CD301b+ dendritic cells suppress T follicular helper cells and antibody responses to protein antigens
Figures
Depletion of CD301b+ DCs leads to enhanced antibody responses.
(a,b) WT and Mgl2-DTR mice were injected i.p. with 0.5 µg DT on days −1, +1 and +3. Mice were immunized with 50 µg papain in 20 µl phosphate-buffered saline (PBS) with or without (No OVA at 1°) 5 µg OVA in the footpad. On day 14, OVA (50 µg in 100 µl PBS) was injected retro-orbitally without adjuvant. Sera were harvested on days −1, +14 and +21. OVA-specific antibody titers were detected by ELISA. Bars indicate mean ± S.E.M. calculated from three (WT and Mgl2-DTR) or two (No OVA at 1°) individual mice. Representative data from three independent experiments are shown. (c,d) Mgl2-DTR mice were treated with DT and immunized with 5 µg OVA and 50 µg papain in the footpad as in a (DT Day −1/+2). Alternatively, WT or Mgl2-DTR mice were immunized with OVA and papain on day 0, then treated with DT on days five and eight (DT Day +5/+8). All mice received a boost immunization with OVA in PBS on day 14. Sera were collected in two independent experiments from total of 5–6 mice per group. (e,f) Mgl2-DTR mice were treated i.p. with DT or PBS and painted with whole cell lysates of house dust mite (HDM) for three times as shown in e. HDM-specific antibody titers were tested by ELISA against the whole cell HDM lysates (f). Data were collected from five animals in each group and are representative of two independent experiments. Bars indicate mean ± S.E.M. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001 by two-tailed Student’s t-test. All statistics indicate comparison to the WT or undepleted control shown in each panel.
Depletion of CD301b+ DCs enhances antibody responses in the non-skin dLNs.
(a,b) Mice were immunized with 10 µg OVA in 100 µl alum i.p. and received i.p. injection of 10 µg OVA without adjuvant on day 14. Sera were harvested on day 21 for OVA-specific antibody ELISA. Bars indicate mean ± S.E.M. calculated from three animals. (c–f) Indicated organs and peritoneal exudate cells were harvested from naïve WT mice and analyzed for surface CD301b expression along with indicated markers. 'Isotype' indicates binding of non-specific rat IgG2a conjugated with the same fluorochrome as the anti-CD301b mAb used.
Phenotype of CD301b+ DCs in LNs and peritoneal cavity.
DT-treated WT or Mgl2-DTR mice were immunized with OVA and papain in the right footpad. Right (dLN) or left (ndLN) popliteal LNs and peritoneal washes were harvested one day after immunization and stained for indicated markers. Non-T/B cells were gated as CD3ε- TCRβ- CD19- B220- cells. In some panels, CD301b+MHCII+ cells are shown as red dots and overlaid onto the total non-T/B cells. The red numbers indicate percentages of the cells in the gate among CD301b+MHCII+ cells, whereas the black numbers indicate percentages among non-T/B cells.
Depletion of CD301b+ DCs leads to enhanced antibody responses to the antigen immunized with weak or no adjuvant.
(a) Immunization and sample collection timeline. (b) Mgl2-DTR mice were treated with 0.5 µg DT or its inactive mutant CRM197 (CRM) on days -1 and +2 and immunized on day 0 with 50 µg papain plus indicated amount of OVA in the footpad. All mice received i.p. injection of 10 µg OVA without adjuvant on day 14 and sera were harvested on day 21 for OVA-specific antibody ELISA. (c) WT and Mgl2-DTR mice were treated with 0.5 µg DT on days −1 and +2 and immunized in the footpad with 5 µg OVA without any adjuvant on day 0 (top). Alternatively, lethally-irradiated WT mice were reconstituted with Mgl2-DTR (DTR only), WT (WT only) or 1:1-mixture of WT and Mgl2-DTR (WT+DTR) BM cells, then immunized with 5 µg OVA without adjuvant (bottom). All mice received i.p. injection of 10 µg OVA without adjuvant on day 14 and day 21, and sera were harvested on day 21 and day 28 for OVA-specific IgG1 ELISA. Bars indicate mean ± S.E.M. calculated from 4–11 individual mice. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 by two-tailed Student’s t-test. All statistics indicate comparison to the undepleted (CRM-treated, WT or WT only BM chimera) control shown in each panel.
