Contrasting roles for parvalbumin-expressing inhibitory neurons in two forms of adult visual cortical plasticity
Figures
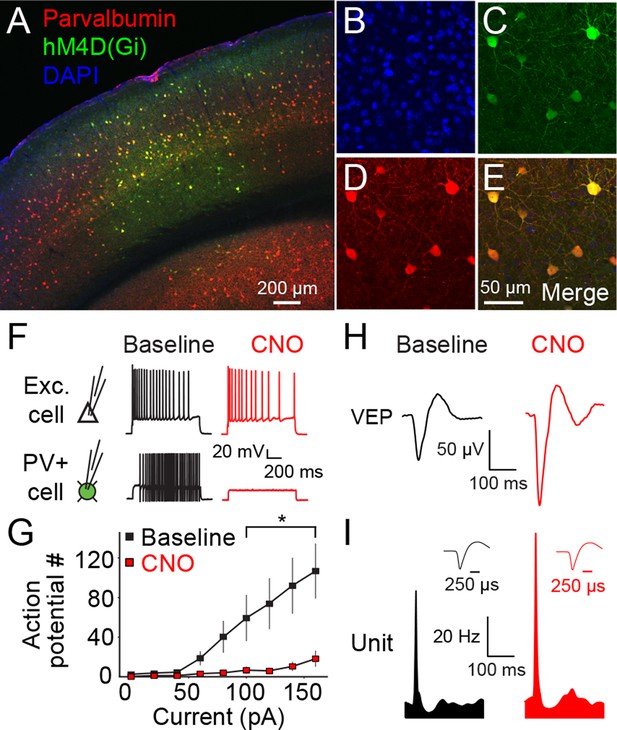
The hM4D(Gi) DREADD system locally inactivates parvalbumin+ neurons in binocular primary visual cortex (V1).
(A) An example of V1 expression of hM4D(Gi) in a parvalbumin (PV)-Cre recombinase (Cre) mouse infected locally in binocular V1 with AAV9-hSyn-DIO-hM4D(Gi)-mCitrine. (B) A DAPI stain for cell nuclei is shown in blue. (C) Infected cells expressing hM4D(Gi) are labeled in green. (D) Immuno-labeled PV+ cells are shown in red. (E) The merged image reveals that hM4D(Gi)-expressing cells are also PV+. (F) Intracellular current clamp recordings of hM4D(Gi)-infected PV+ layer 4 neurons in ex vivo slices of V1 reveal that green-labeled infected cells exhibit a non-adapting fast-spiking phenotype typical of fast-spiking inhibitory neurons (black). These cells do not fire action potentials in the presence of CNO (red), the exogenous ligand for hM4D(Gi) receptors, despite depolarizing current injection. In contrast, neighboring cells that are not mCitrine+ show no impact of CNO application. (G) HM4D(Gi)-mediated inactivation of putative fast-spiking PV+ inhibitory neurons is here summarized as the number of action potentials resulting from a given current injection before (black) and after CNO application (red). (H) The effects of hM4D(Gi)-mediated inactivation of putative PV+ fast-spiking inhibitory neurons in vivo are apparent from electrophysiological recordings from V1 of awake, head-fixed mice viewing phase-reversing sinusoidal grating stimuli. Averaged Visually Evoked Potential (VEP) recordings recorded in layer 4 reveal increased VEP magnitude in the presence of CNO (red), relative to pre-CNO (baseline) recordings (black), indicative of reduced inhibition. (I) Phase reversal-evoked action potentials recorded from neurons in layer 4 also exhibit a similar effect with elevated firing rates in the presence of CNO (red) relative to the baseline recording (black). Labeled scale bars are presented throughout. Error bars are standard error of the mean (S.E.M.).
-
Figure 1—source data 1
Action potential number for current injection.
