Perisomatic GABAergic synapses of basket cells effectively control principal neuron activity in amygdala networks
Figures
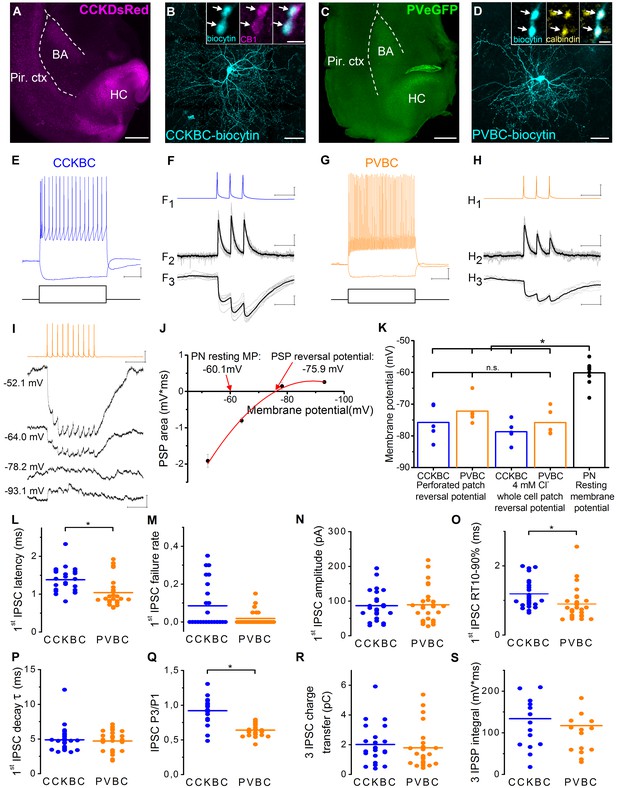
Similar magnitude and decay kinetics characterize the unitary inhibitory connections in CCKBC- and PVBC-PN pairs.
(A, C) Images of horizontal slices taken from a CCK-DsRed and a PV-eGFP mouse, respectively, containing the basolateral amygdala region. Positions of the recorded BCs are shown in Figure 1—figure supplement 2. (B) Maximum z intensity projection image of a biocytin-filled CCKBC. Inset: Axon terminals of CCKBCs (blue) are immunopositive for type one cannabinoid receptor (CB1, magenta). (D) Maximum z intensity projection image of a biocytin-filled PVBC. Inset: Varicosities of PVBCs (blue) are immunopositive for calbindin. For more detailed neurochemical characterization of CCK-DsRed and PV-eGFP cells see Figure 1—figure supplement 1. (E and G) Representative voltage responses of a CCKBC (E, 400, –100 pA) and a PVBC (G, 300, –100 pA) upon step current injections. (F and H) Representative postsynaptic current (PSC, F2, H2) and postsynaptic potential (PSP, F3, H3) recordings in a CCKBC-PN (F) or in a PVBC-PN pair (H) in response to three action potentials evoked at 30 Hz (F1, H1). Ten superimposed consecutive traces in grey, averages in black. (I), PSPs recorded in a PN at different membrane potentials in gramicidin-based perforated patch recordings upon a train of 10 action potentials evoked at 40 Hz in a presynaptic PVBC. (J) The integral of the summed PSPs was calculated at each membrane potential and fitted with a second order polynomial curve to determine the reversal potential of the responses. (K) Comparison of the estimated reversal potential of PSP recorded in perforated patch and whole-cell patch-clamp mode using 4 mM Cl- intrapipette solution and the resting membrane potential of PNs revealed the inhibitory nature of the BC connections (CCKBC: perforated patch mode: −75.74 ± 2.44 mV, whole cell mode: −78.67 ± 1.55 mV, p=0.296, n = 5–5 pairs; PVBC: perforated patch mode: −72.17 ± 1.88 mV; whole cell mode: −75.79 ± 1.90 mV, p=0.403, n = 5–5 pairs, both M-W test; PN resting membrane potential: −60.13 ± 1.09 mV). (L–S) Comparison of basic electrophysiological properties of IPSCs (L–R), and IPSPs (S) at the output synapses of CCKBCs and PVBCs recorded in whole-cell mode. Horizontal line represents mean. For data see Figure 1—source data 1. Scales: A and C: 0.5 mm; B and D: 50 µm, insets: 2 µm; E and G: 10 mV, 200 ms; F and H x: 50 ms, F1 and H1 y: 30 mV, F2 and H2 y: 50 pA, F3 and H3 y: 1 mV, I: 0.5 mV (PN), 30 mV (BC) and 100 ms.
