Cooperation of the ER-shaping proteins atlastin, lunapark, and reticulons to generate a tubular membrane network
Figures
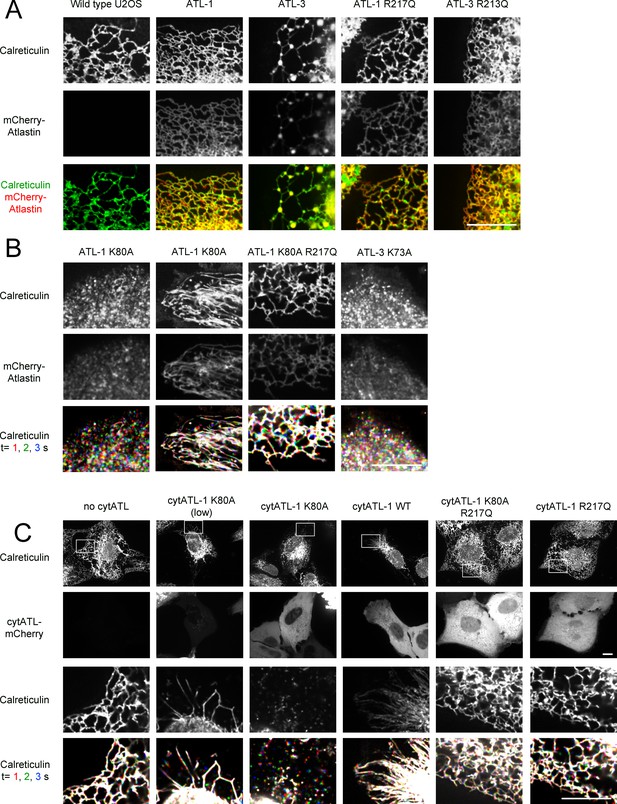
ATL is required to maintain tubules and junctions in mammalian cells.
(A) Peripheral ER network of U2OS cells expressing GFP-calreticulin from the endogenous promoter in wild type cells. Where indicated, the cells also stably expressed mCherry-tagged wild type ATL-1 or ATL-3 or the corresponding dimerization-defective mutants (ATL-1 R217Q or ATL-3 R213Q). Scale bar = 10 µm. (B) As in (A), but with cells stably expressing mCherry-tagged ATL-1 or ATL-3 mutants defective in GTP hydrolysis (ATL-1 K80A or ATL-3 K73A). The first and second columns show ATL-1 K80A-expressing cells, in which the ER is either fragmented or converted into long, unbranched tubules. ATL-1 K80A R217Q is both GTPase- and dimerization- defective. The bottom row shows three time points for calreticulin staining. Stationary pixels appear white, while those moving appear in colors. Scale bar = 10 µm. (C) As in (A), but with cells stably expressing mCherry-tagged cytoplasmic fragments of wild type ATL-1 (cytATL-1 WT), a mutant defective in GTP hydrolysis (cytATL-1 K80A) at low or high level, or a mutant defective in dimerization (cytATL-1 R217Q). CytATL-1 K80A R217Q contains both mutations. The bottom two rows show a close-up image of the peripheral network of the cells depicted in the first two rows. The bottom row shows three time points for calreticulin staining. Stationary pixels appear white, while those moving appear in colors. Scale bar = 10 µm.
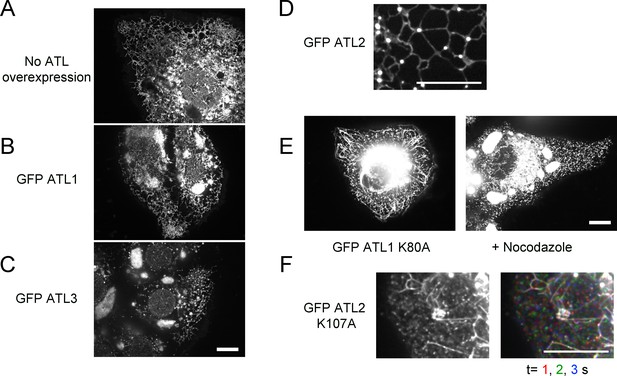
Effect of ATL overexpression on ER morphology in U2OS cells.
(A) Image of a whole U2OS cell expressing GFP-calreticulin. (B) As in (A), but a cell expressing GFP-ATL-1. (C) As in (A), but a cell expressing GFP-ATL-3. Scale bar = 10 µm. (D) Peripheral ER network in a U2OS cell stably expressing GFP-tagged ATL-2. Scale bar = 10 µm. (E) Image of a U2OS cell expressing GFP-ATL-1 K80A, untreated (left panel) or treated with 1 µM nocodazole for 90 min (right panel). Scale bar = 10 µm. (F) Peripheral ER network in a U2OS cell stably expressing a GTPase-defective mutant of ATL-2 (GFP-ATL-2 K107A). The right panel shows three time points. Stationary pixels appear white, while those moving appear in colors. Scale bar = 10 µm.

ATL is required to maintain an ER network in Xenopus egg extracts.
(A) An ER network was generated with a crude interphase Xenopus egg extract and stained with the lipophilic fluorescent dye DiIC18. The sample was imaged with a spinning disk confocal microscope. Scale bar = 10 µm. (B) As in (A), but in the presence of 1 µM cytoplasmic fragment of Xenopus ATL (cytATL). Scale bar = 10 µm. (C) As in (A), but with 2 µM cytATL. Scale bar = 10 µm. (D) As in (A), but with 2 µM of the dimerization-defective mutant fragment cytATL R232Q. Scale bar = 10 µm. (E) As in (A), but with 1.5 mM GTPγS. Scale bar = 10 µm. (F) An ER network was generated with interphase cytosol, light membranes, and an energy-regenerating system. After 30 min, buffer, 2 µM cytATL, 2 µM cytATL R232Q, or 2 mM GTPγS were added. The membranes were stained with octadecyl rhodamine. Scale bar = 20 µm. (G) A mitotic ER network was generated with a crude Xenopus extract containing DiIC18 and 2 µM cytATL R232Q. Scale bar = 10 µm. (H) As in (G), but with 2 µM wild type cytATL. Scale bar = 10 µm.