- https://doi.org/10.7554/eLife.11450.004
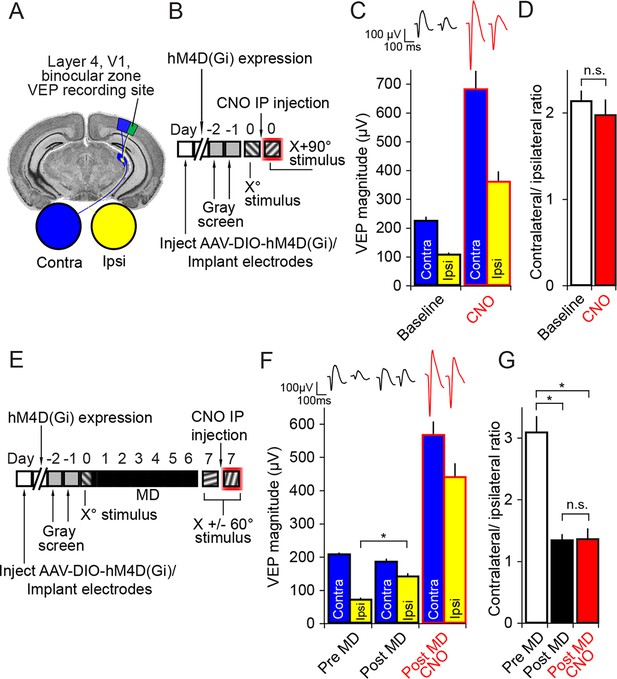
Inactivation of parvalbumin+ neurons has no impact on expression of ocular dominance (OD) or the ocular dominance shift as a result of monocular deprivation (MD) in the adult mouse.
(A) For experiments described in this figure mice were infected across all cortical depths bilaterally in binocular V1 (green). During the same surgical implantation procedure VEP recording electrodes were also positioned in layer 4 and chronically fixed. (B) Mice (P45-60) were infected and implanted with electrodes and then left for 3 weeks for the AAV9 viral vector to reach maximal expression, after which they underwent habituation to head-fixation and a gray screen for two consecutive days. Following this, on experimental day 0, mice were presented with an Xo phase-reversing sinusoidal grating stimulus separately to the left and right eye. VEPs were recorded from each hemisphere in order to determine ocular dominance in binocular V1. Mice were then removed from the recording apparatus and, CNO was delivered systemically half an hour prior to undertaking the same recording procedure, this time using an orthogonal X + 90° visual stimulus. (C) As is well documented, responses in V1 to stimuli viewed through the contralateral eye (blue) were greater in magnitude than those elicited through the ipsilateral eye (yellow), as measured here by VEP magnitude. After application of CNO (red outlines), VEP magnitude dramatically increased. (D) This increase was scaled such that the ratio of Contralateral:Ipsilateral VEP magnitude was maintained before (white) and after CNO (red). (E) In a second group of mice, a similar experimental protocol was observed prior to measuring ocular dominance by recording VEPs in each hemisphere elicited by an Xo stimulus through each eye. One hemisphere was then selected and the contralateral eye was sutured closed. After 7 days of monocular deprivation, the eye was opened and VEPs driven through either eye were again recorded, this time elicited by an X + 60° stimulus. Mice were then systemically injected with CNO and, half an hour later, VEPs driven through either eye were recorded, this time elicited by an X - 60° stimulus. (F) After the adult mice underwent 7 days of MD, there was a significant potentiation of the V1 response to visual input through the ipsilateral eye (yellow). After application of CNO (red outlines), VEPs driven through each eye were elevated in magnitude, but again the increase in VEP magnitude was scaled. (G) As a result of open eye potentiation after MD, the OD ratio shifted dramatically from the contralateral bias of pre MD (white) to an almost equal cortical response through contralateral and ipsilateral eyes (black). This shifted ratio was unaffected by hM4Di-mediated inactivation of putative fast-spiking inhibitory neurons during CNO application (red), indicating that the expression of OD and its shift as a result of MD in adult mice do not require fast-spiking inhibition. Significant comparisons are labeled with an asterisk and non-significant comparisons with n.s. throughout. Error bars are standard error of the mean (S.E.M.).
-
Figure 2—source data 1
Ocular dominance and deprivation effects are maintained after PV neuron inactivation.
