Spatiotemporal regulation of autophagy during Caenorhabditis elegans aging
Figures
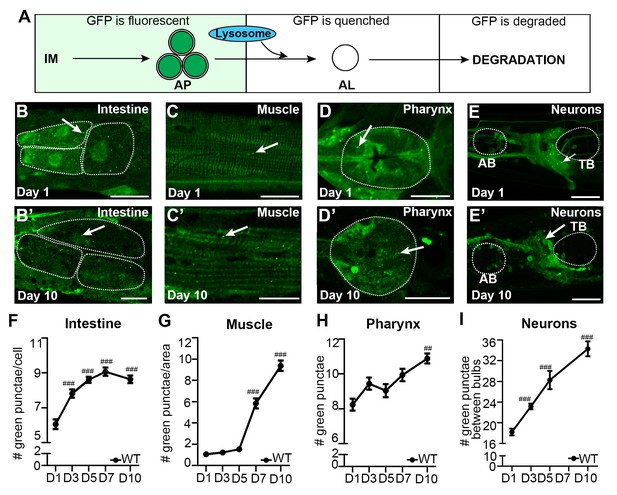
The autophagosome pool size increases with age in C. elegans.
(A) Schematic representation of GFP::LGG-1 fluorescence states in the autophagy pathway. IM, isolation membrane; AP, autophagosome; AL, autolysosome. (B–E’) Adult transgenic WT animals expressing gfp::lgg-1, imaged at Day 1 (B–E) and Day 10 (B’–E’) of adulthood. APs (arrows) can be seen in the intestine (B,B’), body-wall muscle (C,C’), pharynx (D,D’), and nerve-ring neurons (E,E’). Dotted lines outline individual intestinal cells (B,B’) and pharyngeal bulbs (D–E’). AB, anterior pharyngeal bulb; TB, terminal pharyngeal bulb. Scale bars = 20 µm. (F–I) Quantification of autophagosomes (AP; GFP punctae) in the intestine (F), body-wall muscle (G), pharynx (H), and nerve-ring neurons (I) at Days 1, 3, 5, 7, or 10 of adulthood in WT animals. Day 7 was omitted for neurons due to a counting issue at this time point. Data are the mean ± SEM of ≥20 animals combined from three independent experiments per time point. ###p<0.00 and ##p<0.001 for WT control at Day 1, 3, 5, 7, or 10 vs. WT control at Day 1 by Poisson regression.
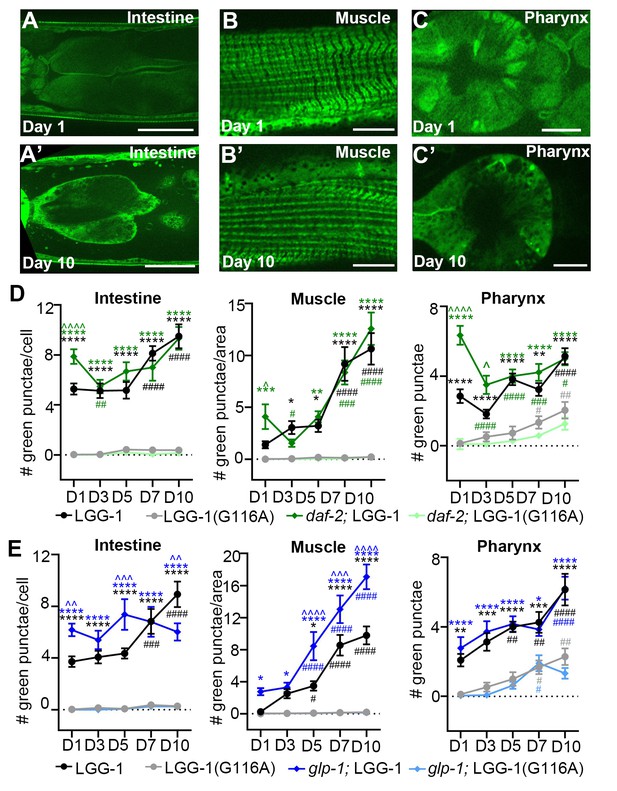
Age-related increase in autophagosome pool size requires lipidation of LGG-1.
(A–C) Adult transgenic wild-type (WT) animals expressing gfp::lgg-1(G116A) imaged at Day 1 (A–C) and 10 (A’–C’) of adulthood in the intestine (A), body-wall muscle (B) and pharynx (C). Scale bars = 150 µm (A), 20 µm (B) and 50 μM (C). (D–E) Quantification of GFP-positive punctae in the intestine, body-wall muscle, and pharynx of WT or daf-2(e1370) animals expressing either gfp::lgg-1 (LGG-1) or gfp::lgg-1(G116A) (LGG-1(G116A)) and raised at 20°C (D), or WT or glp-1(e2141) animals expressing either gfp::lgg-1 or gfp::lgg-1(G116A) (E) and raised at 25°C until Day 1 of adulthood and then grown at 20°C for remainder of life. Data are the mean ± SEM of combined from two independent experiments per time point with ≥20 animals total. Two additional repeats of WT animals showed similar results (data not shown). ∧, WT + control vs. glp-1/daf-2 control at Days 1, 3, 5, 7, and 10; *, glp-1/daf-2 control vs. WT/glp-1/daf-2 + BafA at Days 1, 3, 5, 7, and 10, #, WT/glp-1/daf-2 control at Days 3, 5, 7, and 10 vs. WT/glp-1/daf-2 control at Day 1. ***/∧∧∧/###p<0.0001, **/∧∧/##p<0.001, */∧/#p<0.01 by two-way ANOVA. We note that the pharyngeal counts in WT animals were lower in these experiments compared to others (Figures 3–5 and Figure 4—figure supplement 1); this may be due to the use of different microscopes, strains, and/or experimenters. We note that we also assessed the number of GFP-positive punctae between and near the pharyngeal bulbs to possibly evaluate neurons; however, while we observed very few punctae in this area in animals expressing GFP::LGG-1(G116A) (data not shown), none of the LGG-1 reporters expressed from the endogenous promoter allowed for accurate cell identification, and neurons therefore remain to be fully evaluated.
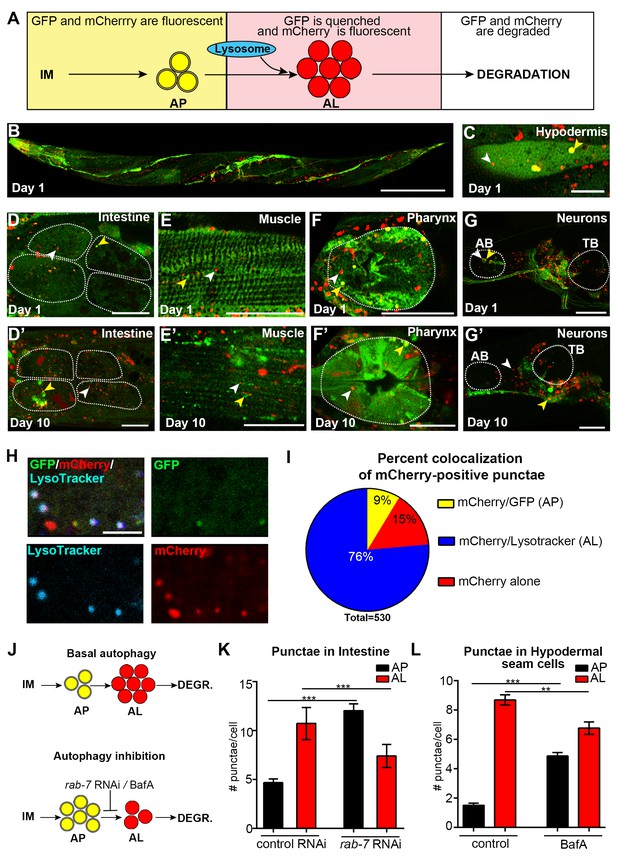
Expression and validation of a novel mCherry::gfp::lgg-1 reporter in C. elegans.
