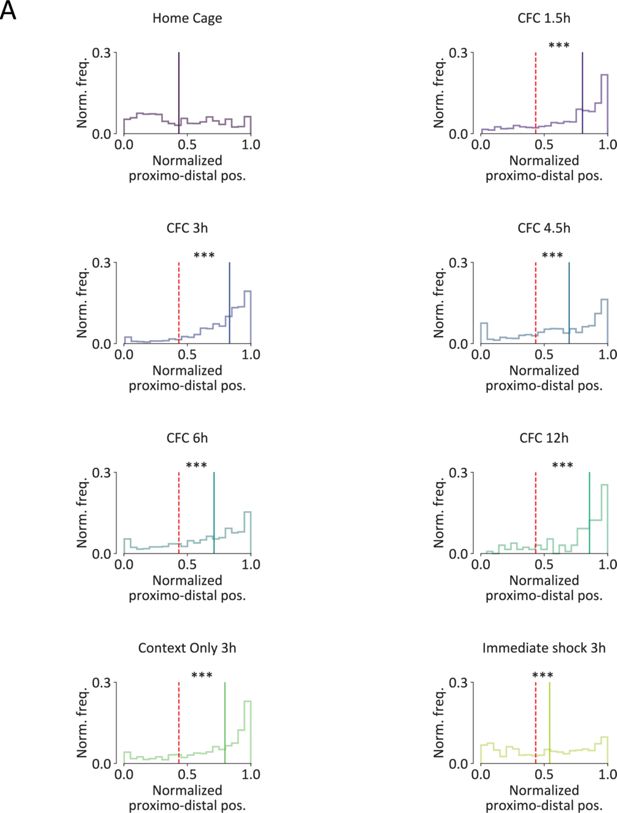
FLEN+ cells distribution along the proximodistal and superficial-to-deep axis.
(A) Normalized proximodistal (x) and superficial-to-deep (y) frequency distribution of the position of CA3 FLEN+ neurons in all behavioral groups. Black dashed lines indicate each group’s median value, while the red dashed line represents the home cage group’s median value. Each plot includes the scattered x–y position of FLEN+ cells within the pyramidal layer, and the frequency distribution along the proximodistal (top) and superficial-to-deep (right) axis. Statistical significance: ***p<0.001.

Electrophysiological analysis of intrinsic properties of CA3 engram neurons.
(A) Experimental outline: mice were injected with either FLEN or RAM construct, subjected to contextual fear conditioning (CFC) and then sacrificed at either 3- or 24-hr post-conditioning to collect acute hippocampal slices. Right: representative images of FLEN+ and RAM+ cells targeted for patch-clamp recordings, with neighboring control FLEN- or RAM− neurons. Scale bar = 20 µm. (B) Membrane potential response. Left panel: representative membrane potential trace in response to incremental injected current in control cells (gray) and engram cells (yellow for FLEN and green for RAM). Right panel: graphs of input resistance for comparison between engram cells and their corresponding control cells. (C) Action potential firing pattern. Left panel: sample traces of long (1-s) above-rheobase current step to analyze the pattern of action potential firing current in control cells (gray) and engram cells (yellow and green). Right panel: average spike frequency graphs. (D) Cumulative spike frequency curve over the 1-s current step shown in (C) showing a similar distribution between FLEN + and FLEN− neurons (top), and a more sustained firing of RAM+ compared to their RAM− neurons (bottom). Statistical signficance: *p<0.05; ***p<0.001.