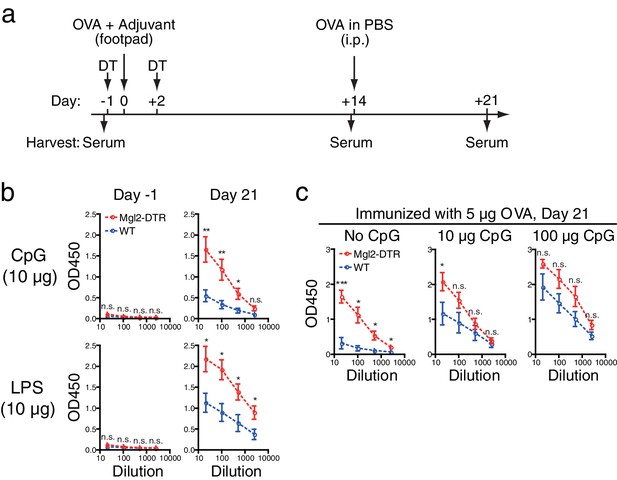
Depletion of CD301b+ DCs enhances antibody responses to type 1 immunization when adjuvant is limited.
WT and Mgl2-DTR mice were treated with 0.5 µg DT on days −1 and +2 and immunized in the footpad with 5 µg OVA plus indicated amount of CpG2216 or LPS as in a. All mice received i.p. injection of 10 µg OVA without adjuvant on day 14 and sera were harvested on day 21 for OVA-specific IgG2b ELISA. Sera were collected in two to four independent experiments. Bars indicate mean ± S.E.M. calculated from 6–7 (b) or 4–11 (c) individual mice. n.s., not significant, *p<0.05, **p<0.01 by two-tailed Student’s t-test. All statistics indicate comparison to the WT control shown in each panel.
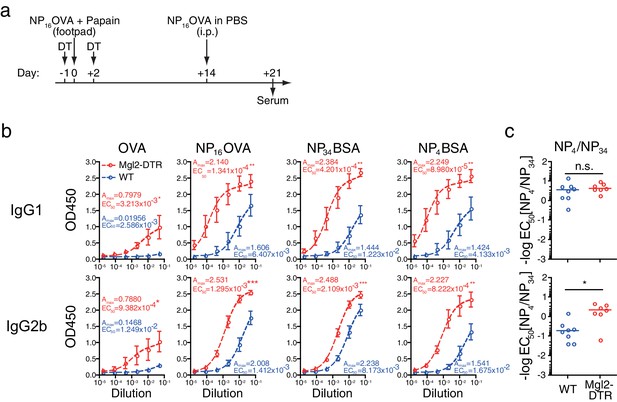
Depletion of CD301b+ DCs enhances the development of antigen-specific B cells with GC B cell phenotype without negatively affecting the affinity of antibodies.
As in a, mice were treated with DT and immunized with 5 µg NP16OVA plus 50 µg papain in the footpad. Two weeks later, all mice received i.p. injection of NP16OVA in PBS. (b) Serum antibodies against indicated antigens were measured by ELISA. n = 7–8 per group collected in two independent experiments. Amax and EC50 are shown in units of OD450 and serum dilution, respectively. (c) The ratio between EC50 for the binding to NP4 epitope and that for NP34 epitope was calculated in each mouse. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001 by Mann-Whitney test. Bars indicate means ± S.E.M. (b) or medians (c).