(A) Schematic representation of mCherry::GFP::LGG-1 fluorescence states in the autophagy pathway. IM, isolation membrane; AP, autophagosome; AL, autolysosome. (B) Whole-body expression of mCherry::GFP::LGG-1 in a wild-type (WT) animal at Day 1 of adulthood. Scale bar = 100 µm. Note that the intensity of red fluorescence compared to green is stronger; thus, the red channel was purposely set lower (see Materials and methods). (C–G’) Adult transgenic WT animals expressing mCherry::gfp::lgg-1, imaged at Day 1 (C–G) or Day 10 (D’–G’) of adulthood. APs (mCherry/GFP; yellow arrowheads) and ALs (mCherry only; white arrowheads) can be seen in the hypodermal seam cells (C), intestine (D,D’), body-wall muscle (E,E’), pharynx (F,F’), and nerve-ring neurons (G,G’). Dotted lines outline individual intestinal cells (D,D’) and pharyngeal bulbs (F,G’). AB, anterior pharyngeal bulb; TB, terminal pharyngeal bulb. Scale bars = 20 µm (D–G’) and (C) = 10 μm. (H) Intestine of Day 1 WT transgenic animals expressing mCherry::gfp::lgg-1 (green, GFP; red, mCherry) and stained with LysoTracker (light blue). Scale bar = 5 µm. (I) Quantification of punctae containing mCherry alone or co-localized with GFP or LysoTracker in the intestine of WT transgenic animals. Data are representative of three independent experiments, each with ≥10 animals. (J) Schematic representation of basal AP and AL pool sizes and the effect of inhibiting autophagy by rab-7 RNAi or Bafilomycin (BafA) treatment. (K) Quantification of APs and ALs in the intestine of Day 1 WT transgenic animals fed from hatching with bacteria expressing empty vector (control) or dsRNA encoding rab-7. Data are the mean ± SEM of ≥40 animals combined from three experiments. ***p<0.0001 by Student’s t-test. (L) Quantification of APs and ALs in the hypodermal seam cells of Day 1 WT transgenic animals injected with BafA or DMSO (control). Data are the mean ± SEM of ≥30 animals combined from three experiments. ***p<0.0001 and **p<0.001 by Student’s t-test.
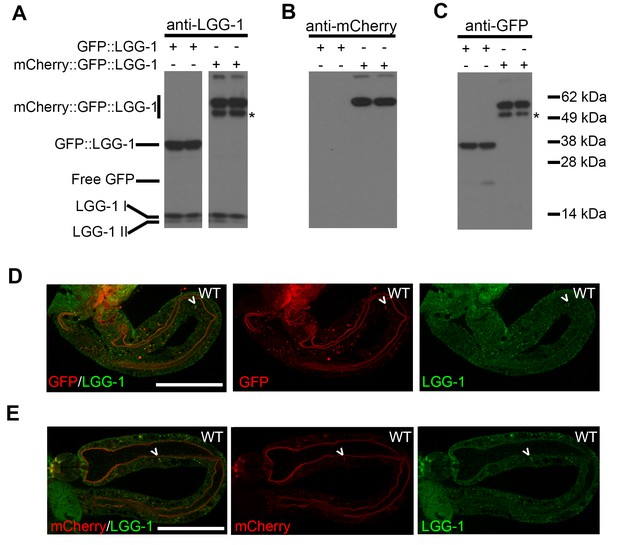
mCherry::GFP::LGG-1 reporter produces a full-length protein.
(A–C) Immunoblot analysis of lysates from Day 1 transgenic animals expressing mCherry::gfp::lgg-1 or gfp::lgg-1. Blots were probed with anti-LGG-1 (A), anti-mCherry (B), and anti-GFP (C) antibodies. Data are representative of at least two experiments. The lower band of the mCherry::GFP::LGG-1 reporter (*) may be due to cleavage of the N-terminus as it has previously been published that the first 11 amino acids of mCherry are susceptible to cleavage resulting in something that is slightly smaller than the full length protein (Huang et al., 2014). (D–E) Immunofluorescence to detect endogenous LGG-1 (green) and GFP (D; red) or mCherry (E; red) in dissected intestines of wild-type (WT) animals. Data are representative of at least two independent experiments, each with ≥5 animals. Scale bars = 20 µm. Full-length protein was detected at Day 10 (data not shown).
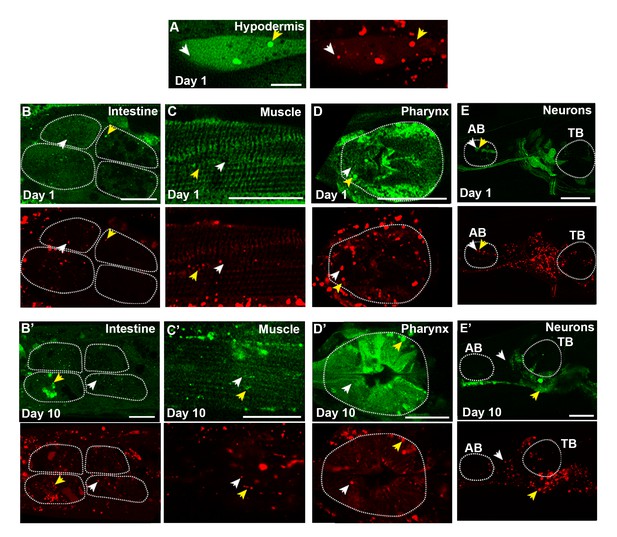
mCherry::GFP::LGG-1 reporter is expressed in multiple tissues.
(A–E’) GFP (green) and mCherry (red) fluorescence images from Day 1 (A–E) and Day 10 (B’–E’) adult wild-type (WT) transgenic animals expressing the mCherry::gfp::lgg-1 reporter. The merged images are shown in Figure 2. Tissues tested were hypodermal seam cells (A), intestine (B,B’), body-wall muscle (C,C’), pharynx (D,D’), and nerve-ring neurons (E,E’). Autophagosomes (AP, mCherry/GFP) are indicated by yellow arrowheads, and autolysosomes (AL, mCherry only) are indicated by white arrowheads. (F–I’) Confocal images of animals with tissue-specific expression of gfp::lgg-1 in the intestine (F,F’), body-wall muscle (G,G’), pharynx (H,H’), and nerve-ring neurons (I,I’) at Day 1 (F–I) and Day 10 (F’–I’) of adulthood. Arrows indicate APs (GFP punctae). Dotted lines outline individual intestinal cells (B,B’,F,F’) and pharyngeal bulbs (D,D’,E,E’,H,H’,I,I’). AB, anterior pharyngeal bulb; TB, terminal pharyngeal bulb. Scale bars = 20 µm except in (A) = 10 µm.
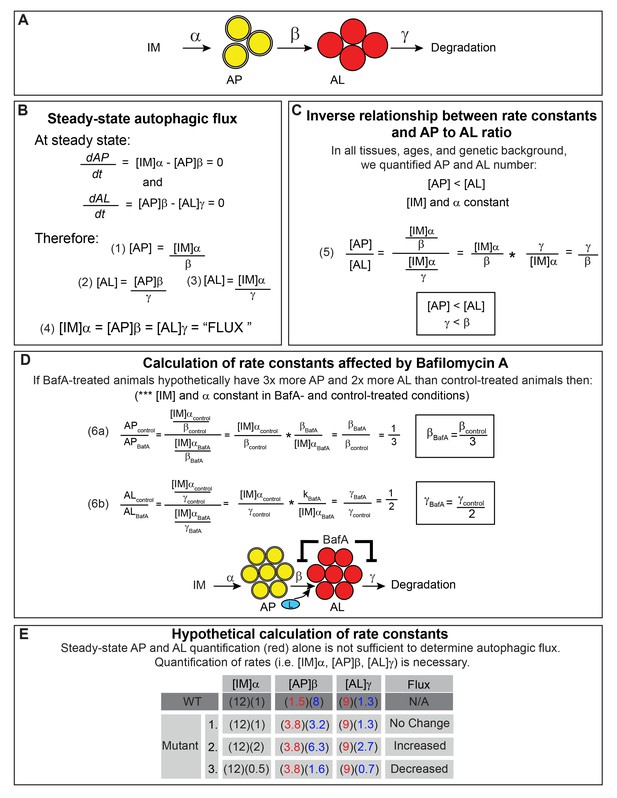
Calculations showing that quantification of autophagic vesicles at steady state provide insufficient information to determine autophagic flux.
