Membrane palmitoylated protein 2 is a synaptic scaffold protein required for synaptic SK2-containing channel function
Figures
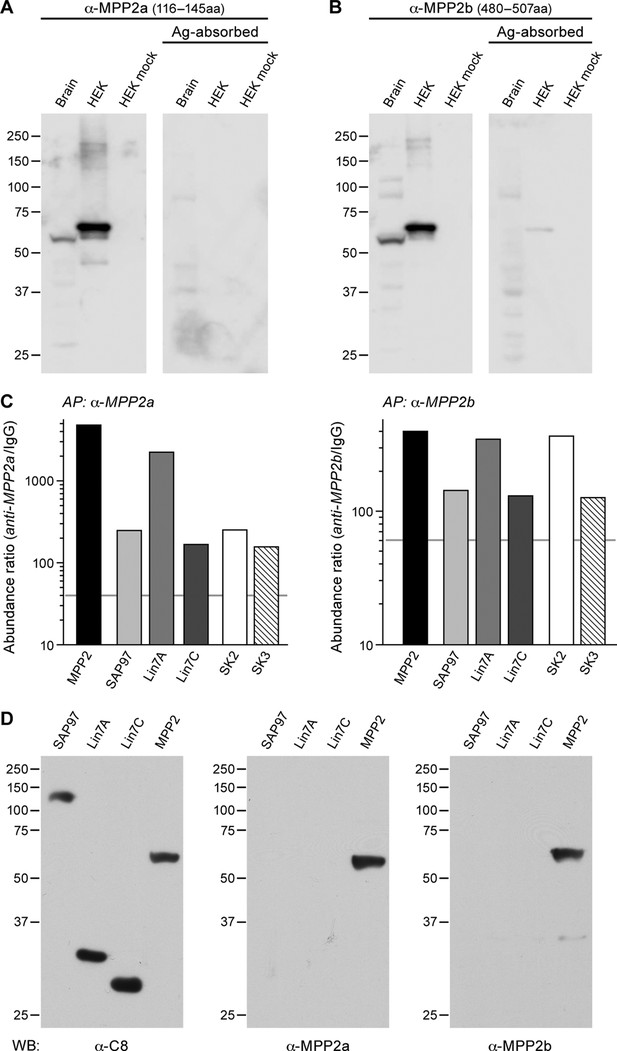
MPP2 interacts with SK2.
(A, B) Western blots were prepared using mouse brain homogenate (Brain; 100 μg) or HEK293 cell extracts (1% of 10 -cm plate), either transfected to express FLAG-MPP2 (HEK) or empty plasmid (HEK mock). Duplicate lanes were prepared and one set was probed with the indicated MPP2 antibody (left panels) while the second set was probed with the same antibody after pre-absorbing with the immunizing antigen (right panels). Native MPP2 and transfected FLAG-MPP2 were detected only in the left panels. (C) Bar graphs illustrating abundance ratios determined for the indicated proteins in APs with two anti-MPP2 antibodies and IgG as a negative control. Horizontal lines denote threshold for specificity of co-purification. (D) Western blots of proteins prepared from HEK293 cells transfected with C8-tagged SAP97, Lin7A, Lin7C, and MPP2, and probed with anti-C8 antibody (left), anti-MPP2a antibody (middle), and anti-MPP2b antibody (right). The MPP2 antibodies recognized only MPP2.

Coverage of the primary sequences of all proteins shown in Figure 1C.
Peptides identified by mass spectrometry are in red; those theoretically accessible but not detected in MS-analyses are in black, and peptides not accessible to MS-analyses under the settings used (peptides with mass values below 738 Da ([absolute lower mass cutoff)] or above 3000 Da ([practical mass limit of the C18 RP-HPLC separation)] are given in grey). The sum of amino acids in red is either related to the accessible primary sequence (relative coverage) or to the entire primary sequence (absolute coverage); transmembrane domains are underlined.

Coverage of the primary sequences of all proteins shown in Figure 1C (continued).
See caption above.
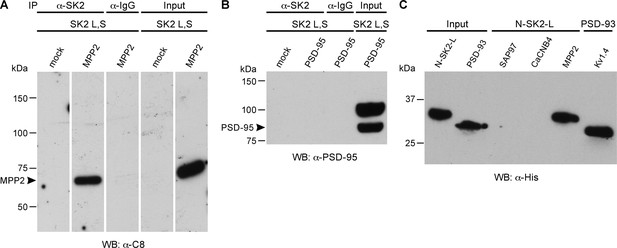
MPP2 interacts with the N-terminal domain of SK2-L.
