Oxytocin improves behavioral and electrophysiological deficits in a novel Shank3-deficient rat
Figures
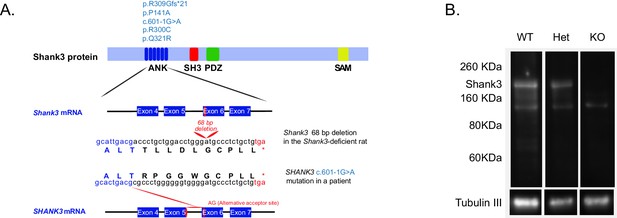
Gene-targeting of the rat Shank3 gene.
(A) The top schematic shows the Shank3 protein domains [ankyrin repeats domain (ANK), a Src homology 3 (SH3) domain, a PDZ domain, and a sterile α-motif (SAM) domain] and indicates the published de novo mutations observed within the ANK domain in PMS patients (light blue text, top schematic) (Durand et al., 2007; Moessner et al., 2007; Hamdan et al., 2011; Boccuto et al., 2013; Yuen et al., 2015). The 68 bp deletion that we introduced in exon 6 of the Shank3 rat gene (middle schematic) produces a stop codon in exon six that truncates the Shank3 protein. In a PMS patient, the c.601–1G>A mutation in intron 5 of the SHANK3 gene abolishes the normal acceptor site and leads to utilization of a cryptic acceptor site introducing a premature stop codon in exon 6 (red lines, bottom schematic), which also results in a similar truncated Shank3 protein (Hamdan et al., 2011). Lower-case letters, genomic sequence; upper-case letters, amino acids; *, premature stop codons. (B) Western blot showing anti-Shank3 staining, using antibodies targeted against the SH3 domain.
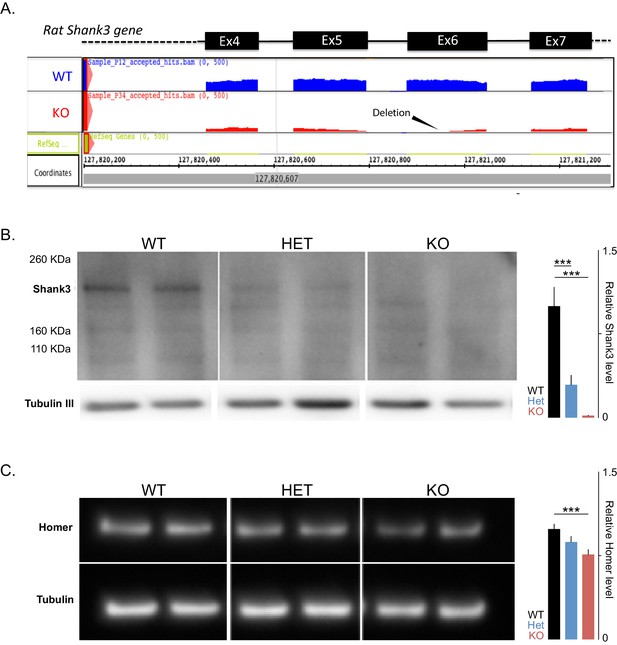
The introduced mutation in rat targets exon 6 and leads to reduced overall Shank3 transcripts and to decreased levels of the full-length Shank3 and Homer proteins.
(A) Schematic based on the integrative genome browser (IGB) output from RNA sequencing (RNAseq) of a wild-type (WT) and a knockout (KO) sample, showing the mapped reads at exons 4 to 7 of the Shank3 gene and the deletion in exon 6. (B) Left; Western blot analyses using anti-Shank3 antibodies (targeted toward amino acids 840–857) and anti-βIII-tubulin in PFC postsynaptic density (PSD) samples in WT and Shank3-heterozygous (Het) and Knockout (KO) rats. Right; quantification of Shank3 protein levels, based on Western blot analysis of three samples per genotype normalized to βIII-tubulin and then to the WT levels. (C) Left; Western blot analyses using anti-Homer-1b/c antibodies and anti-βIII-tubulin in PFC-PSD samples in WT and Shank3-Het and KO rats. Right; quantification of Homer protein levels, based on Western blot analysis of three samples per genotype normalized to βIII-tubulin and then to the WT levels. ***, p<0.001.
