Tetherin is an exosomal tether
Figures
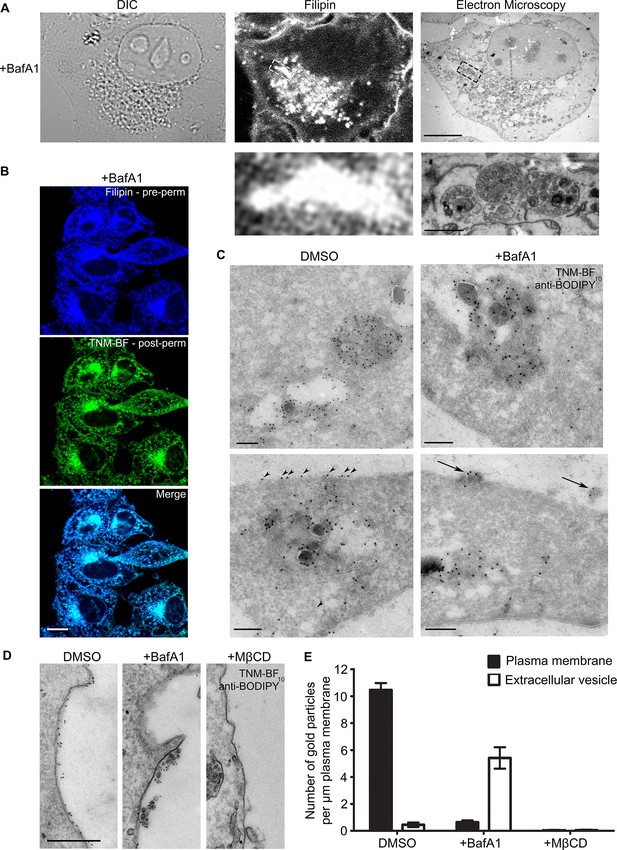
BafA1 treatment causes cholesterol to accumulate in intralumenal vesicles of multivesicular bodies and to be lost from the plasma membrane.
(A) HeLa cells were treated with BafA1 (100 nM, 16 hr), then fixed, stained with the cholesterol probe filipin, and prepared for correlative light and electron microscopy (CLEM). Scale bars: 10 μm (upper) and 1 μm (lower). (B) BafA1-treated cells (100 nM, 16 hr) were stained with filipin, then permeabilised and stained with TNM-BF. Scale bar: 20 μm. (C) Ultrathin cryosections of mock-treated and BafA1-treated HeLa cells (100 nM, 16 hr) were labelled with TNM-BF and stained with rabbit anti-BODIPY followed by 10 nm protein A-gold. There is labelling both in endosomes (upper panels) and at the cell surface (lower panels). Scale bars: 200 nm. (D) Intact mock-treated, BafA1-treated (100 nM, 16 hr), or MβCD-treated (10 mM, 30 min) HeLa cells were labelled with TNM-BF followed by anti-BODIPY and protein A-gold, revealing surface cholesterol localisation. Scale bar: 500 nm. (E) Quantification of TNM/BF/anti-BODIPY gold labeling density for DMSO-treated, BafA1-treated (100 nM, 16 hr), or MβCD-treated (10 mM, 30 min) cells. Graphs show mean ± S.E.M for at least 100 μm of the plasma membrane, over two independent experiments. See also Figure 1—figure supplement 1.
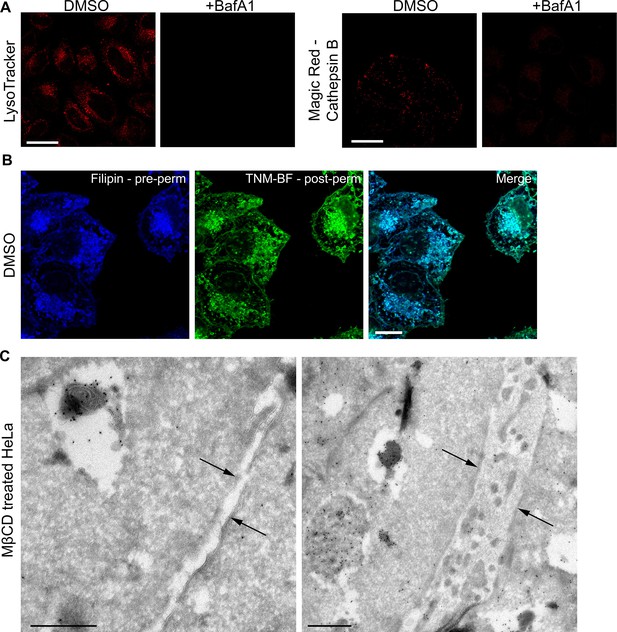
Controls for specificity.