- https://doi.org/10.7554/eLife.11450.006
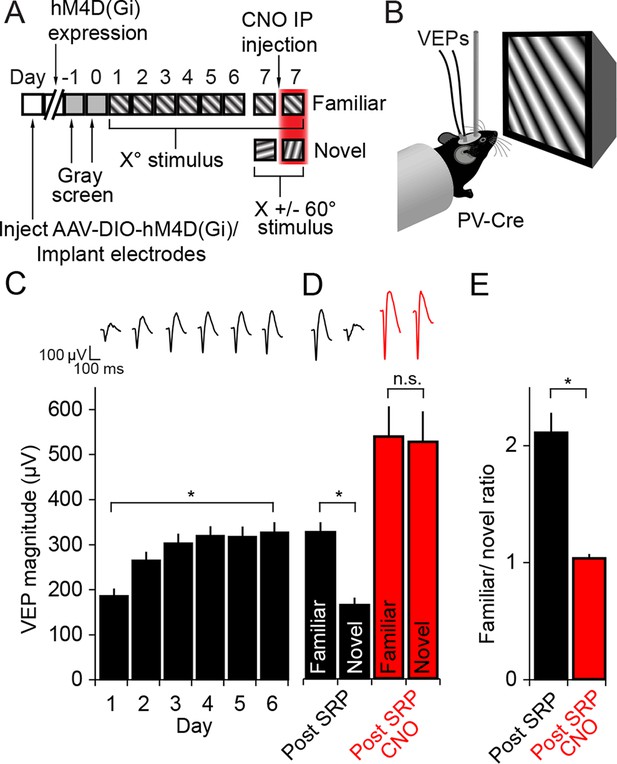
The expression of Stimulus-selective Response Potentiation (SRP) requires activity in PV+ neurons in V1.
(A) Mice expressing hM4D(Gi) receptors in PV+ cells underwent a standard SRP induction protocol. On day 7, mice viewed a novel oriented stimulus in addition to the familiar stimulus; before and after CNO injection. (B) We acquired binocular VEPs from awake, head-fixed mice elicited by the same full-field oriented sinusoidal grating stimulus over several days. (C) As a result of multiple days of experience cortical response was dramatically potentiated such that the familiar stimulus evoked VEPs of significantly greater magnitude than the novel stimulus (black bars). (D) After application of CNO, VEPs underwent a notable increase in magnitude as a result of disinhibition (red bars). Most notably, CNO rendered response to familiar and novel stimuli equivalent in magnitude. (E) This lost discrimination of familiar and novel stimuli is reflected as a drop in the ratio of response to familiar and novel stimuli from approximately 2:1 (black) to approximately 1:1 (red). Significant comparisons are labeled with an asterisk and non-significant comparisons with n.s. throughout. Error bars are standard error of the mean (S.E.M.).
-
Figure 3—source data 1
PV Dreadds SRP induction and expression.
- https://doi.org/10.7554/eLife.11450.008
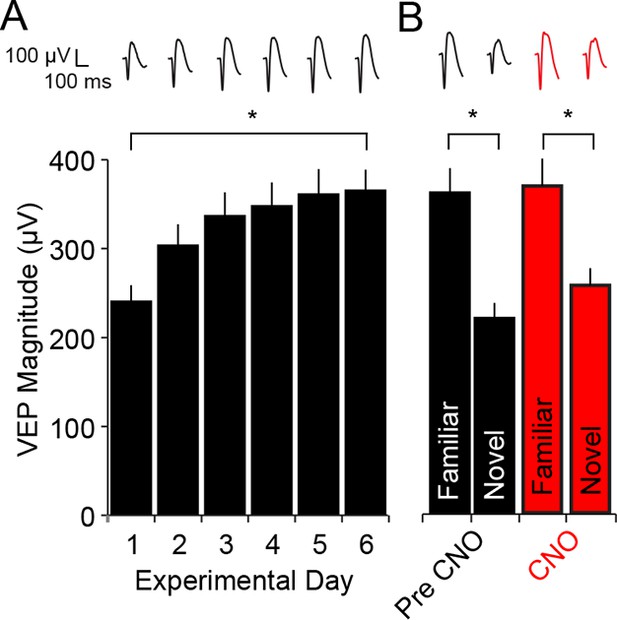
CNO has no impact on SRP expression in WT mice.
(A) C57BL/6 WT mice were bilaterally infected with AAV9-hSyn-DIO-hM4D(Gi)-mCitrine in binocular V1. Because these mice did not express Cre recombinase there was no subsequent expression of hM4D(Gi) DREADDS receptors. After 4 weeks, during which hM4D(Gi) was expressed at high levels in PV-Cre mice, WT mice were taken through a standard SRP protocol of electrode implantation, habituation and visual experience of a single oriented grating. This progressed as usual, resulting in a significant increase in the magnitude of the VEP. (B) Expression of SRP was then tested on day 7 by interleaving presentations of familiar and novel orientations and VEPs elicited by the familiar orientation were of significantly greater magnitude than the novel. After delivery of CNO, VEPs were again tested in response to the familiar orientation and a second novel orientation. The drug had no impact and a similar degree of significant stimulus selectivity was present. Significant comparisons are marked with an asterisk throughout while non-significant comparisons are marked with n.s. Error bars are standard error of the mean (S.E.M.).