(A) Schematic of the autophagy steps with rate constants α, β, and γ. IM, isolation membrane; AP, autophagosome; AL, autolysosome; L, lysosome. (B) Definition of autophagic flux at steady state. The rate of AP formation (dAP/dt) and AL formation (dAL/dt) at steady state is equal to 0 allowing for derivation of equations (1) – (3) for AP pool size ([AP]) and AL pool size ([AL]). The overall autophagic flux (‘FLUX’) at steady state is equivalent to the rates of the individual autophagy steps (equation (4)). (C) Derivation of the relationship between rate constants β and γ and the ratio of AP to AL based on the observation made in this study that [AP] < [AL] in C. elegans (Figure 3). We used the steady-state equations derived in (B). Since AP and AL pool sizes were quantified using the same tissues from animals of the same genotype and age, [IM] and α are equivalent for equations (1) and (3). Thus, the ratio [AP]:[AL] is inversely related to the ratio of the rate constants γ:β, suggesting that γ < β (equation (5)). (D) Relative β and γ rates following Bafilomycin A (BafA) treatment (βBafA and γBafA) compared with control treatment (βcontrol and γcontrol) can be calculated using equations (1) – (3) derived from steady-state conditions in (B). [IM] and α are assumed to be constant in control and BafA-treated animals since AP and AL pool sizes were quantified using animals of the same genotype and age. βBafA and γBafA are inversely related to the change in AP or AL number following BafA treatment (equation 6). Thus, a hypothetical threefold increase in APs and twofold increase in ALs suggests a corresponding threefold decrease in βBafA and twofold decrease in γBafA, demonstrating that BafA inhibits both the AP to AL step and the AL to degradation step, as indicated in the diagram. (E) Hypothetical calculation of rate constants. In WT animals, we arbitrarily set the rate of autophagic flux (i.e. [IM]α) to 12). In daf-2 mutants, we envisioned three scenarios: (1) no change in flux, where [IM]α = 12, similar to WT, (2) a hypothetical twofold increase in flux where [IM]α = 24, and (3) a twofold decrease in flux where [IM]α = 6. We then used our observed AP and AL data from WT animals and daf-2 mutants expressing the mCherry::GFP::LGG-1 reporter (red numbers) to show that different rate constants β and γ (blue numbers) can be calculated for each scenario using the flux equation in (B). Thus, it is necessary to quantify the individual rates (i.e. [IM]α, [AP]β, or [AL]γ) or the rate of overall autophagic degradation to determine autophagic flux. The same conclusion can be reached by solving the equations in (B).
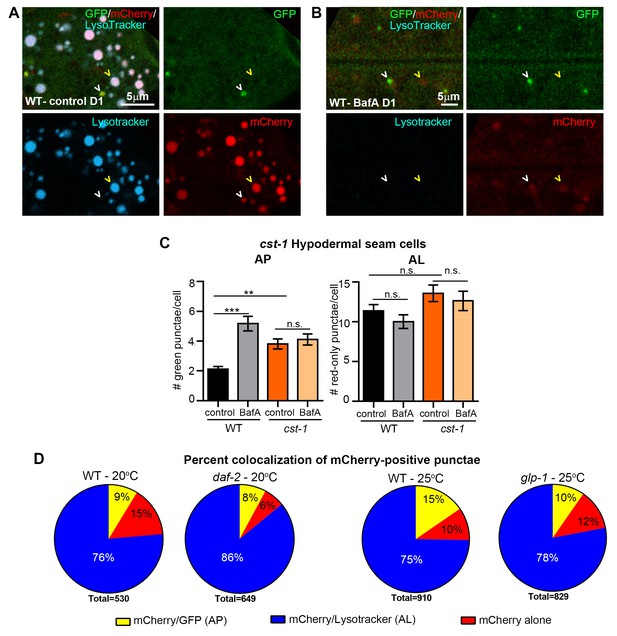
Additional validation of new mCherry::GFP::LGG-1 reporter.
(A,B) Fluorescence microscopy of wild-type (WT) animals expressing mCherry::gfp::lgg-1 treated with LysoTracker and injected with DMSO (control) (A) or Bafilomycin A (BafA) (B) at Day 1 (D1) of adulthood. White arrowhead, mCherry/LysoTracker punctae; yellow arrowhead, mCherry/GFP punctae. (C) Quantification of autophagosomes (AP, mCherry/GFP punctae) and autolysosomes (AL, mCherry-only punctae) in hypodermal seam cells of transgenic WT or cst-1(tm1900) animals expressing mCherry::gfp::lgg-1 and injected with DMSO (control) or BafA. Data are the mean ± SEM of ≥35 animals combined from two experiments. ***p<0.0001 and **p<0.001 by one-way ANOVA. (D) Quantification of punctae containing mCherry alone or co-localized with GFP or LysoTracker in the intestine of Day 1 WT (data from Figure 2) and daf-2(e1730) transgenic animals raised at 20°C or Day 1 WT and glp-1(e2141) transgenic animals raised at 25°C. Data are representative of three independent experiments, each with ≥10 animals.
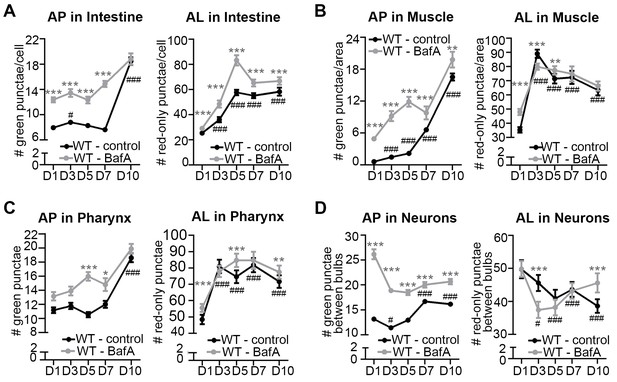
The pool size of autophagic vesicles increases with age in all tissues of wild-type animals.
(A–D) Quantification of autophagosomes (AP) and autolysosomes (AL) in adult Days 1, 3, 5, 7, and 10 wild-type (WT) transgenic animals expressing mCherry::gfp::lgg-1 and injected with DMSO (control, black lines) or Bafilomycin A (BafA, gray lines). Tissues examined were the intestine (A), body-wall muscle (B), pharynx (C), and nerve-ring neurons (D). WT animals were raised at 25°C and incubated at 20°C from Day 1 of adulthood. Data are the mean ± SEM of ≥30 animals combined from three independent experiments per time point. ***p<0.0001, **p<0.001, and *p<0.01 for WT control vs WT + BafA on each day; ###p<0.0001 for WT control at Days 1, 3, 5, 7, or 10 vs. WT control at Day 1 by Poisson regression.