(A) Co-immunoprecipitations. Western blots prepared from HEK cell lysates expressing SK2-S and SK2-L, either alone (mock) or together with C8-MPP2, immunoprecipitated with either anti-SK2 antibody or IgG (control), and probed for C8-MPP2. Adjacent blot shows input of C8-MPP2. (B) A similar experiment except using PSD-95 instead of C8-MPP2. PSD-95 is expressed but does not co-IP with SK2. (C) GST-pull-downs. Blot probed with anti-His antibody shows input prey proteins, His-SK2-L N-terminal domain and His-PSD-93. After exposure to GST-baits representing SH3-HOOK-GK domains of SAP97, CaCNB4, or MPP2, the His-SK2-L N-terminal domain was specifically retained by GST-MPP2. Positive control shows interaction between GST-C-terminal domain of Kv1.4 and His-PSD-93. Co-immunoprecipitations and GST-pull-downs were performed in triplicate.
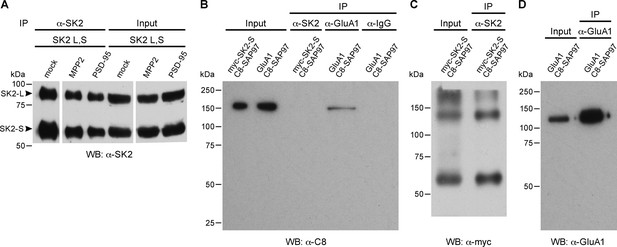
Co-immunoprecipitations.
(A) Immunoprecipitation and input of SK2-S plus SK2-L. Western blot probed with anti-SK2 antibody raised in rabbit. First three lanes show immunoprecipitates from transfected HEK293 cell lysates co-transfected with empty vector (mock), C8-MPP2, or PSD-95 (see Figure 2). Immunoprecipitations were performed using anti-SK2 antibody raised in guinea pig. Last three lanes show input material prior to immunoprecipitation. (B) Co-immunoprecipitation of C8-SAP-97 co-expressed in HEK293 cells with GluA1 but not with myc-SK2-S. Western blot using anti-C8 antibody detects input of C8-SAP-97 co-expressed with myc-SK2-S or GluA1. C8-SAP-97 co-immunoprecipitated with anti-GluA1 antibody but not with anti-SK2 antibody or IgG. (C) Western blot using anti-myc antibody detects myc-SK2-S co-expressed with C8-SAP-97, input and after immunprecipitation with anti-SK2 antibody. Higher MW bands correspond to aggregates of SK2-S. (D) Western blot using mouse monoclonal anti-GluA1 antibody detects GluA1 co-expressed with C8-SAP-97, for GluA1 input and GluA1 immunoprecipitated with rabbit polyclonal anti-GluA1 antibody.
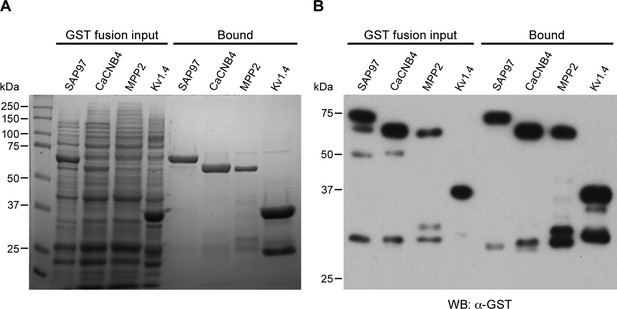
GST-fusion protein expression.
(A) Coomassie stained gel showing input bacterial lysates (left lanes) for the indicated GST-fusion proteins prior to being bound to glutathione agarose beads, and after binding to beads (right lanes). (B) Western blot of the gel in panel A, probed with anti-GST antibody.
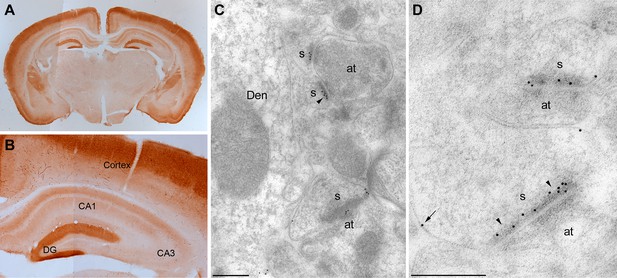
Localization of MPP2 in the hippocampus.