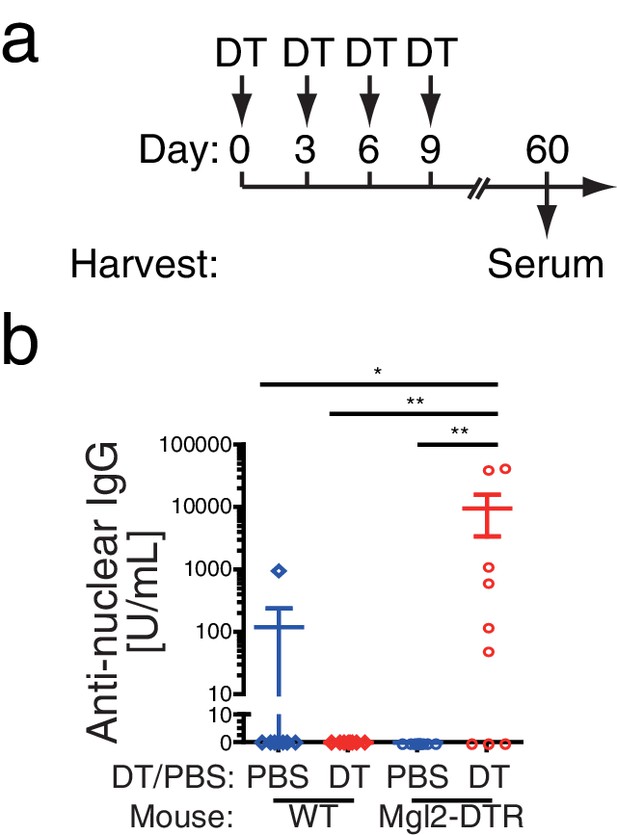
Depletion of CD301b+ DCs enhances autoreactive antibodies.
WT or Mgl2-DTR mice were treated i.p. with PBS or 0.5 µg DT every third day for 9 days as in a. Sera were harvested at day 60 and examined for anti-nuclear IgG antibodies by ELISA (b). Bars indicate mean ± S.E.M. *p<0.05, **p<0.01 by Mann-Whitney test.
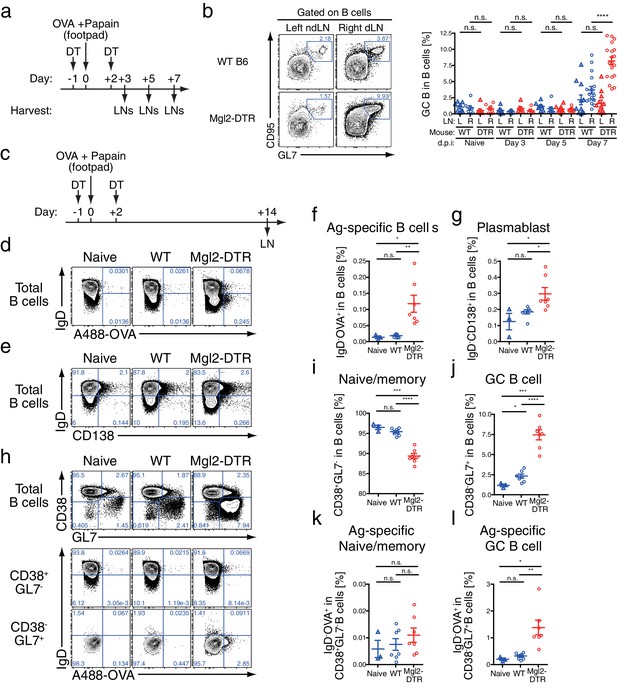
Depletion of CD301b+ DCs leads to GC B cell expansion in response to immunization with OVA and papain.
(a,b) WT and Mgl2-DTR (DTR) mice were treated with DT on days −1 and +2 and immunized with 50 µg papain and 5 µg OVA in 20 µl PBS in the right footpad. The left non-draining (L) or right draining (R) popliteal LNs were harvested on indicated days post immunization (d.p.i) and analyzed for the development of GC B cells (GL7+ CD95+). Mice that were treated with DT but left unimmunzed were analyzed at 7 d.p.i. (eight days from the first DT treatment) and are indicated as ‘Naïve’. Flow-cytometry panels in b are representative of data from day seven. (c) WT and Mgl2-DTR mice were treated with DT on days −1 and +2 and immunized on day 0 with 5 µg OVA with 50 µg papain as in Figure 2. On day 14, dLNs were harvested and cells were stained for the cell-surface markers as well as for their binding to Alexa488-conjugated OVA protein (A488-OVA). (d–g) Intracellular binding to A488-OVA (d,f) and CD138 expression (e,g) by activated IgD- B cells. (h–j) CD38 and GL7 expression in total B cells. (k,l) Cell-surface IgD expression and intracellular A488-OVA binding in naïve/memory (CD38+GL7-) B cells (k) and GC (CD38-GL7+) B cells (l) gated as in e. In all graphs, each dot indicates an individual mouse and the data were pooled from two independent experiments. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 by two-tailed Student’s t-test. Bars indicate means ± S.E.M.