-
Figure 1—source data 1
Basic electrophysiological properties of the output synapses of CCKBCs and PVBCs recorded in whole-cell mode.
- https://doi.org/10.7554/eLife.20721.003
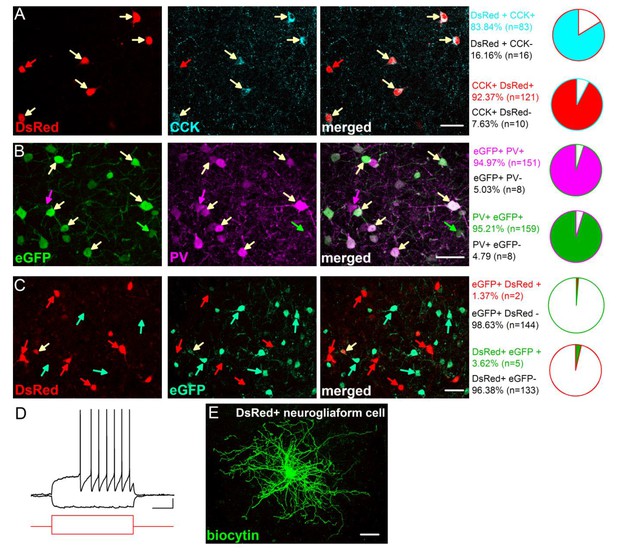
Characterization of the CCK-DsRed and PV-eGFP cells in the BA.
(A) Quantification of the overlap between DsRed and CCK immunostaining in CCK-DsRed mice (n = 3 mice). (B) Quantification of the overlap between eGFP and PV immunostaining in PV-eGFP mice (n = 2 mice). (C) Quantification of the overlap between eGFP and DsRed expression in PV-eGFPxCCK-DsRed double transgenic mice (n = 2 mice). A small portion of DsRed and eGFP expressing cells are neurogliaform cells (2–3% in both populations), which have small soma and can be readily recognized by their characteristic late-response firing pattern (D) and dense dendritic and axonal arbor (E) as described previously (Mańko et al., 2012). These cells were excluded from the study. Scale bar: A-C: 40 µm, E: 50 µm, D: 10 mV and 200 ms, step current injection: + 150 and −100 pA.
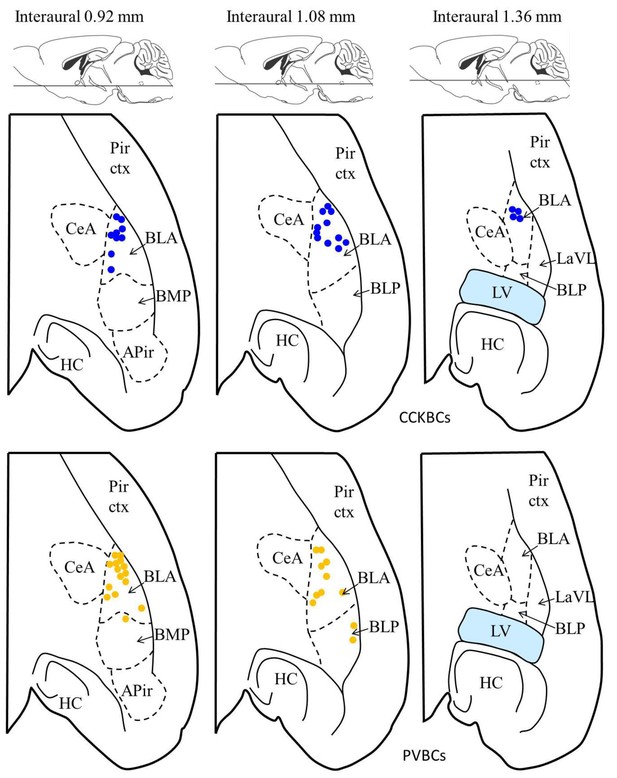
Location of the presynaptic interneurons of the paired recordings in the BA.
Blue dots represent CCKBCs (upper row), orange dots represent PVBCs (lower row). Figures modified from Paxinos and Franklin (2012). Abbreviations: APir: amygdalopiriform transition area, BLA: basolateral amygdaloid nucleus, anterior part, BLP: basolateral amygdaloid nucleus, posterior part, BMP: basomedial amygdaloid nucleus, posterior part, CeA: central amygdaloid nucleus, HC: hippocampus, LaLV: lateral amygdaloid nucleus, ventrolateral part,LV: lateral ventricle, Pir ctx: piriform cortex.