-
Figure 3—figure supplement 1—source data 1
CNO has no effect on SRP expression in WT mice.
- https://doi.org/10.7554/eLife.11450.010
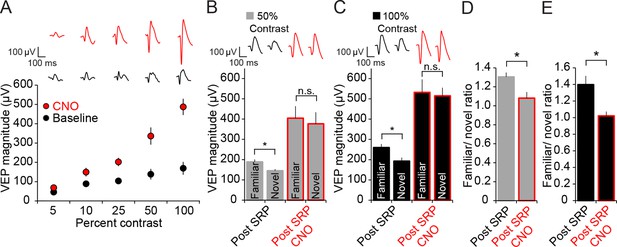
Expression of SRP to two separate contrast values is blocked by hM4D(Gi)-mediated PV+ neuron inactivation, but differential response to contrast is maintained.
(A) CNO delivery to PV-Cre mice that had been infected with AAV9-hSyn-DIO-hM4D(Gi)-mCitrine impacted VEP magnitude (red) significantly across a range of contrast compared to baseline (black). (B) SRP was induced to two differently oriented stimuli, each at different contrast values: 50% (gray) and 100% (black). Modest but significant SRP was expressed at 50% contrast prior to CNO application. After CNO application (red outlines), VEPs increased significantly in magnitude but were no longer significantly different for familiar and a second novel orientation. (C) SRP was also expressed for a different orientation at 100% contrast and, again, VEP magnitude was increased and SRP blocked by delivery of CNO. (D) The blockade of SRP expression by CNO at 50% contrast was apparent in the reduction in the familiar/novel ratio for VEP magnitude. (E) The blockade of SRP expression at 100% contrast was also observed as a significant drop in familiar/novel ratio of VEP magnitude after CNO delivery. Significant comparisons are marked with an asterisk throughout while non-significant comparisons are marked with n.s. Error bars are standard error of the mean (S.E.M.).
-
Figure 4—source data 1
PV-neuron inactivation does not produce ceiling effect.
- https://doi.org/10.7554/eLife.11450.012
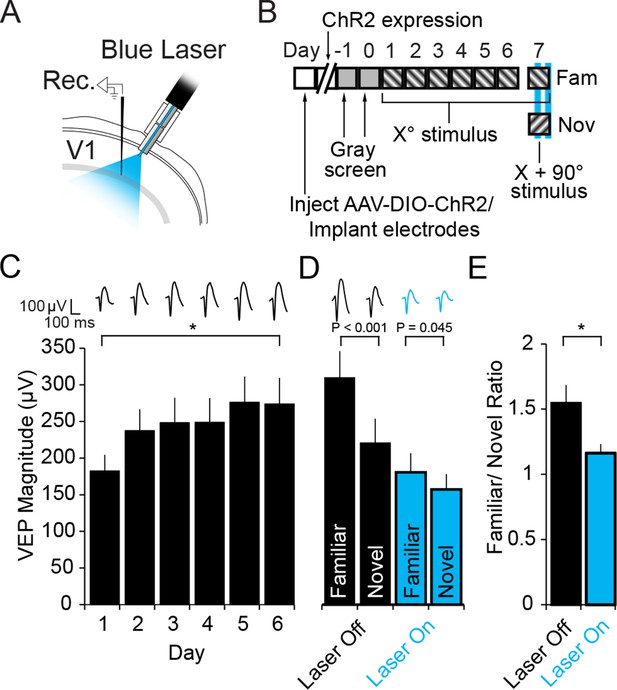
Optogenetic stimulation of PV+ inhibitory neurons prevents SRP expression.