(A) LysoTracker or Magic Red staining of DMSO-treated or BafA1-treated HeLa cells (100 nM, 16 hr). Lysotracker labels acidic organelles, while Magic Red labels organelles containing active acid hydrolases. The loss of signal upon BafA1 treatment shows that organelle acidification has been abolished. Scale bar: 20 μm (B) DMSO-treated HeLa cells were stained with filipin, permeabilised and stained with TNM-BF. Scale bar: 20 μm. (C) Ultrathin cryosections of cells treated with MβCD (10 mM, 30 min), which extracts cholesterol from the plasma membrane, were stained with TNM-BF and labelled with anti-BODIPY followed by 10 nm protein A-gold. Arrows indicate the plasma membrane. Scale bars: 500 nm.
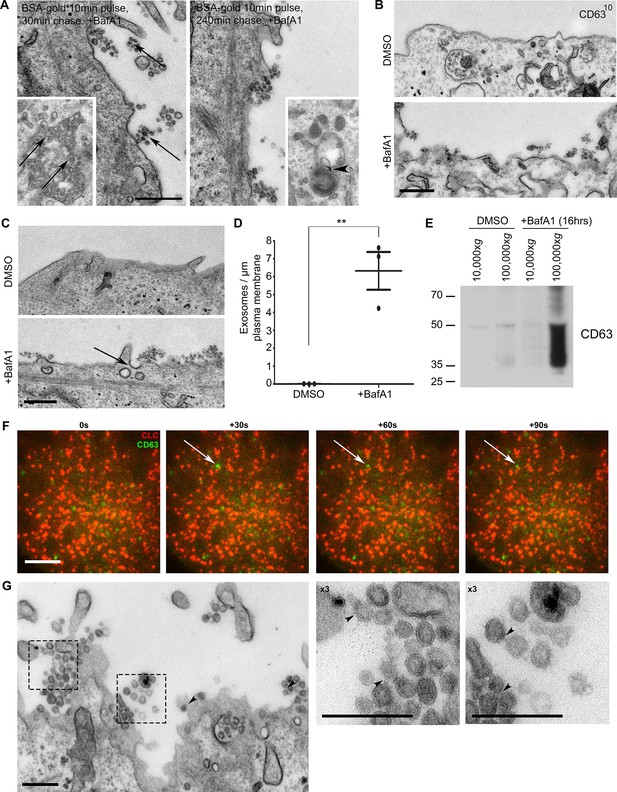
The extracellular vesicles that accumulate in BafA1-treated cells are exosomes.
(A) HeLa cells were incubated with BSA coupled to 5 nm gold for 10 min before being washed several times with PBS to remove any uninternalised label. The cells were then chased in full medium for either 30 min or 4 hr to load BSA-gold into endosomes or lysosomes respectively, then treated with BafA1 (100 nM, 16 hr), fixed, and prepared for conventional EM. Gold could be seen associated with extracellular vesicles from the cells chased for 30 min, but not from the cells chased for 4 hr. Insets: monomeric gold could be found within MVBs from cells chased for 30 min (arrows), and aggregated within lysosomes from cells chased for 4 hr (arrowhead). Scale bar: 200 nm. (B) DMSO- or BafA1-treated HeLa cells (100 nM, 16 hr) were surface-labelled with an antibody against the CD63 lumenal domain to identify exosomes. Scale bar: 500 nm. (C) HeLa cells were treated with DMSO or BafA1 (100 nM, 16 hr) before being fixed and prepared for conventional EM. Exosomes were often associated with clathrin-coated pits (arrow). Scale bar: 500 nm (D) The number of exosomes per μm of plasma membrane was quantified. Data shown are means from three independent experiments, ± S.E.M. ***p<0.001 (E) Culture supernatants were collected from mock-treated and BafA1-treated HeLa cells (100 nM, 16 hr), and centrifuged first at 10,000xg and then at 100,000xg. Western blots of the pellets were probed with anti-CD63. (F) Total internal reflection fluorescence microscopy (TIRF) was performed on HeLa cells transiently expressing both CLC-mCherry and CD63-GFP, following BafA1 treatment (100 nM, 16 hr). Representative stills are shown. Scale bar: 10 μm. (G) Conventional EM of BafA1-treated cells (100 nM, 16 hr), with arrowheads indicating proteinacious material on exosomes. Scale bars: 500 nm. See also Figure 2—figure supplement 1, 2.