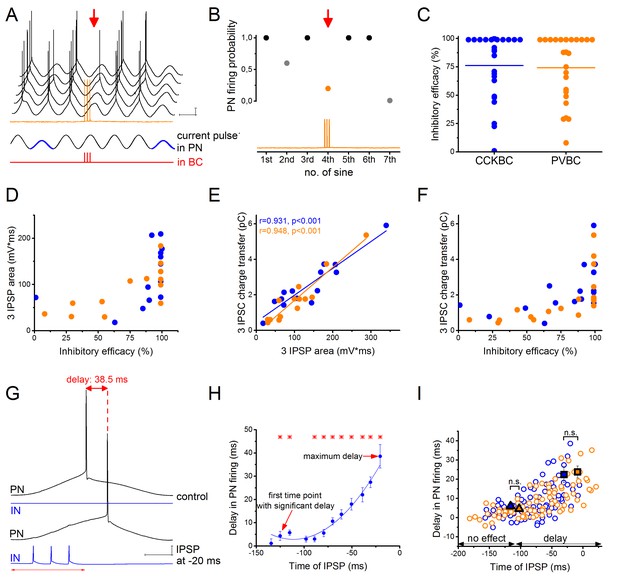
CCKBCs and PVBCs inhibit PN firing with similar efficacy.
(A) Representative experiment for testing the capability of a BC to control spike generation in a PN. Sinusoidal current trains were injected into a PN at a theta frequency (3.53 Hz) to initiate firing and three action potentials were evoked at 30 Hz in the presynaptic PVBC 30–40 ms before the peak of the fourth cycle (for details see Materials and methods). Red arrow points to the cycle, when the presynaptic BC was stimulated. Voltage traces are offset for clarity. (B) Summary data of experiments shown in panel A: the firing probability of the PN was significantly suppressed within the cycle when the action potential train was evoked in the presynaptic PVBC. (C) Comparison of the inhibitory efficacy of CCKBCs and PVBCs. The inhibitory efficacy shows the probability of the suppression of action potential generation in the PN by BC firing (i.e.: 1- [PN firing probability during the control sinusoidal current cycles - firing probability when the BC is stimulated]) CCKBC: 76.2 ± 7.1%, n = 25, PVBC: 75.6 ± 6.5%, n = 25, p=0.88, M-W test. (D) The relationship between the inhibitory efficacy and the area of IPSPs. (E) The area of the IPSPs and the charge transfer of the IPSCs are strongly correlated. (F) The relationship between the inhibitory efficacy and IPSC charge transfer, indicating a larger inhibitory efficacy at a larger charge transfer. (G) Representative experiment for testing BCs ability to postpone PN firing. The timing of the evoked action potential in a BC was systematically shifted relative to the peak of the sinusoidal current injected into the PN. (H) The delay in PN firing as a function of the timing of synaptic inhibition in the pair shown in G. Asterisks indicate significant delay in firing compared to the peak of the cycle during the control period (paired sample Wilcoxon signed rank test p<0.05). (I) Pooled data from 7 CCKBC and 6 PVBC pairs. Squares show the average maximal delay (CCKBC: 22.40 ± 4.14 ms, n = 7, PVBC: 23.76 ± 3.16 ms, n = 6, p=0.72, M-W test) and triangles show the average last time point with significant delay in PN firing (CCKBC: −116.21 ± 14.47 ms, PVBC: −102.41 ± 18.58 ms, p=0.94, M-W test). Color codes are the same as in panel C (blue: CCKBCs, orange: PVBCs). Scales: A: 10 mV, 200 ms; G: 10 mV, 50 ms.
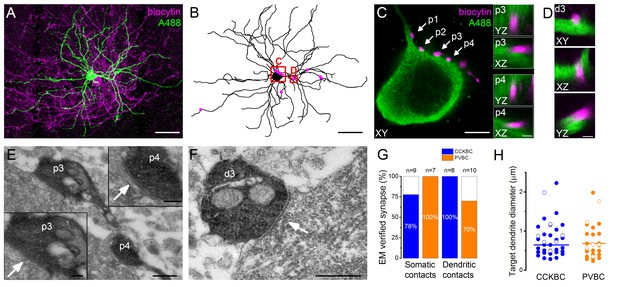
Morphological analysis of the synaptic connections in the recorded pairs.