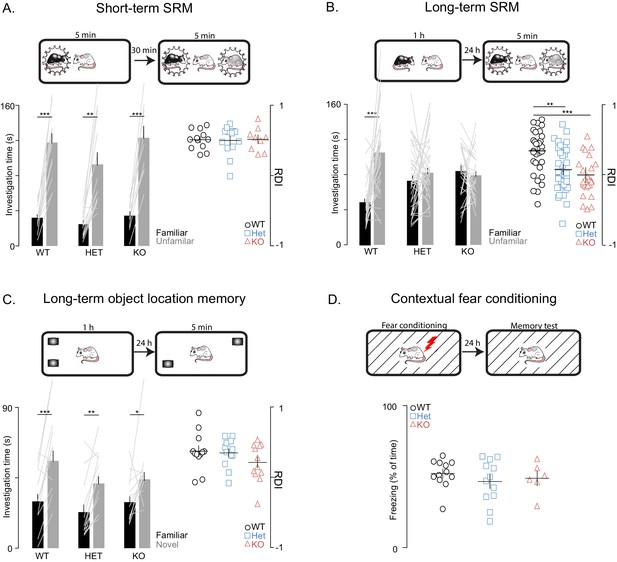
Shank3-deficient rats exhibit deficits in social memory.
(A–B) Above each figure are schematics of the short- and long-term social discrimination (SD) paradigms. The examined adult subject is shown in white, and the juveniles are shown in black and grey. Bar plots show behavior in short-term (WT, n = 12; Het, n = 13; KO, n = 9) and long-term (WT, n = 31; Het, n = 37: KO, n = 25) SD tests. For long-term SD, similar results were observed in two independent cohorts, therefore the results were pooled and presented here as a single cohort. Bars ± SEM at left show test subjects average investigation time of a familiar and unfamiliar juvenile rat. The light overlaid gray lines in A and B show the corresponding individual subject data that comprise each bar. Right scatter plots, presented with mean ± SEM, show the ratio of the investigation time (RDI= (Unfamiliar-Familiar)/(Unfamiliar+Familiar) for individual subjects. (C) Above the figure is a schematic of the long-term object location memory paradigm. The same plotting conventions as bar plots in A and B are used, but here they quantify investigation times of an object (WT, n = 12; Het, n = 12; KO, n = 12) in a novel or familiar location. (D) Above, Contextual fear conditioning paradigm schematic. Scatter plots (mean ± SEM) represent percent time freezing during retrieval of a 1-day-old conditioned fear memory (WT, n = 12; Het, n = 12; KO, n = 6). *, p<0.05, **, p<0.01, ***, p<0.001; see Supplementary file 1 for detailed statistical results.
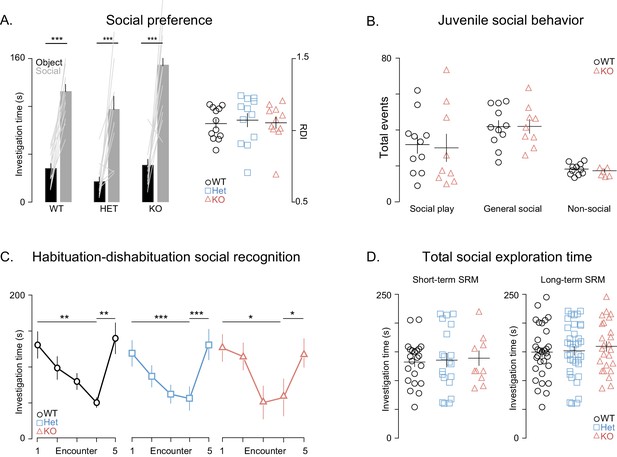
General social behaviors are unaffected in Shank3-deficient rats.
(A) Bars ± SEM show the average investigation times of simultaneously presented juvenile rat (gray bars) and object (black bars) for WT (n = 12), Shank3-Het (n = 12) and KO (n = 11) during the social preference test. The light overlaid gray lines show the corresponding individual subject data that contribute to each bar. Scatter plots on the right show the ratio of these investigation times (RDI = (Social- object)/ (Social+ object) for individual subjects and the group mean ± SEM in thin overlaid black lines. (B) Same plot conventions as in A, but here points show individual rat data for the number of social and non-social behaviors during the juvenile social play assessment in WT (left, n = 11) and KO rats (right, n = 11). (C) Performance of WT (n = 11), Shank3-Het (n = 11) and Shank3-KO (n = 10) rats on the habituation-dishabituation social recognition, see Materials and methods for a detailed description of this task. (D) Identical plotting conventions as in the scatter plots in Figure 2A,B but points show the total investigation time (the sum of investigation time of both the familiar and unfamiliar juvenile rat) for each individual subject on the short-term SRM (left) and long-term SRM (right) paradigms. *p<0.05, **p<0.01, ***p<0.001. See Materials and methods section and Supplementary file 1.
