Cytoplasmic NOTCH and membrane-derived β-catenin link cell fate choice to epithelial-mesenchymal transition during myogenesis
Figures
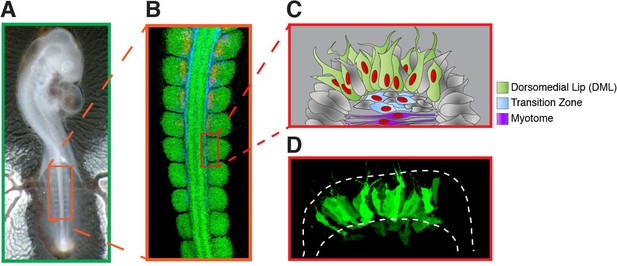
Summary of the experiments performed in this study.
(A) Chick embryos at HH stage 15–16 (24–28 somites) were electroporated in the medial part of the (4–5) newly formed somites (boxed region). (B) Immunostaining of a HH 15 chick embryo with PAX7 (in green), HNK1 (in blue) and MYF5 (in red) to label the dermomyotome, the neural crest and the myotome, respectively. (C) Schematic illustrating regions of the somite that are represented in the confocal stacks of images shown throughout the study. It shows the medial portion of a somite 6–24 hr after its DML was electroporated. Typically, it leads to the mosaic expression (in green) of the electroporated construct(s) in the DML, the transition zone (in blue) and the nascent primary myotome (in purple). (D) is a maximum intensity projection of a confocal stack of a somite electroporated with GFP 6 hr prior to analysis.
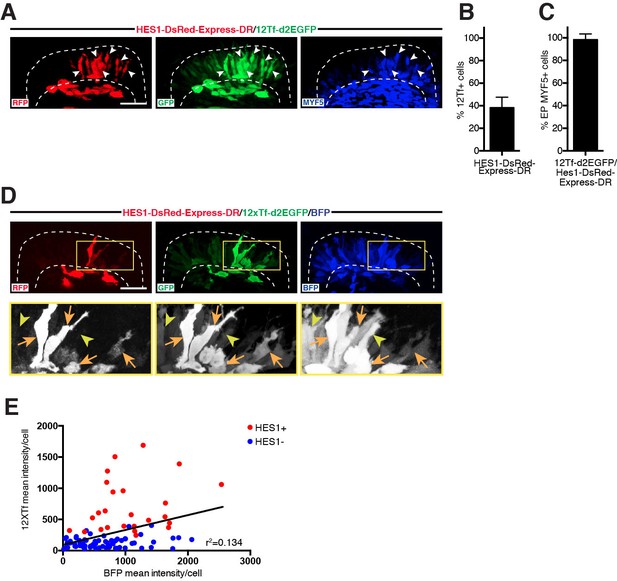
NOTCH signaling is associated with elevated TOPflash activity in early myogenesis.
(A,D) Confocal stacks of somites, in dorsal view, 12 hr after electroporation of HES1-DsRedExpress-DR (NOTCH reporter, in red), 12Tf-d2EGFP (TOPflash reporter, in green), an ubiquitously expressed H2B-BFP (blue in D) or immunostained for MYF5 expression (blue in A). (B–C) Bar charts showing (B) 38% of cells activating the NOTCH reporter within the TOPflash reporter-positive population and (C) 98.5% of cells expressing MYF5 that co-activate the NOTCH and the TOPflash reporters. Lower panels (black and white in D) show enlargements of the yellow boxes (upper panels in D). Orange arrows indicate DML cells positive for the NOTCH reporter and strongly positive for the TOPflash reporter; yellow arrowheads show cells negative for NOTCH and weak or strong for