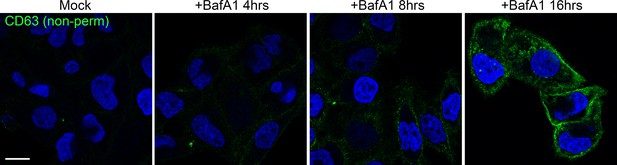
Prolonged treatment with BafA1 induces exosome release.
HeLa cells were treated with BafA1 (100 nM) for 0, 4, 8 or 16 hr before being fixed and prepared for immunofluorescence, without any permeabilisation. Cells were stained with a lumenal anti-CD63 antibody to reveal antigens exposed at the cell surface. Scale bar: 20 μm.
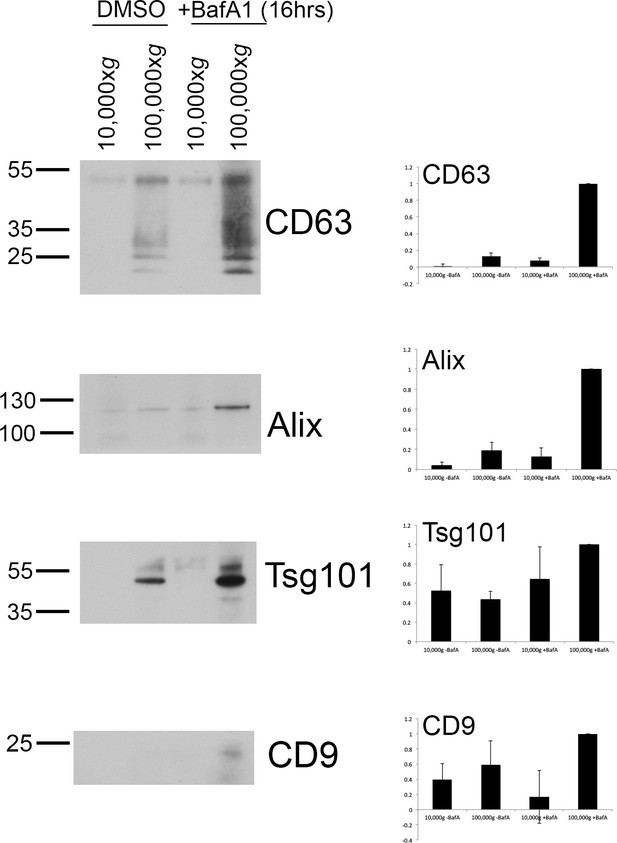
Exosome-enriched preparations probed with antibodies against various extracellular vesicle markers.
Culture supernatants were collected from mock-treated and BafA1-treated HeLa cells (100 nM, 16 hr), and centrifuged first at 10,000xg and then at 100,000xg. Western blots of the pellets were probed with antibodies against CD63, Alix, Tsg101, or CD9. Bands were quantified using ImageJ and normalized to the 100,000xg + BafA1 bands. Representative blots are shown on the left; on the right are the means from three independent experiments, with the error bars showing S.E.M.
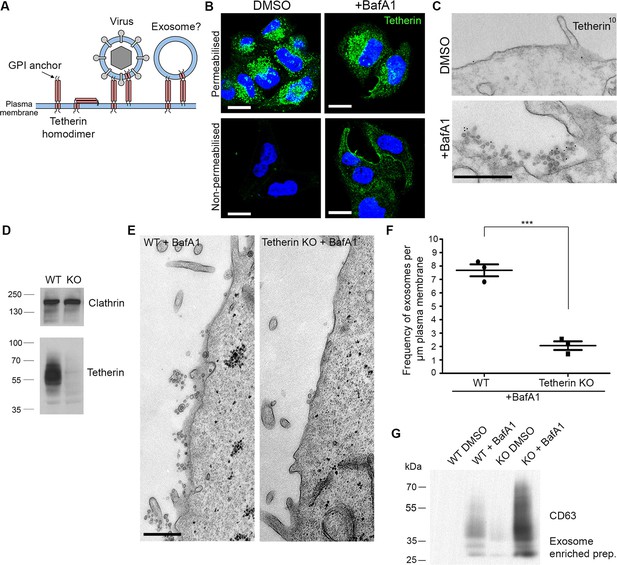
Tetherin localises to exosomes and facilitates exosome tethering.