(A) Blue light was delivered locally into V1 via optic fibers chronically implanted at a 45° angle to target the VEP recording site in layer 4 of binocular V1 of PV-Cre mice infected with AAV5-EF1α-DIO-hChR2(H134R)-eYFP. (B) Experimental timeline showing that after viral infection, electrode implantation, and ChR2 expression; mice were accustomed to head-fixation and gray screen viewing. Subsequently, they underwent a standard SRP induction protocol over 6 days. On day 7, mice viewed a novel oriented stimulus in addition to the familiar stimulus and, on 50% of presentations of each stimulus, blue light (473 nm) was delivered to cortex to optogenetically activate PV+ cells. (C) Significant SRP was induced over 6 days as VEPs underwent a typical potentiation. (D) On day 7, SRP was expressed through significantly larger VEP magnitude in response to the familiar Xo orientation than a novel X + 90° stimulus when blue light was not delivered (Black bars). In the presence of blue light (blue bars), VEPs were suppressed, and there was a significant reduction in the differential magnitude of VEPs driven by familiar and novel stimuli. (E) The ratio of VEP magnitude elicited by familiar/novel stimuli was significantly reduced by optogenetic activation of PV+ neurons, reflecting a decrement in SRP expression. Significant comparisons are marked with an asterisk and post hoc test p values are reported in D to emphasize the impact of laser stimulation on SRP selectivity. Error bars are standard error of the mean (S.E.M.).
-
Figure 5—source data 1
PV-neuronal activation.
- https://doi.org/10.7554/eLife.11450.014
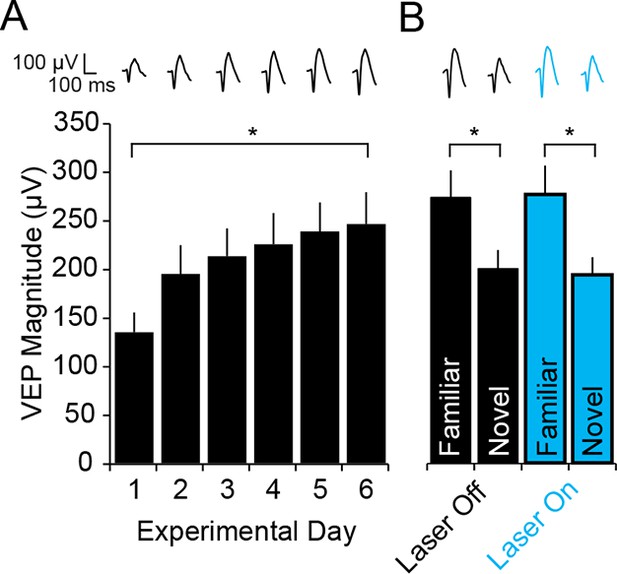
Blue light has no impact on SRP expression.
(A) To check that light itself had no effect on cortical physiology in the optogenetic experiments, WT mice were infected bilaterally with AAV5-EF1α -DIO-hChR2(H134R)-eYFP in binocular V1. After 4 weeks, during which ChR2 was expressed at high levels in PV-Cre mice, WT mice were taken through the standard SRP protocol described above. Again, significant SRP occurred. (B) On test day, during interleaved presentations of a familiar and novel oriented stimulus together with either blue light (473 nm) or no light stimulation delivery to V1, VEPs were significantly greater in magnitude for the familiar than the novel stimulus, regardless of the presence of blue light. Significant comparisons are marked with an asterisk throughout while non-significant comparisons are marked with n.s. Error bars are standard error of the mean (S.E.M.).
-
Figure 5—figure supplement 1—source data 1
Laser does not effect VEPs in WT animals.
- https://doi.org/10.7554/eLife.11450.016
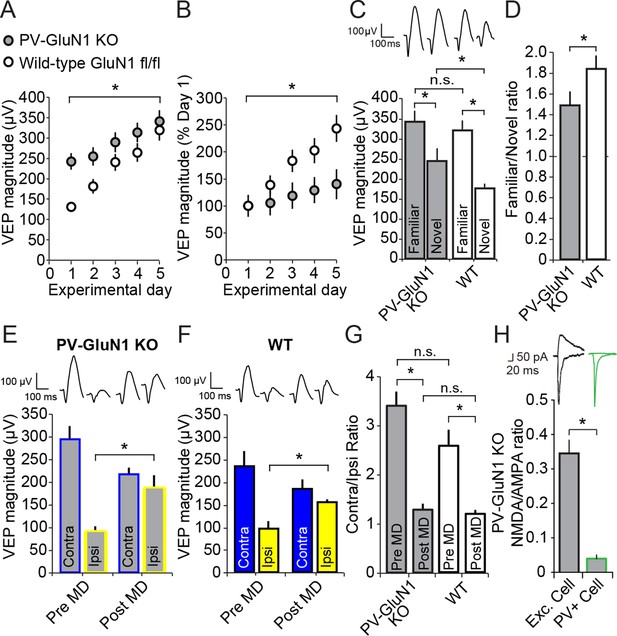
Loss of NMDA receptors selectively from parvalbumin+ cells impacts SRP but not adult OD plasticity.