TOPflash. (E) Linear regression chart plotting mean fluorescence intensity of CAGGS-BFP-positive cells against the mean fluorescence intensity of 12Tf-d2EGFP-positive cells ('Y=0.2378*X+93.61', r(96)=0.134, p<0.0002). Indicated is best-fit regression line for entire population (Hes1+: NOTCH reporter-positive, red dots; Hes1-: NOTCH reporter-negative, blue dots) and r-squared value. In each panel are indicated the antigens that were detected by immunostaining. Abbreviation: EP: electroporation. Scale bars: 50 μm
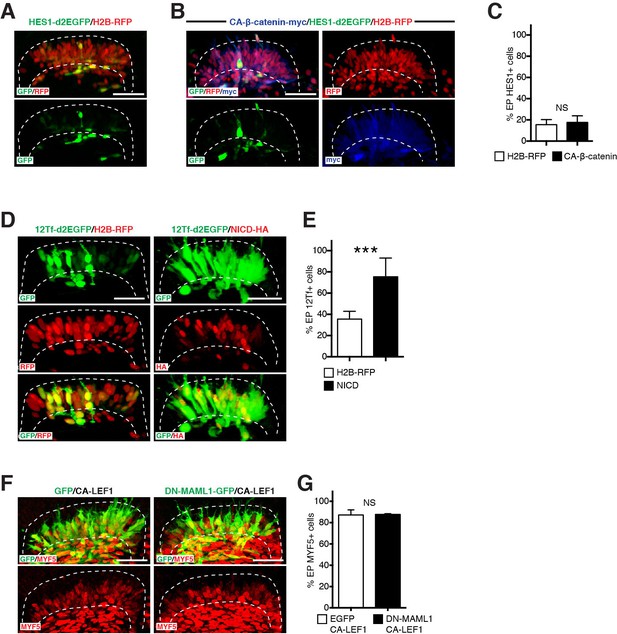
NOTCH regulates regulates TCF/β-catenin transcriptional activity during myogenesis.
(A,B) Confocal stacks, 12 hr after electroporation of (A) the NOTCH reporter HES1-d2EGFP in green, H2B-RFP (in red) and (B) with a constitutively active form of β-catenin (in blue). (C) Bar charts showing 15.6% of HES1-d2EGFP-positive cells in the control (in white) or 17.7% with CA β-catenin (in black). (D) Confocal stacks of somites 6 hr after co-electroporation of 12Tfd2EGFP (TOPflash reporter, in green), and an ubiquitously expressed H2B-RFP (in red, left) or NICD (in red, right). (E) Bar charts showing 35.5% of cells expressing the TOPflash reporter in the controls (in white) or 75.4% with NICD (in black). (F) Confocal stacks of somites 6 hr after co-electroporation of a constitutive active form of LEF1 and GFP (in green, left) or a dominant negative form of MAML1 fused to GFP (in green, right) and a constitutive active form of LEF1; MYF5 expression is shown in red. (G) Bar charts showing 87.2% of MYF5-positive cells in DN-MAML1/CA-LEF1-positive cells and 87.8% MYF5-positive cells when expressing CA-LEF1 alone. In each panel are indicated the antigens that were detected by immunostaining. Abbreviation: EP: electroporation. Scale bars: 50 μm
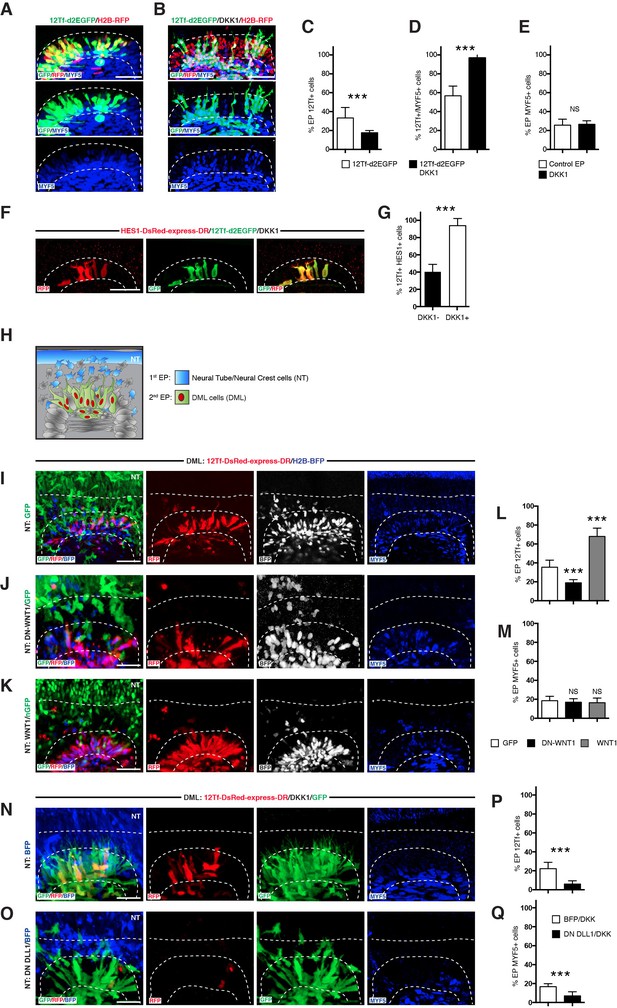
Activation of TCF/β-catenin transcriptional activity and myogenesis by NOTCH is WNT-ligand independent.