(A) Schematic diagram of tetherin. (B) Mock-treated or BafA1-treated HeLa cells (100 nM, 16 hr) were either permeabilised or left intact and stained with an anti-tetherin antibody. Scale bars: 20 μm. (C) Mock-treated or BafA1-treated HeLa cells (100 nM, 16 hr) were surface-labelled using an anti-tetherin antibody followed by 10 nm protein A-gold. Scale bar: 500 nm. (D) The tetherin gene was knocked out using CRISPR/Cas9, and the loss of tetherin in a clonal population was confirmed by Western blotting. (E) Wild-type or tetherin-knockout HeLa cells were treated with BafA1 (100 nM, 16 hr) and processed for EM to analyse exosome frequency. Scale bar: 500 nm. (F) The frequency of exosomes per μm plasma membrane in BafA1-treated (100 nM, 16 hr) wild-type or tetherin-knockout cells was calculated. The mean ± S.E.M are shown from three independent experiments. ***p<0.001. (G) Exosome-enriched preparations were generated from the culture supernatants of both wild-type and tetherin-knockout cells, either with or without BafA1-treatment (100 nM, 16 hr), and Western blots were probed with anti-CD63. See also Figure 3—figure supplements 1, 2, 3.
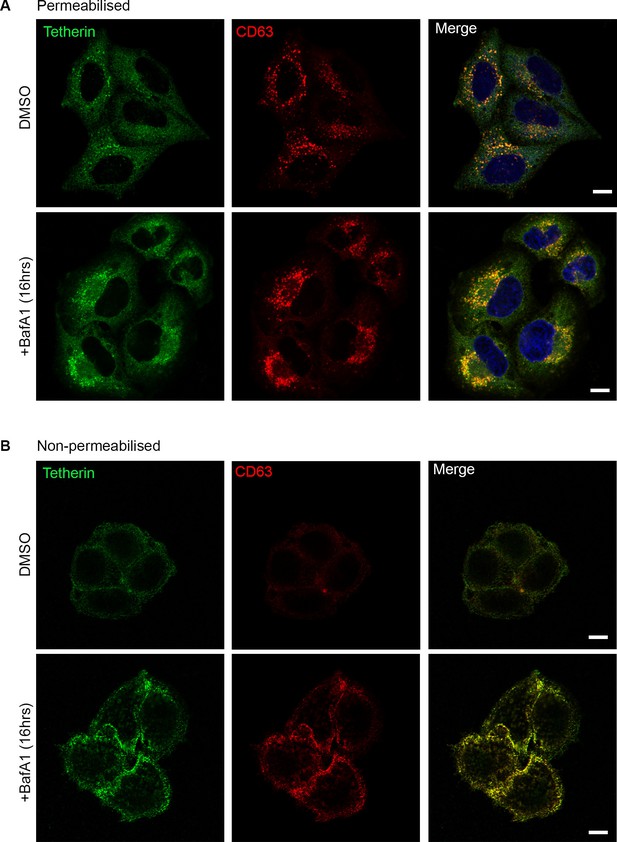
Tetherin and CD63 colocalise in mock-treated and BafA1-treated cells.
HeLa cells were treated with DMSO or BafA1 (100 nM, 16 hr), then fixed and labeled with antibodies against tetherin (rabbit polyclonal) and CD63 (mouse monoclonal), under permeabilised (A) or non-permeabilised (B) conditions. Scale bars: 10 μm.
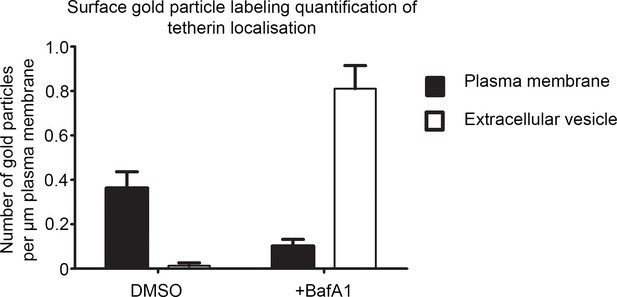
Quantification of surface labeling.