(A) Maximum z intensity projection of a 3D confocal image of a representative pair. Biocytin in the PVBC was visualized with streptavidin-conjugated Alexa 594 (magenta), while Alexa 488 in the PN is shown in green. For the differentiation of Alexa488 and eGFP signals see Figure 3—figure supplement 1. (B) Neurolucida reconstruction of the postsynaptic PN with the contact sites (magenta) originated from the PVBC. (C) High power magnification of the perisomatic region shown in B, which receives multiple contacts (arrows, perisomatic contact #1–4: p1-p4) from the presynaptic PVBC (magenta). Insets: 3D analysis of confocal images of p3 and p4 boutons shows close appositions of the pre- and postsynaptic structures. (D) High power magnification 3D confocal images of the region indicated in panel B, showing a close apposition between a dendrite-targeting bouton (d3) and a PN dendrite. (E) Electron micrographs of p3 and p4 shown in panel C. In both cases synaptic contacts with the soma of the postsynaptic PN (arrows) are visible. (F) An electron micrograph of d3 shown in panel D, confirming the presence of synaptic contact on the dendrite of the postsynaptic PN (arrow). (G) Ratio of close appositions observed at the confocal microscopic level forming synaptic contacts confirmed with electron microscopy. (H) Electron microscopic analysis of the diameters of the dendrites innervated by BCs shows that both BC types form synapses on dendrites with small and large diameters, respectively (CCKBCs: 0.79 ± 0.07 µm, n = 28, PVBC: 0.72 ± 0.08 µm, n = 36, p=0.63, K-S test). Open circles show contacts on the electrophysiologically recorded postsynaptic PNs, filled dots represent synapses on random targets of the biocytin-labeled varicosities of BCs in the sample. Scales (in µm): A and B: 50; C: 5, insets and D: 1; E: 0.5, insets: 0.2; F: 0.5. For the details of the analyzed contact numbers see Figure 3—source datas 1 and 2.
-
Figure 3—source data 1
Number of the pairs and analyzed contacts at the confocal microscopic level.
- https://doi.org/10.7554/eLife.20721.008
-
Figure 3—source data 2
Summary table of the contacts analyzed at the electron microscopic level.
Data is shown as the number of the analyzed contacts, with the number of verified synapses shown in parentheses.
- https://doi.org/10.7554/eLife.20721.009
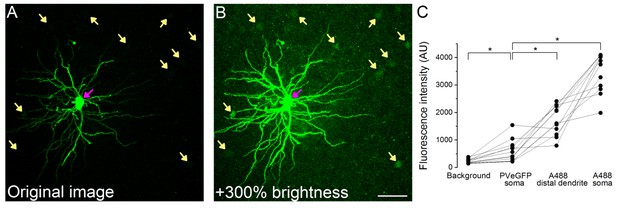
Subcellular compartments of Alexa488 filled and subsequently immunolabeled cells are readily distinguishable from eGFP signal in PV cell somata.
(A) Original maximum z-intensity projection image of an Alexa488 labeled cell (magenta arrow) used for the analysis of the inhibitory contacts (labeled interneuron is not shown for clarity). (B) Same image with +300% brightness correction to show the faint eGFP signal (yellow arrows). (C) Comparison of the fluorescence intensity in the different structures (n = 12 A488 labeled PNs, in each Figure 5 distal dendritic segments, 5 eGFP labeled cells and five background areas were averaged). Note that 4095 AU value represents detector saturating intensity values. Scale bar: 50 µm.
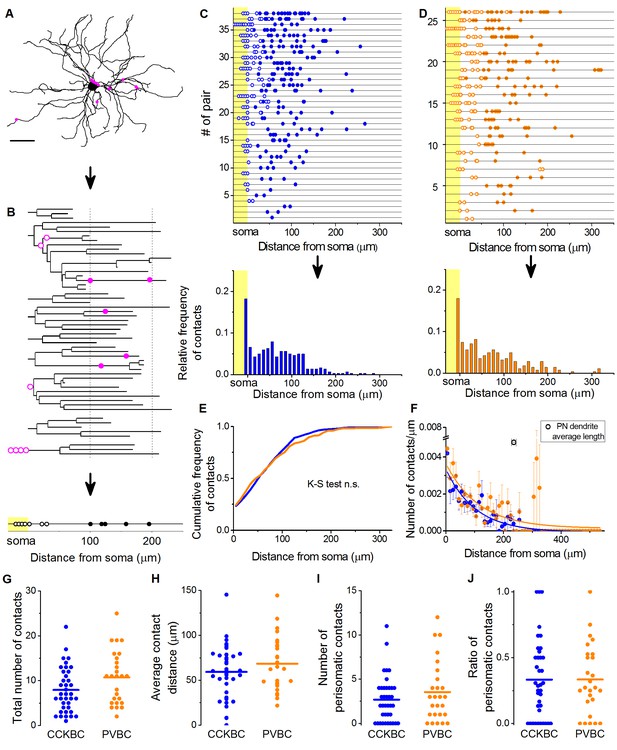
CCKBCs and PVBCs similarly innervate the soma-dendritic membrane surface of PNs.