(A) VEPs recorded from mice in which the mandatory GluN1 subunit of the NMDA receptor was genetically ablated from PV+ cells using Cre recombinase technology (PV GluN1 KO, gray) were significantly greater in magnitude than those recorded from WT littermates (black), suggesting disinhibition of the visual response. In these same PV GluN1 KO mice, SRP was also significantly impacted as there is was significantly less gain in magnitude over days of repeated presentation of an X° stimulus than observed in WT littermates. (B) This significant reduction in the magnitude of SRP was most clearly observed if VEP magnitude was normalized to the magnitude on day 1. (C) After SRP, both PV GluN1 KO mice and their WT littermates exhibited a significantly greater VEP magnitude elicited by the now familiar stimulus than interleaved presentations of a novel oriented stimulus. However, consistent with the observed difference in magnitude on day 1, VEPs elicited by a novel X + 90° stimulus in PV GluN1 KO mice were significantly greater in magnitude than those in WT littermate mice. No significant difference was observed for VEPs elicited by the familiar stimulus. (D) A significant difference in the ratio of VEP magnitude elicited by the familiar and novel stimuli reveals a deficit in SRP expression in PV GluN1 KO mice. (E) In contrast, PV GluN1 KO mice exhibited a normal adult OD shift after 7 days of MD, resulting from open eye potentiation (yellow outlines). (F) Wild-type (WT) littermates exhibited the same significant open eye potentiation (yellow bars) after 7 days of MD. (G) A comparison of the degree of OD shift as a result of 7 days of MD reveals a significant shift in OD ratio in both genotypes but no difference between genotypes, indicating that NMDA receptors in PV+ cells are not required for induction or expression of the OD shift. (H) To confirm genetic ablation of NMDARs selectively from parvalbumin (PV+) neurons in the PV-GluN1 KO; mice were injected with an AAV5 vector to express GFP in a Cre-dependent fashion in PV+ cells only. After 1 month, fluorescence-guided intracellular recordings were performed from ex vivo slices of visual cortex. NMDAR-mediated synaptic transmission was normal in excitatory control cells but abolished in PV+ cells. This is expressed here as the NMDAR EPSC/AMPAR EPSC ratio in excitatory cells (black outline) and PV+ cells (green outline). Sample EPSC traces mediated by the AMPAR (downward) and NMDAR (upward) are shown at the top of the panel. Significant comparisons are marked with an asterisk throughout while non-significant comparisons are marked with n.s. Error bars are standard error of the mean (S.E.M.).
-
Figure 6—source data 1
PV-GluN1 KO
- https://doi.org/10.7554/eLife.11450.018
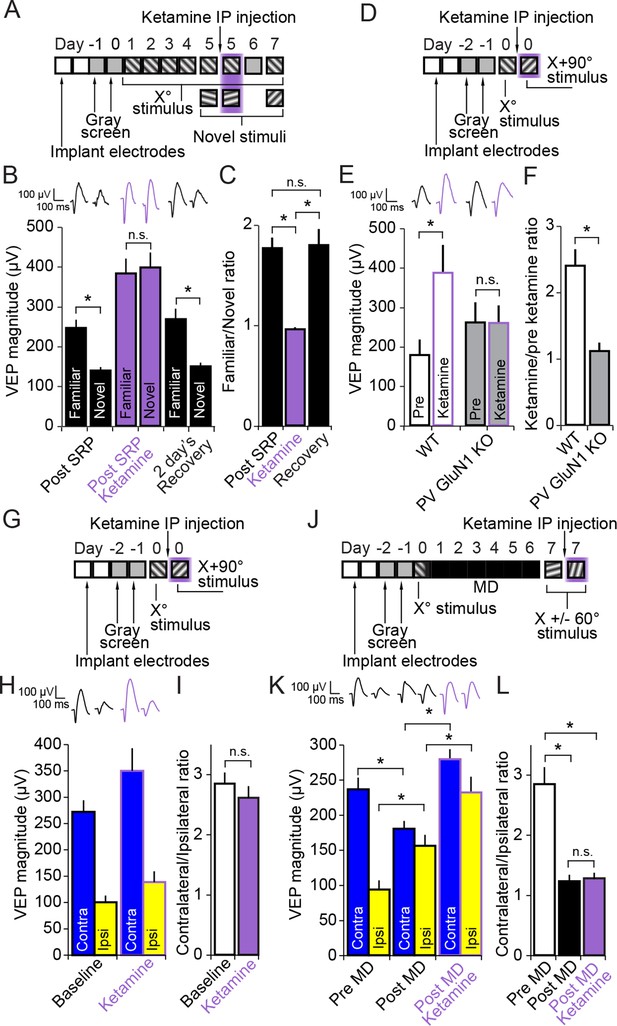
Ketamine prevents expression of SRP through blockade of NMDA receptors expressed in parvalbumin+ neurons but does not impact expression of the adult OD shift.