(A,B) Confocal stacks of somites, 6 hr after co-electroporation of 12Tf-d2EGFP (TOPflash reporter, in green), with H2B-RFP (in red) without (A) or with (B) DKK-1, and immunostained for MYF5 (in blue). (C–E) Bar charts showing (C) 33.3% of TOPflash-positive cells in the controls (in white) or 17.7% with DKK-1 (in black); (D) Bar charts indicating 56.8% of TOPflash-positive cells are MYF5-positive in control embryos, while 96.9% of TOPflash-positive cells are MYF5-positive when DKK1 is present; (E) percentage of electroporated cells that are MYF5-positive in controls: 25.7%; with DKK1: 26.6%. (F) Confocal stacks of somites after co-electroporation of HES1-DsRed-express-DR (NOTCH reporter, in red) or with 12Tf-d2EGFP (TOPflash reporter in green) and DKK1. (G) Bar charts showing the% of electroporated cells co-expressing the TOPflash and the NOTCH reporters when DKK1 is present (91.4%, in black) or absent (39.9%, in white). (H) Schematic representing the design of the double electroporation experiments, with a first electroporation targeting the neural tube (NT) and the migrating neural crest (in blue), followed by electroporation in the DML. (I–K) Confocal stacks of somites showing the expression of 12Tf-DsRed-express-DR (TOPflash reporter, in red) and the electroporation marker H2B-BFP (in blue) in the DML. Dorsal neural tube was electroporated with (I) GFP, (J) dominant-negative WNT1 (in green), or (K) wild-type WNT1 (in green). (L) Bar charts showing 19% of DN-WNT1-positive electroporated cells express the TOPflash reporter (in black), 67.6% of WT-WNT1-positive electroporated cells express the TOPflash reporter (in gray), controls (in white): 35.5%. (M) Bar charts showing 15.04% of DN-WNT1-positive electroporated cells express MYF5 (in black), 15.07% for WT-WNT1-positive electroporated cells (in gray), controls (in white): 15.08%. (N,O) Confocal stacks of somites showing the TOPflash reporter 12Tf-DsRed-express-DR (in red), the electroporation marker GFP (in green) and DKK1, electroporated into the DML. The dorsal neural tube was electroporated with (N) BFP or (O) dominant-negative DLL1 and BFP. (P) Bar charts showing the% of electroporated cells expressing the TOPflash reporter, DN-DLL1 + DKK1: 6%, in black; controls with DKK1: 22.3%, in white. (Q) Bar charts showing the% of electroporated cells expressing MYF5, DN-DLL1 + DKK1: 6.8% in black; controls with DKK1: 16.6%, in white. Dotted lines indicate the borders of the neural tube and somites. Abbreviations: NT: neural tube; DML: Dorso-Medial Lip; EP: electroporation. In each panel are indicated the antigens that were detected by immunostaining with the exception of native BFP blue fluorescence. Scale bars: 50 μm.