(A and B) Neurolucida reconstruction of the postsynaptic PN (same as in Figure 3) and its dendrogram analysis showing the contact sites on the perisomatic region (open magenta dots) and on the dendritic branches (filled magenta dots). Bottom on B: schematic visualization of the innervation pattern compressed along a single horizontal line. Somatic boutons shown in the yellow area, contacts on the perisomatic region are labeled with open dots and dendritic boutons with filled dots along the line indicating their distance from the soma. (C and D) Schematic innervation pattern of the reconstructed 38 CCKBC-PN and 26 PVBC-PN pairs. Bottom: Spatial distribution of the contacts, data from all pairs pooled together. (E) Comparison of the cumulative distribution of the contacts on PNs from the two BC types show no difference in the innervation patterns (p=0.29, K-S test). (F) BCs innervate the whole length of PN dendrites, however, the density of the contacts decreases exponentially towards the end of the dendrites (CCKBCs: r = 0.884, tau: 71.4 ± 8.2 µm; PVBCs: r = 0.557, tau: 79.7 ± 23.4 µm). Boutons contacting the cell bodies were excluded from this analysis. (G–J) Comparison of the total number of boutons (CCKBC: 7.92 ± 0.80, PVBC: 10.73 ± 1.16, p=0.065), the average bouton distance (CCKBC: 59.20 ± 4.80 µm, PVBC: 68.39 ± 5.97, p=0.38), the number of perisomatic boutons (CCKBC: 2.68 ± 0.41, PVBC: 3.53 ± 0.67, p=0.44) and ratio of perisomatic boutons (CCKBC: 0.33 ± 0.04, PVBC: 0.33 ± 0.05, p=0.83) of CCKBC-PN and PVBC-PN connections showed no significant difference (all M-W test, n = 38 CCKBC-PN pairs and n = 26 PVBC-PN pairs). Scale in A: 50 µm.
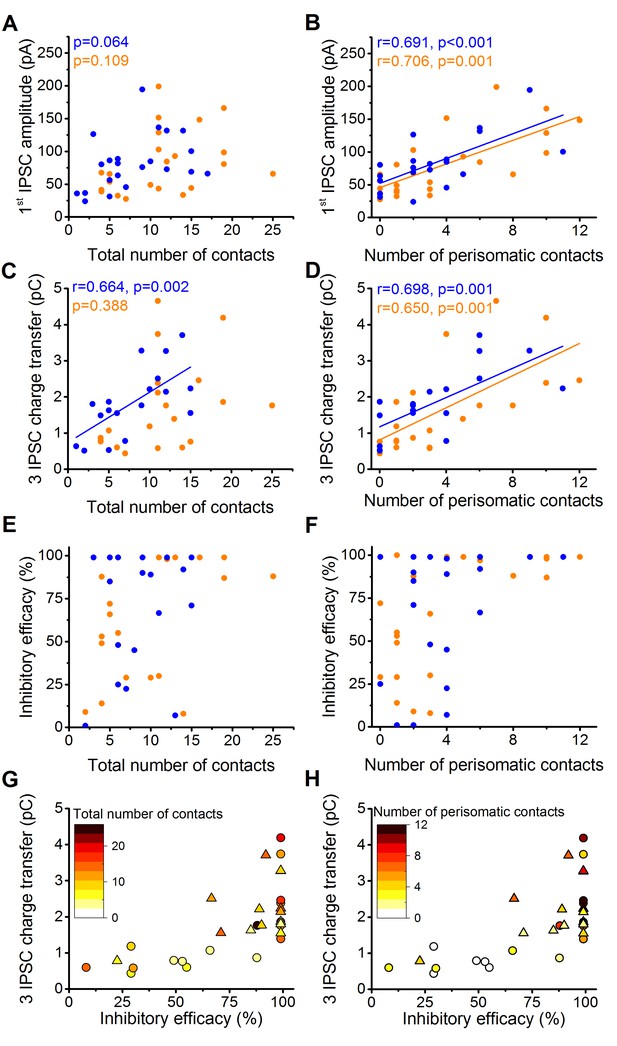
The magnitude of synaptic events and inhibitory efficacy show correlation with the number of the perisomatic contacts but not with the total number of contacts at individual connections.