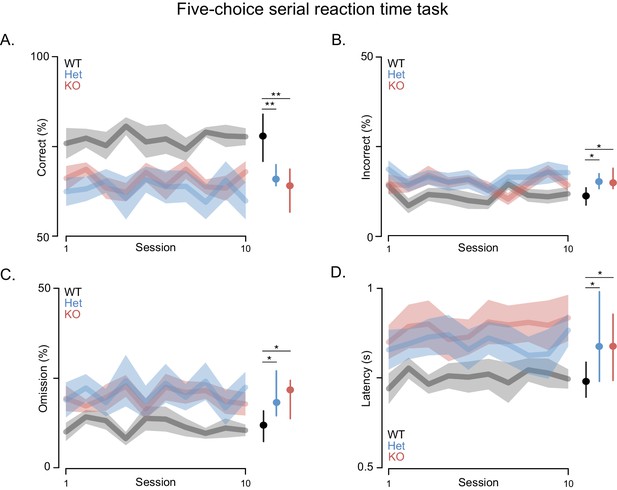
Shank3-deficient rats exhibit deficits in attention.
(A) Traces and clouds indicate mean percentage of trials with a correct response ± SEM (WT, n = 10; Het, n = 13; KO, n = 12) across 10 5-CSRT sessions. The right side in all panels is the cross-rat median (dot) and middle quartiles (vertical lines). (B) Traces represent mean percentage of trials where an incorrect response was made. (C) Mean percentage of trials with no cued response. (D) Average reaction times on trials with a correct response. Results were observed in two independent cohorts; therefore, the results were pooled.
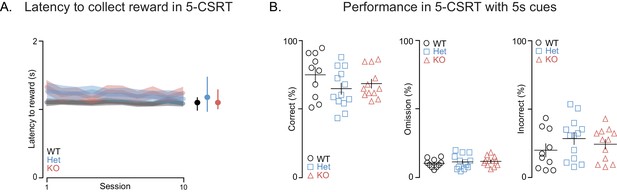
Shank3-deficient rats exhibit normal motivation for reward and task performance is at WT levels when cue durations are long.
(A) Traces and clouds show the average latency ± SEM from a correct screen touch to entry into a reward receptacle for WT (black, n = 10), Shank3 Het (blue, n = 13), or KO rats (red, n = 12) across ten 5-CSRT sessions. (B) Scatter plots, presented with mean ± SEM, show percent of correct, incorrect, or omitted trials for each individual subject during this 5-CSRT test training stage. In this 5-CSRT training stage, the light cues remain on for 5 s. *p<0.05, **p<0.01; See Supplementary file 1 for detailed statistical results.
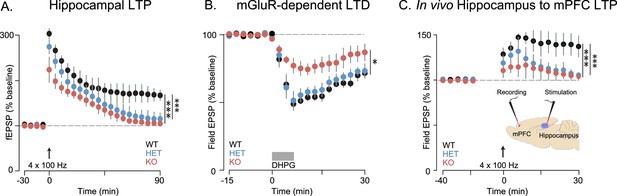
Synaptic plasticity is impaired in Shank3-deficient rats.
(A) High-frequency stimulation (HFS, arrow)-induced long-term potentiation (LTP) at hippocampal Schaffer collateral-CA1 synapses (n = 6 rats/genotype, 1–2 slices per rat). (B) Long-term depression induced by the mGluR agonist DHPG (50 µM, 5 min) is indicated by the horizontal line (n = 6 rats/genotype, six slices per rat). (C) HFS-induced LTP in the prelimbic PFC after stimulation of ipsilateral CA1 in ventral hippocampus of intact anesthetized WT (n = 5), Shank3 Het (n = 6) and KO (n = 6) rats. Inset shows a schematic of the target location of the stimulating and recording electrodes in vivo. Summary data are presented as mean ± SD. *p<0.05, ***p<0.001; See Supplementary file 1 for detailed statistical results.