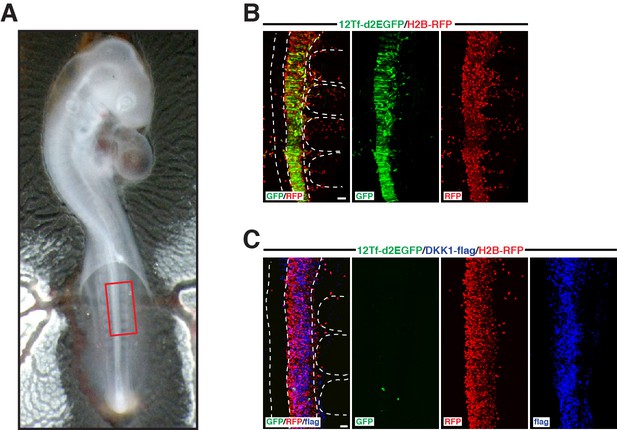
Expression of DKK1 in the neural tube abrogates WNT/β-catenin response.
(A) shows a E2.5 chick embryo at the end of the experiment. The box in (A) indicates the region that is analyzed in B and C. (B) Confocal stacks of a control embryo showing the expression of the TOPflash reporter 12Tf-d2EGFP (in green). In red, RFP immunostaining, identifying all electroporated cells. (C) Neural tube of an embryo in which DKK1 (in blue) was co-electroporated with the TOPflash reporter (in green) and the electroporation marker H2B-RFP (in red). Dotted lines indicate the borders of the neural tube and somites. In each panel are indicated the antigens that were detected by immunostaining. Scale bars: 50 μm.
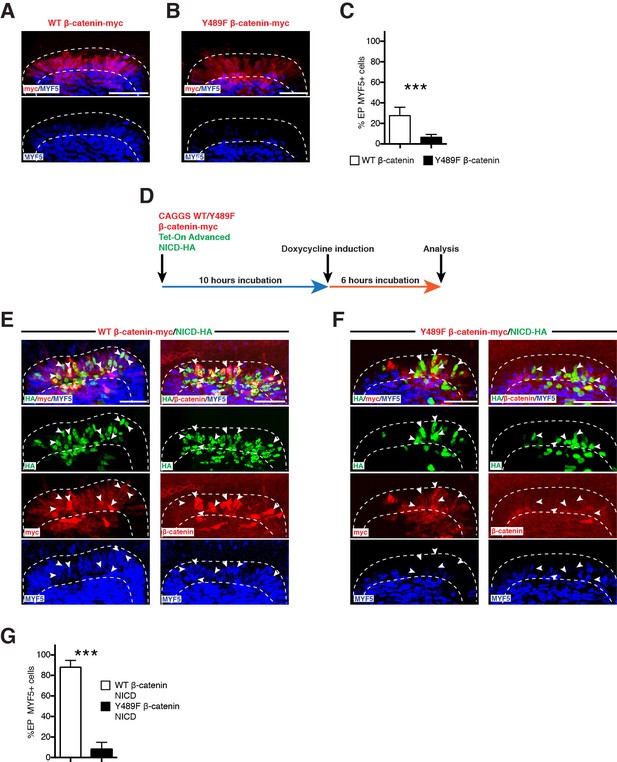
β-catenin from the cell membrane and AJ is required for the NOTCH-mediated activation of MYF5 expression.
(A,B) Confocal stacks of somites after electroporation of WT β-catenin (A) or Y489F β-catenin (B). MYF5 expression is shown (in blue). (C) Bar charts showing the% of electroporated cells that were positive for MYF5 after electroporation of WT β-catenin (27.7%, in white) or Y489F β-catenin (6.4%, in black). (D) Schematic of the experimental design that was followed: (Myc-tagged) WT or a mutant Y489F β-catenin under a CAGGS ubiquitous promoter was electroporated in the DML together with the Tet-On Advanced transactivator (rtTA) and NICD inserted in the response vector. The β-catenin (WT or mutant) was expressed from the start of the experiment; NICD was induced 10 hr later. (E,F) Confocal stacks of somites after electroporation of (E) NICD (in green) and WT β-catenin (in red) or (F) NICD (in green) and Y489F β-catenin (in red). MYF5 expression is shown (in blue). (G) Bar charts showing the% of MYF5-positive cells after electroporation of NICD with WT β-catenin (88%, in white) or Y489F β-catenin (8.1%, in black). In each panel are indicated the antigens that were detected by immunostaining. DML: dorso-medial lip. Scale bars: 50 μm
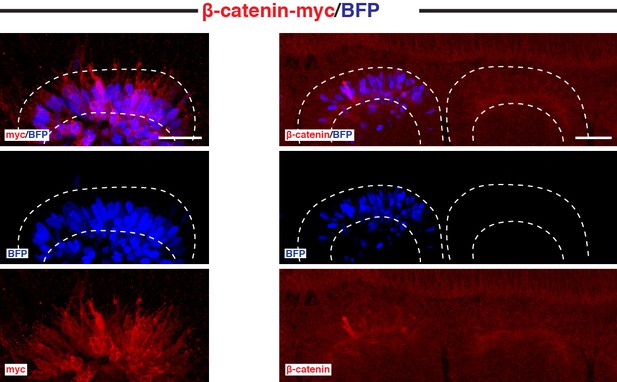
Endogenous and exogenous β-catenin are similarly expressed in the DML.