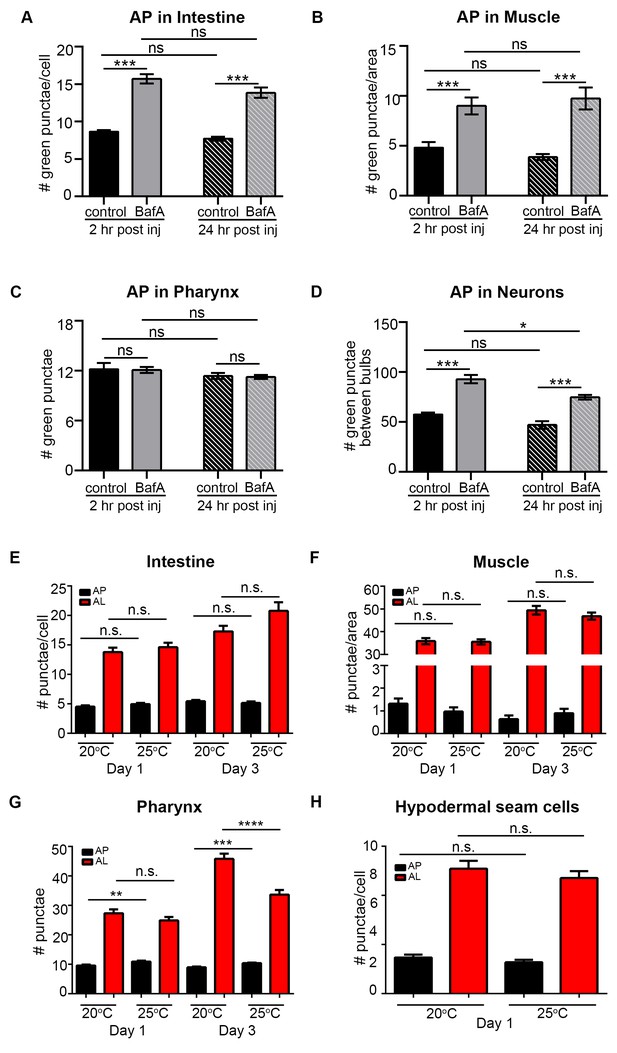
Quantification of autophagosomes at different time points after Bafilomycin A treatment and at different temperatures.
(A–D) Quantification of autophagosomes (AP) in the intestine (A), body-wall muscle (B), pharynx (C), and nerve-ring neurons (D) of Day 1 wild-type (WT) animals expressing gfp::lgg-1 and analyzed at two hours or 24 hours after injection of DMSO (control) or Bafilomycin A (BafA). Data are the mean ± SEM of 15 animals combined from two experiments ***p<0.0001, **p<0.001, *p<0.01 by ANOVA. We note that the BafA treatments in the pharynx were negative in this data set; while other 2 hours incubations caused changes in pharyngeal tissues of WT animals at this and other time points, the response to BafA may be more variable in the pharynx. (E–H) Eggs of WT transgenic animals expressing mCherry::gfp::lgg-1 were allowed to hatch at 20°C overnight and left at 20°C or incubated at 25°C until Day 1 of adulthood. Animals were then moved to 20°C for the rest of their lifespans. Animals were imaged at Day 1 or Day 3 of adulthood. APs (mCherry/GFP punctae) and ALs (autolysosomes; mCherry-only punctae) were quantified in the intestine (E), body-wall muscle (F), pharynx (G), and hypodermal seam cells (H). Data are the mean ± SEM of 15 animals combined from two experiments ***p<0.0001, **p<0.001, *p<0.01 by one-way ANOVA.
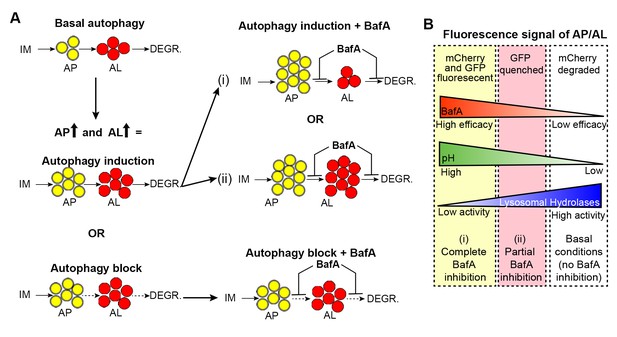
Hypothetical outcomes of autophagosome and autolysosome pool sizes following Bafilomycin A treatment.
(A) Schematic representation of the possible outcomes of Bafilomycin A (BafA) treatment on autophagosomes (AP) and autolysosome (AL) pool sizes. An increase in autophagic vesicles could represent an induction or block in autophagic activity. If a change in either the AP or AL pool size is observed following BafA treatment, this is consistent with active autophagy, whereas no change in pool sizes following BafA indicates a block in autophagy. Possible changes in autophagic vesicles are indicated following (i) complete or (ii) partial inhibition of lysosomal acidification. (B) Diagram illustrating the relationship between changes in fluorescence signal of autophagic vesicles, corresponding pH, and lysosomal hydrolase activity, in response to efficacy of BafA (i and ii).
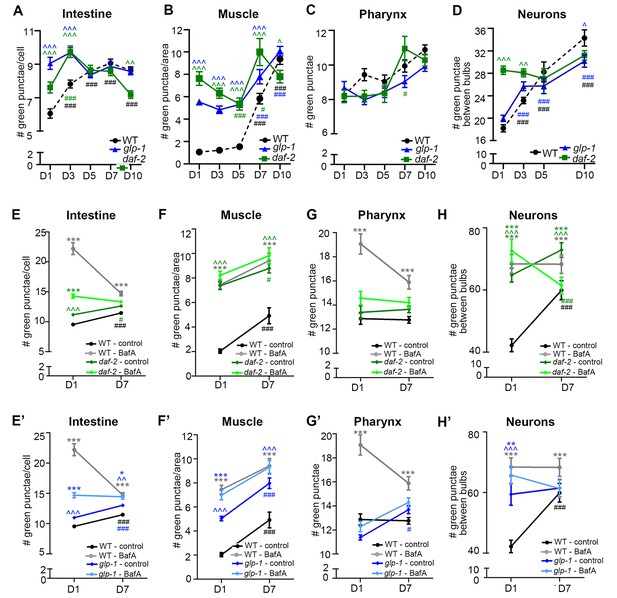
Quantification of GFP::LGG-1 punctae during aging of animals expressing gfp::lgg-1.
(A–D) Quantification of autophagosomes (AP; GFP punctae) in the intestine (A), body-wall muscle (B), pharynx (C), and nerve-ring neurons (D) at Days 1, 3, 5, 7, or 10 of adulthood in wild type (WT; dotted black same as Figure 1F–I), daf-2(e1370) (green), and glp-1(e2141) (blue). Data are the mean ± SEM of ≥20 animals combined from three independent experiments per time point. ***/###p < 0.0001, **/##p < 0.001, and */#p < 0.01. *** for WT control vs glp-1/daf-2 on each day; ### for WT/glp-1/daf-2 control at Days 1, 3, 5, 7, or 10 vs. WT/glp-1/daf-2 control at Day 1 by Poisson regression. (E–H’) Quantification of APs in WT (black line) and daf-2(e1370) animals (E–H, green) and WT and glp-1(e2141) animals (E’–H’, blue) injected with DMSO (control, black/dark green/dark blue) or Bafilomycin A (BafA, gray/light green/light blue) at Day 1 or Day 7 of adulthood. Tissues examined were the intestine (E,E’), body-wall muscle (F,F’), pharynx (G,G’), and nerve-ring neurons (H,H’). Data for WT animals are the same in (E–H) and (E’–H’). Data are the mean ± SEM of ≥25 animals combined from at least three experiments. Day 7 was omitted for neurons due to a counting issue of this time point. , WT control vs. daf-2 or glp-1 control at Days 1 and 7; *, daf-2 or glp-1 control vs. daf-2 or glp-1 + BafA at Days 1 and 7, #, daf-2 or glp-1 control at Day 7 vs. daf-2 or glp-1 control at Day 1. ***//###p < 0.0001, **//##p < 0.001, *//#p < 0.01 by Poisson regression.