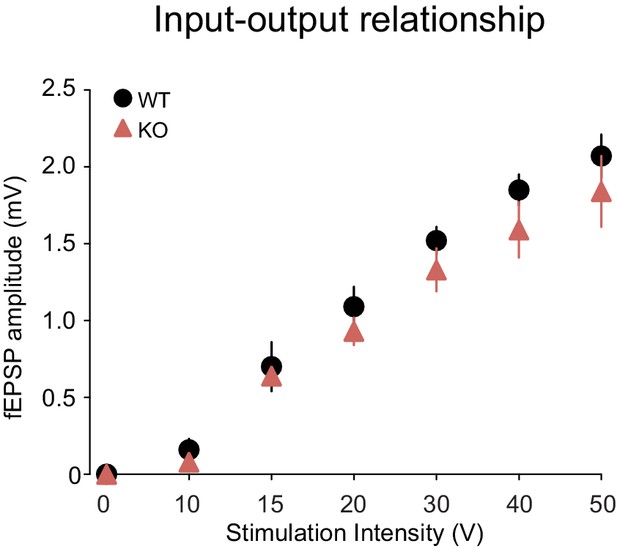
Basal synaptic transmission is intact in Shank3-deficient rats.
Input-output relationship at Schaffer collateral-CA1 synapses (n = 5 rats/genotype, three slices per rat). Summary data are presented as mean ± SD.
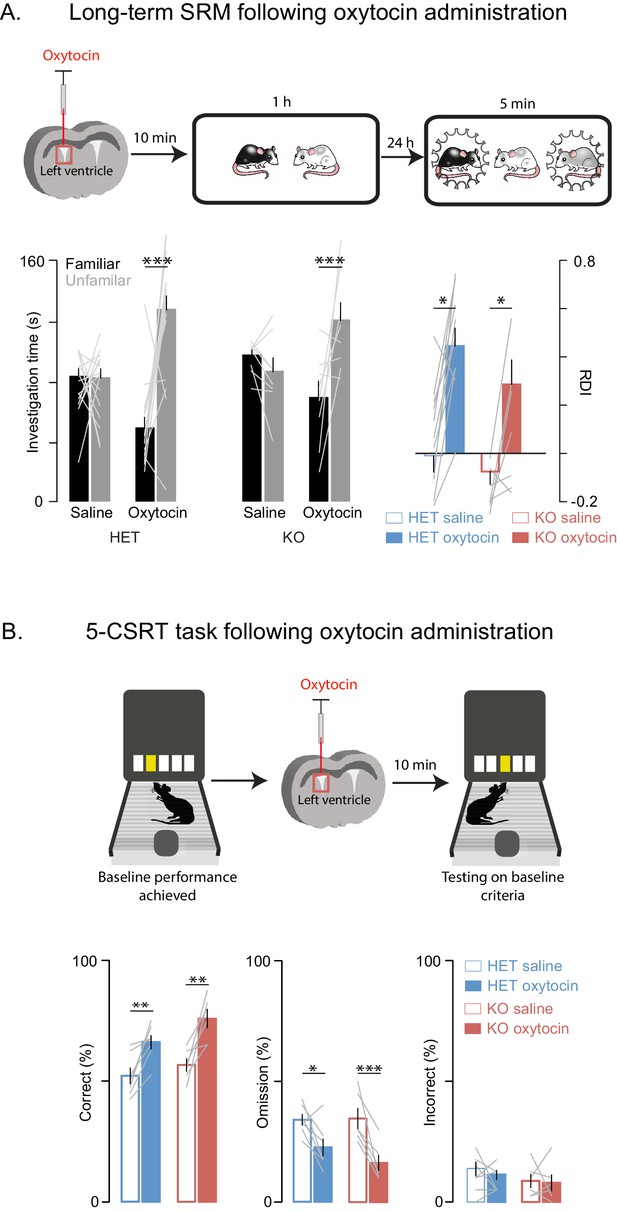
Oxytocin improves social memory and attentional deficits in Shank3-deficient rats.