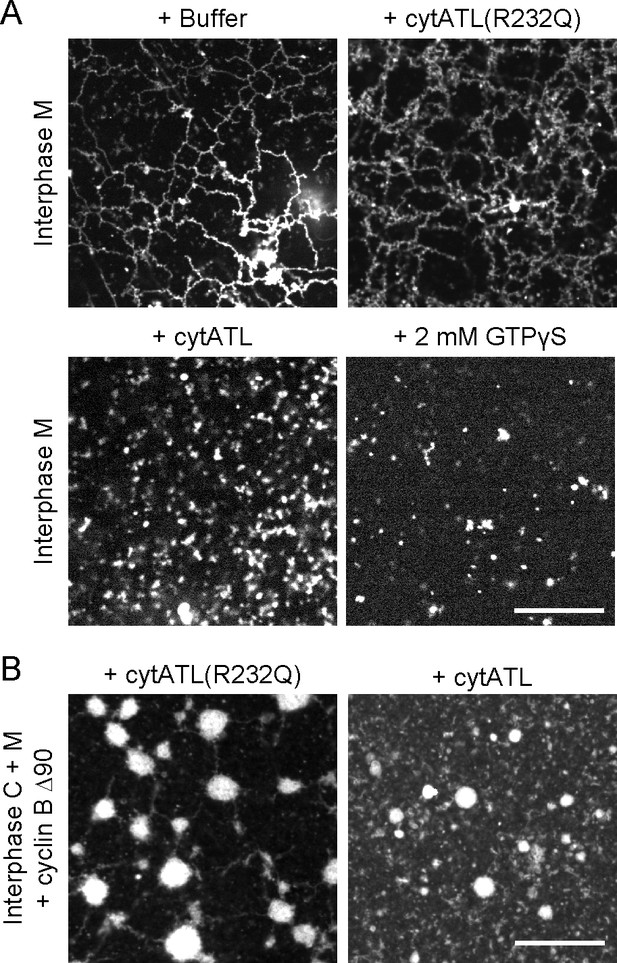
ER network formed with Xenopus extract fractions is disassembled by ATL inactivation.
(A) An ER network was generated with interphase light membranes. Buffer, 2 µM cytATL, 2 µM cytATL R232Q, or 2 mM GTPγS were added subsequently and the membranes were stained with octadecyl rhodamine. (B) A mitotic ER network was formed from the interphase cytosol and light membranes by the addition of non-degradable cyclin B∆90. After addition of 2 µM cytATL or 2 µM cytATL R232Q, the membranes were stained with octadecyl rhodamine. Scale bars = 20 µm.
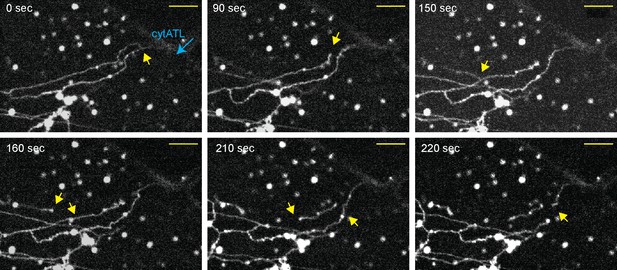
Real-time disassembly of an ER network in the presence of cytATL.
A network was formed from a crude Xenopus egg extract in the presence of the dye DiIC18 in a computer-controlled microfluidics device. CytATL-GFP (5 µM) in cytosol containing an energy regenerating system was then slowly perfused into the chamber at a total laminar flow rate of 0.5 µL/min from multiple ports (one is indicated by a blue arrow). The arrival of cytATL- GFP arrival in the chamber was monitored by GFP fluorescence (not shown). Images represent snapshots of a real-time video (see Video 3). Yellow arrows point to the detachment or breakage of tubules. Scale bar = 10 µm.
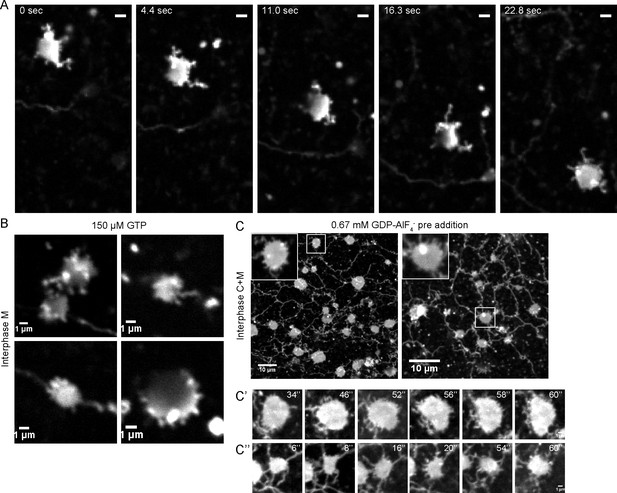
Intermediates during in vitro ER network formation.
(A) DiIC18-prelabeled light membranes were mixed with buffer and an energy regenerating system. The sample was imaged immediately by confocal microscopy. Scale bar = 2 µm. (B) Xenopus egg light membranes were incubated in the presence of 150 µM GTP and octadecyl rhodamine. Scale bar = 1 µm. (C) Interphase Xenopus egg cytosol, light membranes, 0.67 mM GDP-AlF4-, and an energy-regenerating system were incubated for 30 min. The membranes were stained with octadecyl rhodamine. Scale bars = 10 µm. Insets and images in C’ and C’’ show magnified views of small sheets from which short tubules emanate. Scale bars = 1 µm.