The density of surface gold labeling was calculated using an anti-tetherin antibody following 16 hr of DMSO or BafA1 (100 nM). Graphs show mean ± S.E.M for at least 100 μm of plasma membrane. Total lengths of plasma membrane measured were mock 102 μm for mock-treated cells, and 120 μm for BafA1-treated cells.
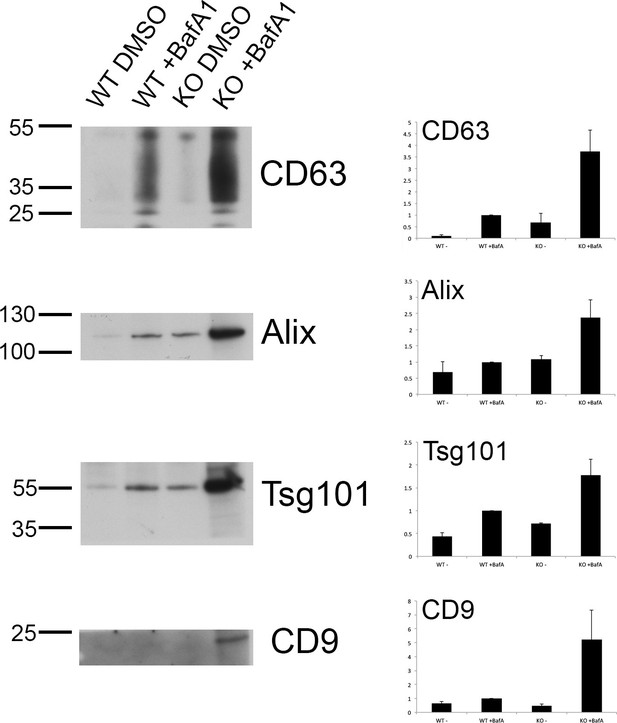
Exosome-enriched preparations probed with antibodies against other extracellular vesicle markers.
Exosome-enriched preparations were generated from the culture supernatants of both wild-type and tetherin-knockout cells, either with or without BafA1-treatment (100 nM, 16 hr), and Western blots were probed with antibodies against CD63, Alix, Tsg101, and CD9. Bands were quantified using ImageJ and normalized to the WT + BafA1 bands. Representative blots are shown on the left; on the right are the means from three independent experiments, with the error bars showing S.E.M.
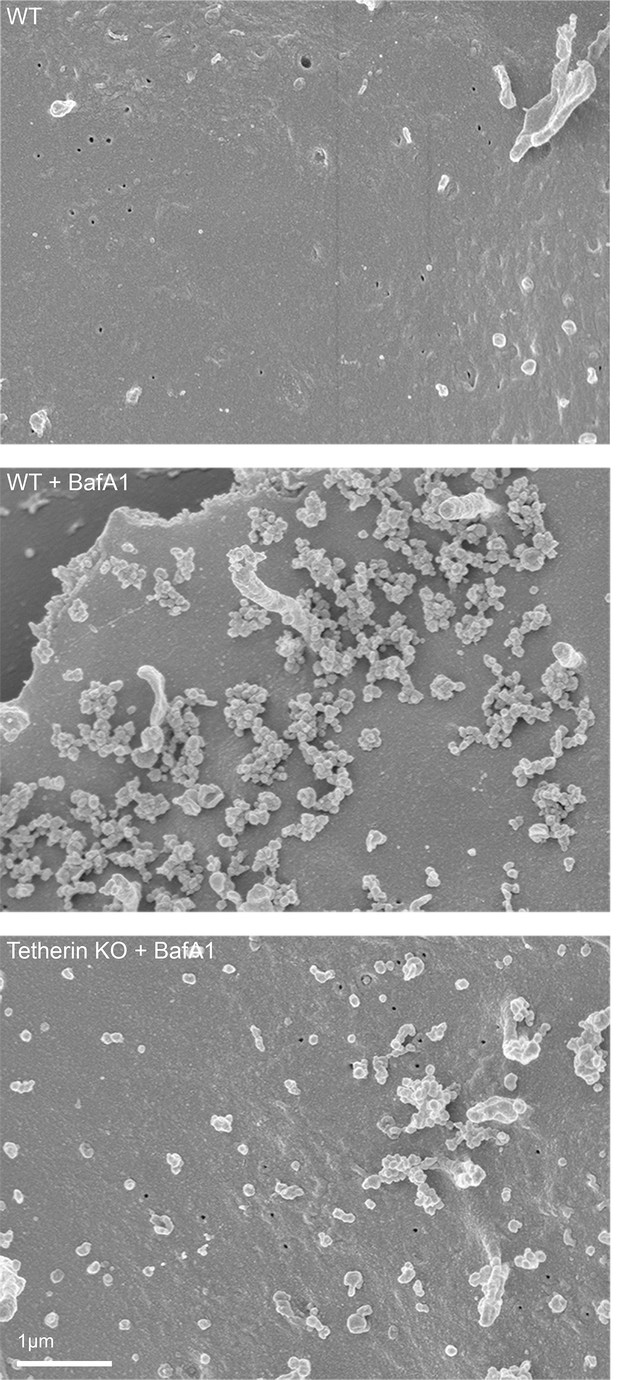
Scanning electron microscopy reveals clustering of exosomes on the cell surface.