Confocal optical slices of somites, 6 hr after electroporation of a DML with CAGGS-WT β-catenin and stained for either c-myc tag or endogenous β-catenin. Shown is a non-electroporated somite as reference. In each panel are indicated the antigens that were detected by immunostaining. Scale bars: 50 μm.
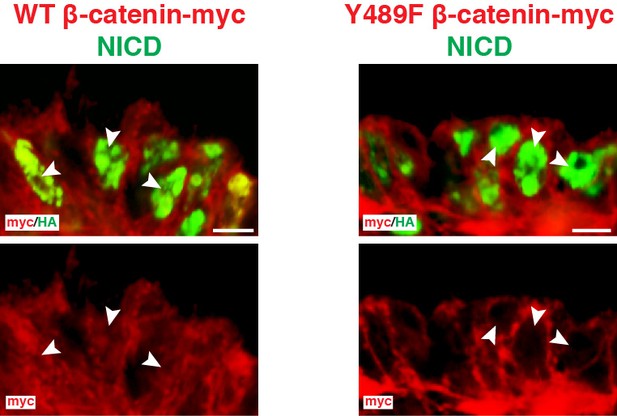
Cellular localization of WT and Y489F β-catenin along with NICD.
Confocal optical slices of DML cells electroporated with (in red) WT β-catenin or Y489F β-catenin, 6 hr after induction of NICD (in green). Arrows in first image show that nuclear NICD co-localizes with prominent β-catenin staining. Arrows in second image show that nuclear NICD does not co-localize with Y489F β-catenin. In each panel are indicated the antigens that were detected by immunostaining. Scale bars: 10 μm.
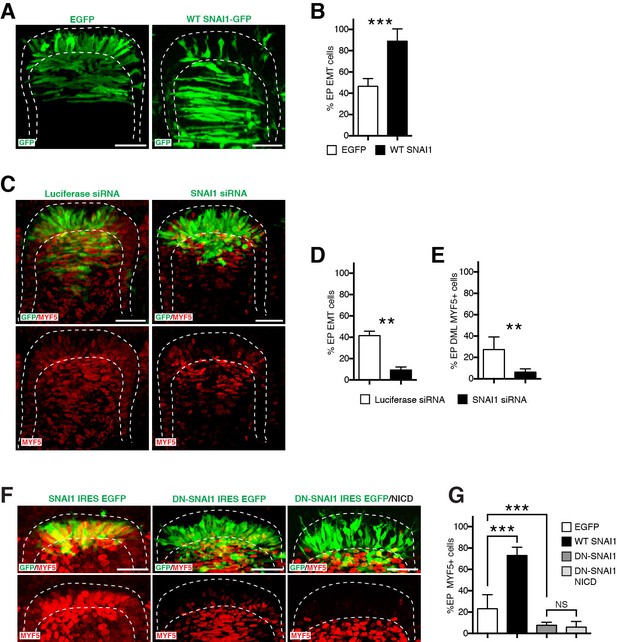
SNAI1 is a necessary and sufficient step for the NOTCH-dependent activation of MYF5.