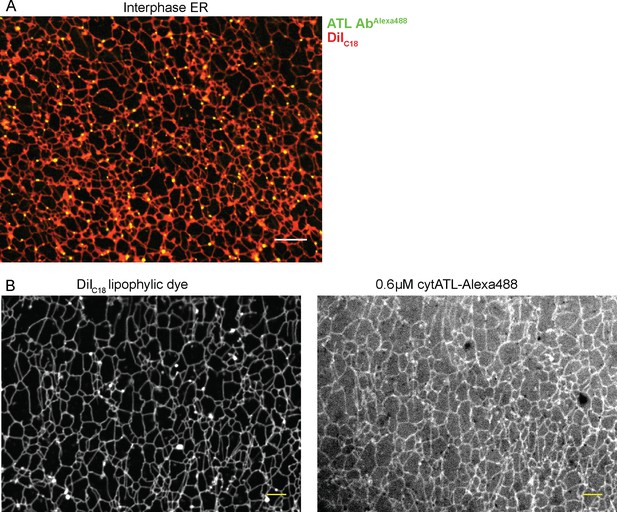
Localization of ATL in an in vitro generated ER network.
(A) An interphase ER network was generated with a crude Xenopus egg extract containing the dye DiIC18. Endogenous ATL was visualized by including 16 nM Alexa488-labeled, affinity-purified antibodies raised against Xenopus ATL (ATL AbAlexa488). Note that ATL localizes preferentially to three-way junctions. Scale bar = 10 µm. (B) An interphase ER network was assembled as in (A) and labeled with DiIC18 and 0.6 µM Alexa488-labeled cytATL. Note that cytATL localizes throughout the tubules, marking the position of inactivated endogenous ATL. Scale bar = 10 µm.
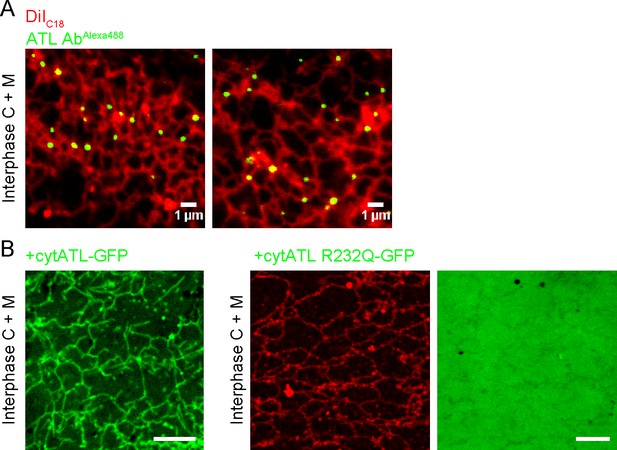
Localization of endogenous and inactivated ATL in a Xenopus ER network.
(A) An interphase ER network was generated with cytosol, DiIC18-prelabeled light membranes, and an energy regenerating system. Endogenous ATL was visualized by including 10.5 nM Alexa488-labeled, affinity-purified antibodies against Xenopus ATL. The sample also contained sperm chromatin to stabilize the network by reducing thermal convection. Scale bars = 1 µm. (B) A mixture of interphase cytosol, light membranes, and an energy regenerating system was incubated in the presence of 0.5 µM cytATL-GFP, but absence of membrane stain,for 30 min (left panel). Parallel reactions were performed with 2 µM cytATL(R232Q)-GFP and the membrane stain octadecyl rhodamine (middle and right panels). Scale bars = 10 µm.
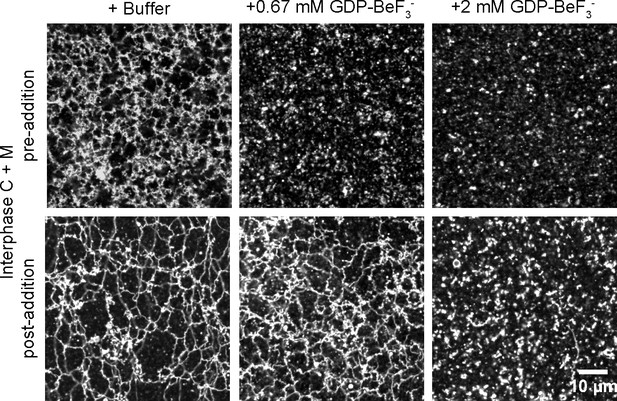
Effect of GTP analogs on ER network formation and maintenance.
GDP-BeF3- was added at 0.67 mM or 2 mM either before (upper panels) or after (lower panels) formation of an ER network from Xenopus egg cytosol (C), light membranes (M), and an energy regenerating system. The membranes were stained with octadecyl rhodamine. Scale bar = 10 µm.
ER network maintenance is less sensitive to ATL inhibition than ER network formation.
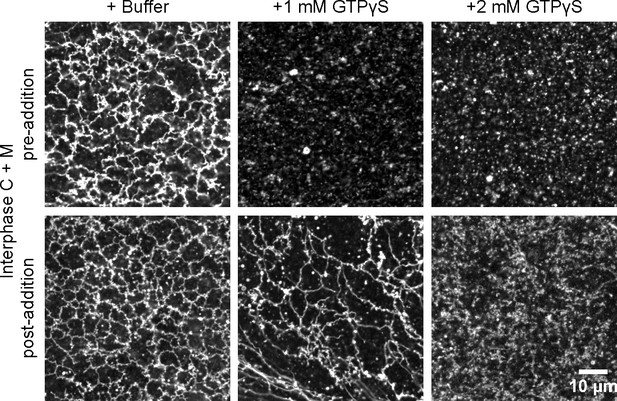
GTPγS was added at 1 mM or 2 mM either before (upper panels) or after (lower panels) formation of an ER network from Xenopus egg cytosol (C), light membranes (M), and an energy regenerating system. The membranes were stained with octadecyl rhodamine. Scale bar = 10 µm.
Interplay between ATL and the reticulons.