Above each figure is a schematic depicting the sequence of oxytocin administration and behavioral testing on the long-term social discrimination (SD) paradigm (in A) or 5-CSRT task (in B). In A, the test subject is shown in white and the juveniles are shown in black and grey. (A) Left bars (± SEM) show the test subject’s average investigation time of a familiar and unfamiliar juvenile rat on long-term SD test following treatment with oxytocin or vehicle for Shank3 Het (n = 15) and KO (n = 8) rats. The light overlaid gray lines show the corresponding individual subject data that comprise each bar. Bars on right show the ratio of the investigation time (RDI= (Unfamiliar-Familiar)/(Unfamiliar+Familiar) for individual subjects with the light overlaid gray lines representing the corresponding individual subject data. (B) Bars (± SEM) represent the percentage of correct (left), omitted (middle), and incorrect (right) trials in saline (solid bars) and oxytocin (open bars) of Shank3 Het (n = 7) and KO (n = 6) rats. Color conventions are identical to those used in Figure 3A–C. The light overlaid gray lines show the corresponding individual subject data that went into each bar. *p<0.05, **p<0.01, ***p<0.001; See Supplementary file 1 for detailed statistical results.
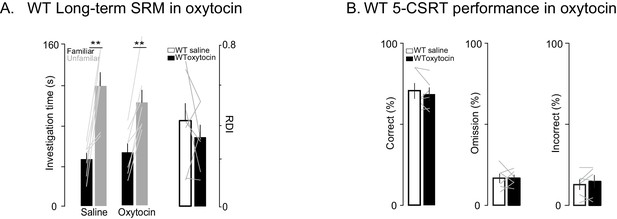
Oxytocin has no effect on social memory or attention in WT rats.
(A) Performance on the long-term social discrimination test following treatment with oxytocin or vehicle in WT rats (n = 7). Plotting conventions are identical to the bar plots in Figure 2A–B. (B) Bars ± SEM represent the percentage of correct (left), omitted (middle), and incorrect (right) trials in saline (solid bars) and oxytocin (open bars) of WT rats (n = 6). Color conventions are identical to those used in Figure 3A–D. The light overlaid gray lines in A and B show corresponding individual subject data. *p<0.05, **p<0.01, ***p<0.001.
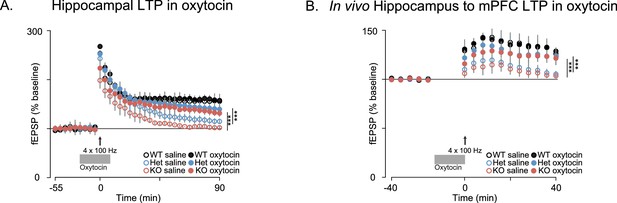
Oxytocin improves synaptic plasticity deficits in Shank3-deficient rats.
(A) Traces depict Hippocampal HFS-induced LTP (4 × 100 Hz) in WT and Shank3-deficient rats (n = 4 rats/genotype, six slices per rat). Application of 1 µM of oxytocin is indicated by the horizontal line. (B) LTP in the hippocampal to prefrontal synaptic pathway recorded in vivo in WT (n = 3), Shank3 Het (n = 4) and KO (n = 4) rats. Oxytocin (2 ng) or saline was administered in the lateral ventricle 5 min before LTP induction. ***p<0.001. See Supplementary file 1 for detailed statistical results.
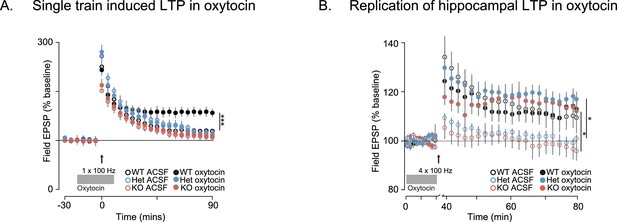
Oxytocin enhances LTP induced by a single pulse at 100 Hz in WT but not in Shank3-deficient rats.
(A) Traces show HFS-induced LTP (1 × 100 Hz) recorded from hippocampal slices in WT and Shank3-deficient rats (n = 4 rats/genotype, six slices per rat). Brain slices were treated with oxytocin (1 µM, indicated by the horizontal line). (B) These traces summarize the results from independent replication of the results shown in Figure 6A. The plotting conventions are identical as in Figure 6A, but here Ns in ACSF and oxytocin are; WT, n = 4 rats/5 slices per rat; Het, n = 4 rats/4 slices per rat; KO, n = 5 rats/6 slices per rat. Application of 1 µM of oxytocin is indicated by the horizontal line. See Supplementary file 1 for summary statistics. *p<0.05, ***p<0.001.
Additional files
-
Supplementary file 1
Summary of all experimental and statistical results.
- https://doi.org/10.7554/eLife.18904.015