(A) Confocal stacks of somites, 17 hr after electroporation of EGFP as control or with SNAI1. (B) Bar charts showing the% of cells that have entered the primary myotome in the control (46.7%, in white) or after electroporation of SNAI1 (89%, in black). (C) Confocal stacks of somites, 17 hr after electroporation of a luciferase-specific siRNA as control or a SNAI1-specific siRNA, immunostained for MYF5 (in red). (D–E) Bar charts showing the% of electroporated cells that have entered the primary myotome (D) in the control (41.6%, in white) of with siRNA SNAI1 (9.3%, in black) and the% of electroporated cells that are MYF5-positive (E) in the control (26.2%, in white) or with siRNA SNAI1 (6.3%, in black). (F) Confocal stacks of somites, 6 hr after electroporation of a WT chicken SNAI1 (in green, left), a dominant negative (DN) form of SNAI1 (in green, right) alone or together with NICD. MYF5 expression is indicated (in red). (G) Bar charts showing the% of MYF5-positive cells after GFP electroporation (23.1%, in white), with chicken SNAI1 (73%, in black), DN SNAI1 alone (7.7%, in dark grey) or together with NICD (6%, in light grey). In each panel are indicated the antigens that were detected by immunostaining. Abbreviation: EP: electroporation. Scale bars: 50 μm.
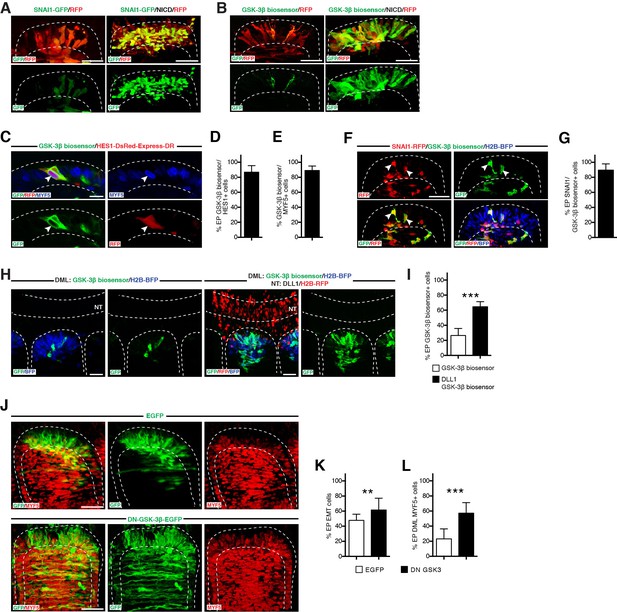
NOTCH regulates SNAI1 degradation through inhibition of GSK-3β activity.
(A) Confocal stacks of somites, 6 hr after electroporation of RFP (in red) and SNAI1-GFP (fusion of SNAI1 and GFP, in green) alone or together with NICD. (B) Confocal stacks of somites, 6 hr after electroporation of GSK-3β biosensor (in green) and RFP (in red) alone (left), or together with NICD (right). (C) Confocal stacks of a somite 6 hr after co-electroporation of the GSK-3β biosensor (green) and the NOTCH reporter (red) and immunostained for MYF5 (blue). (D,E) Bar charts showing the% of GSK-3β biosensor-positive cells that activate NOTCH signaling (86.9%, D) and that are MYF5-positive (88.2%, E). (F) Confocal stacks of a somite 6 hr after co-electroporation of a SNAI fused to RFP (in red) and the GSK-3β biosensor (in green). In blue the electroporated cells, identified by H2B-BFP. (G) Bar charts showing 89.4% of DML electroporated cells are GSK-3β biosensor and SNAI1-positive. (H) Confocal stacks of somites and adjacent neural tube electroporated as described in Figure 4H, only in the DML with the GSK-3β biosensor (in green) and H2B-BFP (in blue, left panels) or double-electroporated in the DML with the GSK-3β biosensor (in green) and H2B BFP (in blue) and in the neural tube with DLL1 under the control of a neural crest-specific promoter (right panels). (I) Bar charts showing the% of GSK-3β biosensor-positive cells in the control (26.4%, in white) or with DLL1 expressed in the neural crest (64.6%, in black). (J) Confocal stacks of somites 17 hr after electroporation of GFP as control or DN-GSK-3β and immunostained for MYF5 (red). (K) Bar charts showing the% of electroporated cells that have entered the primary myotome in the control (45.8%, in white) of with the DN-GSK-3β (61.2%, in black). (L) Bar charts showing the% of electroporated cells that are MYF5-positive in the control (23.1%, in white) of with the DN-GSK-3β (57.2%, in black). In each panel are indicated the antigens that were detected by immunostaining, with the exception of native BFP blue fluorescence. Abbreviation: EP: electroporation; NT: neural tube. Scale bars: 50 μm.