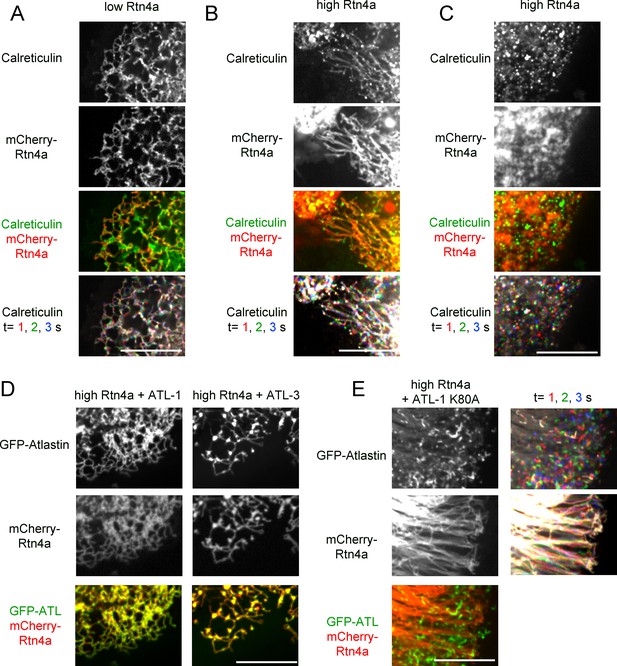
(A) Peripheral ER network in a U2OS cell expressing GFP-calreticulin under the endogenous promoter in wild type cells or in cells stably expressing mCherry-tagged Rtn4a. The bottom row shows three time points. Stationary pixels appear white, while those moving appear in colors. Scale bar = 10 µm. (B) As in (A), but with a higher expression level of mCherry-Rtn4a, resulting in unbranched tubules. (C) As in (B), but showing a cell with fragmented ER. (D) As in (B), but with cells that also stably express GFP-ATL-1 or GFP-ATL-3. (E) As in (B), but with cells that also express a GTPase defective ATL-1 mutant (GFP-ATL-1 K80A). The second column shows three time points for both the ATL and Rtn channels. Stationary pixels appear white, while those moving appear in colors. Scale bar = 10 µm.
Co-overexpression of ATL mutants and Rtn4a in U2OS cells.
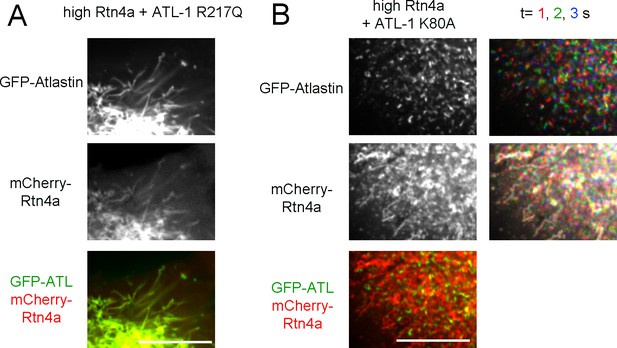
(A) Peripheral ER network in a U2OS cell expressing a high level of mCherry-tagged Rtn4a and GFP-ATL-1 R217Q, a dimerization defective mutant. Scale bar = 10 µm. (B) Peripheral ER network in a U2OS cell expressing a high level of mCherry-tagged Rtn4a and GFP-ATL-1 K80A, a GTPase defective mutant. The second column shows three time points for both the ATL and Rtn channels. Stationary pixels appear white, while those moving appear in colors. Scale bar = 10 µm.
Lnp determines the abundance of three-way tubular junctions in mammalian cells.
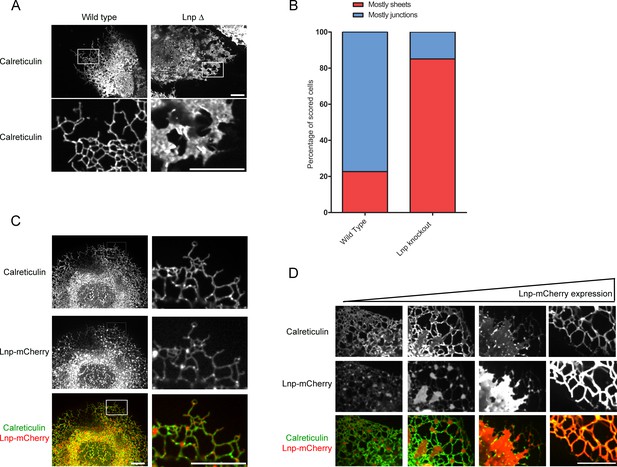
(A) Views of wild type U2OS and Lnp-deleted (LnpΔ) cells expressing GFP-calreticulin from the endogenous promoter. LnpΔ cells were generated by CRISPR targeting the start codon of the LNP gene. The bottom row shows magnifications of the boxed areas of the peripheral ER. Scale bars = 10 µm. (B) Quantification of the LNP deletion phenotype depicted in A. Wild type and LnpΔ cells were scored blindly for the appearance of peripheral ER. (C) Peripheral ER in a LnpΔ cell expressing GFP-calreticulin, as well as stably expressing a low level of Lnp-mCherry. Scale bars = 10 µm. (D) As in (C), but with cells expressing increasing levels of Lnp-mCherry (left to right). Scale bars = 10 µm.
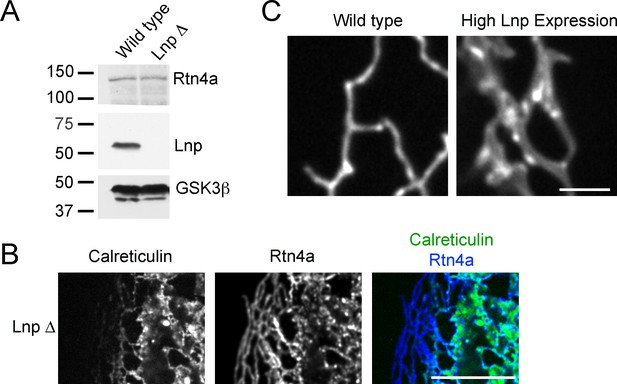
ER morphology in U2OS cells lacking or overexpressing Lnp.