(A) Mice were bilaterally implanted with VEP recording electrodes in layer 4 of binocular V1. After habituation to head-fixation and a gray screen for 2 days, SRP was induced over 4 days by repeatedly presenting sessions of an X° stimulus. On day 5, SRP expression was tested by presenting interleaved blocks of the familiar X° stimulus and a novel X + 60° stimulus. In order to test the acute impact of blocking NMDA receptors on SRP expression, 50 mg/kg of ketamine was then delivered systemically 15 min before re-acquiring VEPs elicited by the familiar X° stimulus and interleaved presentations of a second novel X - 60° stimulus. Mice were then allowed 2 days recovery and a complete washout of ketamine before re-testing SRP expression by again testing VEP magnitude in response to the familiar X° stimulus and a third novel X + 90° stimulus on day 7. (B) Significant SRP was expressed on experimental day 5 prior to ketamine delivery as the familiar X° stimulus elicited VEPs of greater magnitude than the novel stimulus. Delivery of 50 mg/kg ketamine (purple) had two notable impacts on the VEP: First, the overall magnitude of the VEP increased. Second, and most importantly, the significant difference in magnitude of VEPs elicited by familiar and novel stimuli was no longer present. This effect was acute, as SRP expression was again significantly apparent 2 days later. (C) The ratio of VEP magnitude elicited by the familiar stimulus over the novel. This ratio was close to 2 and not significantly different prior to or after recovery from ketamine administration but dropped significantly to approximately 1 during ketamine exposure. (D) We tested whether ketamine had a differential impact on VEP magnitude in PV GluN1 KO mice and WT littermate mice. (E) In the WT littermate mice (white bars) 50 mg/kg ketamine had a significant potentiating effect on VEP magnitude, consistent with our previous observation. In contrast, ketamine had no significant impact on VEP magnitude in the PV GluN1 KO mice (gray bars). (F) The selectivity of ketamine’s impact on the WT mice is observed by comparing the ratio of VEP magnitude during ketamine over baseline, which was significantly greater for WT mice than the PV GluN1 KO mice, in which the ratio was approximately 1. (G) In a separate group of mice, a similar protocol was then used to determine whether the OD ratio is affected by ketamine. (H) Ketamine impacted both the VEPs driven through the contralateral eye (blue) and ipsilateral eye (yellow) equally. (I) This scaled effect is demonstrated by a lack of significant difference between OD ratios prior to (white) and during 50 mg/kg ketamine (purple). (J) We next tested whether ketamine has any impact on the expression of adult OD plasticity by recording VEP magnitudes through either eye in a new group of adult mice before taking them through a standard 7 day MD protocol. (K) As anticipated, 7 days of contralateral eye MD induced a significant ipsilateral eye potentiation (yellow) and ketamine then further potentiated VEPs elicited through both contralateral (blue) and ipsilateral eyes. (L) The OD ratio shifts significantly from a ratio heavily biased towards the contralateral eye, to a less biased ratio. Ketamine administration did not significantly affect the magnitude of the OD shift. Significant comparisons are marked with an asterisk throughout while non-significant comparisons are marked with n.s. Error bars are standard error of the mean (S.E.M.).
-
Figure 7—source data 1
Ketamine impact on cortical plasticity.
