Mechanistic signs of double-barreled structure in a fluoride ion channel
Figures
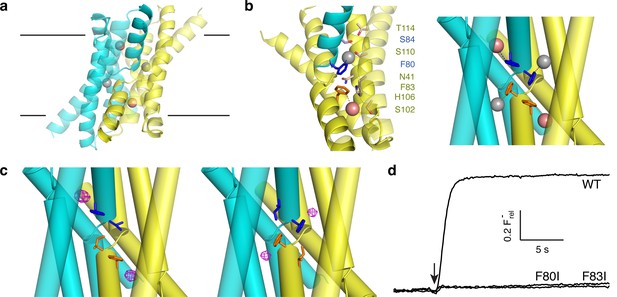
Fluoride ions in Fluc-Ec2 channels.
(a) View of Ec2 channel imagined in membrane (black lines) with subunits colored cyan and yellow and F1-, F2- ions indicated by grey, pink spheres, respectively. From PDB #5A43, rerefined to 2.56 Å resolution. (b) Ion-coordinating region of WT Ec2. 'Polar track' along one pore (left), and detail of the F−-binding Phe-box region (right). The polar track and Phe-box contain residues from both dimer subunits, with sidechains colored accordingly. (c) Phe-box region of F80I (left) or F83I (right) crystal structures (PDB 5KBN, 5KOM, respectively). F−-omit difference densities are shown in magenta, contoured at 4 σ. (d) F− flux traces of indicated Fluc variants. Vertical scale bar represents F− appearing in external medium after addition of valinomycin (arrow).
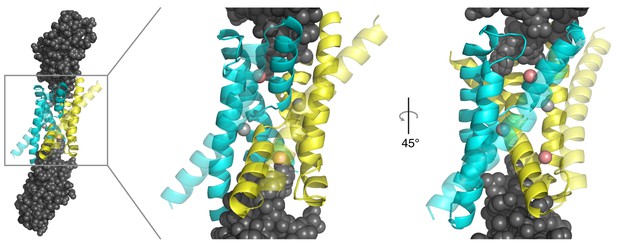
Monobody insertion into Fluc aqueous vestibules.
Structure of WT Ec2 channel (PDB #5A43), also depicting S9 monobodies (grey) bound to each vestibule.
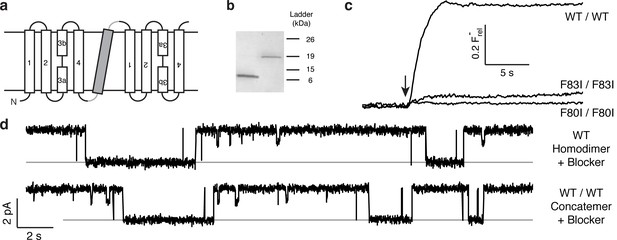
Proper assembly of fused-domain Ec2.
(a) Cartoon of fused-domain construct, showing numbered transmembrane helices in Ec2 domains (open) fused by an 'inversion linker' containing a non-dimerizing glycophorin A helix (grey). (b) Coomassie-stained SDS PAGE gel of purified Ec2 homodimer (left lane) and concatemer (right lane), with soluble-marker ladder positions indicated. (c) Representative F− efflux traces of indicated concatemeric constructs. (d) Representative single-channel recordings in the presence of 75–150 nM monobody, which induces long-lived nonconducting 'blocked' intervals; conducting intervals in these traces represent times when channels are free of monobody, with intrinsic open probability >95% (Stockbridge et al., 2014). Grey lines represent blocked current level.

Amino acid sequence of WT/WT Ec2 concatemer.
Fluc helices are indicated by red residues, and underlined residues indicate the artificial inversion linker, which includes a nondimerizing glycophorin A helix (orange residues). Lower case characters indicate the C-terminal His6 affinity tag and linker.