(A,B) Light microscopic images of anti-MPP2 antibody labelling in hippocampus. MPP2 was present throughout the hippocampus and was prominent in the dendritic arbors of area CA1. (C,D) Electron micrographs of the hippocampus showing immunoparticles for MPP2 in the stratum radiatum of the CA1 region of the hippocampus, as detected using a post-embedding immunogold method. Immunoparticles for MPP2 were detected along the PSD (arrowheads) of dendritic spines (s) of CA1 pyramidal cells establishing asymmetrical synapses with axon terminal (at), as well as at extrasynaptic sites (arrows) of dendritic spines (s). Den, dendritic shaft. Scale bars in C,D: 0.2 μm.
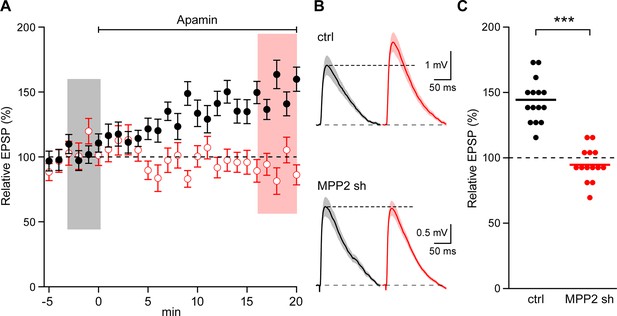
MPP2 is required for synaptic SK2-containing channel function.
(A) Time course of the normalized EPSP amplitude (mean ± s.e.m.) for baseline in control ACSF (Ctrl) and during wash-in of apamin (100 nM) as indicated above in MPP2 sh-transfected cells (open red symbols, n = 14) and non-fluorescent control cells (black symbols, n = 15) mice. (B) Average of 15 EPSPs taken from indicated shaded time points in aCSF (black) and 16–20 min after application of apamin (red); shaded areas are mean ± s.e.m for control non-fluorescent cells (ctrl, upper traces) and MPP2 sh-transfected cells (MPP2 sh, bottom traces). (C) Scatter plot of relative ESPS peak compared to baseline from the individual slices in panel A non-fluorescent control (ctrl, black symbols) and for MPP2 sh transfected (red symbols). Horizontal bar reflects mean response.
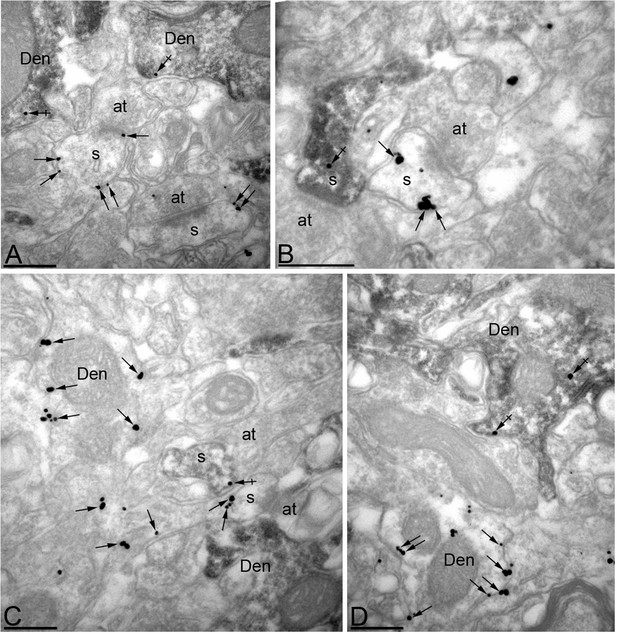
Efficient knock-down of MPP2 expression in CA1 pyramidal neurons.
Immunoreactivity for MPP2 in the CA1 region of the hippocampus, as revealed using a double-labelling pre-embedding method. (A–D) The peroxidase reaction end product (HRP) indicating GFP immunoreactivity filled CA1 pyramidal cells, whereas immunoparticles for MPP2 were mainly located along the plasma membrane and at intracellular sites of pyramidal cells. Immunoparticles for MPP2 were distributed in both GFP-positive (crossed arrows) and GFP-negative (arrows) dendritic spines (s) and dendritic shafts (Den) of pyramidal cells. However, there was a striking reduction of immunoparticles for MPP2 in GFP-positive profiles compared to GFP-negative profiles (see text). at, axon terminal. Scale bars in A-D: 0.2 μm.