(A) Extracts of wild type U2OS cells and a Lnp-deleted U2OS clonal cell line were analyzed by immunoblotting with Lnp and Rtn4 antibodies. Equal amounts of total protein were loaded, and GSK3β was used as a loading control. (B) Peripheral ER in a U2OS cell lacking Lnp expressing GFP-calreticulin from the endogenous promoter. The cells were fixed and analyzed for GFP fluorescence and for endogenous Rtn4a, using specific antibodies and fluorescently labeled secondary antibodies. Scale bar = 10 µm. (C) Peripheral ER in wild type U2OS cells and cells highly expressing Lnp-mCherry. The ER is visualized by GFP-calreticulin expressed under the endogenous promoter. Scale bar = 2 µm.
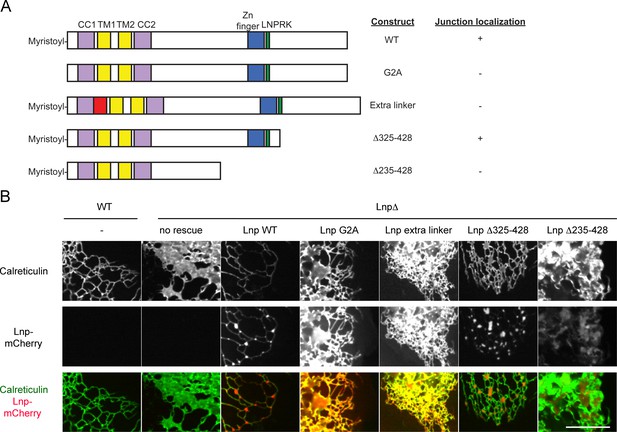
Lnp domains important for junction localization.
(A) Schematic representation of wild type (WT) Lnp and mutants tested for proper localization in the peripheral ER. CC1, CC2, coiled-coil domains 1 and 2, respectively; TM1, TM2, trans-membrane segments 1 and 2, respectively; LNPRK, lunapark motif (single letter code); the segment indicated in red was inserted (extra linker). (B) Peripheral ER in U2OS cells expressing GFP-calreticulin under the endogenous promoter in wild type or Lnp-lacking cells (LnpΔ). Where indicated, the cells also stably expressed wild type or mutant Lnp at low levels. Scale bar = 10 µm.
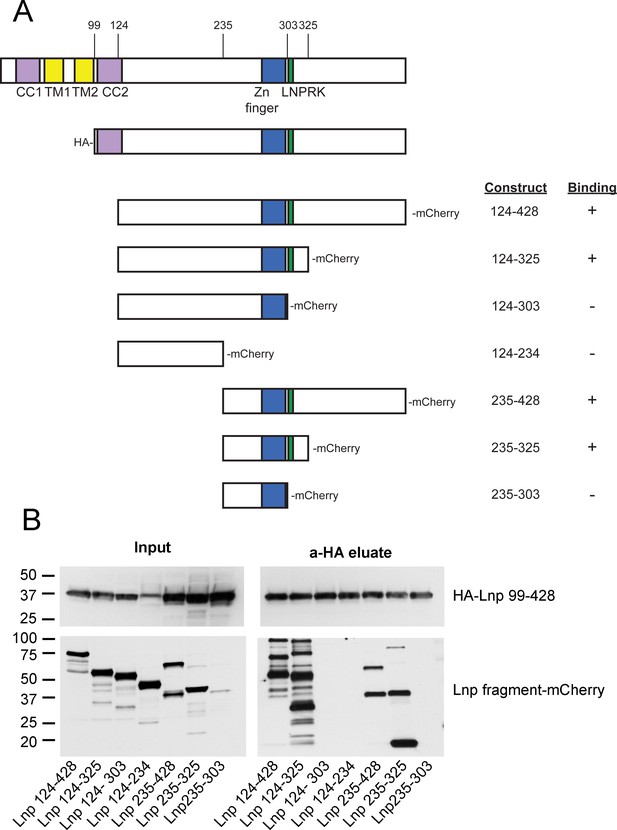
Lnp-Lnp interaction is mediated by the Zn2+-finger domain-containing region.
(A) Schematic representation of Lnp truncation mutants tested for interaction. Hemagglutinin (HA)-tagged Lnp 99–428 was used as bait for pull-downs of mCherry-tagged truncations. (B) Immunoblots of co-immunoprecipitation experiments. Each of the mCherry-tagged fragments and the HA-tagged bait were expressed separately in 293T cells. Cells were lysed, and the levels of mCherry-tagged truncations were normalized according to fluorescence at 590 nm, except Lnp 235–303 which was poorly expressed. After mixing with lysate containing HA-Lnp 99–428, pull-downs were performed with HA-antibodies and the samples were analyzed by immunoblotting with HA- and mCherry- antibodies.
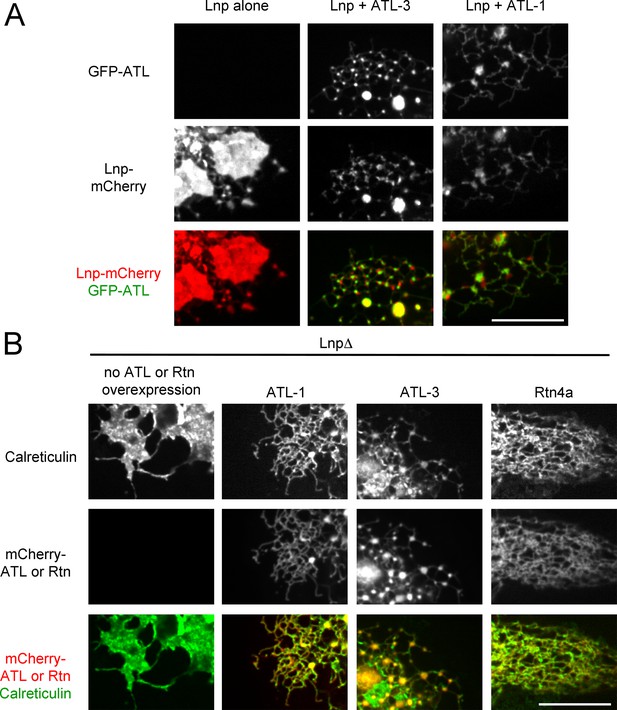
Interplay of ATL, Lnp, and Rtn.