Wild-type and tetherin-knockout cells were treated with or without BafA1 (100 nM, 16 hr) before being fixed and prepared for scanning electron microscopy. Scale bar: 1 μm.
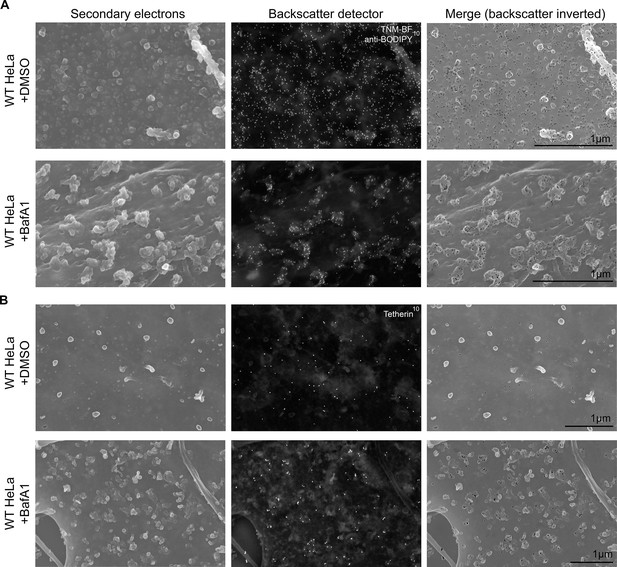
Immuno-SEM reveals the localization of cholesterol and tetherin in 3D.
HeLa cells treated with or without BafA1 (100 nM, 16 hr) were fixed, surface-labelled, and viewed by scanning electron microscopy. The cells in A were probed for cholesterol and the cells in B were probed for tetherin. Small ‘bumps’ observed on the plasma membrane probably represent surface-bound antibody complexes. Scale bars: 1 μm.
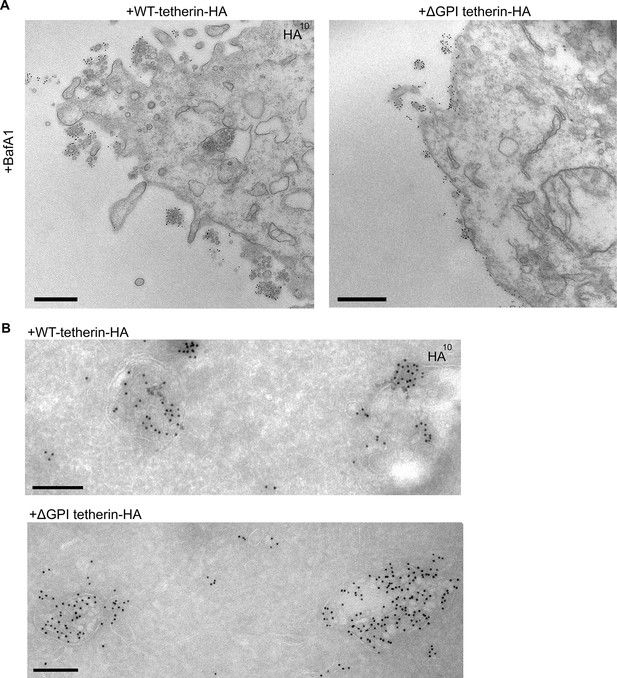
Exosome tethering can be rescued by WT-tetherin, but not by ΔGPI-tetherin.