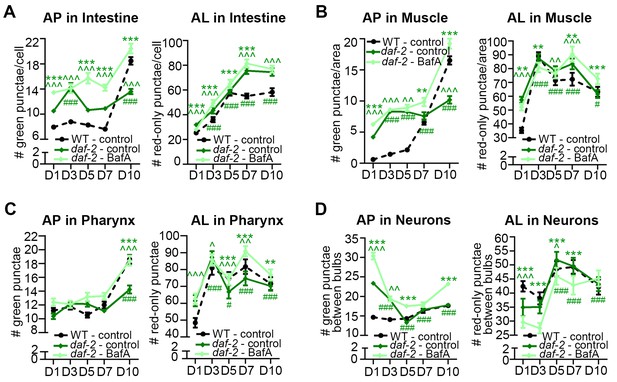
daf-2 insulin/IGF-1 receptor mutants display increased autophagic activity in most tissues.
(A–D) Quantification of autophagosomes (AP) and autolysosomes (AL) in adult Day 1, 3, 5, 7, and 10 daf-2(e1370) animals expressing mCherry::gfp::lgg-1 and injected with DMSO (control, dark green lines) or Bafilomycin A (BafA, light green lines). Tissues examined were the intestine (A), body-wall muscle (B), pharynx (C), and nerve-ring neurons (D). The black dashed lines in (A–C) show data from wild-type (WT) control animals from Figure 3 for comparison (animals were analyzed in parallel). The black dashed line in (D) shows data from WT animals incubated at 20°C for their entire lifespan. Data are the mean ± SEM of ≥25 animals combined from three independent experiments. ∧, WT control vs. daf-2 control at Days 1, 3, 5, 7, and 10; *, daf-2 control vs. daf-2 + BafA at Days 1, 3, 5, 7, and 10; #, daf-2 control at Days 3, 5, 7, and 10 vs. daf-2 control at Day 1. ***/∧∧∧/###p<0.0001, **/∧∧/##p<0.001, */∧/#p<0.01 by Poisson regression. See also Figure 3—figure supplement 1A–H for quantification of APs in gfp::lgg-1 transgenic animals.
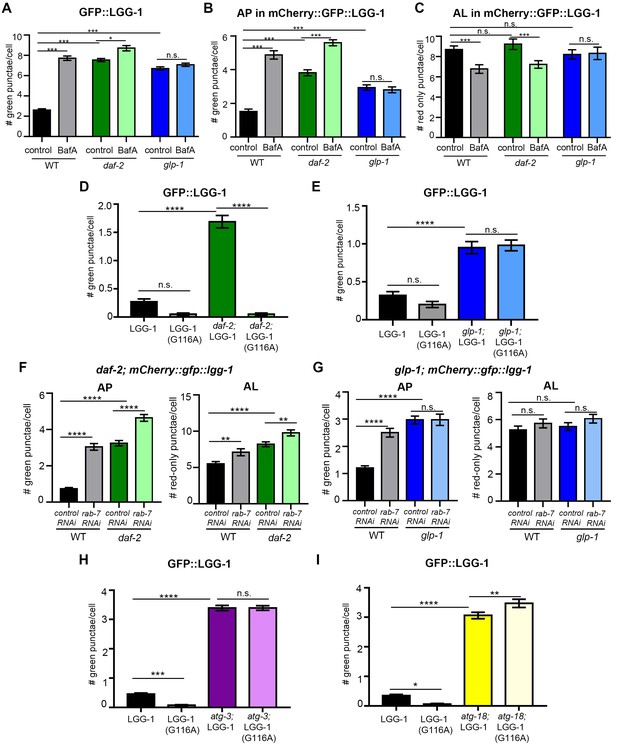
Hypodermal seam cells in daf-2 mutants display increased autophagy, whereas lipidation-independent punctate structures are present in these cells in glp-1 mutants.
(A–C) Quantification of autophagosomes (AP) (A–B) and autolysosomes (AL) (C) in hypodermal seam cells of adult Day 1 wild-type (WT), daf-2(e1370), and glp-1(e2141) transgenic animals expressing gfp::lgg-1 (A) or mCherry::gfp::lgg-1 (B–C) and injected with DMSO (control) or Bafilomycin A (BafA). Data are the mean ± SEM of ≥25 animals combined from at least three independent experiments. ***p<0.0001, *p<0.051 by one-way ANOVA. (D–E) Quantification of GFP-positive punctae in hypodermal seam cells of adult Day 1 WT and daf-2(e1370) animals raised at 20°C (D), and WT and glp-1(e2141) animals raised at 25°C (E) expressing WT gfp::lgg-1 (LGG-1) or mutant gfp::lgg-1 (LGG-1(G116A)). Data are the mean ± SEM from three independent experiments, each with ≥10 animals (one representative experiment shown). ****p<0.0001 by one-way ANOVA. (F–G) Quantification of APs and ALs in the hypodermal seam cells of Day 1 WT and daf-2(e1370 mutants expressing mCherry::gfp::lgg-1 raised at 20°C (F) or Day 1 WT and glp-1(e2141) mutants expressing mCherry::gfp::lgg-1 raised at 25°C (G) and fed from hatching with bacteria expressing empty vector (control) or dsRNA encoding rab-7. Data are the mean ± SEM of ≥40 animals combined from three experiments. ****p<0.0001 and **p<0.005 by one-way ANOVA. (H–I) Quantification of GFP-positive punctae in hypodermal seam cells of adult Day 1 WT, atg-3(bp412) (H) and atg-18(gk378) (I) animals expressing WT gfp::lgg-1 (LGG-1) or mutant gfp::lgg-1 (LGG-1(G116A)). Animals were raised at 20°C. Data are the mean ± SEM combined from three independent experiments for atg-3 with ≥22 animals combined from two independent experiments for atg-18 with ≥16 animals. ****p<0.0001, ***p<0.001, **p<0.01, and *p<0.05 by one-way ANOVA.
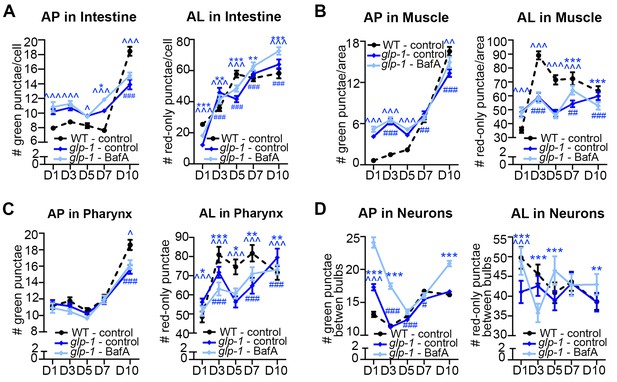
Germline-less glp-1 mutants display a different autophagic activity profile than daf-2 mutants.
(A–D) Quantification of autophagosomes (AP) and autolysosomes (AL) in adult Days 1, 3, 5, 7, and 10 glp-1(e2141) animals expressing mCherry::gfp::lgg-1 and injected with DMSO (control, dark blue lines) or Bafliomycin A (BafA, light blue lines). Tissues examined were the intestine (A), body-wall muscle (B), pharynx (C), and nerve-ring neurons (D). The black dashed lines in (A–D) show data from wild-type (WT) control animals from Figure 3 for comparison (animals were analyzed in parallel). Data are the mean ± SEM of ≥25 animals combined from three independent experiments. ∧, WT + control vs. glp-1 control at Days 1, 3, 5, 7, and 10; *, glp-1 control vs. glp-1 + BafA at Days 1, 3, 5, 7, and 10, #, glp-1 control at Days 3, 5, 7, and 10 vs. glp-1 control at Day 1. ***/∧∧∧/###p<0.0001, **/∧∧/##p<0.001, */∧/#p<0.01 by Poisson regression analysis.