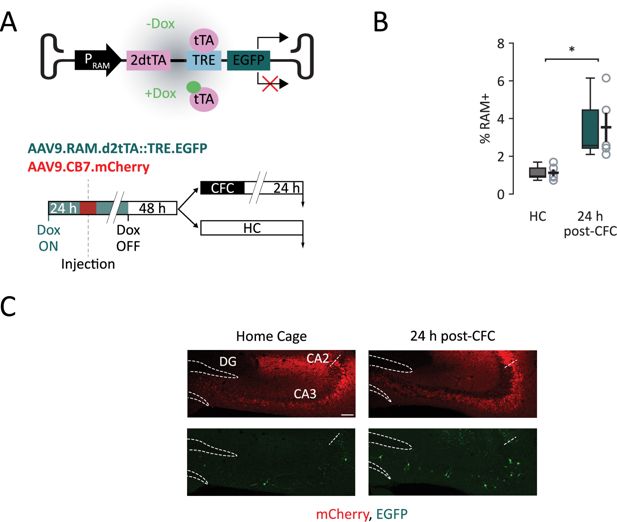
RAM expression 24 hours following CFC.
(A) RAM construct outline and expression mechanism. Top panel: Dox administration prevents binding of tTA protein to the TRE promoter, whereas Dox deprivation allows downstream EGFP transcription. Bottom panel: scheme for in vivo experiments using the RAM system. Dox-fed mice are bilaterally injected in CA3 with viral constructs for RAM and the infection marker AAV-CB7-mCherry. The diet is switched to regular food chow 48 hr prior to the contextual fear conditioning (CFC). Then, mice undergo CFC and sections are collected (downward arrows) 24 hr later and compared to untrained mice that never left their home cage. (B) Percentage of RAM+ CA3 neurons over the total number of mCherry+ neurons in HC and 24 hr post-CFC. (C) Representative sections of CFC-trained mice compared to HC mice. Dashed lines outline the dentate gyrus (DG) cellular layer, while the dashed segment indicates the separation between CA3 and CA2. Scale bar = 100 µm. Statistical signficance: *p<0.05.
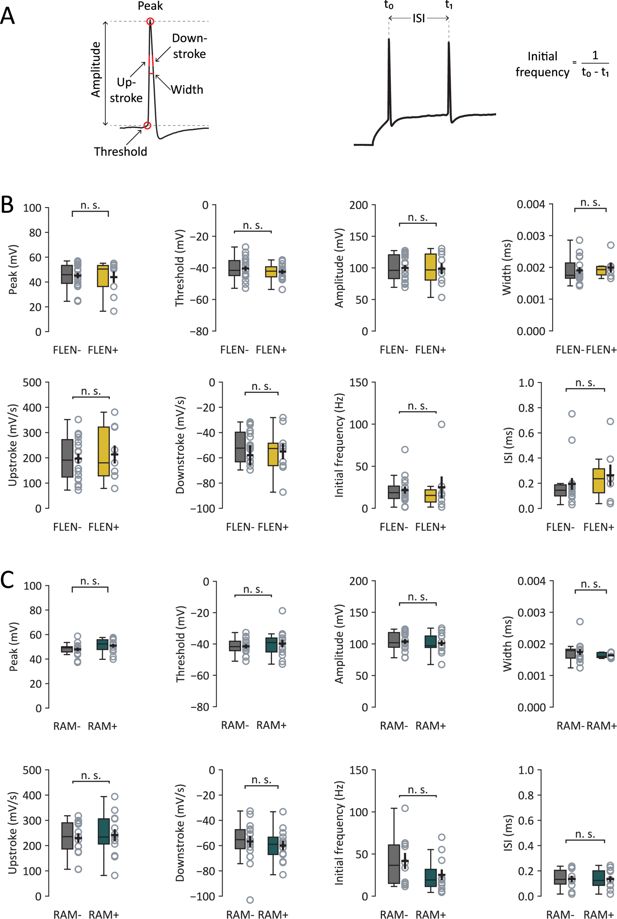
Action potential properties of FLEN+, FLEN-, RAM+ and RAM-neurons.
(A) Left panel: overview of analyzed action potential features. Right panel: initial frequency calculation. Summary of analyzed action potential properties in FLEN+ vs. FLEN− (B) and RAM+ vs. RAM− (C).