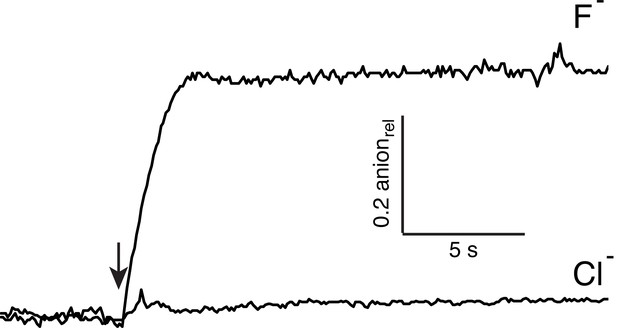
Concatemeric Ec2 retains F− selectivity.
Anion efflux traces shown for WT/WT concatemer, using liposomes loaded with 150 mM KF + 150 mM KCl. Efflux of the different ions was followed by F−- or Cl--selective electrodes.
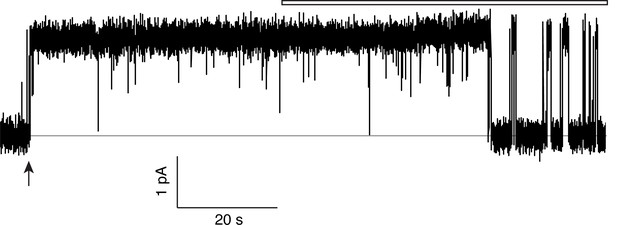
Monobody block to baseline.
Single-channel traces of the WT/WT Ec2 concatemer here are used to demonstrate that monobody fully blocks the concatemer. Planar bilayers were formed and voltage was set to 200 mV. Recordings were begun in the absence of monobody to catch the first channel insertion into the bilayer (arrow). Initial monobody-free measurements show the high open probability of the channel. At the start of the open bar, monobody (200 nM) was added to the solution without stirring, and discrete monobody blocks commenced after the reagent diffused into solution. The level of current before channel insertion is indistinguishable from the level of monobody block, observed over at least four such experiments.
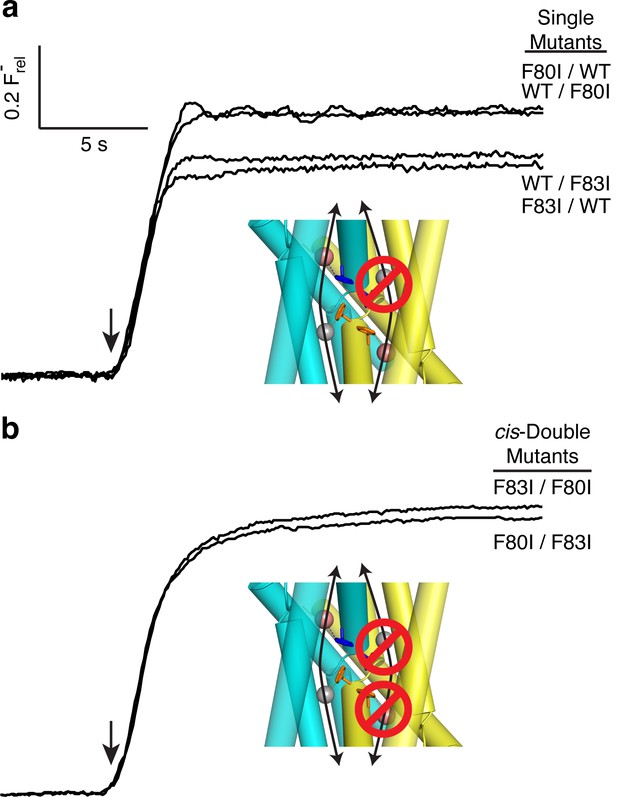
All-or-none assay for active F− channels in concatemers.
F− flux traces are shown for concatemeric constructs containing single (a) or double (b) mutants.
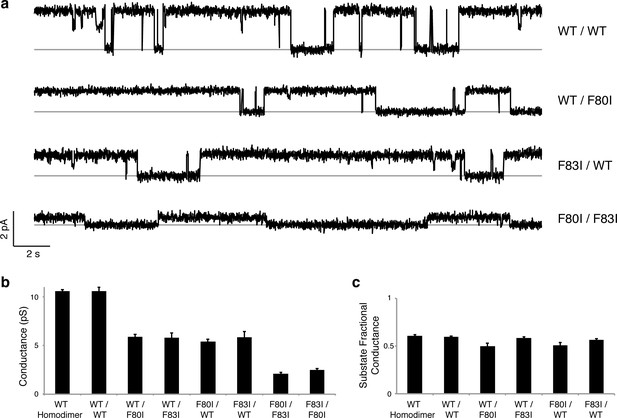
Single channels for single-pore mutants.