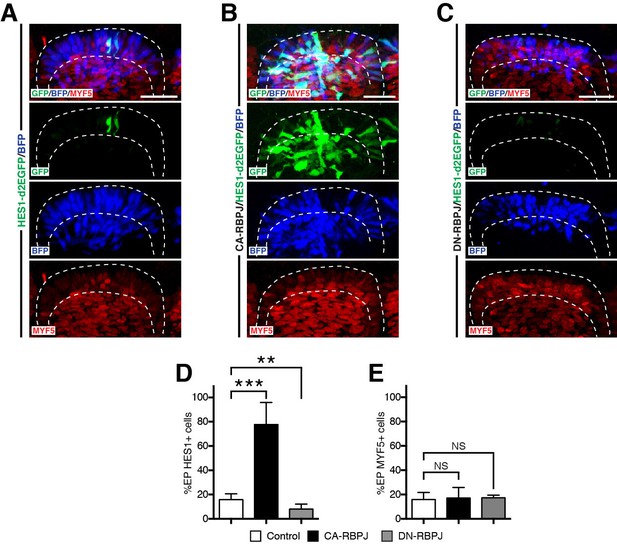
RBPJ does not regulate myogenesis in the DML.
(A–C) Confocal stacks of somites 6 hr after co-electroporation of H2B-BFP (blue) and the NOTCH reporter (green) alone (A) or together with CA-RBPJ (B) or DN-RBPJ (C) and immunostained for MYF5 (red). (D) Bar charts showing the percentage of electroporated cells positive for the NOTCH reporter in the controls (15.8%, in white), with CA-RBPJ (77.6%, in black) or DN-RBPJ (8%, in grey). (E) Bar charts showing the percentage of electroporated cells positive for MYF5 in the controls (15.9%, in white), with CA-RBPJ (17.1%, in black) or DN-RBPJ (17.3%, in grey). In each panel are indicated the antigens that were detected by immunostaining, with the exception of native BFP blue fluorescence. Abbreviation: EP: electroporation. Scale bars: 50 μm.
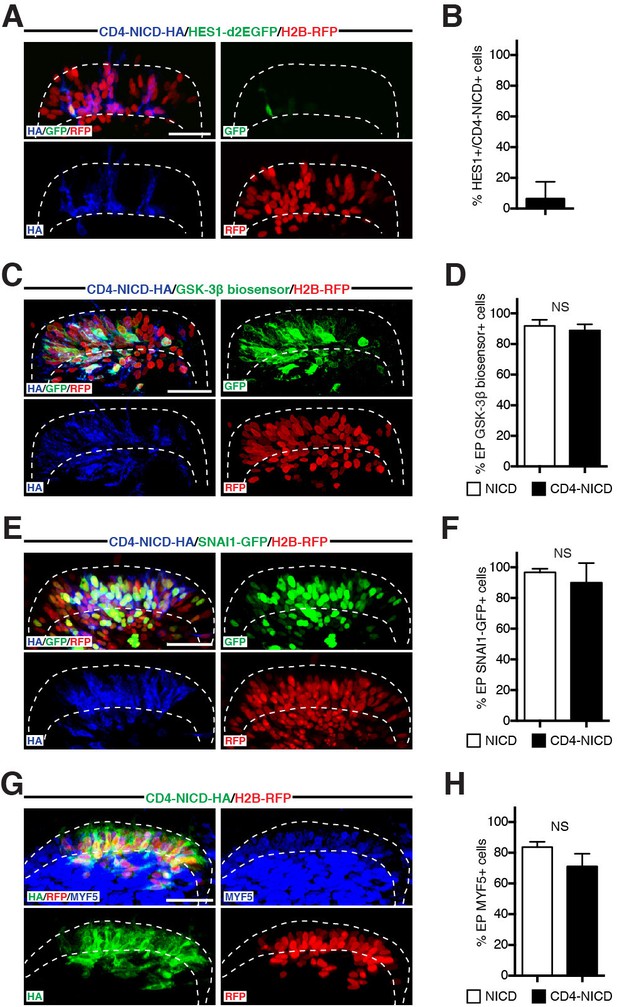
NOTCH1 controls myogenesis independently of its transcriptional role in the nucleus.