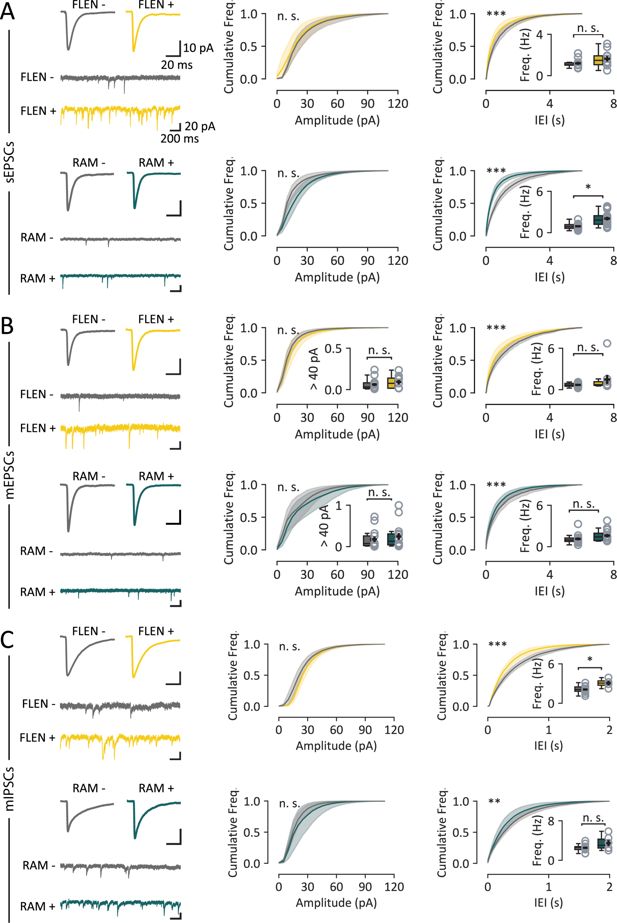
Analysis of EPSCs and IPSCs in CA3 engram neurons.
(A) Sample traces and cumulative distributions of spontaneous excitatory post-synaptic currents (sEPSCs) recorded from CA3 engram neurons (FLEN in yellow, RAM in green). Top panels: comparison between FLEN+ and FLEN− neurons reports no difference in mean sEPSC amplitude cumulative distribution but a higher event frequency in FLEN+ neurons, as seen in the inter-event interval (IEI) cumulative distribution. The inset displays the comparison of the average frequency (Hz) of such events. Bottom panels: RAM+ vs. RAM− neurons show a similar pattern characterized by no change in amplitude and increased frequency in RAM+ neurons. (B) Same analysis as in (A), for miniature EPSCs (mEPSCs). The inset in the amplitude plot shows the fraction of mEPSCs exceeding 40 pA, indicative of giant mossy fiber events. (C) As (A) and (B), for miniature IPSCs (mIPSCs), comparing frequency and amplitude in FLEN+ vs. FLEN− and RAM+ vs, RAM− neurons. Statistical signficance: *p<0.05; ** p<0.01; ***p<0.001.
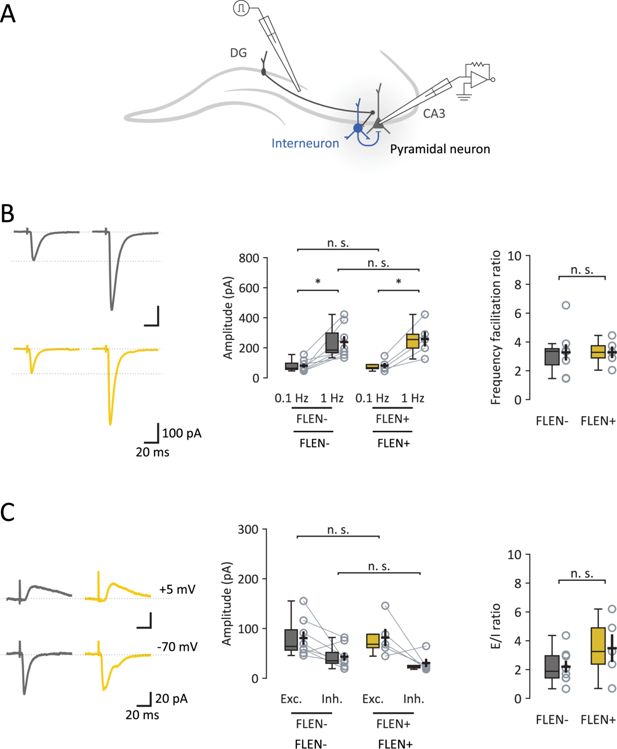
5Properties of Mf-CA3 synapses in CA3 engram neurons.