(A–F) Relationship between the structural and electrophysiological properties of the output synaptic contacts in CCK-PN (blue) and PVBC-PN (orange) pairs. Note that the IPSC amplitude, charge transfer and inhibitory efficacy show stronger correlation with the number of the perisomatic contacts than with the total number of contacts. Linear correlation is labeled with a solid line. Pearson's r values and significance levels of correlations are indicated in the top left corner of the plots. (G and H) Relationship between the inhibitory efficacy and IPSC charge transfer, plotted together with the total number (G) and number of perisomatic contacts (H) as a heat map showing that the inhibitory efficacy is rather dependent on the number of the perisomatic contacts than on the total number of contacts. On panel G and H triangles mark CCKBCs, circles label PVBCs. For additional analysis see Figure 5—figure supplements 1,2.
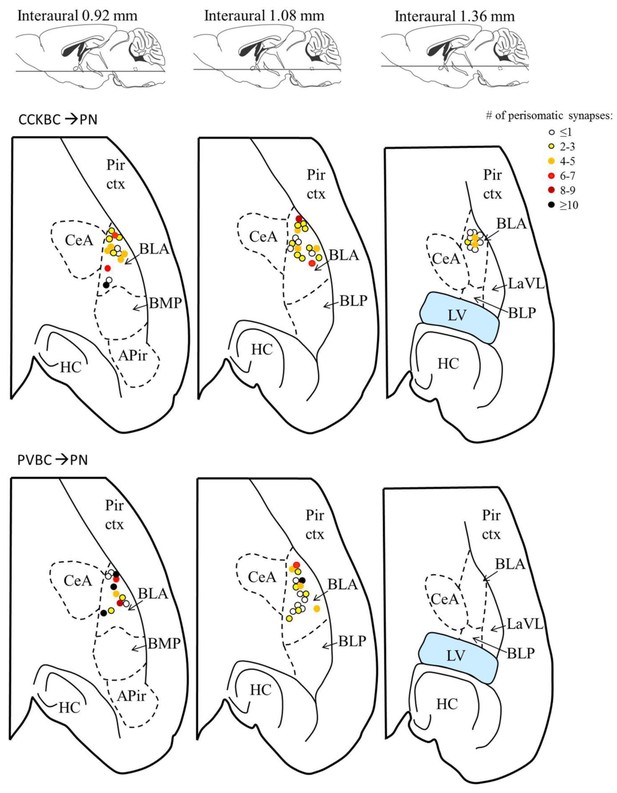
The number of contacts on the perisomatic region of the postsynaptic PN is independent of the location of the target cell within the BA.
Each dot represents the location of the postsynaptic PN sampled using paired recordings (data from Figure 5), the color of a dot codes the number of perisomatic contacts from the presynaptic basket cell. Upper row: CCKBC→PN pairs, lower row: PVBC→ PN pairs. Figures modified from Paxinos and Franklin (2012). Abbreviations: APir: amygdalopiriform transition area, BLA: basolateral amygdaloid nucleus, anterior part, BLP: basolateral amygdaloid nucleus, posterior part, BMP: basomedial amygdaloid nucleus, posterior part, CeA: central amygdaloid nucleus, HC: hippocampus, LaLV: lateral amygdaloid nucleus, ventrolateral part, LV: lateral ventricle, Pir ctx: piriform cortex.
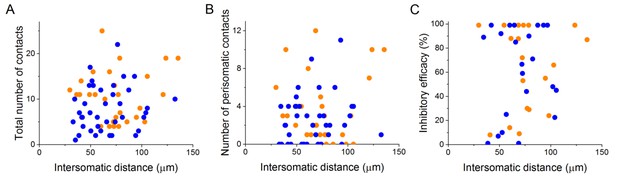
Properties of the connections are independent of the distance between the pre- and postsynaptic cells, at least below 150 µm.
Analysis of the intersomatic distance and the total number of contacts (A), number of perisomatic contacts (B) or inhibitory efficacy (C) shows no correlation (Pearson correlation, p>0.05).