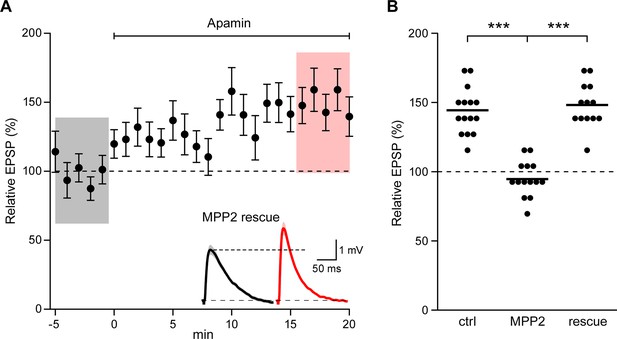
Co-expression of sh-immune MPP2 with MPP2 shRNA rescues synaptic SK2 function.
(A) Time course of the normalized EPSP amplitude (mean ± s.e.m.) for baseline in control ACSF and during wash-in of apamin (100 nM) as indicated above in cells transfected MPP2 sh and MPP2 sh immune (n = 13). Inset: representative cell showing average of 15 EPSPs taken from indicated shaded time points in ACSF (black) and 16–20 min after application of apamin (red); shaded areas are mean ± s.e.m. (B). Scatter plot of relative ESPS peak compared to baseline from the individual slices for non-fluorescent control, MPP2 sh-transfected cells and MPP2 sh transfected with immune MPP2 (rescue). Horizontal bar reflects mean.
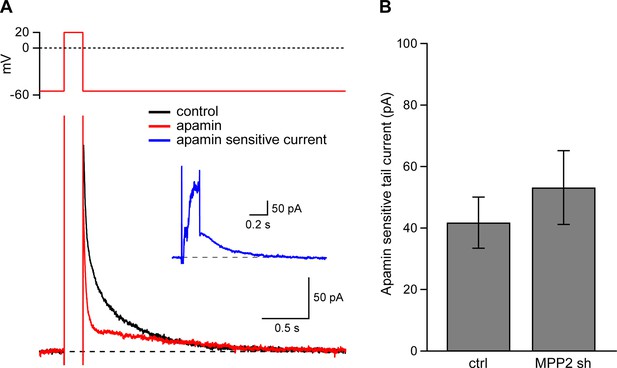
Dendritic SK channel function is not reduced by MPP2 knock-down.
(A) Representative traces of voltage-clamp recordings of IAHP after a 200 ms depolarizing pulse to 20 mV in an MPP2 sh-transfected cell. Apamin (red trace) blocks a component of the IAHP. Inset: subtraction of the traces before and after apamin application yielded the apamin-sensitive ImAHP. (B) Bar graph of apamin-sensitive tail current measured at 100 ms following repolarization to −55 mV. Data presented as mean ± s.e.m. for control non-fluorescent cells (n = 13) and MPP2 sh-transfected cells (n = 7).
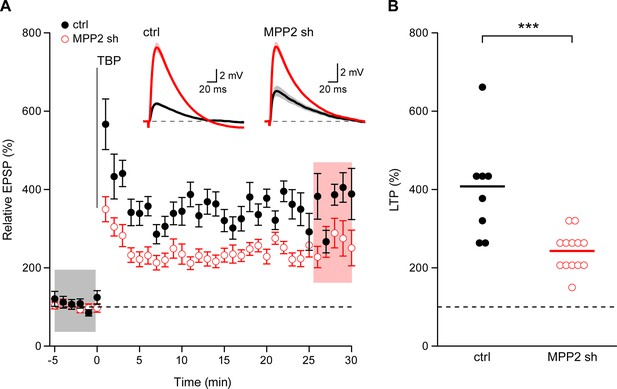
Loss of MPP2 reduces LTP.
(A) Time course of the normalized EPSP amplitude (mean ± s.e.m.) from control non-fluorescent cells (ctrl, closed black symbols, n = 8) and MPP2 sh-transfected cells (MPP2, open red symbols, n = 13). The TBP protocol was delivered at time 0. Inset: representative cell showing average of 15 EPSPs taken from indicated shaded time points in ACSF (black) and 25–30 min after the induction of LTP (red); shaded areas are mean ± s.e.m. (B). Scatter plot of relative ESPS peak compared to baseline from the individual slices for non-fluorescent control (ctrl) and MPP2 sh-transfected cells. Horizontal bar reflects mean response.
Additional files
-
Supplementary file 1
Relative protein abundance in a sample versus control.
- https://doi.org/10.7554/eLife.12637.015