(A) Schematic representation of Mf-CA3 EPSCs and feedforward inhibition recordings. (B) Left panel: representative traces showing EPSC amplitude in FLEN− (gray) and FLEN+ (yellow) cells following low frequency stimulation (0.1 Hz) and corresponding increase evoked with a higher stimulation frequency (1 Hz), due to a presynaptic form of synaptic plasticity characteristics of Mf-CA3 synapses (frequency facilitation). Center panel: amplitude plot for FLEN− and FLEN+ stimulated at 0.1 and 1 Hz. Right panel: frequency facilitation ratio plot shows no difference between FLEN− and FLEN+ neurons. (C) Left panel: representative traces showing Mf-driven IPSCs (feedforward inhibition) and EPSCs (direct excitatory activity) recorded in individual FLEN− (gray) and FLEN+ (yellow) neurons. Center panel: amplitude plot of EPSCs and IPSCs for FLEN− and FLEN+ neurons. Right panel: graph showing excitation-to-inhibition ratio, comparing FLEN− and FLEN+ neurons. Statistical signficance: *p<0.05.
Tables
Summary of AP properties.
| FLEN | RAM | |||||
|---|---|---|---|---|---|---|
| FLEN− | FLEN+ | Test | RAM− | RAM+ | Test | |
| Peak (mV) | 45.19 ± 2.25, n = 19 | 43.92 ± 4.62, n = 9 | Mann–Whitney U, p = 1.00 | 47.93 ± 1.41, n = 15 | 50.91 ± 1.57, n = 13 | T-test, p = 0.17 |
| Threshold (mV) | –40.41 ± 1.54, n = 20 | –42.47 ± 1.36, n = 14 | T-test, p = 0.35 | –41.53 ± 1.35, n = 15 | –39.78 ± 2.46, n = 13 | T-test, p = 0.52 |
| Amplitude (mV) | 99.91 ± 4.63, n = 19 | 98.67 ± 8.73, n = 9 | T-test, p = 0.89 | 103.71 ± 3.93, n = 15 | 101.18 ± 4.53, n = 13 | T-test, p = 0.67 |
| Width (ms) | 0.002 ± 0.001, n = 16 | 0.002 ± 0.001, n = 6 | Mann–Whitney U, p = 0.51 | 0.0017 ± 0.0001, n = 13 | 0.0016 ± 0.00003, n = 7 | Mann–Whitney U, p = 0.36 |
| Upstroke (mV/s) | 197.06 ± 19.54, n = 19 | 213.35 ± 35.48, n = 9 | T-test, p = 0.67 | 229.89 ± 17.86, n = 15 | 241.65 ± 23.2, n = 13 | T-test, p = 0.69 |
| Downstroke (mV/s) | –57.99 ± 7.91, n = 19 | –54.99 ± 6.18, n = 9 | Mann–Whitney U, p = 0.84 | –56.70 ± 4.43, n = 15 | –59.96 ± 3.62, n = 13 | T-test, p = 0.58 |
| Initial frequency (Hz) | 21.58 ± 4.15, n = 16 | 25.04 ± 12.83, n = 7 | Mann–Whitney U, p = 0.62 | 41.74 ± 9.54, n = 10 | 25.30 ± 6.43, n = 11 | Mann–Whitney U, p = 0.25 |
| ISI (ms) | 0.19 ± 0.05, n = 16 | 0.26 ± 0.08, n = 7 | Mann–Whitney U, p = 0.28 | 0.14 ± 0.02, n = 10 | 0.14 ± 0.02, n = 11 | T-test, p = 0.98 |