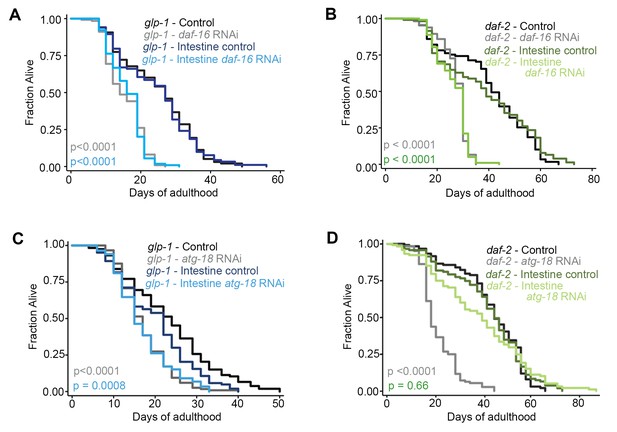
Autophagy genes expressed in intestinal cells are required for lifespan extension of glp-1 mutants, but not of daf-2 mutants.
(A,C) Kaplan–Meier survival curves of glp-1(e2141) single mutants and glp-1(e2141); sid-1(qt9) double mutants expressing sid-1 cDNA in the intestine (vha-6 promoter). Animals were fed from Day 1 of adulthood with bacteria expressing empty vector (control), daf-16/Foxo dsRNA (A), or atg-18/Wipi dsRNA (C). Intestine-specific inhibition of daf-16/Foxo shortened the lifespan of glp-1(e2141) mutants in two out of two experiments ach with ≥100 animals. p<0.0001 for whole body control RNAi vs. whole body daf-16/Foxo RNAi; p<0.0001 for intestine-specific control RNAi vs. intestine-specific daf-16 RNAi by log-rank test. Intestine-specific inhibition of atg-18/Wipi shortened the lifespan of glp-1(e2141) mutants in five out of seven experiments, each with ≥100 animals. p<0.0001 for whole-body control RNAi vs. whole-body atg-18/Wipi RNAi; p=0.0008 for intestine-specific control RNAi vs. intestine-specific atg-18/Wipi RNAi by log-rank test. (B,D) Kaplan–Meier survival curves of daf-2(e1370) single mutants and daf-2(e1370); sid-1(qt9) double mutants expressing sid-1 cDNA in the intestine (vha-6 promoter). Animals were fed from Day 1 of adulthood with bacteria expressing empty vector (control), daf-16/Foxo dsRNA (B), or atg-18/Wipi dsRNA (D). Intestine-specific inhibition of daf-16/Foxo shortened the lifespan of daf-2(e1370) mutants in two out of two experiments, each with ≥100 animals. p<0.0001 for whole body control RNAi vs. whole body daf-16/Foxo RNAi; p<0.0001 for intestine-specific control RNAi vs. intestine-specific daf-16 RNAi by log-rank test. Intestine-specific inhibition of atg-18/Wipi had no significant effect on the lifespan of daf-2(e1370) mutants in all of six experiments, each with ≥100 animals. p<0.0001 for whole-body control RNAi vs. whole-body atg-18/Wipi RNAi; p=0.66 for intestine-specific control RNAi vs. intestine-specific atg-18/Wipi RNAi, by log-rank test. See Supplementary file 1 for details on lifespan analyses and additional repeats.
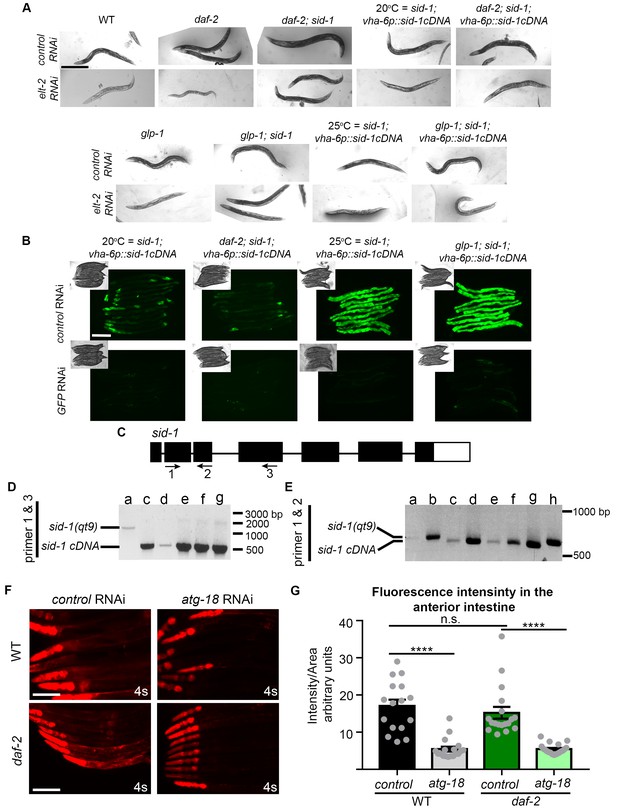
Characterization of intestinal RNAi strains.
(A) Differential interference contrast (DIC) images of Day 3 adult wild-type (WT, (N2), daf-2 (e1370), daf-2(e1370); sid-1(qt9), sid-1(qt9); vha-6p::sid-1cDNA, daf-2(e1370); sid-1(qt9); vha-6p::sid-1cDNA, glp-1(e2141), glp-1(e2141); sid-1(qt9), and glp-1(e2141); sid-1(qt9); vha-6p::sid-1cDNA animals fed from hatching with bacteria containing empty vector (control) or elt-2 dsRNA (elt-2i). Similar results were obtained with animals subjected to pept-1 RNAi, whereas no response was obtained following RNAi knockdown of the muscle-specific gene, unc-112, and hypodermis-specific genes bli-3, bli-4, and lin-26 in sid-1, daf-2; sid-1, or glp-1; sid-1 strains expressing sid-1 in the intestine (data not shown). Scale bar = 400 µm. Data representative of at least two independent experiments. (B) Fluorescence images (GFP), strains express vha-6p::sid-1::sl2::gfp and DIC images (insets) of Day 3 adult animals fed from hatching with bacteria containing empty vector (control) or gfp-encoding dsRNA. Scale bar = 200 µm. Data representative of at least two independent experiments. (C) Schematic of sid-1 gene with primers 1, 2, and 3 indicated by arrows. Black boxes and lines indicate exons and introns, respectively. (D,E) PCR analysis using primers 1 and 3 (D), or 1 and 2 (E) to detect sid-1 transgene expression in (a) glp-1(e2141); sid-1(qt9), (b) daf-2(e1370); sid-1(qt9), (c) glp-1(e2141); sid-1(qt9); myo-3p::sid-1, (d) daf-2(e1370); sid-1(qt9); myo-3p::sid-1, (e) glp-1(e2141); sid-1(qt9); vha-6p::sid-1, (f) daf-2(e1370); sid-1(qt9); vha-6p::sid-1, (g) sid-1(qt9); vha-6p::sid-1, and (h) WT animals. Data are representative of at least two independent experiments. Units are number of base pairs. (F) Fluorescence images (mCherry) of Day 1 adult WT or daf-2(e1370) transgenic animals expressing atg-18p::atg-18::mCherry fed from hatching with bacteria containing empty vector (control) or atg-18/Wipi dsRNA. (G) Quantification of fluorescence intensity in the anterior intestine of Day 1 adult WT or daf-2(e1370) animals expressing atg-18p::atg-18::mCherry fed from hatching with bacteria containing empty vector (control) or atg-18/Wipi-encoding dsRNA. Data are the mean ± SEM and are representative of three independent experiments, each with ≥10 animals. ****p<0.0001 by one-way ANOVA. Scale bar = 100 µm.