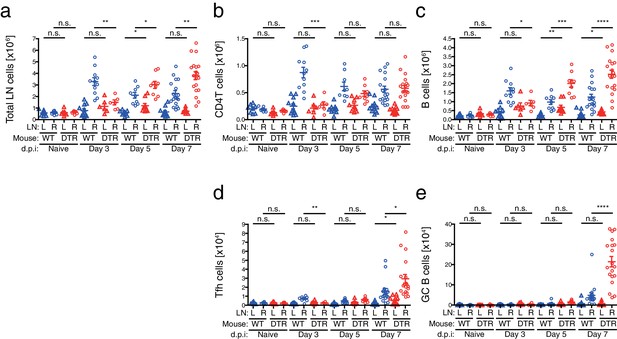
Cellular kinetics in CD301b+ DC-depleted mice immunized with papain and OVA.
Cell numbers of each indicated subset were calculated in mice shown in Figure 7b and Figure 8a and b.
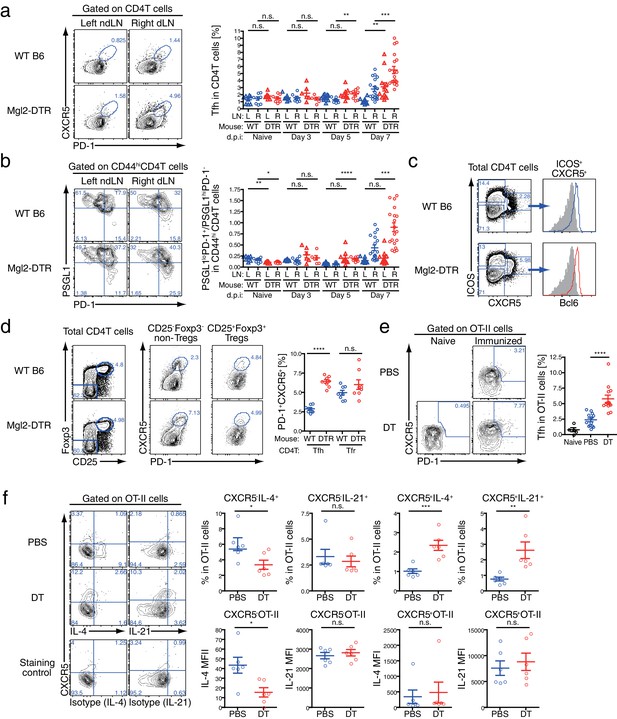
GC B cell expansion in CD301b+ DC-depleted mice is accompanied with Tfh expansion.
(a,b) Mice were immunized as in Figure 7a. Tfh (PD-1+CXCR5+) differentiation (a) and the ratio between follicular (PSGL1loPD-1+) and extrafollicular (PSGL1hiPD-1-) cells within CD44hi CD4+ T cells (b) were examined in the draining (R) or non-draining (L) popliteal LNs. Mice that were treated with DT but left unimmunzed were analyzed at 7 d.p.i (8 days from the first DT treatment) and are indicated as ‘Naïve’. Flow-cytometry panels are representative of data from day seven p.i. (c,d) Mice were immunized as in a. Expression of Bcl6 in ICOS+CXCR5+CD4+ T cells (c) and percentages of PD-1+CXCR5+ cells among Tregs (CD25+Foxp3+CD4+T cells) and non-Tregs (CD25+Foxp3+CD4+T cells) (d) in the dLN at 7 d.p.i are shown. Gray histograms in e indicate binding of non-specific isotype control mAb binding to ICOS+CXCR5+CD4+ T cells pooled from WT and Mgl2-DTR mice. (e,f) Mgl2-DTR mice transferred with CD45.1+ OT-II transgenic CD4+ T cells were treated with DT or PBS, then immunized as in a. On day seven, the draining popliteal LNs were harvested and stained for Tfh markers and cytokines. The donor OT-II cells were identified as CD45.1+ CD4+ cells. Data were pooled from 2–3 individual experiments per group. Each dot indicates an individual mouse. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 by two-tailed Student’s t-test. Bars indicate means ± S.E.M. MFI, mean fluorescent intensity.