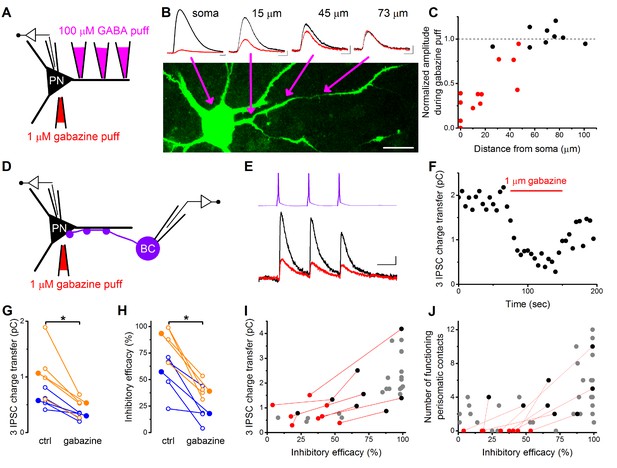
Pharmacological elimination of perisomatic contacts significantly reduces the inhibitory efficacy.
(A) Schematic drawing of the experiment determining the area affected by local somatic gabazine application. (B) Representative recordings of the GABA puff evoked currents at various dendritic locations in control conditions (black) and upon somatic gabazine application (red). (C) Summary of recordings from 22 dendritic locations show that the somatic gabazine puff can significantly reduce GABAergic currents (red dots) in an area of 50 µm radius from the site of the application, which covers the perisomatic region. (D) Schematic drawing of the experiment measuring the effect of the elimination of perisomatic contacts by local somatic gabazine application. (E and F) Representative experiment showing the rapid and substantial drop in IPSC charge transfer upon gabazine application (applied for 75 s in panel F). (G and H) Elimination of perisomatic inhibitory inputs resulted in a significant suppression of the IPSC charge transfer and inhibitory efficacy (p=0.01 and p=0.007, respectively, Wilcoxon Signed Rank test). Blue: CCKBC-PN pairs, orange: PVBC-PN pairs. Open circles represent recorded pairs, filled circles represent median values. (I and J) Relationship between the inhibitory efficacy and IPSC charge transfer (I) or the number of functioning perisomatic contacts (J) before (black dots) and after (red dots) the pharmacological elimination of perisomatic inhibition (individual experiments connected with red line). Grey dots show data from previous recordings for comparison (data from Figures 2F and 5F). Scales: B: 20 pA and 50 ms; 20 µm, E: 8 pA and 20 ms.
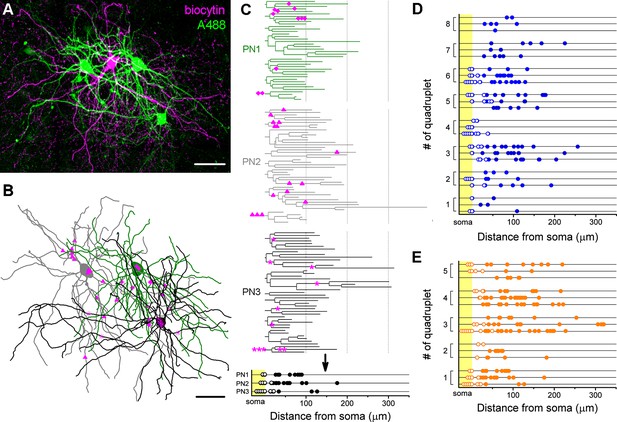
Target distribution of CCKBCs and PVBCs on multiple synaptic partners.
(A) Maximum z intensity projection of a confocal image of a representative quadruplet. A single PVBC was filled with biocytin (visualized by Alexa 594, magenta) and three monosynaptically connected PNs (confirmed with electrophysiological recordings) were consecutively filled with Alexa 488 (green). (B) Neurolucida reconstruction of the three postsynaptic PNs (green) with the contact sites marked in magenta. Diamonds are contacts on PN1, triangles on PN2, stars on PN3. For the reconstructions the same method was used as in Figure 4. (C) On the dendrograms of the three reconstructed postsynaptic PNs the contact sites are marked. Bottom: innervation patterns of the presynaptic PVBC on the three postsynaptic PNs. (D and E) Innervation pattern of the reconstructed quadruplets (n = 8 CCKBC-PNs and 5 PVBC-PN quadruplets). Scales: A and B: 50 µm.