(A) Peripheral ER in U2OS cells stably expressing Lnp-mCherry alone or together with GFP-ATL-3 or GFP-ATL-1. Scale bar = 10 micron. (B) Peripheral ER in Lnp-lacking U2OS cells expressing GFP-calreticulin. Where indicated, the cells also stably expressed mCherry-tagged ATLs or Rtn4a. Scale bar = 10 micron.
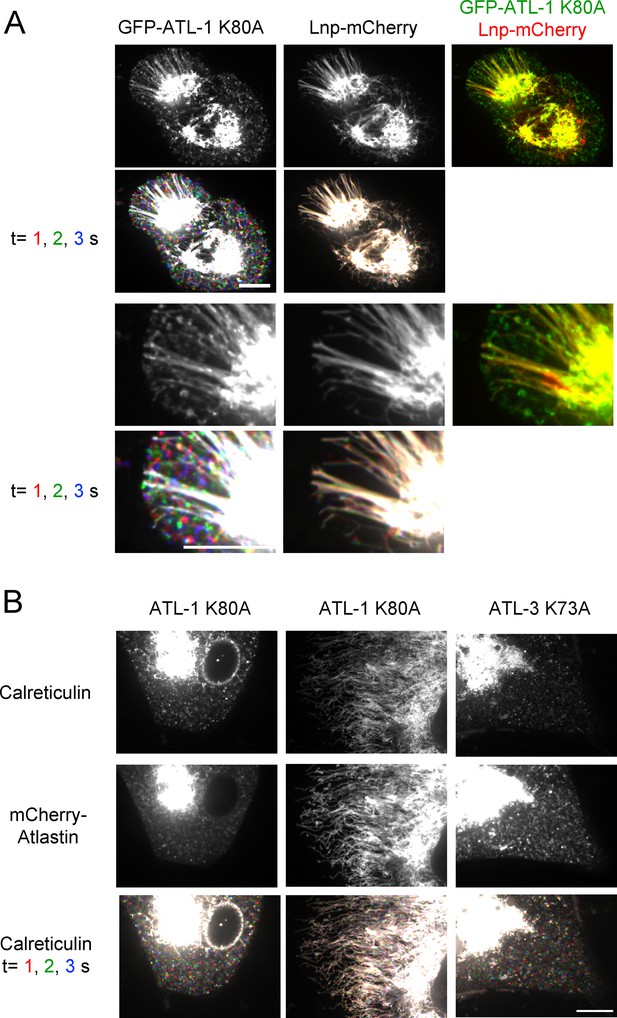
ATL acts upstream of Lnp in U2OS cells.
(A) Peripheral ER in U2OS cells stably expressing Lnp-mCherry and GFP-ATL-1 K80A (GTPase defective mutant). The second and fourth rows show three time points. Stationary pixels appear white, while those moving appear in colors. The top five panels depict two entire cells, while the bottom five panels show magnifications of the peripheral ER of these cells. Scale bars = 10 µm. (B) Peripheral ER in Lnp-lacking U2OS cells expressing GFP-calreticulin. The cells also stably express mCherry-tagged ATL mutants defective in GTPase activity. The bottom row shows three time points. Scale bar = 10 µm.
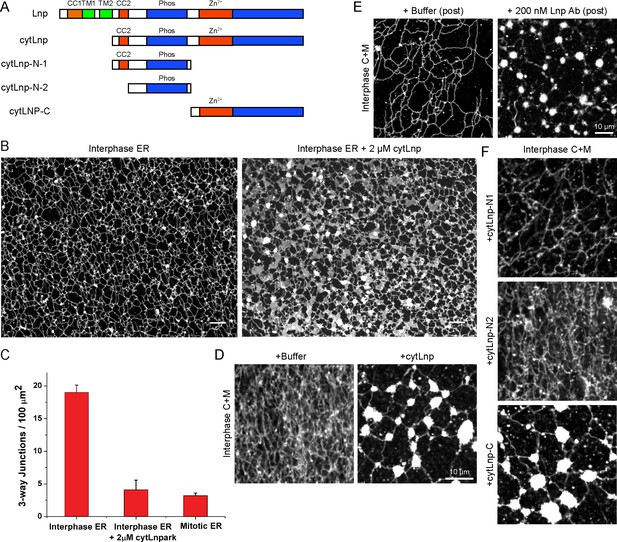
Effect of Lnp inactivation on an in vitro generated ER network.
(A) Schematic representation of wild type and mutant Xenopus Lnp. Phos indicates the domain phosphorylated during mitosis. (B) An ER network was generated for 20 min with crude Xenopus egg extract in the presence of the dye DiIC18 and either buffer (left panel) or 2 µM of a cytoplasmic fragment of Lnp (cytLnp). Scale bar = 10 µm. (C) The number of three-way junctions at different conditions was quantitated. Error bars indicate the mean ± SD of three independent experiments. (D) An interphase ER network was formed with Xenopus egg cytosol (C), light membranes (M), an energy regenerating system, in the presence or absence of 5 µM cytLnp. The network was stained with octadecyl rhodamine. Scale bar = 10 µm. (E) Buffer or 200 nM of affinity-purified Lnp antibodies were added to a preformed interphase network generated with cytosol, membranes, and an energy regenerating system. Scale bar = 10 µm. (F) As in (D), except that 5 µM cytLnp-N1, cytLnp-N2 or cytLnp-C were added at the beginning of the network formation reaction. Scale bars = 10 µm.