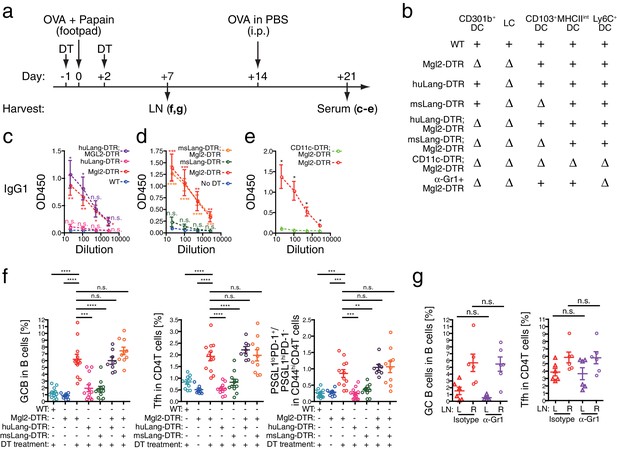
Migratory and inflammatory DC subsets are not required for the enhanced antibody, Tfh and GC B cell development induced by the CD301b+ DC depletion.
(a–d) Mice were treated with 0.5 µg DT and immunized with 50 µg papain and 5 µg OVA in the footpad. At the time of immunization, different DC subsets were either intact (+) or depleted (∆) depending on the host genotype as indicated in b. All mice received an i.p. injection of 10 µg OVA in PBS without adjuvant on day 14. Serum OVA-specific IgG1 on day 21 was detected by ELISA. Bars indicate mean ± S.E.M. calculated from 3–6 individual mice in each group. Representative data from 2–3 independent experiments are shown. (e) Mgl2-DTR and CD11c-DTR;Mgl2-DTR mice were immunized and boosted as in a, except that the mice were treated with a single dose of 125 ng DT on day −1. Sera were harvested on day 21 from three independent experiments and OVA-specific IgG1 was detected by ELISA. Bars indicate mean ± S.E.M. calculated from five (Mgl2-DTR) and three (Mgl2-DTR;CD11c-DTR) individual mice. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 by two-tailed Student’s t-test. All statistics indicate comparison to the undepleted (WT or no DT) control shown in each panel except in b and are shown by color-matched asterisks where applicable. (f) WT mice and mice that carry Mgl2-DTR, huLang-DTR and/or msLang-DTR in their genetic construct were treated with DT and immunized as in a, then LNs were harvested on day seven. Tfh and GC B cell percentages in dLNs. Data were pooled from 2–3 independent experiments per group. (g) Mgl2-DTR mice were treated with DT and immunized as in a. Mice received i.p. injections of 50 µg anti-Gr-1 (α-Gr1) or Rat IgG2a isotype control antibodies every other day starting on day two. Left non-draining (L) or right draining (R) popliteal LNs were harvested on day seven and analyzed for GC B and Tfh cells. Data were pooled from two individual experiments per group. Each dot indicates an individual mouse. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 by two-tailed Student’s t-test. Bars indicate means ± S.E.M.
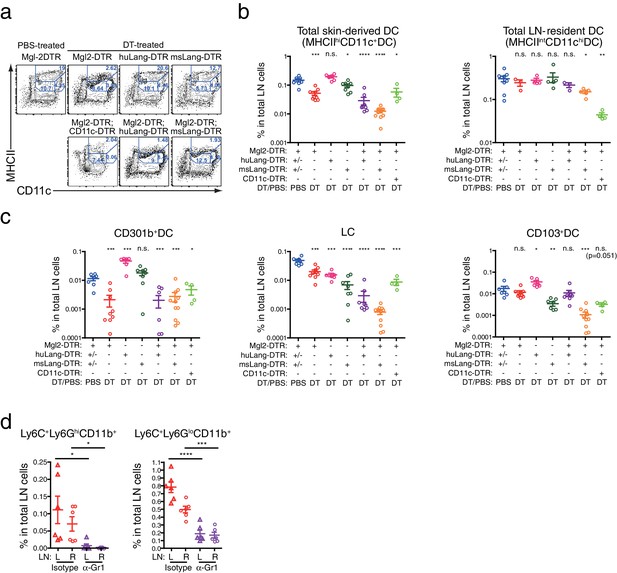
Depletion of various skin-dLN DC subsets in different DTR mice.