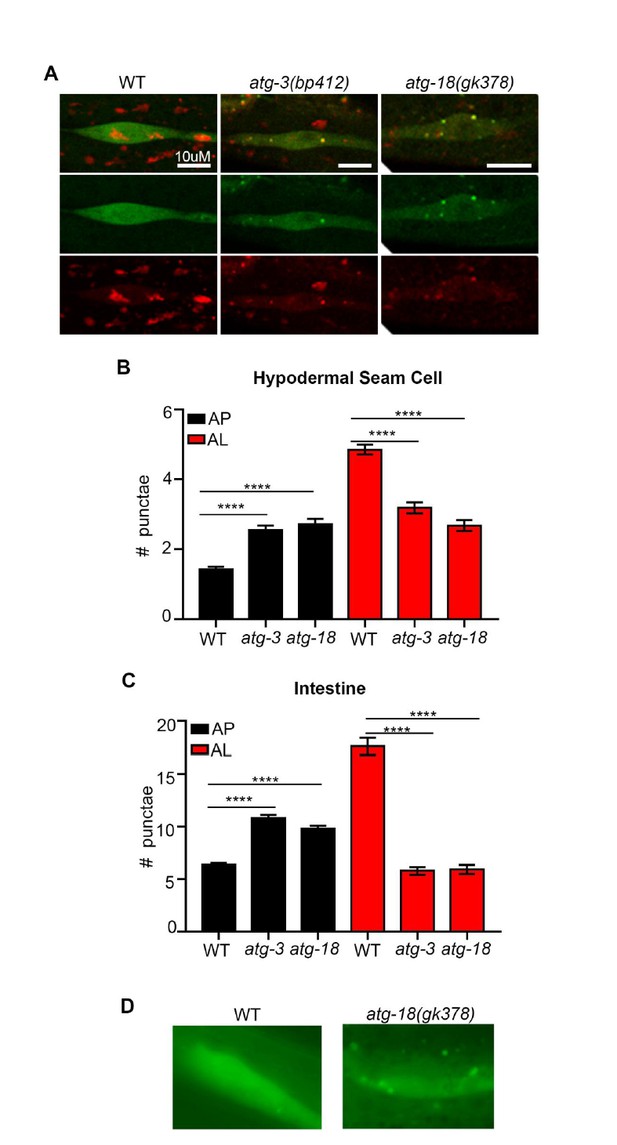
atg-3 and atg-18 mutants have more APs and less ALs compared to wild type.
(A) Hypodermal seam cells in wild-type (WT), atg-3(bp412) and atg-18(gk378) transgenic animals expressing mCherry::gfp::lgg-1, imaged using confocal microscopy at Day 1 of adulthood. (B-C) Quantification of autophagosomes (AP) and autolysosomes (AL) in hypodermal seam cells (B) and intestine (C) of adult Day 1 WT, atg-3(bp412), and atg-18(gk378) animals expressing mCherry::gfp::lgg-1. Data are the mean ± SEM of ≥29 animals combined from three experiments. ****p< 0.0001 by ANOVA. (D) Hypodermal seam cells in WT and atg-18(gk378) transgenic animals expressing gfp::lgg-1 imaged using Zeiss apotome at Day 1 of adulthood. Data courtesy of Dr. Caroline Kumsta, Hansen lab.
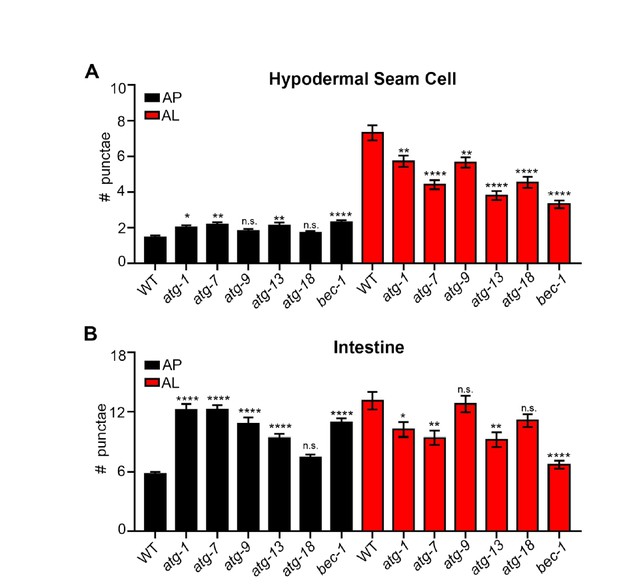
Quantification of autophagosomes and autolysosomes following inhibition of early autophagy genes using RNAi.
(A, B) Quantification of autophagosomes (AP) and autolysosomes (AL) in hypodermal seam cells (A) and intestine (B) of adult Day 1 wild-type animals expressing mCherry::gfp::lgg-1 fed for two generations with bacteria expressing control (empty vector) or atg-1, atg-7, atg-9, atg-13, atg-18 or bec-1 dsRNA. Data are the mean ± SEM of ≥28 animals combined from three experiments. ****p< 0.0001, **p<0.005, * p<0.05 to WT control by ANOVA.
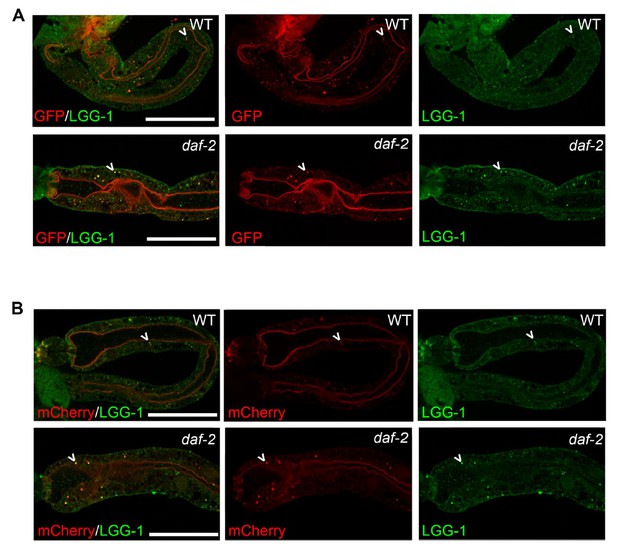
mCherry and GFP co-localize with endogenous LGG-1 in transgenic animals expressing mCherry::gfp::lgg-1.
(A-B) Immunofluorescence to detect endogenous LGG-1 (green, rabbit anti-LGG-1 antibody made by Abgent) and GFP (A; red, mouse anti-GFP from Santa Cruz Biotechnology) or mCherry (B; red, mouse anti-mCherry from Clonetech) in dissected intestines of wild-type (WT, top, data also in Figure 1—figure supplement 1D, E) or daf-2(e1370) animals (bottom). Data representative of ≥2 experiments (N≥5 animals in each). Scale bars = 20 µM.
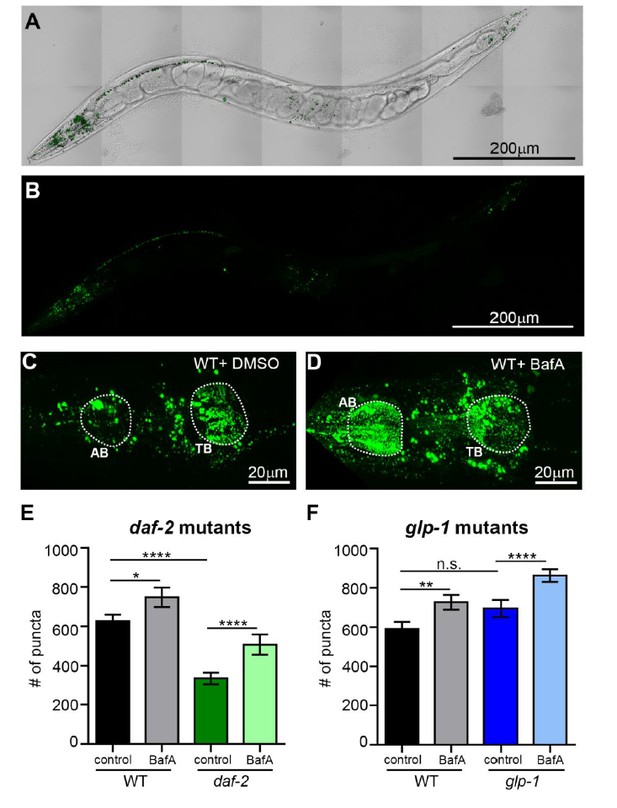
The number of SQST-1/p62 puncta is decreased in daf-2 animals compared to wild type.