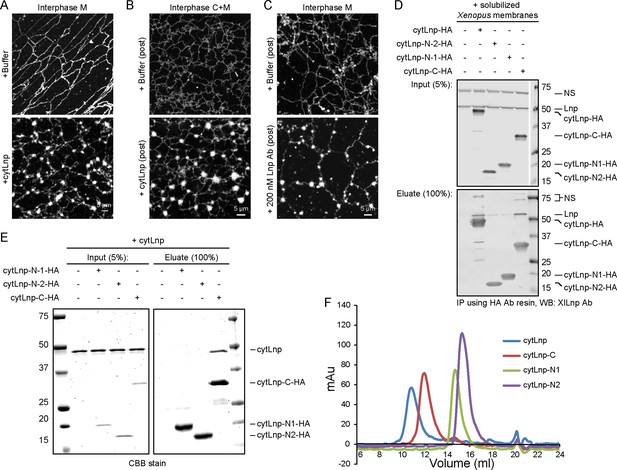
Cytoplasmic fragments of Lnp interfere with the function of endogenous Xenopus Lnp.
(A) Light membranes (M) were incubated with an energy regenerating mix and either buffer or 5 µM cytLnp for 15 min. The membranes were stained with octadecyl rhodamine. Scale bar = 5 µm. (B) Buffer or 5 µM of cytLnp was added to a preformed network formed from cytosol (C) and membranes (M). Scale bar = 5 µm. (C) Buffer or 200 nM of affinity-purified Lnp antibody was added to a preformed network formed from membranes only. Scale bar = 5 µm. (D) Interaction of endogenous Xenopus egg Lnp with various Lnp fragments. A detergent extract of light membranes was incubated with empty resin or hemagglutinin (HA)-tagged Lnp fragments (see Figure 11A). After pull-down with HA antibodies, the bound fractions were analyzed by immunoblotting with antibodies to Xenopus Lnp. (E) Interaction of purified Xenopus cytLnp with various cytLnp fragments. HA-tagged Lnp fragments (see Figure 11A) was incubated with cytLnp, and subsequently HA antibodies. Bound fractions were analyzed by SDS-PAGE and stained with Coomassie blue. (F) Purified cytLnp or fragments of it (see Figure 11A) were analyzed by gel filtration on a Superdex 200 column.
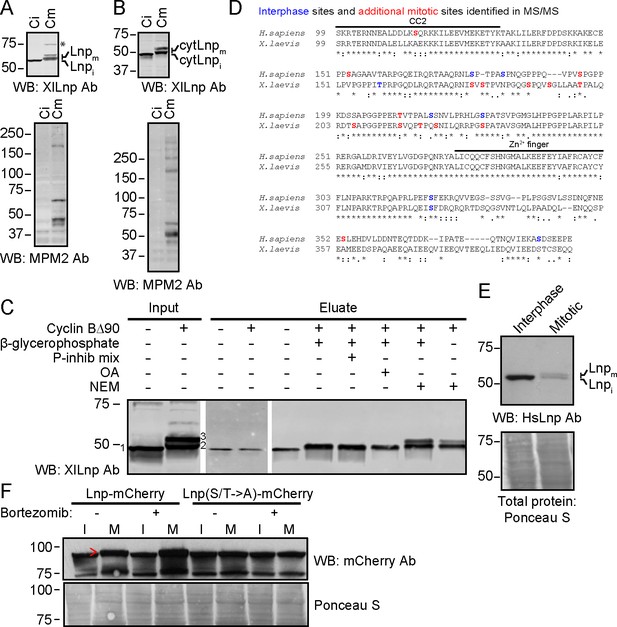
Mitotic phosphorylation of Lnp.
(A) Xenopus egg interphase cytosol, membranes, and an energy regenerating system were incubated with buffer (Ci) or non-degradable cyclin B∆90 (Cm) for 40 min. The samples were analyzed by SDS-PAGE and immunoblotting with Xenopus Lnp antibodies (XlLnp Ab). The lower panel shows an immunoblot with MPM2 antibodies that recognize mitotically phosphorylated proteins. (B) As in (A), but cytLNP was incubated with cytosol in the absence of membranes. (C) Purified HA- and His-tagged cytLnp (HA-cytLnp) was incubated with interphase cytosol with or without cyclin B∆90. An aliquot was analyzed directly by SDS-PAGE (input), while the remainder was incubated with cobalt resin in the presence of various inhibitors (P-inhib is a phosphatase inhibitor cocktail (Sigma); OA, okadaic acid; NEM, N-ethylmaleimide). Material eluted from the beads with imidazole (eluate) was analyzed by SDS-PAGE and immunoblotting with Xenopus Lnp antibodies. Band 1, unmodified HA-cytLnp; bands 2 and 3, mitotically modified Lnp. (D) Alignment of the cytosolic domains of human and Xenopus LNP sequences. Residues in blue are phosphorylated in interphase, as determined by mass spectrometry. Residues in red are additionally phosphorylated during mitosis. (E) U2OS cells were grown in complete medium and left untreated or treated with 100 nM nocodazole overnight. Interphase cells were scraped off, while mitotically arrested cells were collected as non-adherent cells. Equal amounts of total protein were analyzed by SDS-PAGE and immunoblotting with antibodies to human Lnp (HsLnp). Lnpi and Lnpm, interphase and mitotic Lnp, respectively. (F) Lnp-mCherry or Lnp-mCherry with Ser and Thr phosphorylation sites mutated to Ala (S/T ->A) were stably expressed in U2OS cells. Cells were arrested in mitosis by incubation with 100 nM nocodazole overnight. Where indicated, 100 nM bortezomib was present overnight. Equal amounts of total protein from interphase (I) or mitotic (M) cells were analyzed by SDS-PAGE and immunoblotting with mCherry antibodies. The arrowhead indicates the position of mitotically phosphorylated Lnp.