(A) Tetherin-knockout cells were transiently transfected with WT-tetherin-HA or ΔGPI-tetherin-HA, treated with BafA1 (100 nM, 16 hr), and prepared for surface labelling EM. Cells expressing tetherin-HA constructs were identified by surface gold labelling using an anti-HA antibody. Scale bars: 500 nm. (B) Tetherin-knockout HeLa cells were transiently transfected with either WT-tetherin-HA or ΔGPI-tetherin-HA constructs. Cryosections of both were immunolabelled with anti-HA antibodies, revealing localisation to the ILVs of MVBs. Scale bars: 200 nm. See also Figure 6—figure supplement 1.
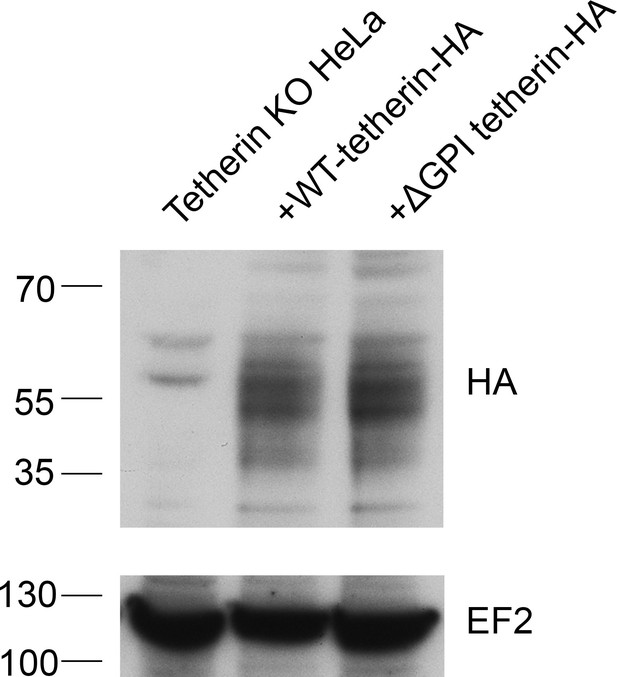
Western blotting of tetherin rescue cells.
Tetherin-knockout HeLa cells were transiently transfected with either WT-tetherin-HA or ΔGPI-tetherin-HA constructs. Lysed cells were analysed by Western blotting using an anti-HA antibody. EF2 is included as a loading control.
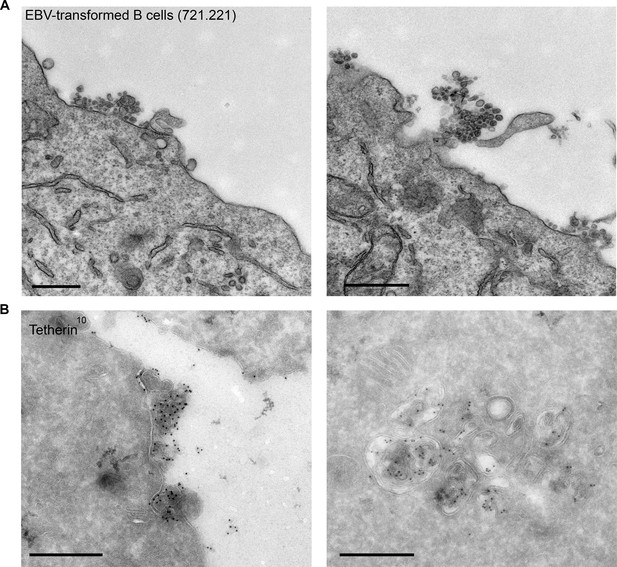
EBV-transformed B cells display clusters of tetherin-positive exosomes.
(A) 721.221 EBV-transformed B cells were prepared for conventional EM. Clusters of exosomes can be seen at the cell surface. Scale bars: 500 nm. (B) Immuno-EM reveals that the clustered exosomes and the ILVs of MVBs are positive for tetherin labeling. Scale bars: 500 nm.