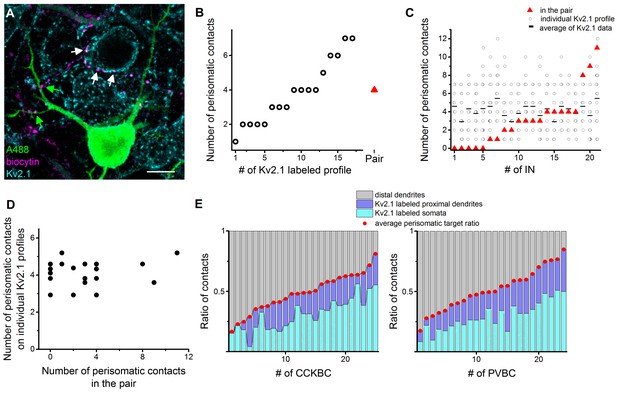
Comparison of the innervation patterns of CCKBCs and PVBCs at single-cell and population levels.
(A) Kv2.1 immunostaining (blue) was used to label the perisomatic region of cells in slices where BC-PN pairs were visualized (biocytin in BC (magenta), and Alexa 488 in PN (green)). Contact sites from the same BC were identified on the intracellularly-labeled PN (green arrows) and on Kv2.1-expressing profiles (white arrows), enabling the investigation of the innervation pattern at both single-cell and population levels. (B) Analysis of the bouton distribution of a representative biocytin-labeled BC (shown in panel A) on 17 Kv2.1-labeled PNs. Data are arranged based on the number of biocytin-filled boutons contacting their perisomatic region (circles). For comparison, the number of the contacts formed on the intracellularly labeled postsynaptic PN is shown with a red triangle. (C and D) Summary data of the innervation patterns of 21 BCs showing that the number of the established contacts at the single-cell level (on the biocytin-filled postsynaptic PN) is independent of the average number of the contacts determined at population levels (i.e., on multiple Kv2.1-labeled PNs). (D) Data in C is arranged as a function of increasing contact number obtained in the pairs. (E) Target distribution analysis of CCKBCs and PVBCs at the population level obtained with Kv2.1 staining: ratio of contacts on the soma, proximal dendrites belonging to the perisomatic region and distal dendrites. Average perisomatic target ratio is shown with red dots. Data is arranged as a function of increasing ratio of contacts on the perisomatic region. Scale: 10 µm. For additional analysis see Figure 8—figure supplement 1.
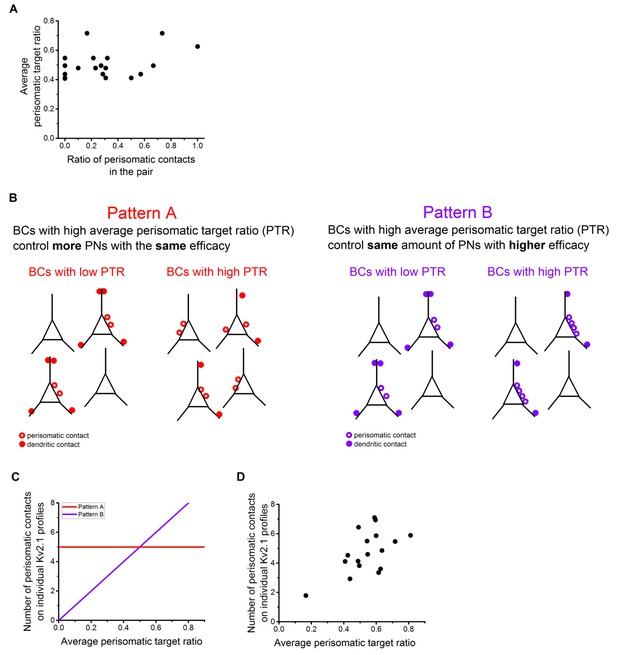
Possible innervation pattern strategies for PVBCs and CCKBCs.
(A) The ratio of perisomatic contacts at single-cell and population levels shows no correlation. (B) Schematics of two different possible innervation pattern strategies. In pattern A, BCs with high perisomatic target ratio (PTR) innervate more PNs than those BCs having low PTR. In pattern B, BCs with high PTR innervate the same amount of PNs than BCs with low PTR, but with more contacts on the perisomatic region. This implies a higher inhibitory efficacy on those PNs. (C) The hypothetical correlation between the number of the perisomatic contacts and average perisomatic target ratio in pattern A (red) and B (purple). (D) Correlation between the number of the perisomatic contacts and average perisomatic target ratio in the experimental dataset. This plot suggests that BCs in the BA might follow innervation pattern ‘B’, implying that those BCs which target the perisomatic region with high ratio at the population level, tend to form more contacts on the perisomatic region of individual PNs, therefore having a more powerful controlling effect on the postsynaptic neuron activity.