Mice that carry Mgl2-DTR, huLangerin-DTR (huLang-DTR), msLangerin-DTR (msLang-DTR) and/or CD11c-DTR in their genetic construct were treated i.p. with 0.5 µg DT or PBS and DC subsets in the skin-dLNs were analyzed on the next day. (a, b) DC subsets defined by MHCII and CD11c expression in the residual CD3-B220- cells in the skin-dLN. (c) Different skin-derived migratory DC subsets within MHCIIhiCD11c+DCs. All statistics indicate comparison to the PBS-treated controls pooled from various DTR strains. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 by two-tailed Student’s t-test. Representative data from two or more independent depletion experiments are shown.
Blockade of PD-L1 at the time of immunization enhances Tfh and GC B cell responses in CD301b+ DC-dependent manner.
(a) Pooled skin-dLN cells from naïve WT mice were stained and gated as indicated. Representative flow cytometry plots from three independent experiments are shown. (b–f) WT and Mgl2-DTR mice were injected i.p. with 250 µg anti-PD-L1 mAb (αPD-L1) or αPD-L2 mAb along with 0.5 µg DT on days −1 and +2, then immunized with 50 µg papain and 5 µg OVA in the footpad on day 0. (c,d) Right draining popliteal LNs were collected on day seven and analyzed for GC B cells (c) and Tfh cells (d). Data were pooled from two to three independent experiments. Each dot indicates an individual mouse. (e,f) Alternatively, mice received additional i.p. injection of OVA without adjuvant on day 14, and OVA-specific serum antibody titers were examined on day 21 by ELISA. n.s., not significant, *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 by two-tailed Student’s t-test. Bars indicate means ± S.E.M.
Depletion of CD301b+ DCs does not reduce PD-1 ligand expression on the residual migratory DCs.
(a) WT or Mgl2-DTR mice were treated with DT, then skin-dLNs were harvested 24 hr later for flow cytometry. (b) Mice were treated with DT and anti-PD-L1 or PD-L2 blocking mAb, and immunized with OVA and papain as in Figure 10b. LNs were collected for analysis on day three. Data were pooled from two independent experiments. Each dot indicates an individual mouse. n.s., not significant, *p<0.05 by two-tailed Student’s t-test. Bars indicate means ± S.E.M.
Differential effects of PD-1 ligand blockade on CD4T and B cell compartments in CD301b+ DC-intact and depleted animals.
(a–c) Tfh and GC B cell development was assessed in the dLN (a,b) or in the contralateral ndLN (c) in mice shown in Figure 10b–d. (d) WT mice were treated with indicated antibodies and immunized as in Figure 10e, and OVA-specific serum antibodies were measured by ELISA.
Model for CD301b+ DC-mediated suppression of humoral immunity.
(a) Summary of the results shown in this study. (b) Model for Tfh and GCB cell suppression by CD301b+ DCs. CD301b+ DCs express high levels of PD-L1 to inhibit the expansion of Tfh. The impaired Tfh expansion in turn leads to suboptimal GC B cell activation and reduced antibody responses. In the absence of CD301b+ DCs, CD301b-LN-resident DCs present soluble antigens to CD4+ T cells to induce Tfh differentiation. This CD301b-LN-resident DC subset is susceptible to depletion in CD11c-DTR mice.
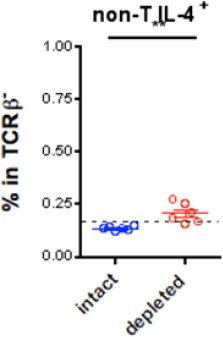
IL-4 production from non-T cells in CD301b+ DC-depleted mice.
Dashed line indicates staining background.
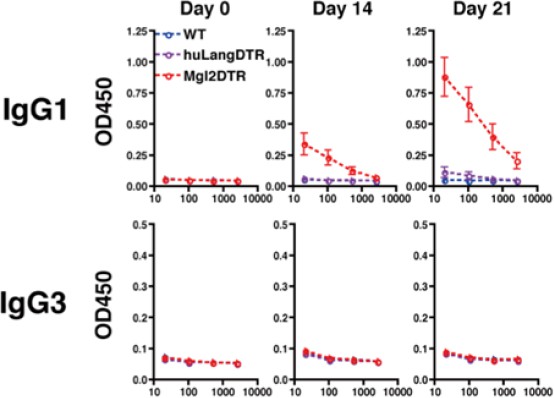
IgG3 production in mice immunized with OVA plus papain in the footpad.
All mice received intraperitoneal injection of OVA without papain on day 14.