- https://doi.org/10.7554/eLife.11450.020
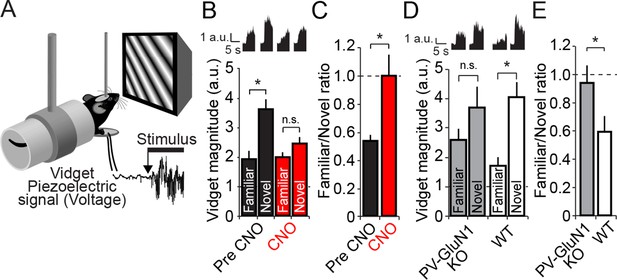
Discrimination of familiar and novel oriented stimuli involves PV+ neurons in V1 and NMDAR expressed within PV+ neurons.
(A) Using the same protocol as described in Figure 3B, mice were progressively familiarized with a specific oriented stimulus. On the test day, as mice viewed familiar and novel stimuli, vidget behavioral responses were measured via a piezoelectric sensor located beneath the forepaws of the head-fixed mouse. (B) After a standard SRP protocol, mice expressing hM4D(Gi) receptors selectively within PV+ cells of binocular V1 were exposed to both familiar and novel stimuli. Prior to application of CNO, mice exhibited significant behavioral evidence of discriminating this familiar orientation from interleaved presentations of a novel oriented stimulus (black bars). After application of CNO, there was no longer successful discrimination of familiar and novel stimuli (red bars). Averaged vidget responses are displayed at the top of this panel. (C) A significant difference was observed in the ratio of response to familiar and novel stimuli from pre CNO (black) to post CNO (red). (D) A deficit in OSH was also apparent in PV GluN1 KO mice as vidget recordings demonstrated a failure to significantly discriminate familiar from novel orientations (gray bars). WT littermates exhibited significantly greater vidget magnitudes for novel than familiar stimuli, indicating unimpaired discrimination of familiarity from novelty (white bars). Averaged behavioral responses are displayed above with accompanying scale bars. (E) The significant deficit of PV GluN1 KO mice in discriminating familiar from novel stimuli is apparent in the ratio of behavior elicited by the familiar over the novel stimulus in comparison to WT littermates. Significant comparisons are marked with an asterisk throughout while non-significant comparisons are marked with n.s. Error bars are standard error of the mean (S.E.M.).
-
Figure 8—source data 1
Novelty detection after PV+ neuronal disruption.
- https://doi.org/10.7554/eLife.11450.022
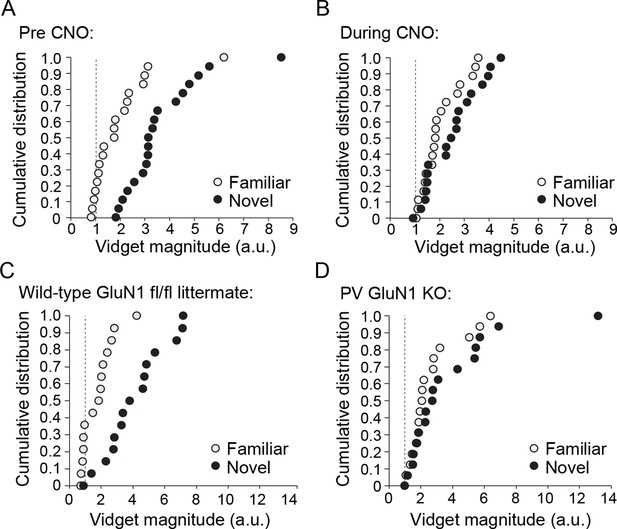
Cumulative distributions of average vidget behavioral response to familiar and novel stimuli for each individual animal included in analysis presented in Figure 8D and Figure 8E.
(A) Prior to CNO administration, average behavioral response to the familiar stimulus (light gray) and the novel stimulus (black) of each individual animal bilaterally expressing hM4D(Gi) in PV+ neurons of binocular V1. Dotted line represents behavior no greater than pre stimulus baseline. (B) Average behavioral response of the same animals to the familiar and a new novel stimulus during PV+ neuron inactivation via CNO delivery, revealing consequent deficit in discrimination of familiar and novel stimuli. (C) Cumulative distributions of average vidget behavioral response to the familiar stimulus (light gray) and the novel stimulus (black) of each individual WT GluN1 fl/fl mouse. (D) Average vidget behavioral response of each PV-GluN1 KO mouse to the familiar and a new novel stimulus, revealing deficit in discrimination of familiar and novel stimuli. Dotted line represents behavior no greater than pre stimulus baseline.
-
Figure 8—figure supplement 1—source data 1
Per animal plots of Novelty detection after PV+ neuronal disruption.
- https://doi.org/10.7554/eLife.11450.024