(A) Confocal stacks of somites, 6 hr after co-electroporation of a HA-tagged, membrane-tethered NICD, CD4-NICD (in blue), a NOTCH reporter (in green) and H2B-RFP (in red). (B) Bar charts showing 6.4% of NOTCH reporter-positive cells electroporated with CD4-NICD. (C) Confocal stacks, 6 hr after electroporation of CD4-NICD (in blue), GSK-3β biosensor (in green) and H2B-RFP (in red). (D) Bar charts showing the percentage of GSK-3β biosensor-positive cells after electroporation with NICD (91.6%, in white) or with CD4-NICD (88.6%, in black). (E) Confocal stacks, 6 hr after co-electroporation of CD4-NICD (in blue), SNA1-GFP (in green) and H2B-RFP (in red). (F) Bar charts showing the percentage of SNAI1-GFP-positive cells after electroporation with NICD (96.7%, in white) or with CD4-NICD (90%, in black). (G) Confocal stacks, 6 hr after co-electroporation of CD4-NICD (in green), H2B-RFP (in red) and immunostained for MYF5 (in blue). (H) Bar charts showing the percentage of MYF5-positive cells after electroporation with NICD (83.7%, in white) or with CD4-NICD (71.1%, in black). In each panel are indicated the antigens that were detected by immunostaining. Abbreviation: EP: electroporation. Scale bars: 50 μm
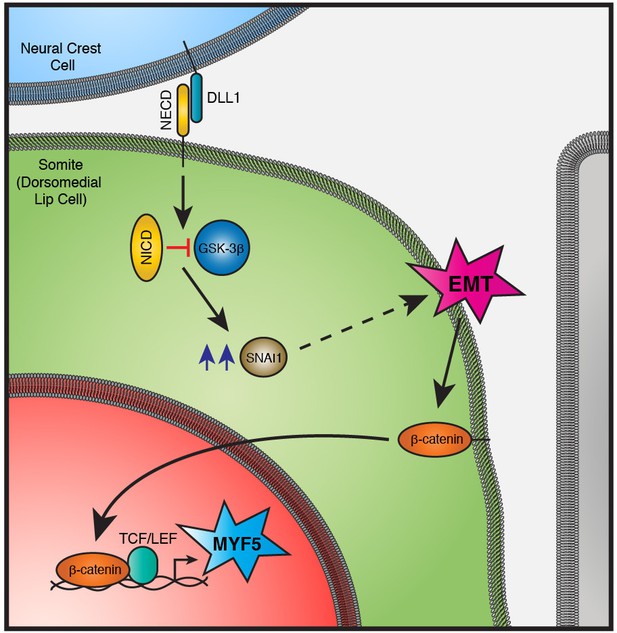
A signaling module in the DML that combines EMT and cell fate change.
The physical contact with DLL1-positive neural crest cells triggers the cleavage of NOTCH receptor and the release of NICD in DML cells. NICD represses the activity of GSK-3β, independent of its transcriptional activity. The accumulation of SNAI1 (blue arrows) allows its translocation in the nucleus (not shown) where it activates EMT. This releases β-catenin from the adherens junction and/or membrane, leading it to enter into the nucleus where, together with TCF/LEF co-activator, it activates MYF5 expression. Abbreviations: DLL1: Delta-like 1; NECD: NOTCH extracellular domain; NICD: NOTCH intracellular domain; EMT: Epithelial to Mesenchymal Transition.