(a) Illustrative single-channel traces in the presence of monobody blocker for indicated concatemers with two WT pores, and with three classes of single-pore mutants. Grey lines mark blocked state. (b) Summary of single-channel conductances of all channels scoring active in (Figure 3 ). (c) Summary for indicated constructs of substate conductance normalized to the full conductance level of the individual constructs. cis-Double-mutant channels were too small for substate analysis. Bars represent mean + s.e. of 10–14 separate channels.
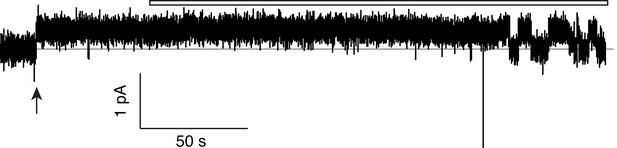
Monobody block to baseline.
Single channel trace of F80I/F83I concatemer initially in the absence and later in the presence (open bar) of 200 nM monobody, as in Figure 2—figure supplement 3. The double phenylalanine single-pore mutant recaptures the WT/WT double-pore protein’s high open probability in the absence of monobody, and complete block by monobody.
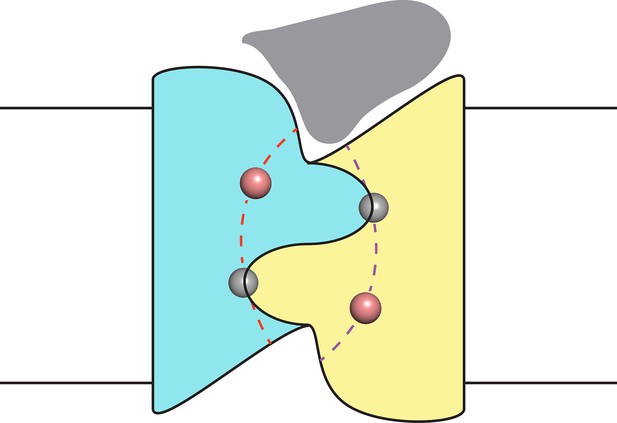
Proposal for trajectory of F− permeation pathways.
Cartoon envisions the dual-topology homodimeric channel with F− ions (spheres) in the selective pores (dashed curves) connecting the vestibules. Monobody is shown (grey) bound in one of the vestibules, as in blocking experiments here, such that it occludes both pores simultaneously.
Tables
Data collection and refinement statistics.
| F80I (PDB 5KBN) | F83I (PDB 5KOM) | |
|---|---|---|
| P41 | P41 | |
| cell dimensions | ||
| α, β, γ (°) | ||
| Resolution (Å) | 37.9–2.48 (2.58–2.48) | 38.0–2.69 (2.82–2.69) |
| Rmerge | 0.085 (1.42) | 0.083 (1.04) |
| I/σ | 20.4 (2.0) | 15.9 (2.0) |
| CC1/2 | 1.00 (0.851) | 1.00 (0.819) |
| Completeness | 0.999 (1.00) | 0.999 (1.00) |
| Multiplicity | 15.0 (15.2) | 7.4 (7.5) |
| Refinement Statistics | ||
| Resolution (Å) | 37.9–2.48 | 38.0–2.69 |
| No. Reflections | 36356 | 28695 |
| Rwork/Rfree | 0.219 / 0.239 | 0.217 / 0.230 |
| Ramachandran Favored | 0.972 | 0.982 |
| Ramachandran Outliers | 0 | 0 |
| RMS deviations | ||
| Bond Lengths (Å) | 0.0080 | 0.0084 |
| Bond Angles (°) | 1.19 | 1.28 |