(A, B) Whole-body expression of new sqst-1p::sqst-1::gfp reporter in a wild-type (WT) animal at Day 1 of adulthood. Scale bar = 200 µm. Overlay of GFP and bright-field images (A) and GFP only (B). (C, D) Images of the head region quantified (between pharyngeal bulbs indicated by dotted lines) in WT animals treated with DMSO (C) or BafA (D). AB = anterior bulb; TB = terminal bulb. Scale bar = 20 µM. (E, F) Quantification of the number of SQST-1::GFP puncta in daf-2(e1470) and WT animals raised at 20oC (E) or glp-1(e2141) and WT animals raised at 25oC (F) and injected with DMSO or BafA at Day 1 of adulthood. Data are the mean ± SEM of ≥30 animals combined from three experiments. ****p< 0.0001, **p<0.005, * p<0.05 by ANOVA.
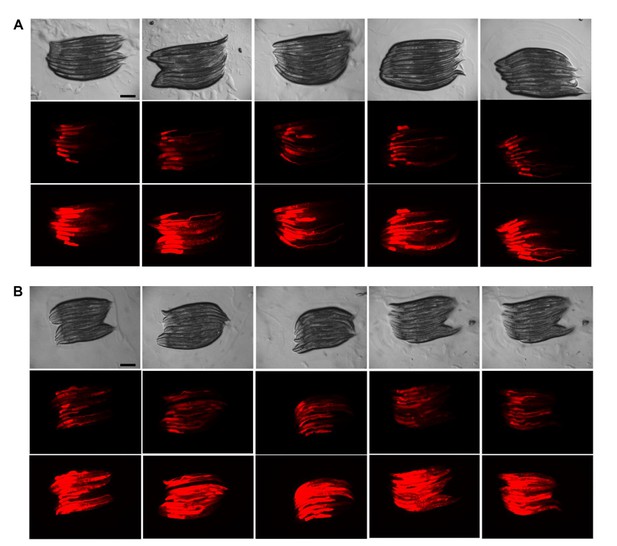
Texas Red Dextran injections are very reproducible.
(A, B) Injection of Texas Red Dextran (5000 Daltons) into wild-type (WT) animals. Images (8x magnification) from 2 experiments (A and B) with 50 injected animals analyzed 10 at a time (i.e., 5 panels of 10 animals) per experiment. Top row = Brightfield, middle row = 0.25 second exposure, bottom row = 1 second exposure. Scale bars = 200 µM.
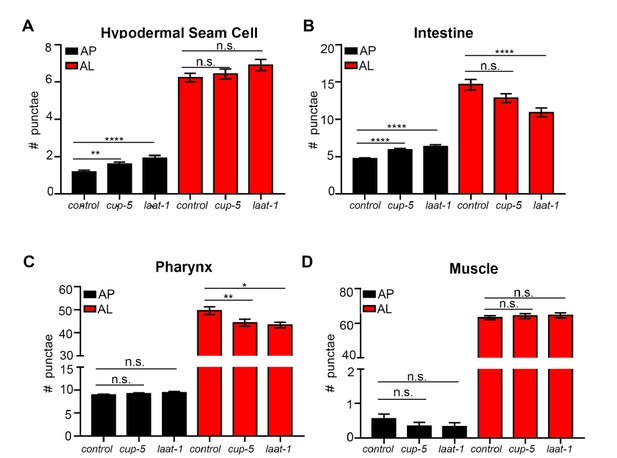
Quantification of autophagosomes and autolysosomes following Inhibition of lysosomal genes cup-5 and laat-1.
(A-D) Quantification of autophagosomes (AP) and autolysosomes (AL) in hypodermal seam cells (A), intestine (B), pharynx (C), and muscle (D) of adult Day 1 wild-type (WT) animals expressing mCherry::gfp::lgg-1 fed for two generations with bacteria expressing empty vector (control) or cup-5 or laat-1 dsRNA. Data are the mean ± SEM of ≥45 animals combined from three experiments. ****p< 0.0001, **p<0.005, *p<0.05 by ANOVA.
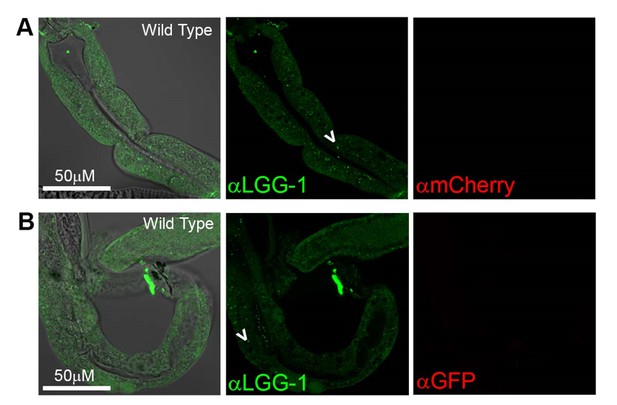
Antibody staining in N2 (Wild Type) animals.
(A-B) Day 1 WT animals stained with rabbit anti-LGG-1 (green middle) and either mouse anti-mCherry (A), or mouse anti-GFP (B) (which here functioned as negative controls). Secondary antibody control had no fluorescence in either green or red channels (data not shown). Left column is merged image DIC, anti-LGG-1 and anti-mCherry or anti-GFP, middle is anti-LGG-1 only (green), and right is either anti-mCherry (A) or anti-GFP (B). Data representative of one experiment (N≥5 animals in each condition).
Additional files
-
Supplementary file 1
(A–B) Lifespan analysis of daf-2 and glp-1 animals subjected to whole-body or tissue-specific RNAi of autophagy genes during adulthood.
Lifespan analysis of glp-1(e2141) and glp-1(e2141); sid-1(qt9) (A), and daf-2(e1370) and daf-2(e1370); sid-1(qt9) (B) animals carrying tissue-specific arrays to re-establish RNAi by expression of sid-1 transgene from intestinal-specific (vha-6) or muscle-specific (myo-3) promoters. WT and daf-2 animals were incubated at 20°C and fed from Day 1 of adulthood with bacteria containing empty vector (control) or expressing dsRNA encoding autophagy genes atg-18/Wipi, lgg-1/Atg8 or daf-16/Foxo. glp-1(e2141) animals were raised at 25°C and moved to 20°C at Day 1 of adulthood for the rest of their lifespan. Individual experiments are numbered (Exp#). We note that lgg-1 RNAi did not significantly shorten the lifespan of glp-1(e2141); sid-1(qt9); vha-6p::sid-1 transgenic animals, but this treatment had relatively weak effects in glp-1(e2141) single mutants analyzed in parallel experiments. Moreover, the glp-1; sid-1; vha-6p::sid-1 strain was, for unexplainable reasons, short-lived in experiment number 5. Whole-body, muscle-specific, and intestinal-specific inhibition of autophagy genes generally had no effect or slightly decreased the lifespan of wild-type N2 animals (JTC and MH, unpublished results). Data show the average lifespan (avg. LS), the number of events (# animals/Total; number of dead animals/total number of animals analyzed, the percentage lifespan extension (% change) of animals subjected to gene-specific vs control RNAi, and the p value for this comparison. p values were calculated using the Mantel–Cox log-rank test. *data shown in Figure 6.
- https://doi.org/10.7554/eLife.18459.018
-
Supplementary file 2
C. elegans strains used in this study.
- https://doi.org/10.7554/eLife.18459.019
-
Supplementary file 3
Primers used in this study.
- https://doi.org/10.7554/eLife.18459.020