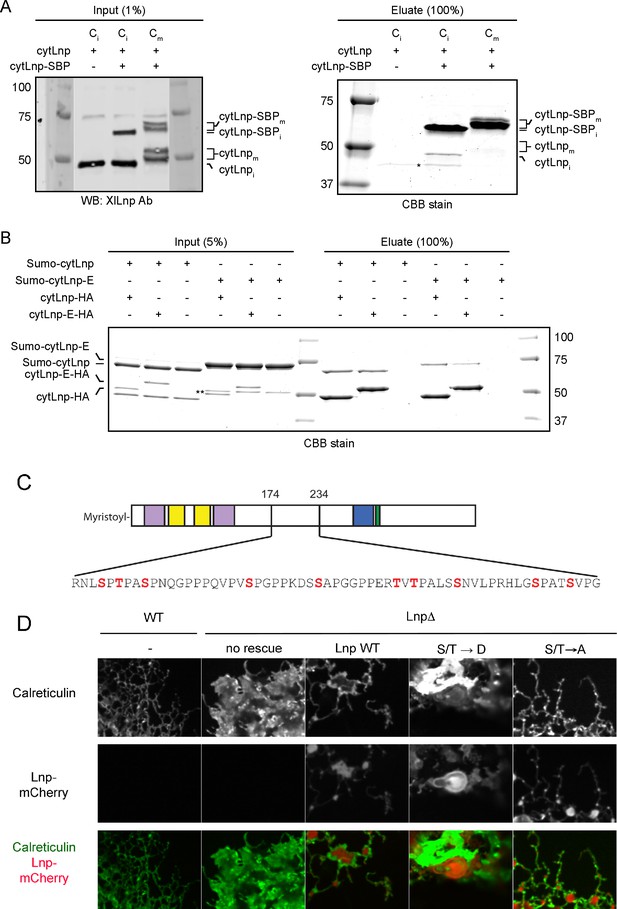
Mitotic phosphorylation weakens Lnp-Lnp interaction.
(A) Lnp-Lnp interaction is weakened during mitosis. Purified cytLnp and SBP-tagged cytLnp (cytLnp-SBP) were incubated with interphase cytosol in the absence or presence of cyclin B∆90. The samples were incubated with streptavidin resin, bound material was eluted with biotin, and analyzed by SDS-PAGE and staining with Coomassie blue. The left panels show the input material. (B) Purified cytLnp-HA or a phosphomimetic version of it (cytLnp-E-HA) were incubated with Sumo-tagged cytLnp (Sumo-cytLnp) or a phosphomimetic version of it (Sumo-cytLnp-E). Pull-downs were performed with anti-HA resin and the bound material was analyzed by SDS-PAGE and staining with Coomassie blue. (C) Lnp residues that were mutated to aspartate or alanine to generate phosphomimetic (S/T → D) or unphosphorylatable (S/T → A) mutants are shown in red. (D) Peripheral ER in Lnp-lacking U2OS cells (Lnp-/-) expressing GFP-calreticulin. Where indicated, the cells also stably express at low level mCherry-tagged wild type Lnp, or phosphomimetic or unphosphorylatable mutants. Scale bar = 10 µm.
Videos
ER in U2OS cells expressing dominant-negative ATL-1.
U2OS cell stably expressing GFP-ATL-1 K80A (GTPase-defective mutant) with most of the peripheral ER fragmented. Images were acquired with a spinning disk confocal microscope at 0.05 sec intervals for 5 sec. The video is displayed at the same rate it was acquired (20 frames per sec). Image scale: 87 × 66 µm.
ER in U2OS cells expressing dominant-negative ATL-1.
U2OS cell stably expressing GFP-ATL-1 K80A (GTPase-defective mutant) with many unbranched tubules and a fragmented peripheral ER. Images were acquired with a spinning disk confocal microscope at 0.05 sec intervals for 5 sec. The video is displayed at the same rate it was acquired (20 frames per sec). This cell is also depicted in Figure 1— figure supplement 1A, left panel. Image scale: 87 × 66 µm.
Disassembly of an in vitro generated ER network by cytATL.
An ER network was formed in a microfluidics chamber from Xenopus crude extract containing DiIC18. Xenopus egg cytosol containing 5 µM cytATL-GFP was slowly perfused into the chamber using a computer-controlled pneumatic pump (blue arrow indicates perfusion port) while images were acquired with a spinning disk confocal microscope at 10-sec intervals for 9 min. The video is shown at 3 frames per sec. Yellow arrowheads indicate ER disassembly events. Still images of this video were used for Figure 3. Scale bar = 5 µm.
Intermediates of an in vitro generated ER network at early time points.
An ER network assembly reaction was prepared by mixing DiIC18-prelabeled Xenopus egg light membranes with buffer and an energy regenerating system. The sample was imaged immediately using a spinning-disk confocal microscope at 0.5-sec time intervals for 39 sec. The video is shown at 10 frames per sec. Scale bar = 2 µm.
Intermediates of an in vitro generated ER network at early time points.
As in Video 4, but the image was acquired at 0.2-sec time intervals for 5 sec. Scale bar = 5 µm. The video is shown at 5 frames per sec.
ER morphology in U2OS cells after overexpression of Rtn4a.
U2OS cells expressing GFP-calreticulin under the endogenous promoter as well as stably expressing mCherry- Rtn4a. Left panel: calreticulin signal, right panel: mCherry-Rtn4a signal. Images were acquired with a spinning disk confocal microscope at 0.4 sec intervals for 4 sec. The video is displayed at the same rate it was acquired (2.5 frames per sec). Image scale: 87 × 66 µm for each panel.





